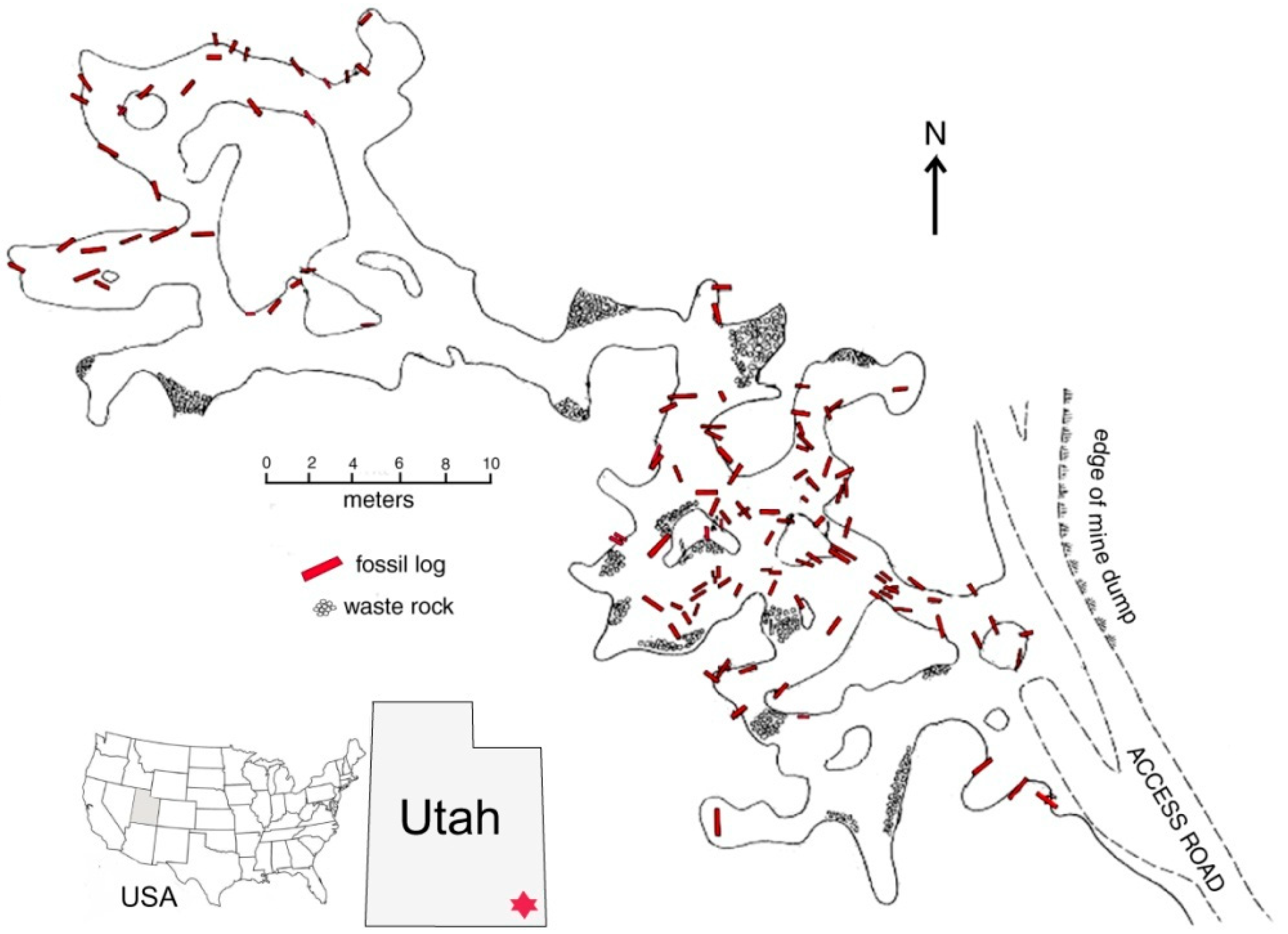
Figure 1.
Fossil logs in Verdure Mine, Montezuma Canyon, San Juan County (UT, USA). Adapted from [
6].
Figure 1.
Fossil logs in Verdure Mine, Montezuma Canyon, San Juan County (UT, USA). Adapted from [
6].
Figure 2.
Woody debris layer exposed in the wall of an inactive uranium mine in the White Valley District, San Juan County (UT, USA). 2019 photo, courtesy of Mark Hart.
Figure 2.
Woody debris layer exposed in the wall of an inactive uranium mine in the White Valley District, San Juan County (UT, USA). 2019 photo, courtesy of Mark Hart.
Figure 3.
Reflected light optical photomicrographs of polished specimens of uraninite-mineralized fossil wood from San Juan County (UT, USA). (
A) Wood with calcite (
C) filling cell lumina and uraninite replacing cell walls. (
B) Coffinite (Co) filling lumina, uraninite (U) replacing cell walls.
A,
B from Mi Vida Mine, Lisbon-Big Indian Wash District [
9]. (
C) Carbonized wood fragment partially mineralized with chalcopyrite and uraninite. Chalcopyrite (white) has filled lumina, uraninite (gray) has replaced cell walls. W = unmineralized carbonized wood. Happy Jack Mine, White Canyon District [
10]. (
D) Coalified wood from Mi Vida Mine, with cell walls replaced by uraninite (U), open fractures and lumina interior regions mineralized with calcite (Ca) [
11].
Figure 3.
Reflected light optical photomicrographs of polished specimens of uraninite-mineralized fossil wood from San Juan County (UT, USA). (
A) Wood with calcite (
C) filling cell lumina and uraninite replacing cell walls. (
B) Coffinite (Co) filling lumina, uraninite (U) replacing cell walls.
A,
B from Mi Vida Mine, Lisbon-Big Indian Wash District [
9]. (
C) Carbonized wood fragment partially mineralized with chalcopyrite and uraninite. Chalcopyrite (white) has filled lumina, uraninite (gray) has replaced cell walls. W = unmineralized carbonized wood. Happy Jack Mine, White Canyon District [
10]. (
D) Coalified wood from Mi Vida Mine, with cell walls replaced by uraninite (U), open fractures and lumina interior regions mineralized with calcite (Ca) [
11].
Figure 4.
Uranium minerals in fossil wood from mines in the American Southwest. (A) Uraninite, Henry Mountains (UT). (B) Uraninite with secondary alteration, Reuben Weeks Mine, Cainville Wash (UT). (C) Uranophane, Happy Jack Mine, San Juan County (UT). (D), Carnotite, La Sal Valley, (CO). (E) Carnotite, Grand County (UT). (F) Zippeite, Happy Jack Mine, San Juan County (UT).
Figure 4.
Uranium minerals in fossil wood from mines in the American Southwest. (A) Uraninite, Henry Mountains (UT). (B) Uraninite with secondary alteration, Reuben Weeks Mine, Cainville Wash (UT). (C) Uranophane, Happy Jack Mine, San Juan County (UT). (D), Carnotite, La Sal Valley, (CO). (E) Carnotite, Grand County (UT). (F) Zippeite, Happy Jack Mine, San Juan County (UT).
