A Monosodium Iodoacetate Osteoarthritis Lameness Model in Growing Pigs
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethical Statement
2.2. Animals
2.3. Housing
2.4. Handling and Habituation
2.5. Experimental Design
2.5.1. Intra-Articular Injection
2.5.2. Locomotion and Gait Measurements
Visual Scoring
Pressure Mat Analysis
2.5.3. Euthanasia
2.5.4. Necropsy and Histopathology
2.6. Data Preparation
2.7. Statistical Analysis
3. Results
3.1. Pathology
3.1.1. Gross Pathology
3.1.2. Histopathology of Target Joints
3.2. Gait Analyses
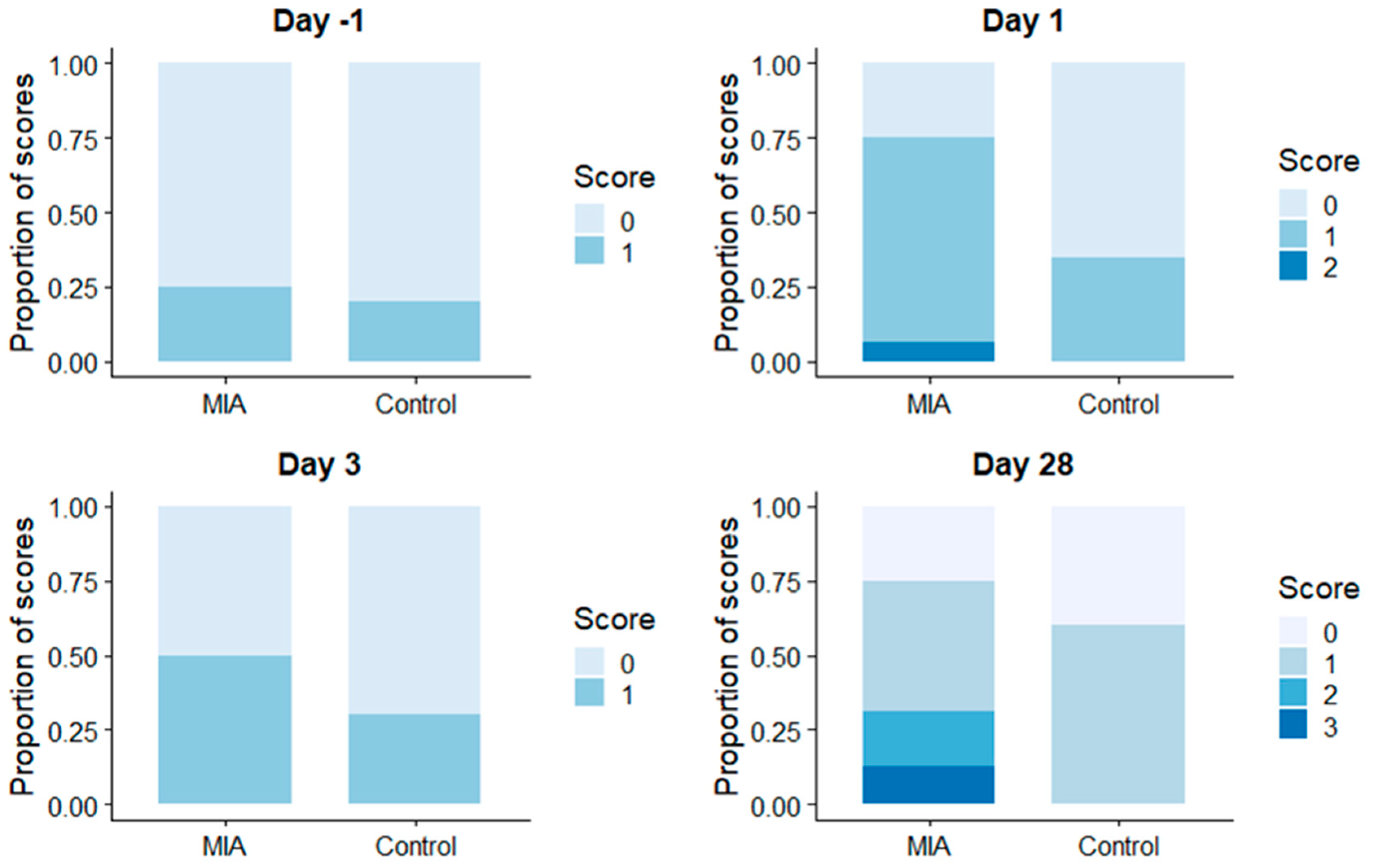
3.2.1. Visual Scoring
3.2.2. Pressure Mat Analyses
4. Discussion
4.1. Face Validity
4.1.1. Histopathological Presentation of Joint Changes
4.1.2. Altered Weight Bearing (Lameness Assessment)
Visual Scoring
Kinetic Data
4.2. Predictive Validity
4.3. Construct Validity
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kilbride, A.L.; Gillman, C.E.; Green, L.E. A cross-sectional study of the prevalence of lameness in finishing pigs, gilts and pregnant sows and associations with limb lesions and floor types on commercial farms in England. Anim. Welf. 2009, 18, 215–224. [Google Scholar]
- Anil, S.; Anil, L.; Deen, J. Effect of Lameness in Pigs in Terms of “Five Freedoms”. J. Appl. Anim. Welf. Sci. 2009, 12, 144–145. [Google Scholar] [CrossRef]
- Heinonen, M.; Peltoniemi, O.; Valros, A. Impact of lameness and claw lesions in sows on welfare, health and production. Livest. Sci. 2013, 156, 2–9. [Google Scholar] [CrossRef]
- Jensen, T.B.; Baadsgaard, N.P.; Houe, H.; Toft, N.; Østergaard, S. The effect of lameness treatments and treatments for other health disorders on the weight gain and feed conversion in boars at a Danish test station. Livest. Sci. 2007, 112, 34–42. [Google Scholar] [CrossRef]
- Jensen, T.B.; Kristensen, H.H.; Toft, N. Quantifying the impact of lameness on welfare and profitability of finisher pigs using expert opinions. Livest. Sci. 2012, 149, 209–214. [Google Scholar] [CrossRef]
- Lane, N.E.; Brandt, K.; Hawker, G.; Peeva, E.; Schreyer, E.; Tsuji, W.; Hochberg, M.C. OARSI-FDA initiative: defining the disease state of osteoarthritis. Osteoarthritis Cartilage 2011, 19, 478–482. [Google Scholar] [CrossRef] [PubMed]
- Jensen, T. Causes of and predisposing risk factors for leg disorders in growing-finishing pigs. CAB Rev. Perspect. Agric. Vet. Sci. Nutr. Nat. Resour. 2009, 4, 1–8. [Google Scholar] [CrossRef]
- Macfadyen, M.A.; Daniel, Z.; Kelly, S.; Parr, T.; Brameld, J.M.; Murton, A.J.; Jones, S.W. The commercial pig as a model of spontaneously-occurring osteoarthritis. BMC Musculoskelet. Disord. 2019, 20, 70. [Google Scholar] [CrossRef]
