Effects of a Nutritional Supplement on Cognitive Function in Aged Dogs and on Synaptic Function of Primary Cultured Neurons
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Clinical Scores, Haematology and Serum Biochemistry
2.3. Spatial Navigation Test
2.3.1. Determination of Navigation Strategy
2.3.1.1. Experimental Setting
2.3.1.2. Familiarization
2.3.1.3. Side Choice Trial
2.3.1.4. Learning Phase
2.3.1.5. Strategy Phase
2.3.1.6. Classification of the Preferred Strategy
2.3.2. Reversal Learning Phase
2.3.2.1. From Egocentric to Allocentric Strategy
2.3.2.2. From Allocentric to Egocentric Strategy
2.3.3. Data Collection and Analysis
2.4. Primary Hippocampal Neuron Cultures
2.4.1. β-Amyloid42 and DìSeniorTM Preparation
2.4.2. DiOlistic Labeling
3. Results
3.1. Clinical Scores and Biochemical Analysis
3.2. Spatial Navigation Test
3.2.1. Determination of Navigation Strategy
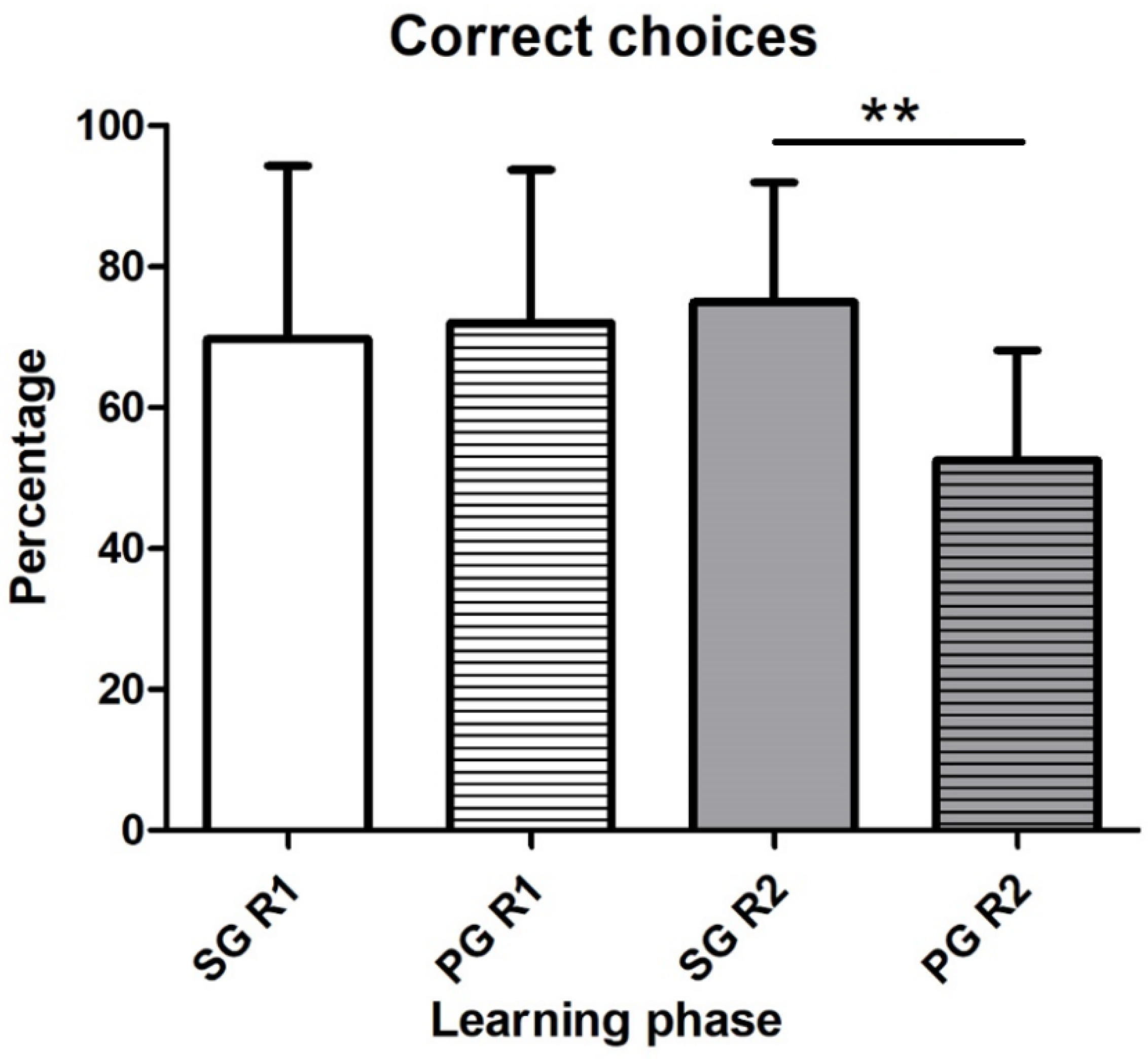
3.2.2. Reversal Learning Strategy
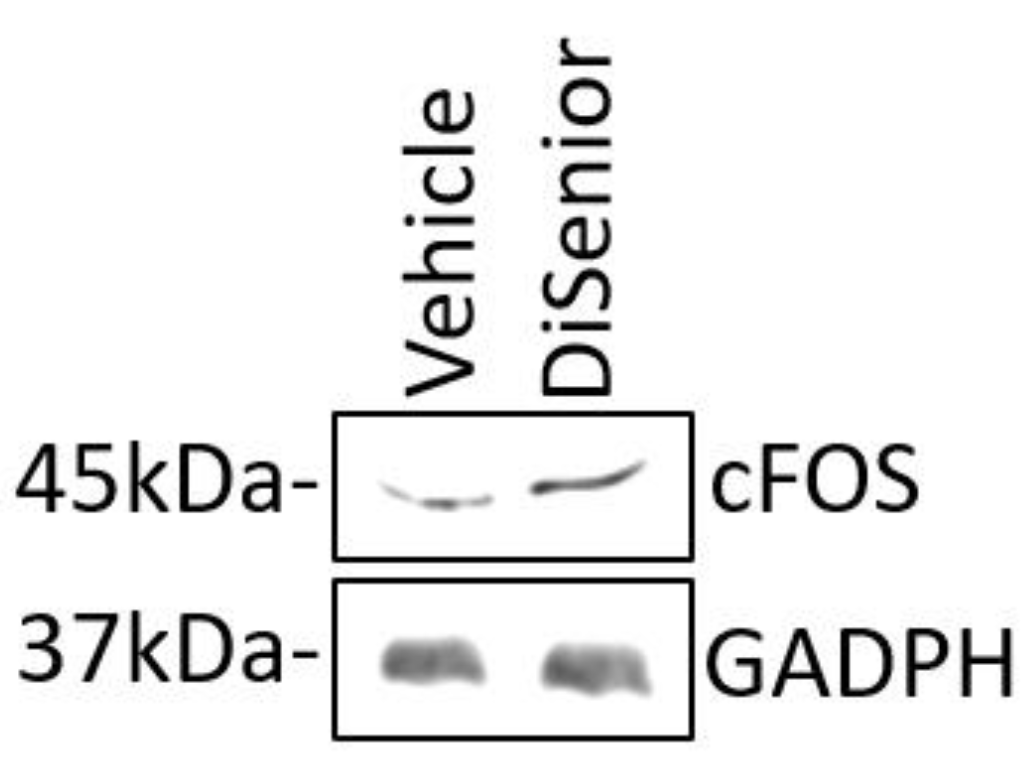
3.3. Effects on Cultured Hippocampal Neurons
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Landsberg, G.M.; Nichol, J.; Araujo, J.A. Cognitive dysfunction syndrome: A disease of canine and feline brain aging. Vet. Clin. N. Am. Small Anim. Pract. 2012, 42, 749–768. [Google Scholar] [CrossRef] [PubMed]
- Bosch, M.N.; Pugliese, M.; Gimeno-Bayon, J.; Rodriguez, M.J.; Mahy, N. Dogs with cognitive dysfunction syndrome: A natural model of Alzheimer’s disease. Curr. Alzheimer Res. 2012, 9, 298–314. [Google Scholar] [CrossRef] [PubMed]
- Davis, P.R.; Head, E. Prevention approaches in a preclinical canine model of Alzheimer’s disease: Benefits and challenges. Front. Pharmacol. 2014, 5, 47. [Google Scholar] [CrossRef] [PubMed]
- Milgram, N.W.; Head, E.; Weiner, E.; Thomas, E. Cognitive functions and aging in the dog: Acquisition of nonspatial visual tasks. Behav. Neurosci. 1994, 108, 57–68. [Google Scholar] [CrossRef] [PubMed]
- Adams, B.; Chan, A.; Callahan, H.; Milgram, N.W. The canine as a model of human cognitive aging: Recent developments. Prog. Neuropsychopharmacol. Biol. Psychiatry 2000, 24, 675–692. [Google Scholar] [CrossRef]