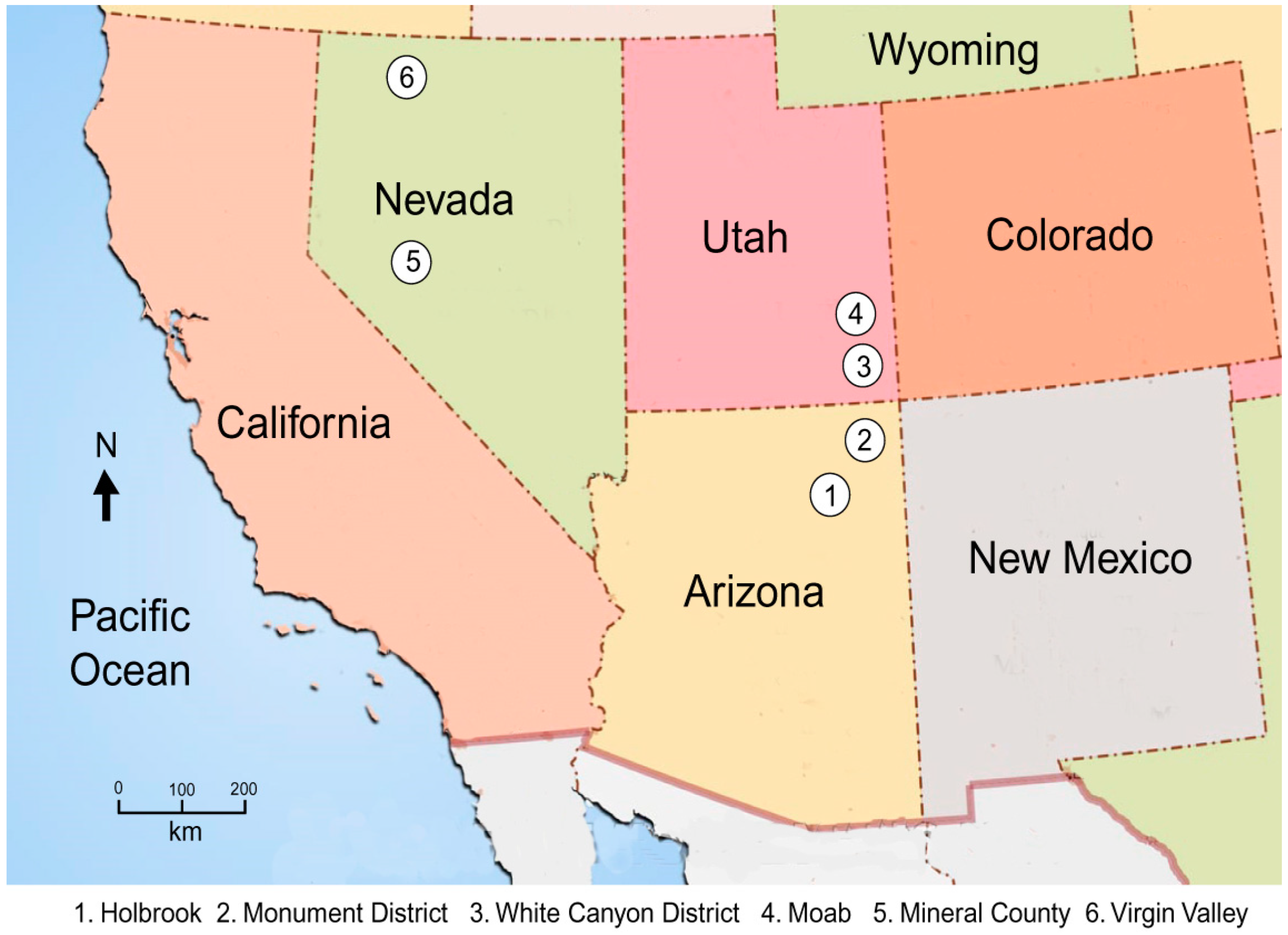
Figure 5.
Location map for the samples used in this study.
Figure 5.
Location map for the samples used in this study.
Figure 6.
Uraniferous silicified wood, Holbrook (AZ, USA). (A) Light brown wood and dark gray uranium rich zone. (B) Fracture planes in dark gray zone contain thin coatings of a secondary uranium mineral, possibly tyuyamunite.
Figure 6.
Uraniferous silicified wood, Holbrook (AZ, USA). (A) Light brown wood and dark gray uranium rich zone. (B) Fracture planes in dark gray zone contain thin coatings of a secondary uranium mineral, possibly tyuyamunite.
Figure 7.
SEM views of silicified wood in light brown zone. (A) Longitudinal view shows tracheid cells that have been replaced by silica, secondary electron image. (B) Carbonaceous material (labeled C) is preserved as brittle material in areas of cell walls, quartz-mineralized wood is marked with Q. Thin stringers of coffinite (unlabelled) appear as white zones in this backscatter electron image.
Figure 7.
SEM views of silicified wood in light brown zone. (A) Longitudinal view shows tracheid cells that have been replaced by silica, secondary electron image. (B) Carbonaceous material (labeled C) is preserved as brittle material in areas of cell walls, quartz-mineralized wood is marked with Q. Thin stringers of coffinite (unlabelled) appear as white zones in this backscatter electron image.
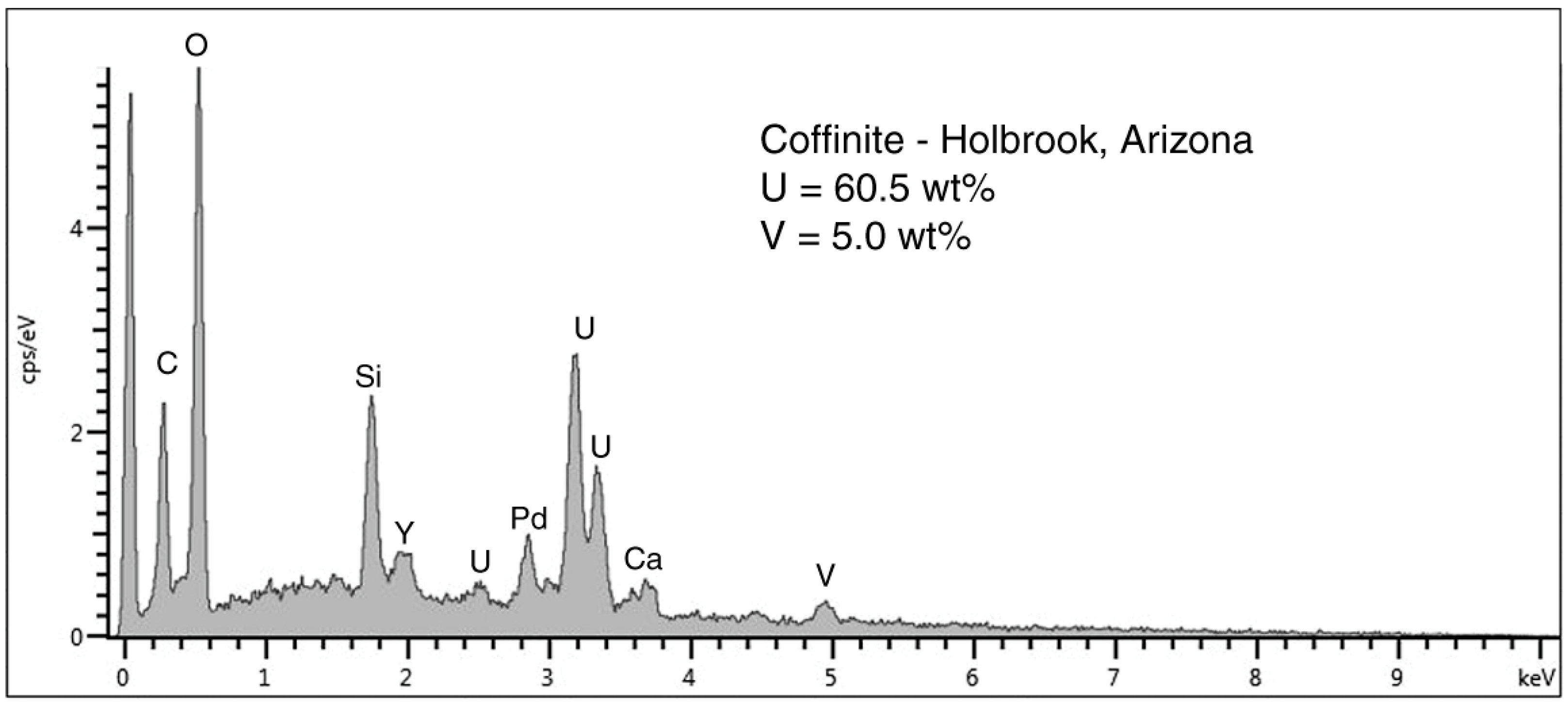
Figure 8.
SEM/EDS spectrum of coffinite in the dark gray zone shown in
Figure 6. Peak at 0 KeV is detector noise, Pd is coating applied to provide electrical conductivity. The large Si peak probably comes partly from from silicified wood cells, in addition to Si in coffinite, a uranium silicate. Y is a common minor element in uranium ores.
Figure 8.
SEM/EDS spectrum of coffinite in the dark gray zone shown in
Figure 6. Peak at 0 KeV is detector noise, Pd is coating applied to provide electrical conductivity. The large Si peak probably comes partly from from silicified wood cells, in addition to Si in coffinite, a uranium silicate. Y is a common minor element in uranium ores.
Figure 9.
SEM BSE images. (A) Coffinite is revealed as bright white zones in the silicified wood. (B) Coffinite in a region of relatively low uranium mineralization, longitudinal view of silicified wood. (C) Coffinite occupying cell lumina, and as fine disseminations in silicified cell walls in a zone of relatively high uranium abundance.
Figure 9.