- Deveza, L.A.; Hunter, D.J. Editorial: Unraveling Osteoarthritis Pathogenesis: New Insights Into Preradiographic Disease and Patient Phenotypes. Arthritis Rheumatol. 2015, 67, 3097–3100. [Google Scholar] [CrossRef]
- Van der Staay, F.J. Animal models of behavioral dysfunctions: basic concepts and classifications, and an evaluation strategy. Brain Res. Rev. 2006, 52, 131–159. [Google Scholar] [CrossRef]
- Maximino, C.; van der Staay, F.J. Behavioral models in psychopathology: epistemic and semantic considerations. Behav. Brain Funct. BBF 2019, 15, 1. [Google Scholar] [CrossRef] [PubMed]
- Dieppe, P.A.; Lohmander, L.S. Pathogenesis and management of pain in osteoarthritis. Lancet 2005, 365, 965–973. [Google Scholar] [CrossRef]
- Ishikawa, G.; Nagakura, Y.; Takeshita, N.; Shimizu, Y. Efficacy of drugs with different mechanisms of action in relieving spontaneous pain at rest and during movement in a rat model of osteoarthritis. Eur. J. Pharmacol. 2014, 738, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Ison, S.H.; Clutton, R.E.; Di Giminiani, P.; Rutherford, K.M.D. A Review of Pain Assessment in Pigs. Front. Vet. Sci. 2016, 3, 108. [Google Scholar] [CrossRef] [PubMed]
- Meijer, E.; van Nes, A.; Back, W.; van der Staay, F.J. Clinical effects of buprenorphine on open field behaviour and gait symmetry in healthy and lame weaned piglets. Vet. J. Lond. Engl. 1997 2015, 206, 298–303. [Google Scholar] [CrossRef] [PubMed]
- Ferreira-Gomes, J.; Adães, S.; Mendonça, M.; Castro-Lopes, J.M. Analgesic effects of lidocaine, morphine and diclofenac on movement-induced nociception, as assessed by the Knee-Bend and CatWalk tests in a rat model of osteoarthritis. Pharmacol. Biochem. Behav. 2012, 101, 617–624. [Google Scholar] [CrossRef] [PubMed]
- Clinical Examination of Farm Animals: Jackson/Clinical; Jackson, P.G.G., Cockcroft, P.D., Eds.; Blackwell Science Ltd.: Oxford, UK, 2002; ISBN 978-0-470-75242-5. [Google Scholar]
- Arkell, M.; Archer, R.M.; Guitian, F.J.; May, S.A. Evidence of bias affecting the interpretation of the results of local anaesthetic nerve blocks when assessing lameness in horses. Vet. Rec. 2006, 159, 346–349. [Google Scholar] [CrossRef] [PubMed]
- D’Eath, R.B. Repeated locomotion scoring of a sow herd to measure lameness: consistency over time, the effect of sow characteristics and inter-observer reliability. Anim. Welf. 2012, 21, 219–231. [Google Scholar] [CrossRef]
- Petersen, H.H.; Enøe, C.; Nielsen, E.O. Observer agreement on pen level prevalence of clinical signs in finishing pigs. Prev. Vet. Med. 2004, 64, 147–156. [Google Scholar] [CrossRef]
