Insights on the Origin of Vitrified Rocks from Serravuda, Acri (Italy): Rock Fulgurite or Anthropogenic Activity?
Abstract
:1. Introduction
2. Materials and Methods
2.1. Geologic Setting of the Outcrop of Fused Rocks at Serravuda Hill
2.2. Sample Collection
- (a)
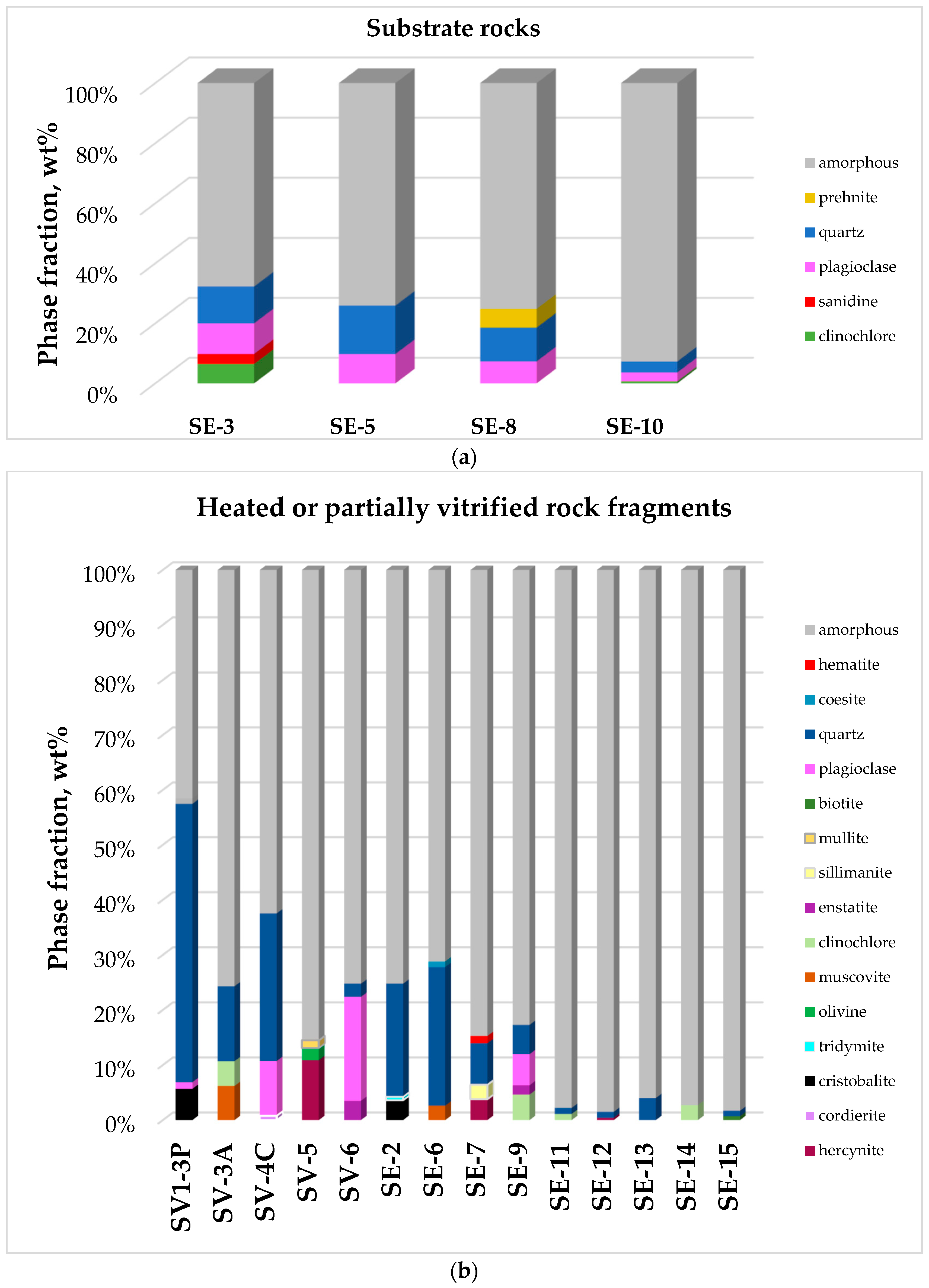
- Substrate rocks outcropping on the top and on the flanks of the Serravuda hill. Field observations show that rocks outcropping on the top of the Serravuda hill consist of whitish-light colored minerals macroscopically identified as quartz and plagioclase. Substrate rocks show limited melting or heat-alteration processes. Sample SE 3 is a reddish weathered granitoid rock collected on the top of a neighboring hill. SE 5, SE 8, and SE 10 samples are medium-large grained granitoid rocks that were previously identified by Bertolani [1] as migmatites.