- Tapp, P.D.; Siwak, C.T.; Estrada, J.; Holowachuk, D.; Milgram, N.W. Effects of age on measures of complex working memory span in the beagle dog (Canis familiaris) using two versions of a spatial list learning paradigm. Learn. Mem. 2003, 10, 148–160. [Google Scholar] [CrossRef]
- Mongillo, P.; Araujo, J.A.; Pitteri, E.; Carnier, P.; Adamelli, S.; Regolin, L.; Marinelli, L. Spatial reversal learning is impaired by age in pet dogs. Age 2013, 35, 2273–2282. [Google Scholar] [CrossRef]
- Gunn-Moore, D.; Moffat, K.; Christie, L.A.; Head, E. Cognitive dysfunction and the neurobiology of ageing in cats. J. Small Anim. Pract. 2007, 48, 546–553. [Google Scholar] [CrossRef]
- Osella, M.C.; Re, G.; Odore, R.; Girardi, C.; Badino, P.; Barbero, R.; Bergamasco, L. Canine cognitive dysfunction syndrome: Prevalence, clinical signs and treatment with a neuroprotective nutraceutical. Appl. Anim. Behav. Sci. 2007, 105, 297–310. [Google Scholar] [CrossRef]
- Azkona, G.; Garcia-Belenguer, S.; Chacon, G.; Rosado, B.; Leon, M.; Palacio, J. Prevalence and risk factors of behavioural changes associated with age-related cognitive impairment in geriatric dogs. J. Small Anim. Pract. 2009, 50, 87–91. [Google Scholar] [CrossRef]
- Gonzalez-Martinez, A.; Rosado, B.; Pesini, P.; Suarez, M.L.; Santamarina, G.; Garcia-Belenguer, S.; Villegas, A.; Monleon, I.; Sarasa, M. Plasma beta-amyloid peptides in canine aging and cognitive dysfunction as a model of Alzheimer’s disease. Exp. Gerontol. 2011, 46, 590–596. [Google Scholar] [CrossRef] [PubMed]
- Landsberg, G.M.; Hunthausen, W.; Ackerman, L.A. The effect of aging on behavior in senior pets. In Behavior Problems of the Dog and Cat, 3rd ed.; Saunders: Edinburgh, UK, 2013; pp. 211–235. [Google Scholar]
- Madari, A.; Farbakova, J.; Katina, S.; Smolek, T.; Novak, P.; Weissova, T.; Novak, M.; Zilka, N. Assessment of severity and progression of canine cognitive dysfunction syndrome using the CAnine DEmentia Scale (CADES). Appl. Anim. Behav. Sci. 2015, 171, 138–145. [Google Scholar] [CrossRef]
- Rofina, J.E.; Van Ederen, A.M.; Toussaint, M.J.M.; Secrève, M.; van der Spek, A.; van der Meer, I.; Van Eerdenburg, F.J.; Gruys, E. Cognitive disturbances in old dogs suffering from the canine counterpart of Alzheimer’s disease. Brain Res. 2006, 1069, 216–226. [Google Scholar] [CrossRef] [PubMed]