- Etterlin, P.E.; Morrison, D.A.; Österberg, J.; Ytrehus, B.; Heldmer, E.; Ekman, S. Osteochondrosis, but not lameness, is more frequent among free-range pigs than confined herd-mates. Acta Vet. Scand. 2015, 57, 63. [Google Scholar] [CrossRef]
- Quinn, M.M.; Keuler, N.S.; Lu, Y.; Faria, M.L.E.; Muir, P.; Markel, M.D. Evaluation of agreement between numerical rating scales, visual analogue scoring scales, and force plate gait analysis in dogs. Vet. Surg. VS 2007, 36, 360–367. [Google Scholar] [CrossRef] [PubMed]
- Karriker, L.A.; Abell, C.E.; Pairis-Garcia, M.D.; Holt, W.A.; Sun, G.; Coetzee, J.F.; Johnson, A.K.; Hoff, S.J.; Stalder, K.J. Validation of a lameness model in sows using physiological and mechanical measurements. J. Anim. Sci. 2013, 91, 130–136. [Google Scholar] [CrossRef] [PubMed]
- Pairis-Garcia, M.D.; Johnson, A.K.; Abell, C.A.; Coetzee, J.F.; Karriker, L.A.; Millman, S.T.; Stalder, K.J. Measuring the efficacy of flunixin meglumine and meloxicam for lame sows using a GAITFour pressure mat and an embedded microcomputer-based force plate system. J. Anim. Sci. 2015, 93, 2100–2110. [Google Scholar] [CrossRef] [PubMed]
- Guingamp, C.; Gegout-Pottie, P.; Philippe, L.; Terlain, B.; Netter, P.; Gillet, P. Mono-iodoacetate-induced experimental osteoarthritis: a dose-response study of loss of mobility, morphology, and biochemistry. Arthritis Rheum. 1997, 40, 1670–1679. [Google Scholar] [CrossRef] [PubMed]
- Bove, S.E.; Calcaterra, S.L.; Brooker, R.M.; Huber, C.M.; Guzman, R.E.; Juneau, P.L.; Schrier, D.J.; Kilgore, K.S. Weight bearing as a measure of disease progression and efficacy of anti-inflammatory compounds in a model of monosodium iodoacetate-induced osteoarthritis. Osteoarthritis Cartilage 2003, 11, 821–830. [Google Scholar] [CrossRef]
- Combe, R.; Bramwell, S.; Field, M.J. The monosodium iodoacetate model of osteoarthritis: A model of chronic nociceptive pain in rats? Neurosci. Lett. 2004, 370, 236–240. [Google Scholar] [CrossRef] [PubMed]
- Kalbhen, D.A. Chemical model of osteoarthritis—A pharmacological evaluation. J. Rheumatol. 1987, 14, 130–131. [Google Scholar] [PubMed]
- Van der Kraan, P.M.; Vitters, E.L.; van de Putte, L.B.; van den Berg, W.B. Development of osteoarthritic lesions in mice by “metabolic” and “mechanical” alterations in the knee joints. Am. J. Pathol. 1989, 135, 1001–1014. [Google Scholar]
- Main, D.C.J.; Clegg, J.; Spatz, A.; Green, L.E. Repeatability of a lameness scoring system for finishing pigs. Vet. Rec. 2000, 147, 574–576. [Google Scholar] [CrossRef]