SEM BSE images. (A) Coffinite is revealed as bright white zones in the silicified wood. (B) Coffinite in a region of relatively low uranium mineralization, longitudinal view of silicified wood. (C) Coffinite occupying cell lumina, and as fine disseminations in silicified cell walls in a zone of relatively high uranium abundance.
Figure 10.
Carbonaceous sample containing light-colored ncrustrations. (A) β-Radiation intensity of the upper surface is low, β=~100 CPM (B) Reflected light photomicrograph showing white patches of aluminum sulfate.
Figure 10.
Carbonaceous sample containing light-colored ncrustrations. (A) β-Radiation intensity of the upper surface is low, β=~100 CPM (B) Reflected light photomicrograph showing white patches of aluminum sulfate.
Figure 11.
SEM images of uraniferous wood from White Canyon District (UT). (A) Silicified wood, oblique longitudinal view showing unmineralized spaces between some adjacent cells. (B) BSE image showing prismatic crystals of zippeite on wood surface. (C) Layer of zippeite encrusting specimen surface, BSE image. (D) High magnification SE image showing semi-botryoidal zippeite (Z) adjacent to gypsum crystal cluster (G).
Figure 11.
SEM images of uraniferous wood from White Canyon District (UT). (A) Silicified wood, oblique longitudinal view showing unmineralized spaces between some adjacent cells. (B) BSE image showing prismatic crystals of zippeite on wood surface. (C) Layer of zippeite encrusting specimen surface, BSE image. (D) High magnification SE image showing semi-botryoidal zippeite (Z) adjacent to gypsum crystal cluster (G).
Figure 12.
SEM/EDS spectrum of microcrystals shown in
Figure 11B. The Pd peak is caused by the metal layer applied to the specimen surface to provide electrical conductivity. The peak at 0KeV is detector noise related to the low count rate. The peak at 3.3KeV is a combination of the M orbital peak for uranium and the Kα peak for potassium. The presence of the latter element can be confirmed by the smaller Kβ peak at 3.59 KeV.
Figure 12.
SEM/EDS spectrum of microcrystals shown in
Figure 11B. The Pd peak is caused by the metal layer applied to the specimen surface to provide electrical conductivity. The peak at 0KeV is detector noise related to the low count rate. The peak at 3.3KeV is a combination of the M orbital peak for uranium and the Kα peak for potassium. The presence of the latter element can be confirmed by the smaller Kβ peak at 3.59 KeV.
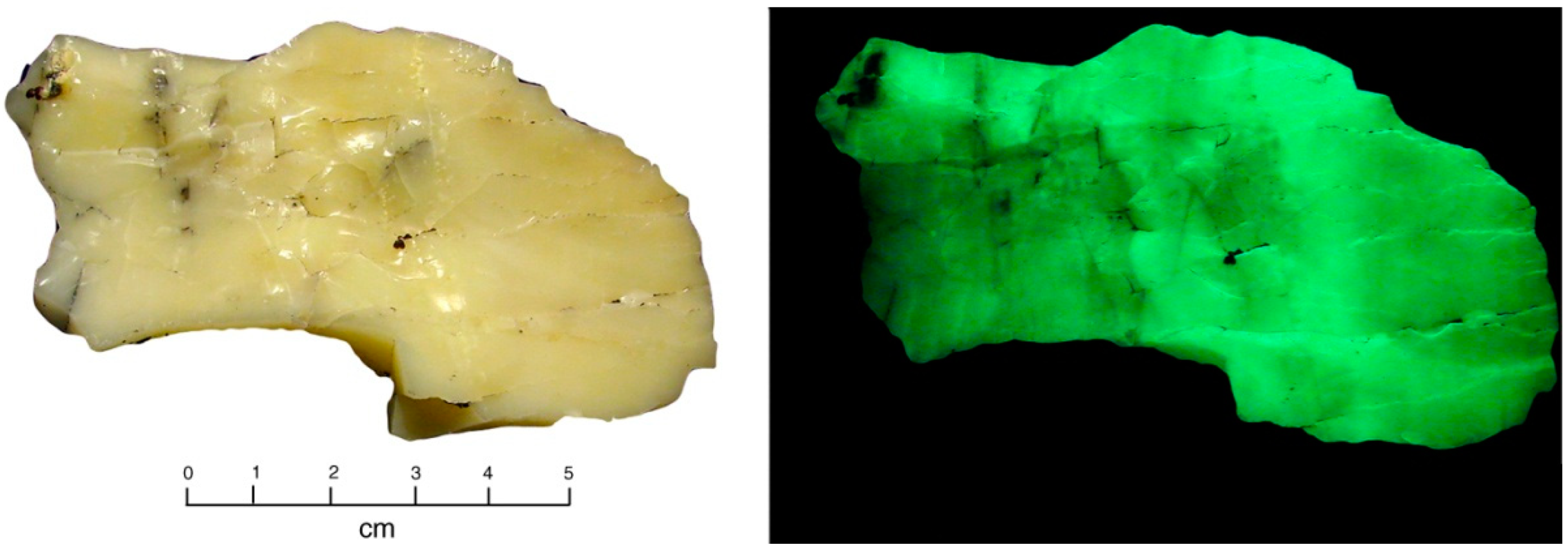
Figure 13.
Flat silty surface of White Canyon sample, containing thin coatings of zippeite. (A) Ordinary light. (B) Fluorescence under short-wave UV light.
Figure 13.
Flat silty surface of White Canyon sample, containing thin coatings of zippeite. (A) Ordinary light. (B) Fluorescence under short-wave UV light.
Figure 14.
SEM BSE images of light-colored encrustations reveal two forms. (A) Prismatic crystals of aluminum sulfate. (B) porous coral-like ferruginous aluminum sulfate.
Figure 14.
SEM BSE images of light-colored encrustations reveal two forms. (A) Prismatic crystals of aluminum sulfate. (B) porous coral-like ferruginous aluminum sulfate.
Figure 15.
Carnotite-bearing fossil wood from Judd Claim, White Canyon District (UT).
Figure 15.
Carnotite-bearing fossil wood from Judd Claim, White Canyon District (UT).
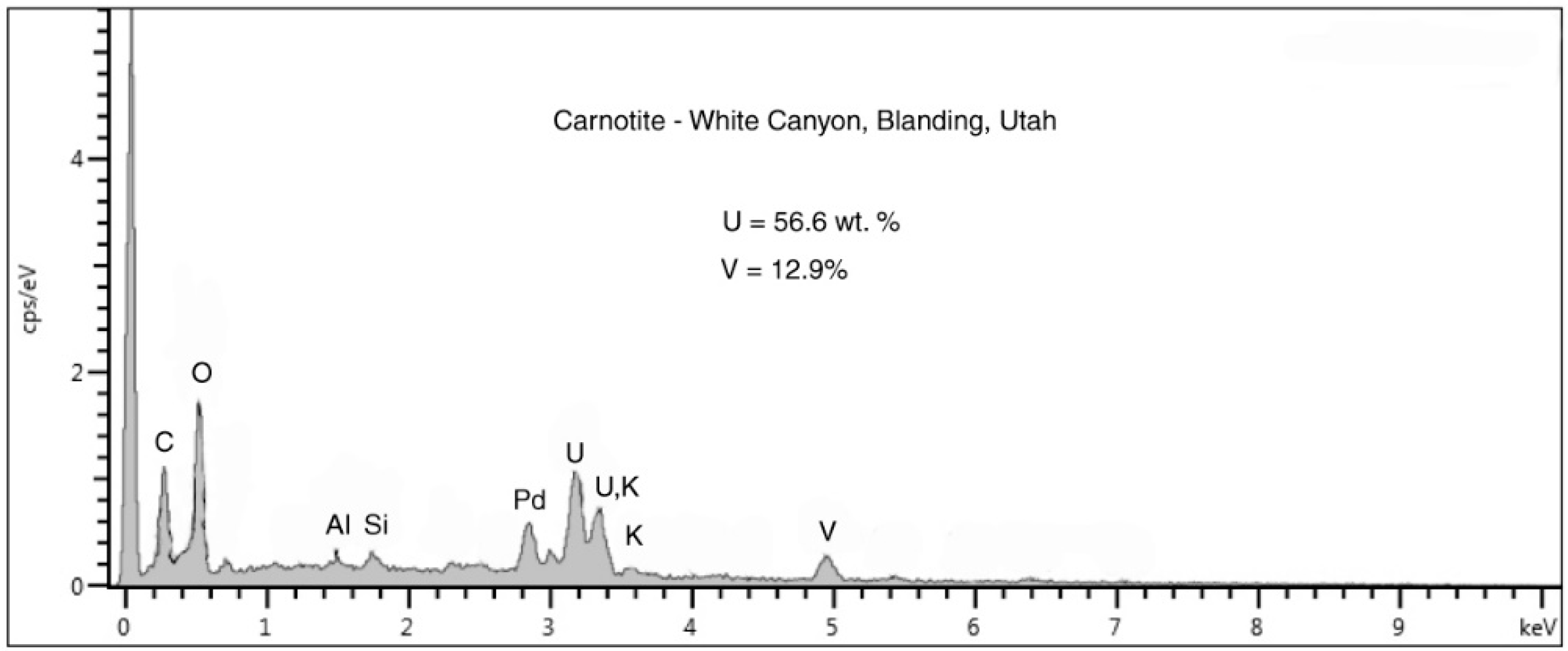
Figure 16.