- Landsberg, G.M.; DePorter, T.; Araujo, J.A. Clinical signs and management of anxiety, sleep- lessness, and cognitive dysfunction in the senior pet. Vet. Clin. N. Am. Small Anim. Pract. 2011, 41, 565–590. [Google Scholar] [CrossRef] [PubMed]
- Salvin, H.E.; McGreevy, P.D.; Sachdev, P.S.; Valenzuela, M.J. Growing old gracefully-behavioral changes associated with “successful aging” in the dog, Canis familiaris. J. Vet. Behav. Clin. Appl. Res. 2011, 6, 313–320. [Google Scholar] [CrossRef]
- Hasegawa, D.; Yayoshi, N.; Fujita, Y.; Fujita, M.; Orima, H. Measurement of interthalamic adhesion thickness as a criteria for brain atrophy in dogs with and without cognitive dysfunction (dementia). Vet. Radiol. Ultrasound 2005, 46, 452–457. [Google Scholar] [CrossRef]
- Su, M.Y.; Tapp, P.D.; Vu, L.; Chen, Y.F.; Chu, Y.; Muggenburg, B.; Chiou, J.Y.; Chen, C.; Wang, J.; Bracco, C.; et al. A longitudinal study of brain morphometrics using serial magnetic resonance imaging analysis in a canine model of aging. Progr. Neuro-Psychopharmacol. Biol. Psychiatry 2005, 29, 389–397. [Google Scholar] [CrossRef] [PubMed]
- Tapp, P.D.; Siwak, C.T.; Gao, F.Q.; Chiou, J.Y.; Black, S.E.; Head, E.; Muggenburg, B.A.; Cotman, C.W.; Milgram, N.W.; Su, M.Y. Frontal lobe volume, function, and β-amyloid pathology in a canine model of aging. J. Neurosci. 2004, 24, 8205–8213. [Google Scholar] [CrossRef] [PubMed]
- Tapp, P.D.; Head, K.; Head, E.; Milgram, N.W.; Muggenburg, B.A.; Su, M.Y. Application of an automated voxel-based morphometry technique to assess regional gray and white matter brain atrophy in a canine model of aging. Neuroimage 2006, 29, 234–244. [Google Scholar] [CrossRef] [PubMed]
- Heiblum, M.; Labastida, R.; Gris, G.C.; Tejeda, A. “Didy”, a clinical case of cognitive dysfunction syndrome. J. Vet. Behav. 2007, 2, 68–72. [Google Scholar] [CrossRef]
- Milgram, N.W.; Head, E.; Zicker, S.C.; Ikeda-Douglas, C.; Murphey, H.; Muggenberg, B.A.; Siwak, C.T.; Tapp, P.D.; Lowry, S.R.; Cotman, C.W. Long-term treatment with antioxidants and a program of behavioral enrichment reduces age-dependent impairment in discrimination and reversal learning in beagle dogs. Exp. Gerontol. 2004, 39, 753–765. [Google Scholar] [CrossRef]