- Meijer, E.; Oosterlinck, M.; van Nes, A.; Back, W.; van der Staay, F.J. Pressure mat analysis of naturally occurring lameness in young pigs after weaning. BMC Vet. Res. 2014, 10, 193. [Google Scholar]
- R Development Core Team. R: A Language and Environment for Statistical Computing; R. Foundation for statistical Computing: Vienna, Austria, 2008. [Google Scholar]
- Pinheiro, J.; Bates, D.; DebRoy, S.; Sarkar, D. nlme: Linear and Nonlinear Mixed Effects Models. Available online: https://cran.r-project.org/web/packages/nlme/index.html (accessed on 1 July 2019).
- Swindle, M.M.; Makin, A.; Herron, A.J.; Clubb, F.J.; Frazier, K.S. Swine as Models in Biomedical Research and Toxicology Testing. Vet. Pathol. Online 2012, 49, 344–356. [Google Scholar] [CrossRef] [PubMed]
- Teeple, E.; Jay, G.D.; Elsaid, K.A.; Fleming, B.C. Animal Models of Osteoarthritis: Challenges of Model Selection and Analysis. AAPS J. 2013, 15, 438–446. [Google Scholar] [CrossRef] [PubMed]
- Lampropoulou-Adamidou, K.; Lelovas, P.; Karadimas, E.V.; Liakou, C.; Triantafillopoulos, I.K.; Dontas, I.; Papaioannou, N.A. Useful animal models for the research of osteoarthritis. Eur. J. Orthop. Surg. Traumatol. 2014, 24, 263–271. [Google Scholar] [CrossRef] [PubMed]
- Guzman, R.E.; Evans, M.G.; Bove, S.; Morenko, B.; Kilgore, K. Mono-iodoacetate-induced histologic changes in subchondral bone and articular cartilage of rat femorotibial joints: An animal model of osteoarthritis. Toxicol. Pathol. 2003, 31, 619–624. [Google Scholar] [CrossRef] [PubMed]
- Stobie, D.; Lipowitz, A.J.; Greer, N.L. Failure of monoiodoacetate to induce lameness in dogs. Vet. Comp. Orthop. Traumatol. VCOT 1994, 7, 91–93. [Google Scholar] [CrossRef]
- Galois, L.; Etienne, S.; Grossin, L.; Watrin-Pinzano, A.; Cournil-Henrionnet, C.; Loeuille, D.; Netter, P.; Mainard, D.; Gillet, P. Dose-response relationship for exercise on severity of experimental osteoarthritis in rats: A pilot study. Osteoarthritis Cartilage 2004, 12, 779–786. [Google Scholar] [CrossRef] [PubMed]
- Fallah Mohammadi, M.; Hajizadeh Moghaddam, A.; Mirkarimpur, H. The Effects of a Moderate Exercise Program on Knee Osteoarthritis in Male Wistar Rats. Iran. J. Basic Med. Sci. 2013, 16, 683–688. [Google Scholar] [PubMed]
- Waxman, A.S.; Robinson, D.A.; Evans, R.B.; Hulse, D.A.; Innes, J.F.; Conzemius, M.G. Relationship between objective and subjective assessment of limb function in normal dogs with an experimentally induced lameness. Vet. Surg. VS 2008, 37, 241–246. [Google Scholar] [CrossRef] [PubMed]
- Fitzgerald, M. What do we really know about newborn infant pain? Exp. Physiol. 2015, 100, 1451–1457. [Google Scholar] [CrossRef] [PubMed]