- (b)
- Heated or partially vitrified rock fragments collected along the northern and north-west flanks of the flat top of the Serravuda hill. This rock set includes a variety of different materials (about 10 cm average size) present as loose fragments on the top of the Serravuda hill. Colorless or greenish vitreous material was observed as thin crust covering some pebbles. SV 4-C, SE 6, SE 11, SE 12, SV 1-3P samples are granitoid that were subjected to intense heating effects with mineral fracturing and partial fusion of the rock. SV 6, SE 9, SV 3-A, SV 4-C, SV 4-V, SV 5, SV 1-3P, SV 1-3V were extracted from two distinct cemented blocks labelled 1 (12 kg, Figure 1a) and C (25 kg, Figure 1b) collected at the NW flanks of the Serravuda hill. SV4C and SV1-3P represent mostly or poorly molted material from SV4 and SV1 fragments, SV 4-V is mainly glassy material present as a crust partially covering SV4 fragment. SE 9, SE 14, and SE 15 samples of dark-brown medium-high grade biotite rich-gneisses were collected along the N-W flanks of the Serravuda hill. The products of the fusion consist of reddish-black vitreous materials in veins veinlets inside the fragments and external crusts or pockets. Lithologies of the heated or partially vitrified rock fragments are mixed from parent rocks not outcropping on the top of the hill such as hypersilicic rocks (quartzite, SE 13), mica-rich gneisses (SV 3-A, SE 7, SE 9, SE 14, SE 15) and amphibolite (SV 5, SV 6).
- (c)
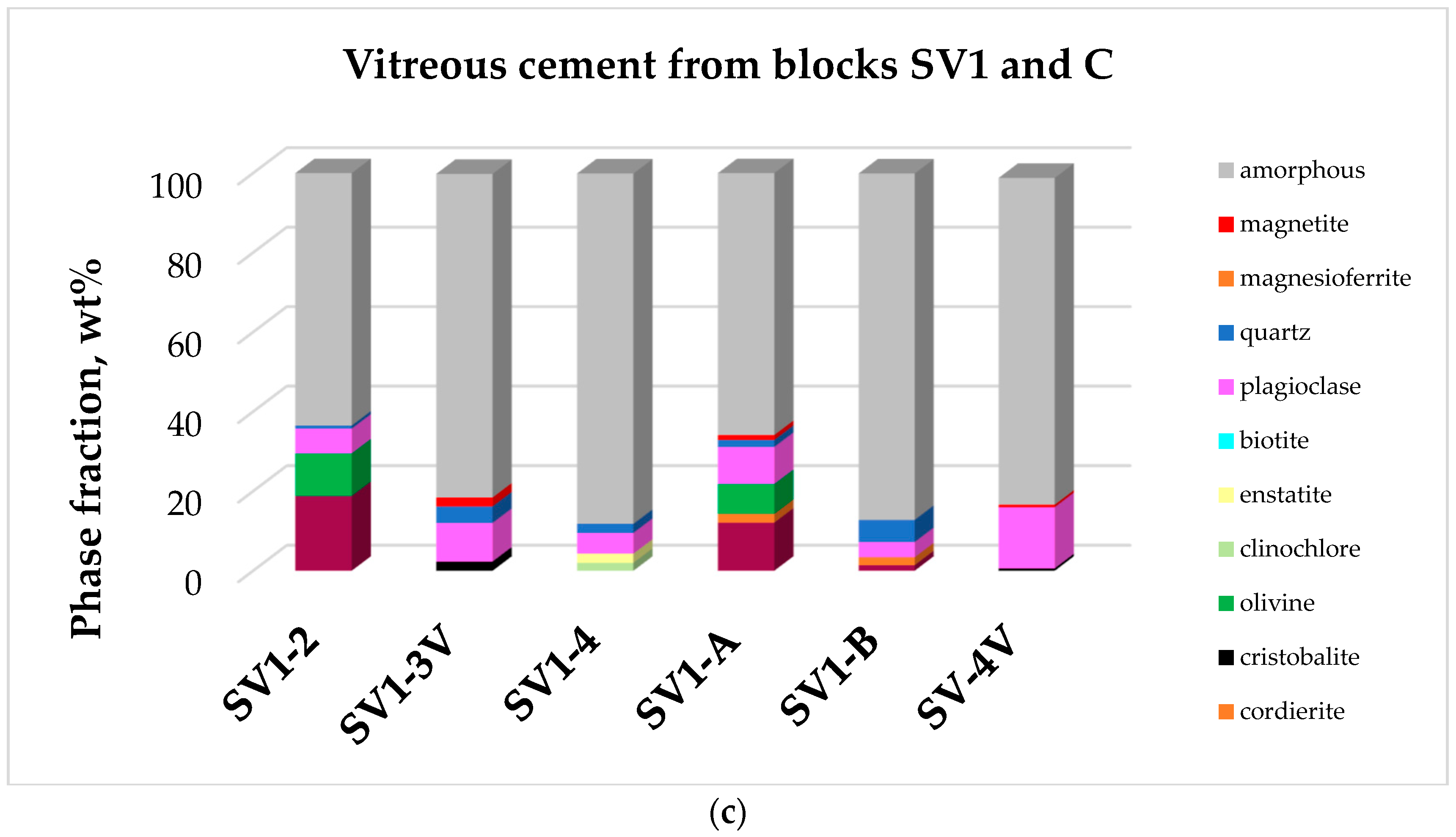
- Dark–vitreous materials present as large veins or pockets (5–10 cm average size) cementing SV 1 and C rock fragments. Samples in this set were extracted (separated) from SV 1 and C where they are present as veins and pockets. Samples labelled SV 1-3V and SV 4-V are glassy crusts covering SV 1 and SV 4 loose rock fragments. Sample SV 1-A is a massive black-glass with few vesicles. SV 1-B is a vesicular glass rich with crystalline inclusions.
2.3. Sample Preparation
2.4. X-ray Powder Diffraction (XRPD)
2.5. Scanning Electron Microscopy/Energy Dispersive X-ray Spectroscopy (SEM/EDS)
3. Results
4. Discussion
5. Prospective Origin of Serravuda Vitrified Rocks
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bertolani, M. An enigmatic outcrop of vitrified rocks near Acri (Cosenza). Boll. Della Soc. Geol. Ital. 1972, 91, 683–692. [Google Scholar]
- Bertolani, M.; Foggia, F. La formazione kinzigitica della Sila Greca. Ital. J. Geosci. 1975, 94, 329–345. [Google Scholar]
- Doglioni, C.; Flores, G. An Introduction to the Italian Geology; Il Salice: Potenza, Italy, 1994. [Google Scholar]