- Head, E. Combining an antioxidant-fortified diet with behavioral enrichment leads to cognitive improvement and reduced brain pathology in aging canines. Ann. N. Y. Acad. Sci. 2007, 1114, 398–406. [Google Scholar] [CrossRef]
- Siwak-Tapp, C.T.; Head, E.; Muggenburg, B.A.; Milgram, N.W.; Cotman, C.W. Region specific neuron loss in the aged canine hippocampus is reduced by enrichment. Neurobiol. Aging 2008, 29, 39–50. [Google Scholar] [CrossRef] [PubMed]
- Head, E.; Nukala, V.N.; Fenoglio, K.A.; Muggenburg, B.A.; Cotman, C.W.; Sullivan, P.G. Effects of age, dietary, and behavioral enrichment on brain mitochondria in a canine model of human aging. Exp. Neurol. 2009, 220, 171–176. [Google Scholar] [CrossRef]
- Valenzuela, M.; Sachdev, P.S. Harnessing brain and cognitive reserve for the prevention of dementia. Ind. J. Psychiatry 2009, 51, 16–21. [Google Scholar]
- Chapagain, D.; Range, F.; Huber, L.; Virányi, Z. Cognitive aging in dogs. Gerontology 2018, 64, 165–171. [Google Scholar] [CrossRef] [PubMed]
- Ruehl, W.W.; Bruyette, D.S.; DePaoli, A.; Cotman, C.W.; Head, E.; Milgram, N.W.; Cummings, B.J. Canine cognitive dysfunction as a model for human age-related cognitive decline, dementia and Alzheimer’s disease: Clinical presentation, cognitive testing, pathology and response to 1-deprenyl therapy. Progr. Brain Res. 1995, 106, 217–225. [Google Scholar] [CrossRef]
- Head, E.; Hartley, J.; Kameka, A.M.; Mehta, R.; Ivy, G.O.; Ruehl, W.W.; Milgram, N.W. The effects of L-deprenyl on spatial short term memory in young and aged dogs. Progr. Neuro-Psychopharmacol. Biol. Psychiatry 1996, 20, 515–530. [Google Scholar] [CrossRef]
- Campbell, S.; Trettien, A.; Kozan, B. A noncomparative open-label study evaluating the effect of selegiline hydrochloride in a clinical setting. Vet. Ther. 2001, 2, 24–39. [Google Scholar]
- Siwak, C.T.; Gruet, P.; Woehrlé, F.; Muggenburg, B.A.; Murphey, H.L.; Milgram, N.W. Comparison of the effects of adrafinil, propentofylline, and nicergoline on behavior in aged dogs. Am. J. Vet. Res. 2000, 61, 1410–1414. [Google Scholar] [CrossRef]
- Penaliggon, J. Proceedings of the First International Conference on Behavioural Medicine; Mills, D.S., Heath, S.E., Harrington, L.J., Eds.; UFAW Herts: Great Britain, UK, 1997; pp. 37–41. [Google Scholar]