- Fuller, C.J.; Bladon, B.M.; Driver, A.J.; Barr, A.R.S. The intra- and inter-assessor reliability of measurement of functional outcome by lameness scoring in horses. Vet. J. Lond. Engl. 1997 2006, 171, 281–286. [Google Scholar] [CrossRef]
- Hewetson, M.; Christley, R.M.; Hunt, I.D.; Voute, L.C. Investigations of the reliability of observational gait analysis for the assessment of lameness in horses. Vet. Rec. 2006, 158, 852–857. [Google Scholar] [CrossRef] [PubMed]
- Kaler, J.; Wassink, G.J.; Green, L.E. The inter- and intra-observer reliability of a locomotion scoring scale for sheep. Vet. J. Lond. Engl. 1997 2009, 180, 189–194. [Google Scholar] [CrossRef] [PubMed]
- Keegan, K.G.; Wilson, D.A.; Wilson, D.J.; Smith, B.; Gaughan, E.M.; Pleasant, R.S.; Lillich, J.D.; Kramer, J.; Howard, R.D.; Bacon-Miller, C.; et al. Evaluation of mild lameness in horses trotting on a treadmill by clinicians and interns or residents and correlation of their assessments with kinematic gait analysis. Am. J. Vet. Res. 1998, 59, 1370–1377. [Google Scholar] [PubMed]
- Rodriguez, N.A.; Cooper, D.M.; Risdahl, J.M. Antinociceptive activity of and clinical experience with buprenorphine in swine. Contemp. Top. Lab. Anim. Sci. Am. Assoc. Lab. Anim. Sci. 2001, 40, 17–20. [Google Scholar]
- Hermansen, K.; Pedersen, L.E.; Olesen, H.O. The analgesic effect of buprenorphine, etorphine and pethidine in the pig: A randomized double blind cross-over study. Acta Pharmacol. Toxicol. (Copenh.) 1986, 59, 27–35. [Google Scholar] [CrossRef] [PubMed]
- Tranquilli, W.J.; Thurmon, J.C.; Grimm, K.A. Chapter 29- Swine. In Veterinary Anesthesia and Analgesia; 4th ed.; Blackwell Puyblishing: Oxford, UK, 2007; pp. 747–763. ISBN 978-0-7817-5471-2. [Google Scholar]
- Dowling, B.; Dart, A.; Matthews, S. Chemical arthrodesis of the distal tarsal joints using sodium monoiodoacetate in 104 horses. Aust. Vet. J. 2004, 82, 38–42. [Google Scholar] [CrossRef] [PubMed]
- Goranov, N. Clinical changes in sodium monoiodoacetate induced stifle osteoarthritis model in dogs. Vet. World 2012, 5, 138–144. [Google Scholar] [CrossRef]
- Ivanavicius, S.P.; Ball, A.D.; Heapy, C.G.; Westwood, F.R.; Murray, F.; Read, S.J. Structural pathology in a rodent model of osteoarthritis is associated with neuropathic pain: Increased expression of ATF-3 and pharmacological characterisation. PAIN 2007, 128, 272–282. [Google Scholar] [CrossRef]
- Pomonis, J.D.; Boulet, J.M.; Gottshall, S.L.; Phillips, S.; Sellers, R.; Bunton, T.; Walker, K. Development and pharmacological characterization of a rat model of osteoarthritis pain. Pain 2005, 114, 339–346. [Google Scholar] [CrossRef]