- Degen, T.; Sadki, M.; Bron, E.; König, U.; Nénert, G. The Highscore suite. Powder Diffr. 2014, 29, S13–S18. [Google Scholar] [CrossRef] [Green Version]
- Whitney, D.L.; Evans, B.W. Abbreviations for names of rock-forming minerals. Am. Mineral. 2010, 95, 185–187. [Google Scholar] [CrossRef]
- Matano, F.; Di Nocera, S. Weathering patterns in the Sila Massif (Northern Calabria Italy). Ital. J. Quat. Sci. 1999, 12, 141–148. [Google Scholar]
- Le Pera, E.; Critelli, S.; Sorriso-Valvo, M. Weathering of gneiss in Calabria, Southern Italy. CATENA 2001, 42, 1–15. [Google Scholar] [CrossRef]
- Scarciglia, F.; Saporito, N.; La Russa, M.F.; Le Pera, E.; Macchione, M.; Puntillo, D.; Crisci, G.M.; Pezzino, A. Role of lichens in weathering of granodiorite in the Sila Uplands (Calabria, Southern Italy). Sediment. Geol. 2012, 280, 119–134. [Google Scholar] [CrossRef]
- Scarciglia, F.; Critelli, S.; Borrelli, L.; Coniglio, S.; Muto, F.; Perri, F. Weathering profiles in granitoid rocks of the Sila Massif Uplands, Calabria, Southern Italy: New insights into their formation processes and rates. Sediment. Geol. 2016, 336, 46–67. [Google Scholar] [CrossRef]
- Biondino, D.; Borrelli, L.; Critelli, S.; Muto, F.; Gullà, G. The interplay of structural pathway and weathering intensity in forming mass-wasting processes in deeply weathered gneissic rocks (Sila Massif, Calabria, Italy). J. Maps 2018, 14, 242–256. [Google Scholar] [CrossRef] [Green Version]
- Schipper, C.I.; Rickard, W.D.A.; Reddy, S.M.; Saxey, D.W.; Castro, J.M.; Fougerouse, D.; Quadir, Z.; Conway, C.; Prior, D.J.; Lilly, K. Volcanic SiO2-cristobalite: A natural product of chemical vapor deposition. Am. Mineral. 2020, 105, 510–524. [Google Scholar] [CrossRef]
- Horwell, C.J.; Williamson, B.J.; Llewellin, E.W.; Damby, D.E.; Le Blond, J.S. The nature and formation of cristobalite at the Soufrière Hills Volcano, Montserrat: Implications for the petrology and stability of silicic lava domes. Bull. Volcanol. 2013, 75, 696. [Google Scholar] [CrossRef] [Green Version]
- Darling, R.S.; Chou, I.-M.; Bodnar, R.J. An occurrence of metastable cristobalite in high-pressure garnet granulite. Science 1997, 276, 91–93. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Holmquist, S.B. Conversion of quartz to tridymite. J. Am. Ceram. Soc. 1961, 44, 82–86. [Google Scholar] [CrossRef]
- Cole, S.S. The conversion of quartz into cristobalite below 1000 °C and some properties of cristobalite formed. J. Am. Ceram. Soc. 1935, 18, 149–154. [Google Scholar] [CrossRef]