- Denenberg, S.; Landsberg, G. Current pharmacological and non-pharmacological approaches for therapy of feline and canine dementia. In Canine and Feline Dementia. Molecular Basis, Diagnostics and Therapy, 1st ed.; Landsberg, G.M., Madari, A., Žilka, N., Eds.; Springer International Publishing: Berlin, Germany, 2017; pp. 129–145. [Google Scholar]
- Gamoh, S.; Hashimoto, M.; Sugioka, K.; Shahdat Hossain, M.; Hata, N.; Misawa, Y.; Masumura, S. Chronic administration of docosahexaenoic acid improves reference memory-related learning ability in young rats. Neuroscience 1999, 93, 237–241. [Google Scholar] [CrossRef]
- Wibrand, K.; Berge, K.; Messaoudi, M.; Duffaud, A.; Panja, D.; Bramham, C.R.; Burri, L. Enhanced cognitive function and antidepressant-like effects after krill oil supplementation in rats. Lipids Health Dis. 2013, 12, 6. [Google Scholar] [CrossRef]
- Forouzanfar, F.; Hosseinzadeh, H.; Bideskan, E.A.; Sadeghnia, H.R. Aqueous and ethanolic extracts of Boswellia serrata protect against focal cerebral ischemia and reperfusion injury in rats. Phytother. Res. 2016, 30, 1954–1967. [Google Scholar] [CrossRef] [PubMed]
- Tan, M.S.; Yu, J.T.; Tan, C.C.; Wang, H.F.; Meng, X.F.; Wang, C.; Jiang, T.; Zhu, X.C.; Tan, L. Efficacy and adverse effects of Ginkgo Biloba for cognitive impairment and dementia: A systematic review and meta-analysis. J. Alzheimer Dis. 2015, 43, 589–603. [Google Scholar] [CrossRef] [PubMed]
- Macpherson, K.; Roberts, W.A. Spatial memory in dogs (Canis familiaris) on a radial maze. J. Comp. Phys. 2010, 124, 47–56. [Google Scholar] [CrossRef]
- Craig, M.; Rand, J.; Mesch, R.; Shyan-Norwalt, M.; Morton, J.; Flickinger, E. Domestic dogs (Canis familiaris) and the radial arm maze: Spatial memory and serial position effects. J. Comp. Psychol. 2012, 126, 233–242. [Google Scholar] [CrossRef] [PubMed]
- Parsons, R.L.; Ellinwood, N.M.; Zylstra, T.; Greiner, A.; Johnson, B.; Millman, S. Use of the T-maze to assess cognition in normal and MPS IIIB affected dogs. Mol. Genet. Metab. 2016, 117, S14–S124. [Google Scholar] [CrossRef]
- Scandurra, A.; Marinelli, L.; Lõoke, M.; D’Aniello, B.; Mongillo, P. The effect of age, sex and gonadectomy on dogs’ use of spatial navigation strategies. Appl. Anim. Behav. Sci. 2018, 205, 89–97. [Google Scholar] [CrossRef]