- Fernihough, J.; Gentry, C.; Malcangio, M.; Fox, A.; Rediske, J.; Pellas, T.; Kidd, B.; Bevan, S.; Winter, J. Pain related behaviour in two models of osteoarthritis in the rat knee. Pain 2004, 112, 83–93. [Google Scholar] [CrossRef]
- Gorissen, B.M.C.; Uilenreef, J.J.; Bergmann, W.; Meijer, E.; van Rietbergen, B.; van der Staay, F.J.; van Weeren, P.R.; Wolschrijn, C.F. Effects of long-term use of the preferential COX-2 inhibitor meloxicam on growing pigs. Vet. Rec. 2017, 181, 564. [Google Scholar] [CrossRef] [PubMed]
- Pountos, I.; Georgouli, T.; Calori, G.M.; Giannoudis, P.V. Do nonsteroidal anti-inflammatory drugs affect bone healing? A critical analysis. Sci. World J. 2012, 2012, 606404. [Google Scholar] [CrossRef]
- Ishii, H.; Kohno, T.; Yamakura, T.; Ikoma, M.; Baba, H. Action of dexmedetomidine on the substantia gelatinosa neurons of the rat spinal cord. Eur. J. Neurosci. 2008, 27, 3182–3190. [Google Scholar] [CrossRef] [PubMed]
- Christoph, T.; Schiene, K.; Englberger, W.; Parsons, C.G.; Chizh, B.A. The antiallodynic effect of NMDA antagonists in neuropathic pain outlasts the duration of the in vivo NMDA antagonism. Neuropharmacology 2006, 51, 12–17. [Google Scholar] [CrossRef] [PubMed]
- Arendt-Nielsen, L.; Egsgaard, L.L.; Petersen, K.K.; Eskehave, T.N.; Graven- Nielsen, T.; Hoeck, H.C.; Simonsen, O. A mechanism-based pain sensitivity index to characterize knee osteoarthritis patients with different disease stages and pain levels. Eur. J. Pain 2015, 19, 1406–1417. [Google Scholar] [CrossRef] [PubMed]
- Neogi, T. The epidemiology and impact of pain in osteoarthritis. Osteoarthritis Cartilage 2013, 21, 1145–1153. [Google Scholar] [CrossRef] [PubMed]
- Thakur, M.; Dawes, J.M.; McMahon, S.B. Genomics of pain in osteoarthritis. Osteoarthritis Cartilage 2013, 21, 1374–1382. [Google Scholar] [CrossRef] [PubMed]
- King, C.D.; Sibille, K.T.; Goodin, B.R.; Cruz-Almeida, Y.; Glover, T.L.; Bartley, E.; Riley, J.L.; Herbert, M.S.; Sotolongo, A.; Schmidt, J.; et al. Experimental pain sensitivity differs as a function of clinical pain severity in symptomatic knee osteoarthritis. Osteoarthritis Cartilage 2013, 21, 1243–1252. [Google Scholar] [CrossRef] [PubMed]
- Creamer, P.; Lethbridge-Cejku, M.; Hochberg, M.C. Factors associated with functional impairment in symptomatic knee osteoarthritis. Rheumatology 2000, 39, 490–496. [Google Scholar] [CrossRef]
- Dimitroulas, T.; Duarte, R.V.; Behura, A.; Kitas, G.D.; Raphael, J.H. Neuropathic pain in osteoarthritis: A review of pathophysiological mechanisms and implications for treatment. Semin. Arthritis Rheum. 2014, 44, 145–154. [Google Scholar] [CrossRef]