- Dapiaggi, M.; Pagliari, L.; Pavese, A.; Sciascia, L.; Merli, M.; Francescon, F. The formation of silica high temperature polymorphs from quartz: Influence of grain size and mineralising agents. J. Eur. Ceram. Soc. 2015, 35, 4547–4555. [Google Scholar] [CrossRef] [Green Version]
- Davis, R.F. Mullite. In Concise Encyclopedia of Advanced Ceramic Materials; Brook, R., Ed.; Pergamon: Oxford, UK, 1991; pp. 315–317. [Google Scholar]
- Ban, T.; Okada, K. Cristallization of mullite and immiscibility in SiO2–Al2O3 system. In Advanced Materials ’93; Mizutani, N., Akashi, K., Kimura, T., Ohno, S., Yoshimura, M., Maruyama, T., Saito, Y., Przybylski, K., Stringer, J., Kawamura, H., et al., Eds.; Elsevier: Amsterdam, The Netherlands, 1994; pp. 483–486. [Google Scholar]
- Li, X.; Wang, H.; Zhou, Q.; Qi, T.; Liu, G.; Peng, Z.; Wang, Y. Reaction behavior of kaolinite with ferric oxide during reduction roasting. Trans. Nonferrous Met. Soc. China 2019, 29, 186–193. [Google Scholar] [CrossRef]
- Minowa, S.; Yamada, M.; Kato, M. The study on the formation and reduction of hercynite (FeAl2O4). Tetsu-to-Hagane 1965, 51, 2309–2321. [Google Scholar] [CrossRef] [Green Version]
- Sedmale, G.; Sperberga, I.; Sedmalis, U.; Valancius, Z. Formation of high-temperature crystalline phases in ceramic from illite clay and dolomite. J. Eur. Ceram. Soc. 2006, 26, 3351–3355. [Google Scholar] [CrossRef]
- de Brito, I.P.; de Almeida, E.P.; de Araújo Neves, G.; de Lucena Lira, H.; Menezes, R.R.; da Silva, V.J.; de Lima Santana, L.N. Development of cordierite/mullite composites using industrial wastes. Int. J. Appl. Ceram. Technol. 2021, 18, 253–261. [Google Scholar] [CrossRef]
- Grapes, R.H. (Ed.) Quartzofeldspathic Rocks. In Pyrometamorphism; Springer: Berlin/Heidelberg, Germany, 2006; pp. 41–113. ISBN 978-3-540-29454-2. [Google Scholar]
- Baboolal, A.A.; Knight, J.; Wilson, B. Petrography and mineralogy of pyrometamorphic combustion metamorphic rocks associated with spontaneous oxidation of lignite seams of the Erin formation, Trinidad. J. S. Am. Earth Sci. 2018, 82, 181–192. [Google Scholar] [CrossRef]
- Jung, R.; Pacciarelli, M.; Zach, B.; Klee, M.; Thanheiser, U. Punta di Zambrone (Calabria)—A bronze age harbour site. First preliminary report on the recent bronze age (2011–2012 campaigns). Archaeol. Austriaca 2015, 99, 53–110. [Google Scholar] [CrossRef]
- Chen, B.; Wang, Y.; Franceschi, M.; Duan, X.; Li, K.; Yu, Y.; Wang, M.; Shi, Z. Petrography, mineralogy, and geochemistry of combustion metamorphic rocks in the Northeastern Ordos Basin, China: Implications for the origin of “white sandstone”. Minerals 2020, 10, 1086. [Google Scholar] [CrossRef]