- Mongillo, P.; Bertotto, D.; Pitteri, E.; Stefani, A.; Marinelli, L.; Gabai, G. Peripheral leukocyte populations and oxidative stress biomarkers in aged dogs showing impaired cognitive abilities. Age 2015, 37, 39. [Google Scholar] [CrossRef]
- Jean, Y.Y.; Ribe, E.M.; Pero, M.E.; Moskalenko, M.; Iqbal, Z.; Marks, L.J.; Greene, L.A.; Troy, C.M. Caspase-2 is essential for c-Jun transcriptional activation and Bim induction in neuron death. Biochem. J. 2013, 455, 15–25. [Google Scholar] [CrossRef]
- Qu, X.; Yuan, F.N.; Corona, C.; Pasini, S.; Pero, M.E.; Gundersen, G.G.; Shelanski, M.L.; Bartolini, F. Stabilization of dynamic microtubules by mDia1 drives Tau-dependent Aβ1-42 synaptotoxicity. J. Cell Biol. 2017, 216, 3161–3178. [Google Scholar] [CrossRef] [PubMed]
- Murman, D.L. The impact of age on cognition. In Seminars in Hearing; Thieme Medical Publishers: Stuttgart, Germany, 2015; Volume 36, pp. 111–121. [Google Scholar]
- Gore, F.; Schwartz, E.C.; Brangers, B.C.; Aladi, S.; Stujenske, J.M.; Likhtik, E.; Russo, M.J.; Gordon, J.A.; Salzman, C.D.; Axel, R. Neural representations of unconditioned stimuli in basolateral amygdala mediate innate and learned responses. Cell 2015, 162, 134–145. [Google Scholar] [CrossRef] [PubMed]
- Tonegawa, S.; Liu, X.; Ramirez, S.; Redondo, R. Memory Engram Cells Have Come of Age. Neuron 2015, 87, 918–931. [Google Scholar] [CrossRef] [PubMed]
- de Hoz, L.; Gierej, D.; Lioudyno, V.; Jaworski, J.; Blazejczyk, M.; Cruces-Solís, H.; Beroun, A.; Lebitko, T.; Nikolaev, T.; Knapska, E.; et al. Blocking c-Fos expression reveals the role of auditory cortex plasticity in sound frequency discrimination learning. Cereb. Cortex 2018, 28, 1645–1655. [Google Scholar] [CrossRef] [PubMed]


| Item | Unit | GROUP | P | RMSE | ||||
|---|---|---|---|---|---|---|---|---|
| Supplement (SG) | Placebo (PG) | |||||||
| Round 1 | Round 2 | Round 1 | Round 2 | GROUP | TIME | |||
| HCT | % | 40.5 | 40.4 | 41.8 | 41.1 | 0.282 | 0.713 | 2.98 |
| HB | g/dL | 15.2 | 14.9 | 15.3 | 15.8 | 0.435 | 0.938 | 2.03 |
| RBC | 106/mL | 7.0 | 6.7 | 7.1 | 6.8 | 0.652 | 0.314 | 0.992 |
| WBC | 103/mL | 9.1 | 9.4 | 8.3 | 8.6 | 0.099 | 0.504 | 1.47 |