SEM/EDS spectrum showing carnotite composition. Palladium peak is an artifact from specimen coating. The high peak at the left side of the spectrum is detector noise related to the low count rate.
Figure 16.
SEM/EDS spectrum showing carnotite composition. Palladium peak is an artifact from specimen coating. The high peak at the left side of the spectrum is detector noise related to the low count rate.
Figure 17.
SEM images from Judd Claim sample. (A,B) carnotite occurs in layers within carbonized wood, visible as bright zones in these BSE images. Note the constellation of tiny bright spots in the fractured carbonized wood in image B. (C) Transverse view, showing carnotite filling lumina. Cell walls are carbonized. (D) Longitudinal view, showing wood cells encrusted with patchy carnotiite. (E) Pure carnotite showing crystal habit. (F) Platy clay minerals sowing disseminated carnotite.
Figure 17.
SEM images from Judd Claim sample. (A,B) carnotite occurs in layers within carbonized wood, visible as bright zones in these BSE images. Note the constellation of tiny bright spots in the fractured carbonized wood in image B. (C) Transverse view, showing carnotite filling lumina. Cell walls are carbonized. (D) Longitudinal view, showing wood cells encrusted with patchy carnotiite. (E) Pure carnotite showing crystal habit. (F) Platy clay minerals sowing disseminated carnotite.
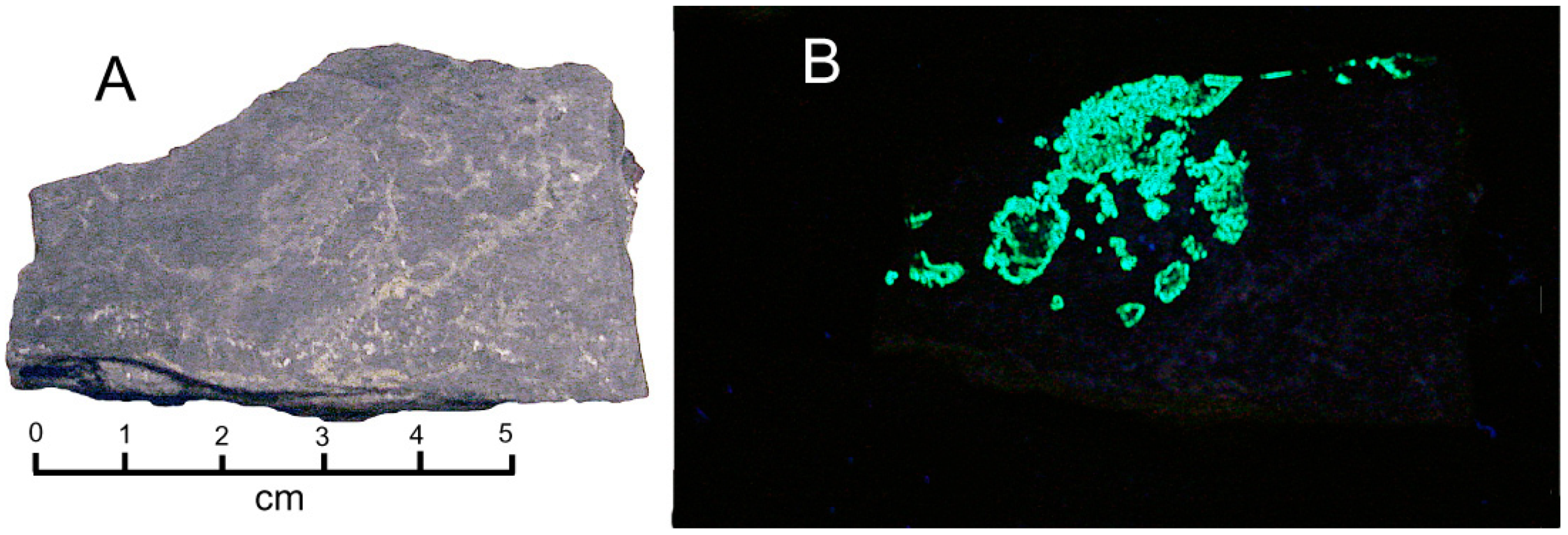
Figure 18.
Silicified wood containing uranium and vanadium, Monument #2 Mine, Monument Valley, AZ. (A) Tyuyamunite coatings occur in fracture plane surfaces, and within silicified wood tissue. (B) Reflected light photomicrograph.
Figure 18.
Silicified wood containing uranium and vanadium, Monument #2 Mine, Monument Valley, AZ. (A) Tyuyamunite coatings occur in fracture plane surfaces, and within silicified wood tissue. (B) Reflected light photomicrograph.
Figure 19.
SEM backscatter electron images show bright crystals of tyuyamanite (labeled U) within dark silicified wood (labeled W). (A,B) Radiating crystals of tyuyamunite on fossil wood. (C) Mass of pure tyuyamunite. (D) Tyuyamunite and a vanadium mineral (V) incorporated within fossil wood.
Figure 19.
SEM backscatter electron images show bright crystals of tyuyamanite (labeled U) within dark silicified wood (labeled W). (A,B) Radiating crystals of tyuyamunite on fossil wood. (C) Mass of pure tyuyamunite. (D) Tyuyamunite and a vanadium mineral (V) incorporated within fossil wood.
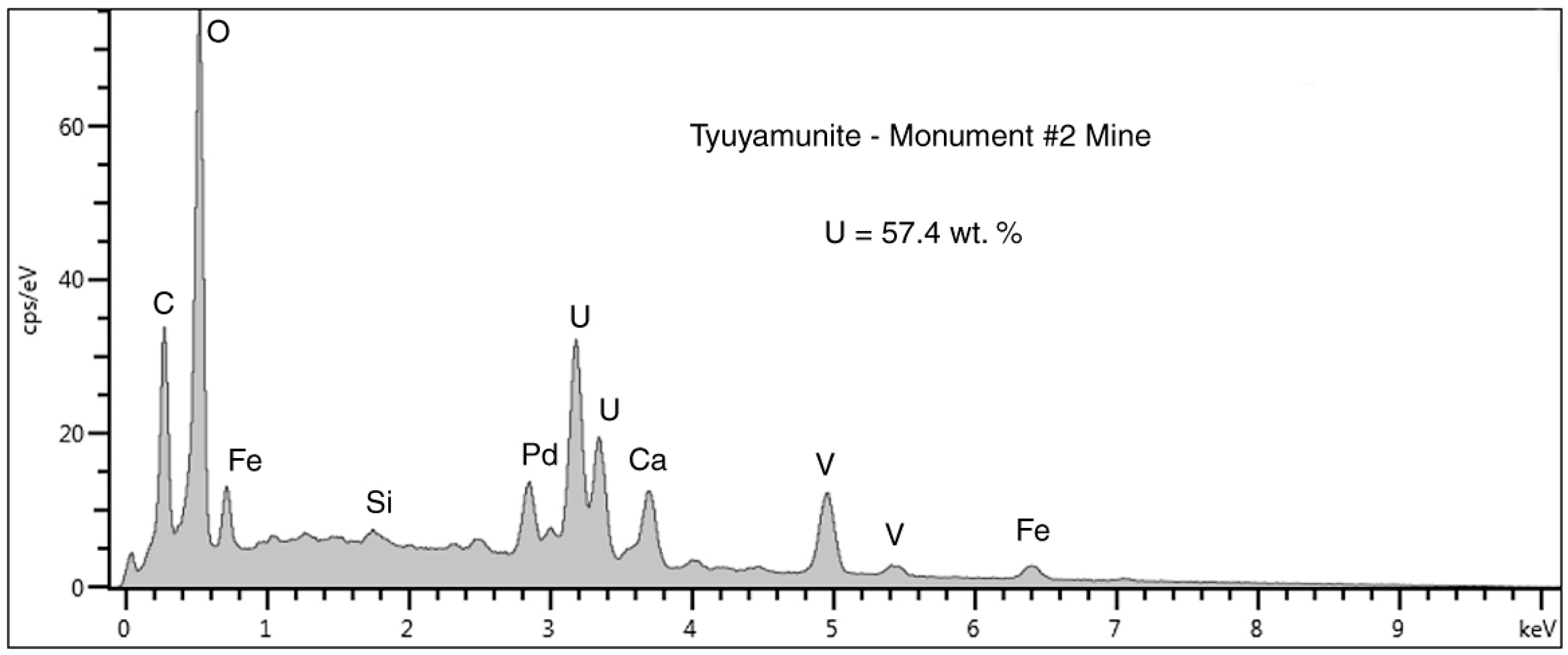
Figure 20.