- Shimura, Y.; Kurosawa, H.; Sugawara, Y.; Tsuchiya, M.; Sawa, M.; Kaneko, H.; Futami, I.; Liu, L.; Sadatsuki, R.; Hada, S.; et al. The factors associated with pain severity in patients with knee osteoarthritis vary according to the radiographic disease severity: A cross-sectional study. Osteoarthritis Cartilage 2013, 21, 1179–1184. [Google Scholar] [CrossRef] [PubMed]

| ID | Litter | Gender | Group |
|---|---|---|---|
| 1 | 1 | F 1 | MIA 3 |
| 2 | 1 | M 2 | MIA |
| 3 | 2 | F | MIA |
| 4 | 2 | F | MIA |
| 5 | 3 | M | MIA |
| 6 | 3 | F | MIA |
| 7 | 4 | F | MIA |
| 8 | 4 | F | MIA |
| 9 | 5 | F | MIA |
| 10 | 5 | F | MIA |
| 11 | 1 | M | SC 4 |
| 12 | 1 | F | SC |
| 13 | 2 | M | SC |
| 14 | 2 | M | SC |
| 15 | 3 | F | SC |
| 16 | 3 | M | SC |
| 17 | 4 | F | SC |
| 18 | 4 | M | SC |
| 19 | 5 | M | SC |
| 20 | 5 | F | SC |
| Day Relative to Joint Injection | Age of Animals (days) | Performed Procedures |
|---|---|---|
| –21 | 28 | Selection of litters and piglets; Weaning and transportation to experimenteal facility |
| –21 to –1 | 28–49 | Habitation and training (to personnel and being alone in the different test set-ups) |
| –1 | 49 | Baseline measurements:
|
| 0 | 50 | General anaesthesia for intra-articular injection:
|
| ||
| 1 | 51 | Day 1 measurements:
|
| 3 | 53 | Day 3 measurements:
|
| 7 | 57 | Day 7 measurements:
|
| 14 | 64 | Day 14 measurements:
|
| 21 | 71 | Day 21 measurements:
|
| 28 | 78 | Day 28 measurements:
|
| 56 | 106 | Day 56 measurements:
|
| 68 | 118 | General anaesthesia for euthanasia; necropsy |
| Lameness Score | Standing Posture | Gait |
|---|---|---|
| 0 | Pig stands squarely on 4 legs. | Even strides1. Caudal body sways slightly while walking. Pig is able to accelerate and change direction rapidly |
| 1 | As for score 0. | Abnormal stride length (not easily identified). Movements no longer fluent (pig appears stiff). Pig still able to accelerate and change direction. |
| 2 | Uneven posture. | Shortened stride. Lameness detected. Swagger of caudal body while walking. No hindrance in pig’s agility. |
| 3 | Uneven posture. Will not bear weight on affected limb (appears to be standing on toes). | Shortened stride. Minimum weight-bearing on affected limb. Swagger of caudal body while walking. Will still trot and gallop. |
| 4 | Affected limb elevated off floor. Pig appears visibly distressed. | Pig may not place affected limb on the floor while moving. |
| 5 | Will not stand unaided. | Does not move. |
| Day | Parameter | MIA | SC |
|---|---|---|---|
| 1 | PVF | 2.94 ± 20.59 | −0.27 ± 18.88 |
| VI | 7.36 ± 24.55 | 1.52 ± 20.30 | |