- Butler, J.R. Pyrometamorphism. In Petrology. Encyclopedia of Earth Science; Springer: Boston, MA, USA, 1990; pp. 491–492. [Google Scholar]
- Ippolito, F. Before the Iron Age: The Oldest Settlements in the Hinterland of the Sibaritide (Calabria, Italy). Ph.D. Thesis, Rijksuniversiteit Groningen, Groningen, The Netherlands, 2016. [Google Scholar]
- Sevink, J.; de Neef, W.; Vito, M.A.D.; Arienzo, I.; Attema, P.A.; van Loon, E.E.; Ullrich, B.; den Haan, M.; Ippolito, F.; Noorda, N. A multidisciplinary study of an exceptional prehistoric waste dump in the mountainous inland of Calabria (Italy): Implications for reconstructions of prehistoric land use and vegetation in Southern Italy. Holocene 2020, 30, 1310–1331. [Google Scholar] [CrossRef]
- Gualtieri, M. Fortifications and settlement organization: An example from pre-roman Italy. World Archaeol. 1987, 19, 30–46. [Google Scholar] [CrossRef]
- Berrocal-Rangel, L.; García-Giménez, R.; Ruano, L.; Vigil de la Villa, R. Vitrified walls in the iron age of Western Iberia: New research from an archaeometric perspective. Eur. J. Archaeol. 2019, 22, 185–209. [Google Scholar] [CrossRef]
- McCloy, J.S.; Marcial, J.; Clarke, J.S.; Ahmadzadeh, M.; Wolff, J.A.; Vicenzi, E.P.; Bollinger, D.L.; Ogenhall, E.; Englund, M.; Pearce, C.I.; et al. Reproduction of melting behavior for vitrified hillforts based on amphibolite, granite, and basalt lithologies. Sci. Rep. 2021, 11, 1272. [Google Scholar] [CrossRef]
- Youngblood, E.; Fredriksson, B.J.; Kraut, F.; Fredriksson, K. Celtic vitrified forts: Implications of a chemical-petrological study of glasses and source rocks. J. Archaeol. Sci. 1978, 5, 99–121. [Google Scholar] [CrossRef]
- Ralston, I. The Yorkshire television vitrified wall experiment at East Tullos, city of Aberdeen District. In Proceedings of the Society of Antiquaries of Scotland; Society of Antiquaries of Scotland: Edinburgh, UK, 1987; Volume 116, pp. 17–40. [Google Scholar]
- Friend, C.R.L.; Dye, J.; Fowler, M.B. New field and geochemical evidence from vitrified forts in South Morar and Moidart, NW Scotland: Further insight into melting and the process of vitrification. J. Archaeol. Sci. 2007, 34, 1685–1701. [Google Scholar] [CrossRef]
- Wadsworth, F.B.; Heap, M.J.; Dingwell, D.B. Friendly fire: Engineering a fort wall in the iron age. J. Archaeol. Sci. 2016, 67, 7–13. [Google Scholar] [CrossRef]