SEM/EDS spectrum of a surface crust of tyuyamunite on fossil wood from the Monument #2 Mine. The large carbon peak may be caused by small amounts of calcium carbonate. The Pd peak is a artifact from specimen coating to provide electrical conductivity. The small peak at 0 KeV is caused by detector noise.
Figure 20.
SEM/EDS spectrum of a surface crust of tyuyamunite on fossil wood from the Monument #2 Mine. The large carbon peak may be caused by small amounts of calcium carbonate. The Pd peak is a artifact from specimen coating to provide electrical conductivity. The small peak at 0 KeV is caused by detector noise.
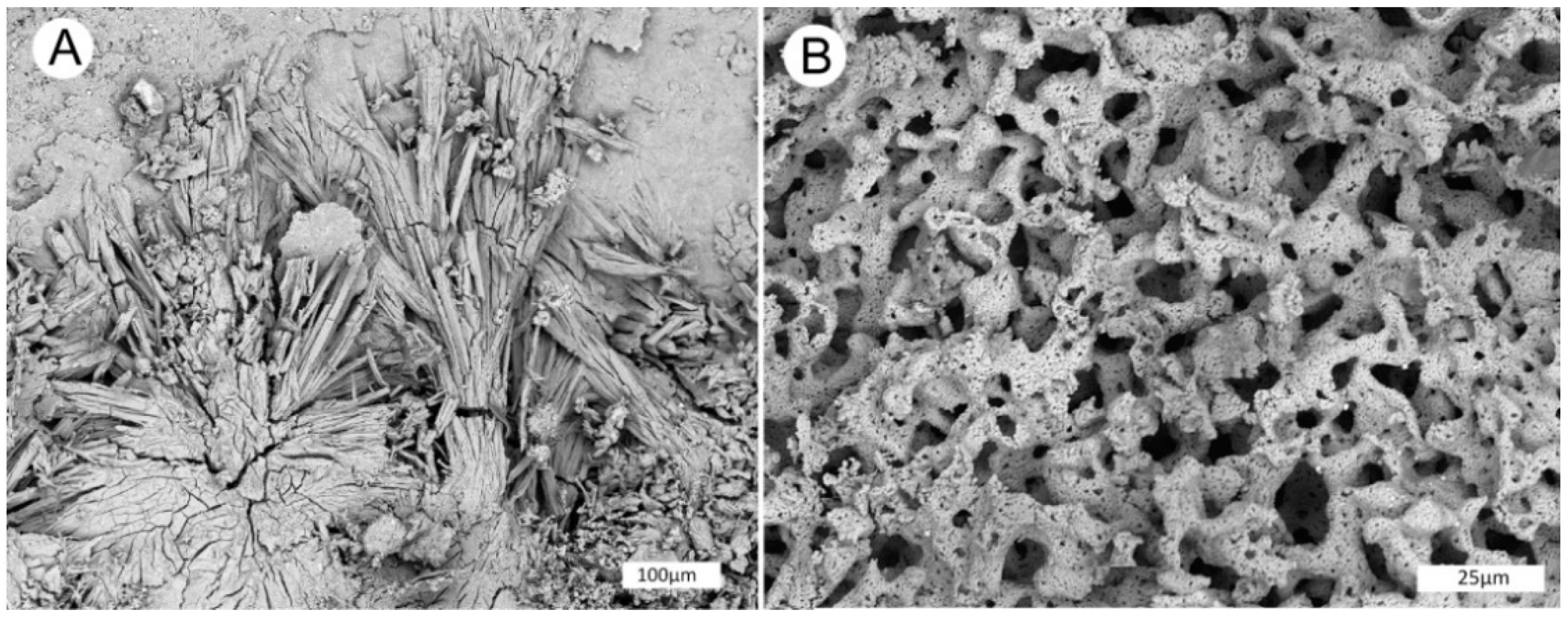
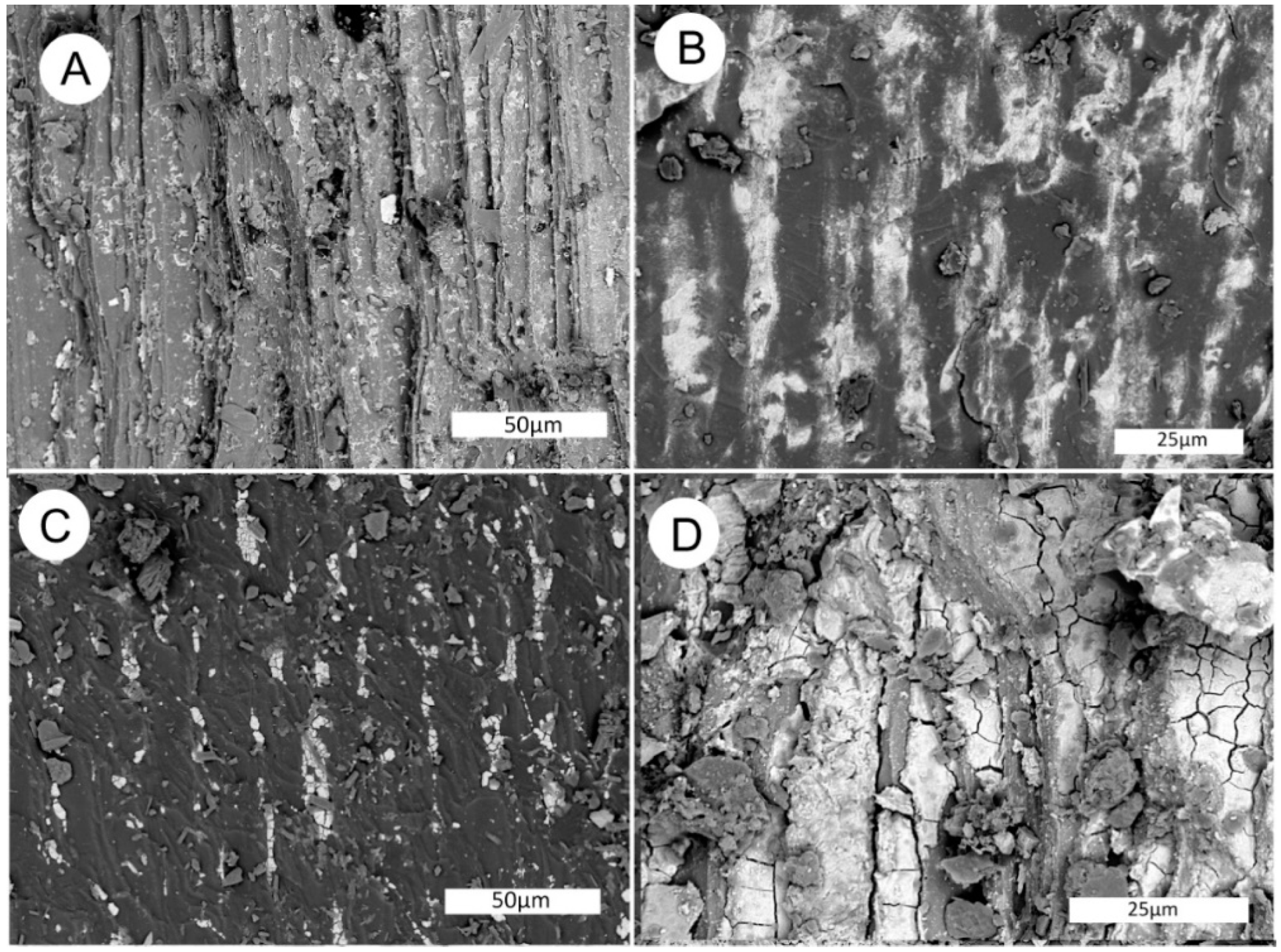
Figure 21.