| 1 | PVF | −36.79 ± 22.94 | −28.55 ± 25.42 |
| VI | −43.47 ± 22.98 | −28.06 ± 27.59 | |
| 3 | PVF | −27.08 ± 25.41 1 | -0.38 ± 16.77 |
| VI | −33.16 ± 19.28 1 | −3.44 ± 19.10 | |
| 7 | PVF | −18.32 ± 30.12 | −3.64 ± 25.02 |
| VI | −19.90 ± 30.58 | −4.99 ± 22.05 | |
| 14 | PVF | 32.84 ± 23.58 1 | 2.31 ± 17.82 |
| VI | −33.46 ± 26.89 1 | 4.21 ± 18.63 | |
| 28 | PVF | −32.84 ± 32.67 1 | 14.69 ± 12.03 |
| VI | −32.58 ± 34.30 1 | 16.84 ± 8.63 | |
| 56 | PVF | −44.14 ± 50.09 1 | −3.10 ± 14.70 |
| VI | −40.03 ± 56.93 1 | −3.83 ± 17.06 |
| Explanatory Variable | Estimate | Standard Error | p-Value |
|---|---|---|---|
| (Intercept) 1 | 2.94 | 8.71 | 0.737 |
| Day 1 | −39.73 | 11.58 | 0.001 |
| Day3 | −30.01 | 11.58 | 0.011 |
| Day7 | −21.26 | 11.58 | 0.070 |
| Day 14 | −35.77 | 11.58 | 0.003 |
| Day28 | −35.78 | 11.58 | 0.003 |
| Day56 | −47.08 | 11.58 | 0.000 |
| SC group | −3.21 | 11.69 | 0.787 |
| Day 1 × SC group | 11.45 | 15.54 | 0.463 |
| Day 3 × SC group | 29.90 | 15.54 | 0.057 |
| Day 7 × SC group | 17.89 | 15.54 | 0.253 |
| Day14 × SC group | 38.35 | 15.54 | 0.015 |
| Day 28 × SC group | 50.75 | 15.54 | 0.002 |
| Day56 × SC group | 44.25 | 15.54 | 0.005 |
| Explanatory Variable | Estimate | Standard Error | p-Value |
|---|---|---|---|
| (Intercept) 1 | 8.382.94 | 8.50 | 0.326 |
| Day 1 | −50.83 | 11.97 | 0.000 |
| Day3 | −40.52 | 11.98 | 0.001 |
| Day7 | −27.26 | 12.02 | 0.026 |
| Day 14 | −40.83 | 12.16 | 0.001 |
| Day28 | −39.94 | 12.66 | 0.002 |
| Day56 | −47.40 | 14.46 | 0.002 |
| SC group | −1.96 | 11.69 | 0.869 |
| Day 1 × SC group | 21.24 | 16.06 | 0.189 |
| Day 3 × SC group | 35.54 | 16.08 | 0.029 |
| Day 7 × SC group | 20.74 | 16.13 | 0.202 |
| Day14 × SC group | 43.51 | 16.31 | 0.009 |
| Day 28 × SC group | 55.25 | 16.99 | 0.002 |
| Day56 × SC group | 42.04 | 19.41 | 0.033 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Uilenreef, J.; van der Staay, F.J.; Meijer, E. A Monosodium Iodoacetate Osteoarthritis Lameness Model in Growing Pigs. Animals 2019, 9, 405. https://doi.org/10.3390/ani9070405
Uilenreef J, van der Staay FJ, Meijer E. A Monosodium Iodoacetate Osteoarthritis Lameness Model in Growing Pigs. Animals. 2019; 9(7):405. https://doi.org/10.3390/ani9070405
Chicago/Turabian StyleUilenreef, Joost, Franz Josef van der Staay, and Ellen Meijer. 2019. "A Monosodium Iodoacetate Osteoarthritis Lameness Model in Growing Pigs" Animals 9, no. 7: 405. https://doi.org/10.3390/ani9070405
APA StyleUilenreef, J., van der Staay, F. J., & Meijer, E. (2019). A Monosodium Iodoacetate Osteoarthritis Lameness Model in Growing Pigs. Animals, 9(7), 405. https://doi.org/10.3390/ani9070405