| PLT | 103/L | 275.4 | 285.8 | 287.4 | 273.6 | 0.994 | 0.971 | 44.4 |
| TP | g/L | 6.4 | 6.4 | 6.5 | 6.5 | 0.766 | 0.081 | 1.1 |
| Urea | mg/dL | 41.5 | 39.7 | 47.7 | 50.3 | 0.087 | 0.959 | 14.76 |
| Crea | mg/dL | 1.4 | 1.3 | 1.2 | 1.2 | 0.780 | 0.111 | 0.009 |
| GLU | mg/dL | 94.2 | 93.4 | 92.3 | 93.1 | 0.888 | 0.868 | 12.31 |
| ALT | U/L | 55.1 | 72 | 54.7 | 53.0 | 0.384 | 0.440 | 34.69 |
| ALP | U/L | 223.3 | 224.1 | 242 | 249 | 0.652 | 0.944 | 155 |
| BIL | mg/dL | 0.3 | 0.3 | 0.1 | 0.1 | 0.059 | 0.808 | 0.296 |
| CHOL | mg/dL | 188.1 | 182.5 | 185.3 | 209.4 | 0.136 | 0.796 | 49.88 |
| TRI | mg/dL | 59.9 | 58.2 | 72.4 | 71.2 | 0.087 | 0.837 | 22.92 |
| TT4 | g/dL | 2.1 | 2.1 | 2.3 | 2.2 | 0.360 | 0.888 | 0.446 |
| fT4 | ng/dL | 1.1 | 1.2 | 1.3 | 1.3 | 0.151 | 0.578 | 0.351 |
| TSH | IU/L | 22.7 | 23.4 | 24.7 | 21.8 | 0.918 | 0.579 | 5.37 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pero, M.E.; Cortese, L.; Mastellone, V.; Tudisco, R.; Musco, N.; Scandurra, A.; D’Aniello, B.; Vassalotti, G.; Bartolini, F.; Lombardi, P. Effects of a Nutritional Supplement on Cognitive Function in Aged Dogs and on Synaptic Function of Primary Cultured Neurons. Animals 2019, 9, 393. https://doi.org/10.3390/ani9070393
Pero ME, Cortese L, Mastellone V, Tudisco R, Musco N, Scandurra A, D’Aniello B, Vassalotti G, Bartolini F, Lombardi P. Effects of a Nutritional Supplement on Cognitive Function in Aged Dogs and on Synaptic Function of Primary Cultured Neurons. Animals. 2019; 9(7):393. https://doi.org/10.3390/ani9070393
Chicago/Turabian StylePero, Maria Elena, Laura Cortese, Vincenzo Mastellone, Raffaella Tudisco, Nadia Musco, Anna Scandurra, Biagio D’Aniello, Giuseppe Vassalotti, Francesca Bartolini, and Pietro Lombardi. 2019. "Effects of a Nutritional Supplement on Cognitive Function in Aged Dogs and on Synaptic Function of Primary Cultured Neurons" Animals 9, no. 7: 393. https://doi.org/10.3390/ani9070393
APA StylePero, M. E., Cortese, L., Mastellone, V., Tudisco, R., Musco, N., Scandurra, A., D’Aniello, B., Vassalotti, G., Bartolini, F., & Lombardi, P. (2019). Effects of a Nutritional Supplement on Cognitive Function in Aged Dogs and on Synaptic Function of Primary Cultured Neurons. Animals, 9(7), 393. https://doi.org/10.3390/ani9070393