SEM images of fossil wood from Monment #2 Mine. (A–C) Longitudinal view of tracheid cells containing fossil wood (W) and a vanadium mineral (V). (D–E) Fossil wood containing a vanadium mineral and tyuyamunite (U), a uranium mineral.
Figure 21.
SEM images of fossil wood from Monment #2 Mine. (A–C) Longitudinal view of tracheid cells containing fossil wood (W) and a vanadium mineral (V). (D–E) Fossil wood containing a vanadium mineral and tyuyamunite (U), a uranium mineral.
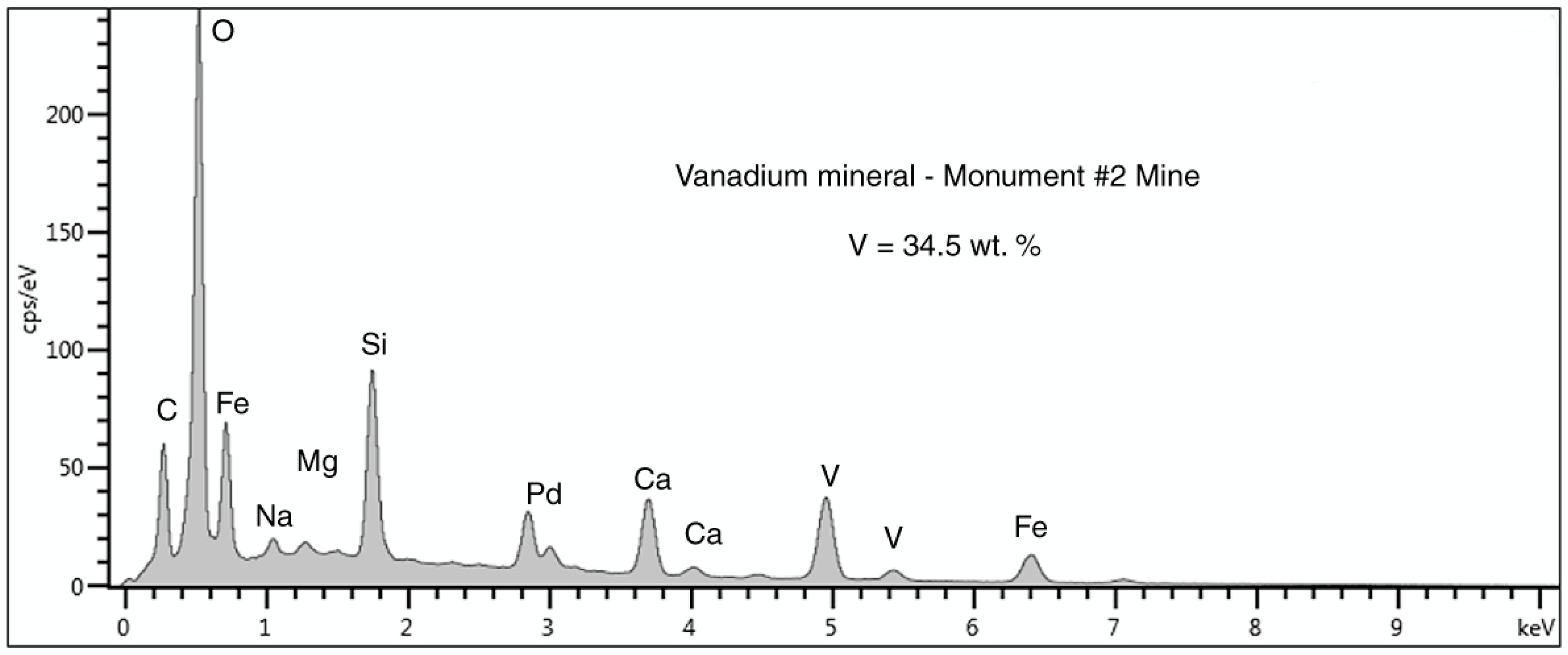
Figure 22.
SEM/EDS spectrum of the vanadium mineral n silicified wood from the Monument #2 Mine. The carbon peak is perhaps indicative of small amounts calcium carbonate. The large Si peak is probably from the silicified wood. The Pd peak is an artifact from specimen coating to provide electrical conductivity.
Figure 22.
SEM/EDS spectrum of the vanadium mineral n silicified wood from the Monument #2 Mine. The carbon peak is perhaps indicative of small amounts calcium carbonate. The large Si peak is probably from the silicified wood. The Pd peak is an artifact from specimen coating to provide electrical conductivity.
Figure 23.
Navajoite in silty sandstone, Monument # 2 Mine, Apache County, AZ.
Figure 23.
Navajoite in silty sandstone, Monument # 2 Mine, Apache County, AZ.
Figure 24.
(A) SEM/EDS spectrum of silica filling cell lumina. Pd peak is an artifact from metal coating of the specimen to provide electrical conductivity, Peak at 0 KeV is detector noise. (B) SEM BSE image showing bright areas where U and V minerals have replaced cell walls and filled intracellular spaces. The uranium mineral is probably tyuyamunite.
Figure 24.
(A) SEM/EDS spectrum of silica filling cell lumina. Pd peak is an artifact from metal coating of the specimen to provide electrical conductivity, Peak at 0 KeV is detector noise. (B) SEM BSE image showing bright areas where U and V minerals have replaced cell walls and filled intracellular spaces. The uranium mineral is probably tyuyamunite.
Figure 25.
Uranium ore from Moab, Utah area, consisting of carbonized wood in a sandstone matrix. (A) Upper zone contains uranium-bearing carbonized wood, lower zone contains sandstone with local iron oxide staining. (B) Rhombic white calcite crystals are abundant within the wood layer. (C) Thin fracture networks in wood zone are filled with yellow barite.
Figure 25.
Uranium ore from Moab, Utah area, consisting of carbonized wood in a sandstone matrix. (A) Upper zone contains uranium-bearing carbonized wood, lower zone contains sandstone with local iron oxide staining. (B) Rhombic white calcite crystals are abundant within the wood layer. (C) Thin fracture networks in wood zone are filled with yellow barite.
Figure 26.
SEM images of carbonized wood layer, Moab, Utah specimen. (A) Secondary electron image showing relict cell structure. (B,C) Backscattered electron images, longitudinal view of tracheid cells showing cabonized tissue (dark), and the uranium mineral coffinite (light). Coffinite primarily occurs within cell lumina. (D) BSE image shows carbonized cell walls, with luminafileld with zippeite, probably formed as an alteration product of coffinite. (E) Surface crust containing nearly pure zippeite. (F)Surface curst containing tabular gypsum crystals (dark) and microcrystalline Na-zippeite (light).
Figure 26.
SEM images of carbonized wood layer, Moab, Utah specimen. (A) Secondary electron image showing relict cell structure. (B,C) Backscattered electron images, longitudinal view of tracheid cells showing cabonized tissue (dark), and the uranium mineral coffinite (light). Coffinite primarily occurs within cell lumina. (D) BSE image shows carbonized cell walls, with luminafileld with zippeite, probably formed as an alteration product of coffinite. (E) Surface crust containing nearly pure zippeite. (F)Surface curst containing tabular gypsum crystals (dark) and microcrystalline Na-zippeite (light).