- Wadsworth, F.B.; Heap, M.J.; Damby, D.E.; Hess, K.-U.; Najorka, J.; Vasseur, J.; Fahrner, D.; Dingwell, D.B. Local geology controlled the feasibility of vitrifying iron age buildings. Sci. Rep. 2017, 7, 40028. [Google Scholar] [CrossRef] [Green Version]
- Lock, G.; Ralston, I. (Eds.) Hillforts: Britain, Ireland and the Nearer Continent: Papers from the Atlas of Hillforts of Britain and Ireland Conference, June 2017; Archaeopress Publishing Ltd.: Oxford, UK, 2019; ISBN 978-1-78969-227-3. [Google Scholar]



| Sample | Locality | Lithology | Description |
|---|---|---|---|
| Substrate Basement Rocks | |||
| SE 3 | top of an elevation 50 m N-E from the Serravuda hill | granitoid | Reddish fine-grained weathered granitoid |
| SE 5 | top of Serravuda hill | granitoid | Light-gray coarse-grained granitoid, partially weathered |
| SE 8 | top of the Serravuda hill | granitoid | Light gray medium-grained granitoid, with whitish hydrothermal veins (calcite?) |
| SE 10 | top of Serravuda hill | granitoid | Light-gray coarse-grained, partially weathered granitoid |
| Heated or Partially Vitrified Rock Fragments | |||
| SV 3A | Sample from cemented block C | gneiss | Medium-sized (10 cm) reddish, coarse-grained mica rich-gneiss pebble with minor fracturing. |
| SV 4C | Sample from cemented block C | granitoid | SV4-C is a coarse-grained strongly brecciated rock fragment (quartz-rich foliated granitoid) with thin black veins (glass) localized in the fracture plains. |
| SV 5 | Sample from cemented block C | amphibolite | SV5 is a large (>15 cm) dark-grey, strongly fractured rock pebble apparently unaffected by melting. |
| SV 6 | Large (>15 cm) fragment from cemented block C | amphibolite | Light-gray coarse-grained strongly fractured pebble with reddish coating. |
| SE 2 | top of Serravuda hill | granitoid | Loose large pebble (>20 cm) light-gray, quartz-rich rock showing intense fracturing and pervasive veins of dark vitreous material. |
| SE 6 | top of Serravuda hill | granitoid | Loose medium-sized (<15 cm) light-gray feldspar-rich pegmatitic granitoid pebble, intensely fractured but no evidence of melting |
| SE 7 | top of Serravuda hill | gneiss | Loose, medium-sized (<15 cm), dark heavy foliated gneiss pebble, intensely fractured but with no evidence of melting |
| SE 9 | NE flank of the Serravuda hill | gneiss | Greenish/gray foliated rock rich in phyllosilicates (biotite-chlorite gneiss?). |
| SE 11 | top of Serravuda hill | granitoid | Loose, medium-sized (10 cm) reddish pebble of granitoid with minor fracturing and no evidence of melting |
| SE 12 | top of Serravuda hill | granitoid | Loose, medium-sized (5–10 cm) dark-grey pebble, intensively fractured |
| SE 13 | top of Serravuda hill | quartzite | Loose, medium-sized (5–10 cm) reddish, coarse-grained quartz-rich pebble, totally covered by a fusion crust. |
| SE 14 | NE flank of the Serravuda hill | gneiss | Dark-gray foliated phyllosilicate-rich rock (kinzigitic gneiss) |
| SE 15 | NE flank of the Serravuda hill | gneiss | Dark-gray foliated phyllosilicate-rich rock (kinzigitic gneiss) |
| SV1 3P | Sample from cemented block SV1 | granitoid | SV1-3P is a medium sized (5–10 cm), light-colored, coarse-grained quartz-rich granitoid, intensely fractured pebble covered by a dark vitreous crust. |
| Vitreous Cements from Block SV1 and C | |||
| SV 1-2 | Selected from cemented block SV1 | glass | Black colored, slightly vesicular glass containing abundant relics of light-colored minerals (plagioclase and quartz) or newly formed coarse-grained (pegmatitic) mineral phases. |
| SV 1-3V | Sample from cemented block SV1. | glass | Dark vitreous matrix englobing sample SV1-3P. |
| SV 1-4 | Sample from cemented block SV1 | glass | Black, vesicular glass containing abundant relics of light-colored coarse-grained mineral phases (plagioclase and quartz). |
| SV 1-A | Sample from cemented block SV1. | glass | Composite sample of dark, dense, not vesicular vitreous material accumulated in a large pocket (up to 15 cm diameter) or deposited along void channels, containing abundant light-colored crystals (plagioclase and quartz). |
| SV 1-B | Sample from cemented block SV1. | glass | Composite sample of dark, strongly vesicular vitreous material usually deposited as pockets and incrustations in cavities within rock fragments. |
| SV 4-V | Sample from cemented block C | glass | Dark, strongly vesicular vitreous material associated to and partially covering sample SV4-C. |
| Sample | Mineral Concentration (wt%) | Amorphous Concentration (wt%) |
|---|---|---|
| Substrate rocks | ||
| SE 3 | Qz 12.3, Pl 10.2, Clc 6.5, Sa 3.3 | 67.7 |
| SE 5 | Qz 16.1, Pl 9.7, Ms 0.5, Clc 0.1 | 73.7 |
| SE 8 | Qz 11.3, Pl 7.3, Prh 6.2, Clc 0.1 | 75.2 |
| SE 10 | Qz 5.3, Pl 5.6, Clc 0.7, Bt 0.1 | 75.2 |
| Heated of Partially Vitrified Rock Fragments | ||
| SV 1-3P | Qz 50.9, Crs 5.7, Pl 1.2, Crd 0.2, Hr 0.1 | 41.8 |
| SV 3-A | Qz 13.6, Ms 6.2, Clc 4.5 | 75.7 |
| SV 4-C | Qz 26.8, Pl 9.9, Cdr 0.8 | 62.4 |
| SV 5 | Hr 10.9, Ol 2.2, Mul 1.4 | 82.2 |
| SV 6 | Pl 18.8, Aug 3.5, Qz 2.4, Ky 0.4 | 75.0 |
| SE 2 | Qz 20.4, Crs 3.6, Trd 0.6, Crd 0.4, Hr 0.3 | 74.7 |
| SE 6 | Qz 25.1, Ms 0.5, Coe 1.0, Bt 0.2, Crs 0.1 | 71.0 |
| SE 7 | Qz 7.5, Hr 3.8, Sil 2.6, Hem 1.3, Crs 0.3 | 84.4 |