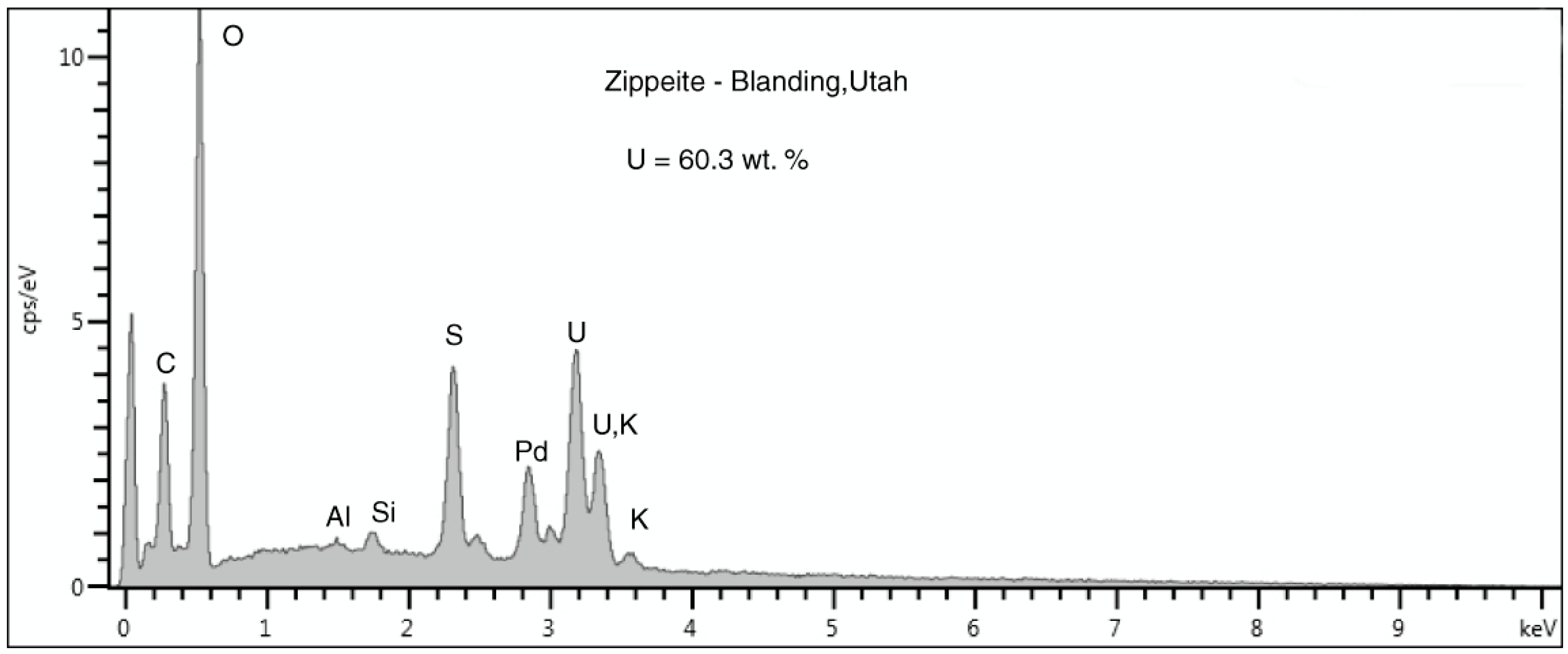
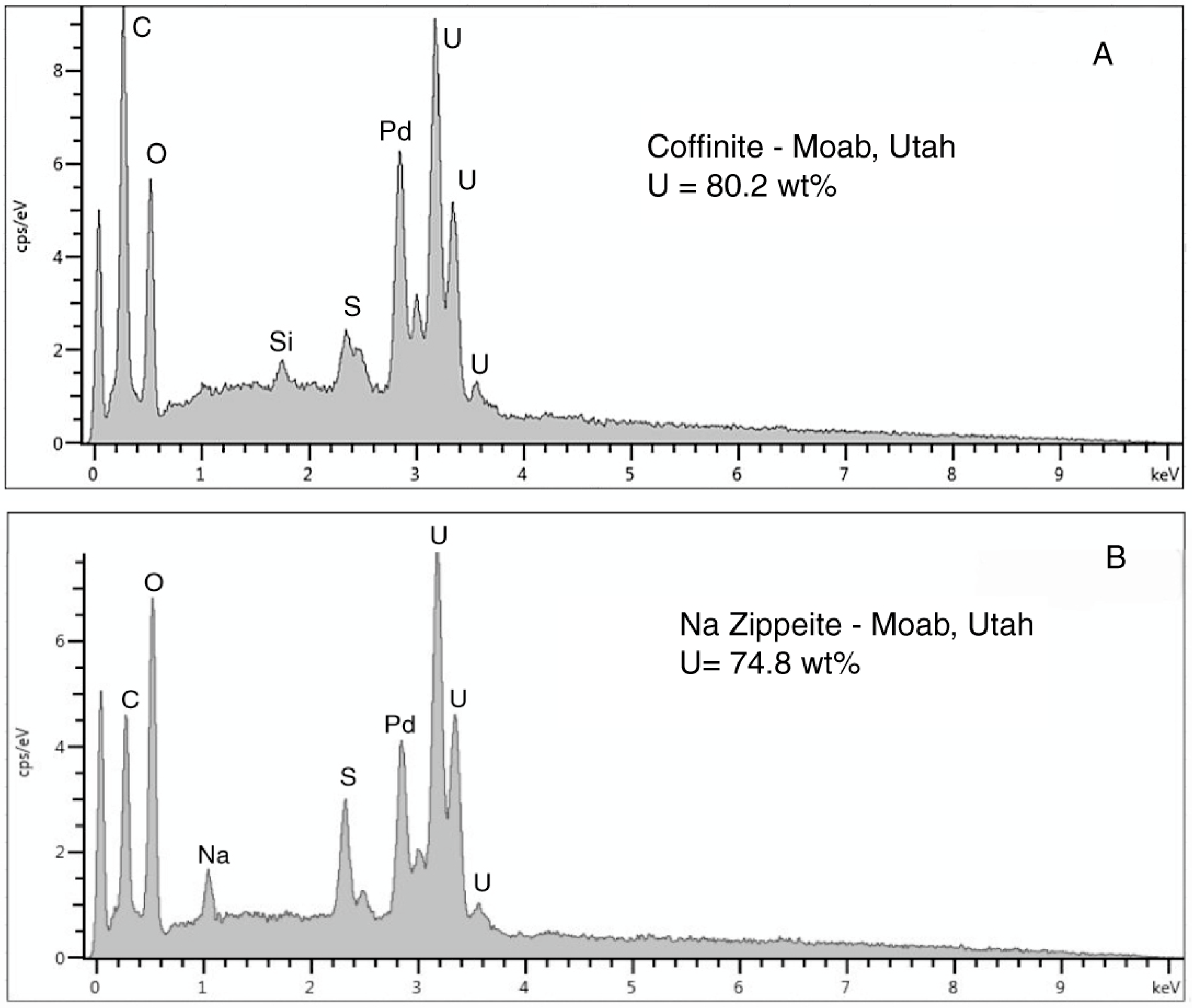
Figure 27.
SEM/EDS spectra of the three uranium minerals present in the Moab, Utah specimen. (A) Coffinite is the primary uranium mineral, typically occurring in lumina of carbonized wood cells. (B) Zippeite appears to be an alteration product that occurs both as surface crusts and within the carbonized wood zone. Na-Zippeite is associated with gypsum crystal clusters that are present as surface crusts.
Figure 27.
SEM/EDS spectra of the three uranium minerals present in the Moab, Utah specimen. (A) Coffinite is the primary uranium mineral, typically occurring in lumina of carbonized wood cells. (B) Zippeite appears to be an alteration product that occurs both as surface crusts and within the carbonized wood zone. Na-Zippeite is associated with gypsum crystal clusters that are present as surface crusts.
Figure 28.
Moab (UT) specimen, SEM backscatter electron images. (A) Barite (light) occurring as a cell replacement for carbonized wood. (B) Barite filling fracture in carbonized wood. (C) Barite inclusion within carbonized wood. (D) Unidentified uranium mineral (light) within a calcite crystal in wood layer.
Figure 28.
Moab (UT) specimen, SEM backscatter electron images. (A) Barite (light) occurring as a cell replacement for carbonized wood. (B) Barite filling fracture in carbonized wood. (C) Barite inclusion within carbonized wood. (D) Unidentified uranium mineral (light) within a calcite crystal in wood layer.
Figure 29.
Uraniferous fossil wood from Bubbles Claim. Mineral County (NV). Photos on right show fluorescence under short-wave UV light. (A) Opalized wood showing well-preserved grain pattern. (B) Assemblage of small wood fragments cemented by common opal. (C) Wood mineralized with common opal.
Figure 29.
Uraniferous fossil wood from Bubbles Claim. Mineral County (NV). Photos on right show fluorescence under short-wave UV light. (A) Opalized wood showing well-preserved grain pattern. (B) Assemblage of small wood fragments cemented by common opal. (C) Wood mineralized with common opal.
Figure 30.
Reflected light photomicrographs of uraniferous wood from Mineral County, Nevada USA. (A) Fragmental wood silicified with opal cement. (B) Bright yellow uranium-rich opal following wood grain. (C) Wood replaced by opal, with color zones parallel to wood grain. (D) Fossil wood with uranium-rich opal filling fractures that are perpendicular to wood grain.
Figure 30.
Reflected light photomicrographs of uraniferous wood from Mineral County, Nevada USA. (A) Fragmental wood silicified with opal cement. (B) Bright yellow uranium-rich opal following wood grain. (C) Wood replaced by opal, with color zones parallel to wood grain. (D) Fossil wood with uranium-rich opal filling fractures that are perpendicular to wood grain.
Figure 31.
SEM images of specimen shown in
Figure 29B. (
A) SE image of small wood fragments in a matrix of common opal. (
B) longitudinal view of wood cells encased in botryoidal opal. (
C) High magnification SE image of opal-coated wood cells, Arrow shows end view of a diatom frustule. Crystalline structure of Opal-CT is visible on background surfaces. (
D) Fractures in dessicated opal are filled with microcrystalline quartz. (
E) BSE image of botryoidal opal on exterior surface of specimen. (
F) BSE view showing an unidentified vanadium minera deposited on the surface of a plagioclase feldspar clast. Similar vanadium inclusions occur in the adjacent opalized wood.
Figure 31.
SEM images of specimen shown in
Figure 29B. (
A) SE image of small wood fragments in a matrix of common opal. (
B) longitudinal view of wood cells encased in botryoidal opal. (
C) High magnification SE image of opal-coated wood cells, Arrow shows end view of a diatom frustule. Crystalline structure of Opal-CT is visible on background surfaces. (
D) Fractures in dessicated opal are filled with microcrystalline quartz. (
E) BSE image of botryoidal opal on exterior surface of specimen. (
F) BSE view showing an unidentified vanadium minera deposited on the surface of a plagioclase feldspar clast. Similar vanadium inclusions occur in the adjacent opalized wood.
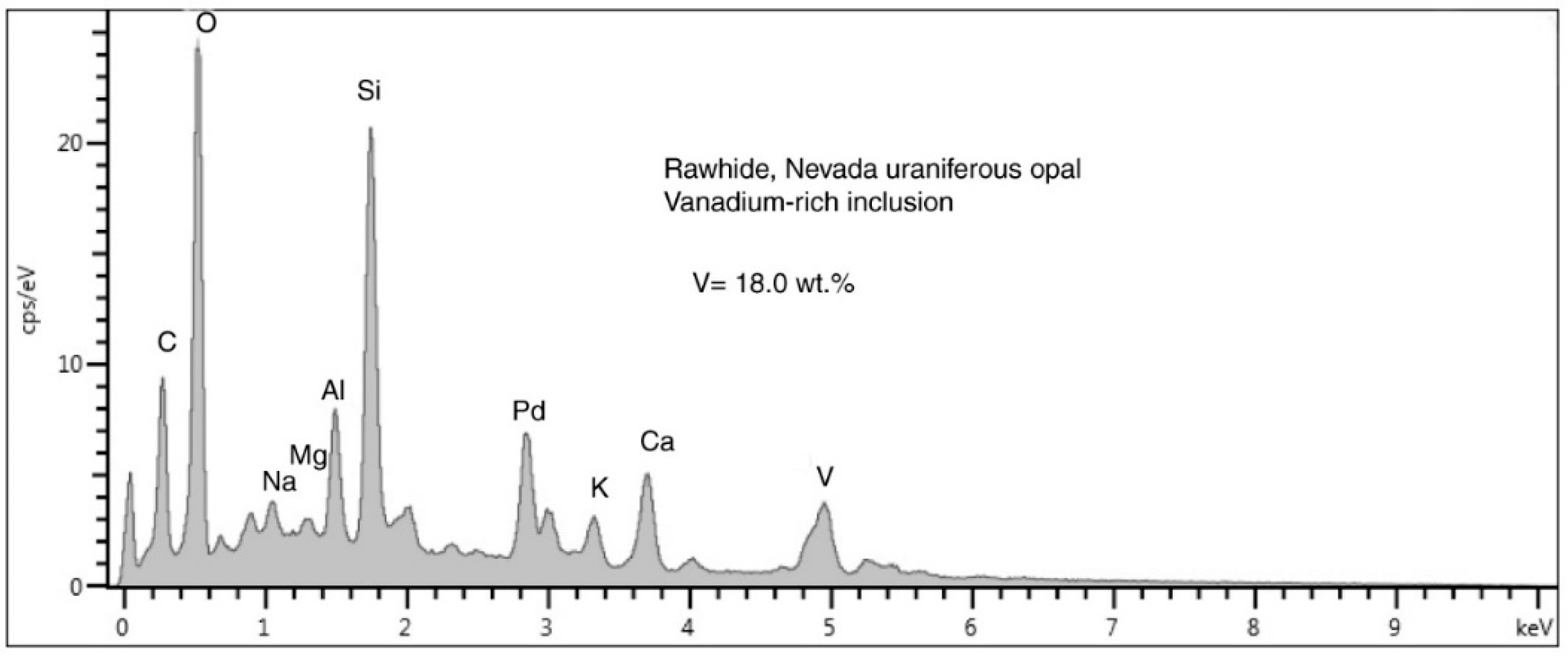
Figure 32.
SEM/EDS spectrum of a single small vanadium mineral bleb lying on a plagioclase clast (
Figure 22F). The tiny size of the vanadium-rich inclusion allows the electron beam to excite elemnts in the adjacent matrix. The Pd peak is an artifact from specimen coating. The unlabelled peak ner 0 KeV is detector noise resulting form the low count rate.
Figure 32.
SEM/EDS spectrum of a single small vanadium mineral bleb lying on a plagioclase clast (
Figure 22F). The tiny size of the vanadium-rich inclusion allows the electron beam to excite elemnts in the adjacent matrix. The Pd peak is an artifact from specimen coating. The unlabelled peak ner 0 KeV is detector noise resulting form the low count rate.
Figure 33.
Uraniferous opal from uranium claim on hillside above Royal Peacock Mine headquarters. Location details canbe found in [
21,
22].
Figure 33.
Uraniferous opal from uranium claim on hillside above Royal Peacock Mine headquarters. Location details canbe found in [
21,
22].
Table 1.
Common Uranium Minerals.
Table 1.
Common Uranium Minerals.
| Mineral | Formula |
|---|
| Uraninite | UO2 |
| Autunite | Ca(UO2)2(PO4)·H2O |
| Carnotite | K2(UO2)2(VO4)2·3H2O |
| Tyuyamunite | Ca(UO2)2(VO4)2·5–8H2O |
| Coffinite | U(SiO4)1-8(OH)4X |
| Zippeite | K4(UO2)6(SO4)3(OH)10·4H2O |
| Uranopilite | (UO2)6(SO4)2(OH)6·14H2O |
Table 2.
SEM/EDS results for uranium mineral from Monument Mine #2.
Table 2.
SEM/EDS results for uranium mineral from Monument Mine #2.
| Element | Wt% | Atom % |
|---|
| O | 17.6 | 63.5 |
| Si | 0.4 | 0.7 |
| K | 0.04 | 0.06 |
| Ca | 3.8 | 5.5 |
| V | 11.1 | 12.5 |
| Fe | 27.1 | 13.8 |
| U | 65.5 | 15.9 |
| U:V: ratio | | 1.15 |
Table 3.
SEM/EDS results for vanadium mineral from Monument Mine #2.
Table 3.
SEM/EDS results for vanadium mineral from Monument Mine #2.
| Element | Wt% | Atom % |
|---|
| O | 33.8 | 59.9 |
| Na | 0.64 | 0.78 |
| Mg | 0.32 | 0.38 |
| Si | 6.17 | 6.22 |
| K | 0.04 | 0.03 |
| Ca | 8.32 | 5.98 |
| V | 23.5 | 13.1 |
| Fe | 27.1 | 13.8 |
| V:Fe ratio | | 1.05 |
Table 4.
Feric vanadium minerals with Fe:V = 1.
Table 4.
Feric vanadium minerals with Fe:V = 1.
| Mineral | Composition | Type Locality: District | Reference |
|---|
| Montroseite | (V3+Fe3+)O(OH) | Colorado, USA | [11] |
| Fervanite | Fe3+4V5+4O16·5H2O | Colorado, USA | [17] |
| Navajoite | (Fe3+V5+)10O24·12H2O | Arizona, USA | [18] |
| Schubenelite | Fe3+VO4·H2O | Gabon, Africa | [19] |
| Ziminaite | Fe3+6(VO4)6 | Kamchatka, Russia | [20] |