| SE 9 | Qz 5.3, Pl 5.6, Clc 4.6, En 1.7, B 0.3 | 82.3 |
| SE 11 | Qz 1.1, Clc 1.1, Bt 0.5 | 97.3 |
| SE 12 | Qz 1.1, Hr 0.4, Crs 0.2, Cdr 0.2 | 98.2 |
| SE 13 | Qz 4.0, Crs 0.2 | 95.8 |
| SE 14 | Clc 2.7, Bt 0.4, Hc 0.1 | 96.9 |
| SE 15 | Qz 1.00, Bt 0.7, Clc 0.1 | 98.1 |
| Vitreous Cements from Blocks SV1 and C | ||
| SV 1-2 | Hc 18.8, Ol 10.7, Pl 6.2, Qtz 0.8 | 63.5 |
| SV 1-3V | Pl 9.7, Qz 4.2, Crs 2.3, Mag 2.2 | 81.4 |
| SV 1-4 | Pl 5.2, Qz 2.3, Ens 2.4, Bt 0.1 | 88.1 |
| SV 1-A | Hr 12.1, Pl 9.3, Ol 7.5, Crd 2.2, Qz 1.8, Mag 1.2 | 65.9 |
| SV 1-B | Qz 5.6, Pl 3.8, Crd 2.1, Hr 1.4, Crs 0.1 | 87.1 |
| SV 4-V | Pl 15.4, Qz 0.6, Crs 0.6, Mag 0.6, Mfr 0.6 | 82.2 |
| Plagioclase | Olivine | Plagioclase | Olivine | |||
|---|---|---|---|---|---|---|
| wt% | apfu | |||||
| n | 6 | 4 | ||||
| SiO2 | 65.91 | 39.41 | Si | 3.13 | 1.24 | |
| TiO2 | 5.33 | 2.22 | Ti | 0.19 | 0.05 | |
| Al2O3 | 16.67 | 18.01 | Al | 0.93 | 0.67 | |
| FeO(tot) | 4.11 | 20.22 | Fe3+ | 0.00 | 0.00 | |
| MgO | 1.35 | 10.48 | Fe2+ | 0.16 | 0.53 | |
| CaO | 1.17 | 0.23 | Mg | 0.10 | 0.49 | |
| Na2O | 3.76 | 0.00 | Ca | 0.06 | 0.01 | |
| K2O | 1.51 | 10.05 | Na | 0.35 | 0.00 | |
| Σ | 99.8 | 100.62 | K | 0.09 | 0.40 | |
| An | 11.94 | Fo | 47.66 | |||
| Ab | 69.63 | Fa | 51.59 | |||
| Or | 18.43 | Ca-Ol | 0.75 | |||
| Chemical Composition | Concentration (wt%) |
|---|---|
| n = 6 | |
| SiO2 | 32.26 |
| Al2O3 | 12.45 |
| MgO | 1.14 |
| K2O | 1.22 |
| CaO | 1.33 |
| TiO2 | 1.32 |
| MnO | 0.42 |
| FeO (total) | 34.11 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Elmi, C.; Cipriani, A.; Lugli, F.; Sighinolfi, G. Insights on the Origin of Vitrified Rocks from Serravuda, Acri (Italy): Rock Fulgurite or Anthropogenic Activity? Geosciences 2021, 11, 493. https://doi.org/10.3390/geosciences11120493
Elmi C, Cipriani A, Lugli F, Sighinolfi G. Insights on the Origin of Vitrified Rocks from Serravuda, Acri (Italy): Rock Fulgurite or Anthropogenic Activity? Geosciences. 2021; 11(12):493. https://doi.org/10.3390/geosciences11120493
Chicago/Turabian StyleElmi, Chiara, Anna Cipriani, Federico Lugli, and Giampaolo Sighinolfi. 2021. "Insights on the Origin of Vitrified Rocks from Serravuda, Acri (Italy): Rock Fulgurite or Anthropogenic Activity?" Geosciences 11, no. 12: 493. https://doi.org/10.3390/geosciences11120493
APA StyleElmi, C., Cipriani, A., Lugli, F., & Sighinolfi, G. (2021). Insights on the Origin of Vitrified Rocks from Serravuda, Acri (Italy): Rock Fulgurite or Anthropogenic Activity? Geosciences, 11(12), 493. https://doi.org/10.3390/geosciences11120493







