Dietary Pomegranate Pulp: Effect on Ewe Milk Quality during Late Lactation
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Dried Pomegranate Pulp
2.2. Animals and Diet
2.3. Sampling and Analyses
2.3.1. Feed
2.3.2. Milk
2.3.3. Total Antioxidant Capacity of Feeds and Milk
2.4. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Viuda-Martos, M.; Fernández-Lopéz, J.; Pérez-Álvarez, J.A. Pomegranate and its many functional components as related to human health: A review. Compr. Rev. Food Sci. Food Saf. 2010, 9, 635–643. [Google Scholar] [CrossRef]
- Gil, M.; Tomás-Barberán, F.A.; Hess-Pierce, B.; Holcroft, D.M.; Kader, A.A. Antioxidant activity of pomegranate juice and its relationship with phenolic composition and processing. J. Agric. Food Chem. 2000, 48, 4581–4589. [Google Scholar] [CrossRef] [PubMed]
- Akhtar, S.; Ismail, T.; Fraternale, D.; Sestili, P. Pomegranate peel and peel extracts: chemistry and food features. Food Chem. 2015, 174, 417–425. [Google Scholar] [CrossRef]
- Aruna, P.; Venkataramanamma, D.; Singh, A.K.; Singh, R.P. Health benefits of punicic acid: A review. Compr. Rev. Food Sci. Food Saf. 2015, 15, 16–27. [Google Scholar] [CrossRef]
- Vasta, V.; Luciano, G. The effects of dietary consumption of plants secondary compounds on small ruminants’ products quality. Small Rum. Res. 2011, 101, 150–159. [Google Scholar] [CrossRef]
- Liu, H.; Li, K.; Mingbin, L.; Zhao, J.; Xiong, B. Effects of chestnut tannins on the meat quality, welfare, and antioxidant status of heat-stressed lambs. Meat Sci. 2016, 116, 236–242. [Google Scholar] [CrossRef]
- Shaani, Y.; Eliyahu, D.; Mizrahi, I.; Yosef, E.; Ben-Meir, Y.; Nikbachat, M.; Solomon, R.; Mabjeesh, S.J. Miron. J. Effect of feeding ensiled mixture of pomegranate pulp and drier feeds on digestibility and milk performance in dairy cows. J. Dairy Res. 2016, 83, 35–41. [Google Scholar] [CrossRef]
- Modaresi, J.; Fathi-Nasri, M.H.; Rashidi, L.; Dayani, O.; Kebreab, E. Effects of supplementation with pomegranate seed pulp on concentrations of conjugated linoleic acid and punicic acid in goat milk. J. Dairy Sci. 2011, 94, 4075–4080. [Google Scholar] [CrossRef]
- Emami, M.H.; Fathi-Nasri, F.; Ganjkhanlou, M.; Rashidi, L.; Zali, A. Effect of pomegranate seed oil as a source of conjugated linolenic acid on performance and milk fatty acid profile of dairy goats. Livest. Sci. 2016, 193, 1–7. [Google Scholar] [CrossRef]
- Kotsampasi, B.; Christodoulou, C.; Tsiplakou, E.; Mavrommatis, A.; Mitsiopoulou, C.; Karaiskou, C.; Dotasa, V.; Robinsonc, P.H.; Bampidisd, V.A.; Christodouloua, V.; Zervasb, G. Effects of dietary pomegranate pulp silage supplementation on milk yield and composition, milk fatty acid profile and blood plasma antioxidant status of lactating dairy cows. Anim. Feed Sci. Technol. 2017, 234, 228–236. [Google Scholar] [CrossRef]
- Kotsampasi, B.; Christodouloua, V.; Zotosb, A.; Liakopoulou-Kyriakidesc, M.; Goulasd, P.; Petrotosd, K.; Natasc, P.; Bampidis, V.A. Effects of dietary pomegranate byproduct silage supplementation on performance, carcass characteristics and meat quality of growing lambs. Anim. Feed Sci. Technol. 2014, 197, 92–102. [Google Scholar] [CrossRef]
- Cannas, A.; Tedeschi, L.O.; Fox, D.G.; Pell, A.N.; Van Soest, P.J. A mechanistic model for predicting the nutrient requirements and feed biological values for sheep. J. Anim. Sci. 2004, 82, 149–169. [Google Scholar] [CrossRef]
- D’Urso, G.; Avondo, M.; Bordonaro, S.; Marletta, M.; Guastella, A.M. Effect of sustained-release somatotropin on performance and grazing behavior of ewes housed at different stocking rates. J. Dairy Sci. 1998, 81, 958–965. [Google Scholar] [CrossRef]
- AOAC International. Official Methods of Analysis, 19th ed.; AOAC International: Rockvile, MD, USA, 1995. [Google Scholar]
- Van Soest, P.J.; Robertson, J.B.; Lewis, B.A. Methods for dietary fibre, neutral detergent fiber, and nonstarch polysaccharides in relation to animal nutrition. J. Dairy Sci. 1991, 74, 3583–3597. [Google Scholar] [CrossRef]
- Makkar, H.P.S.; Blümmel, M.; Borowy, N.K.; Becker, K. Gravimetric determination of tannins and their correlations with chemical and protein-precipitation methods. J. Sci. Food Agric. 1993, 61, 161–165. [Google Scholar] [CrossRef]
- Valenti, B.; Pagano, R.I.; Avondo, M. Effect of diet at different energy levels on milk casein composition of Girgentana goats differing in CSN1S1 genotype. Small Rum. Res. 2012, 105, 135–139. [Google Scholar] [CrossRef]
- Clément, P.; Agboola, S.O.; Bencini, R. A study of polymorphism in milk proteins from local and imported dairy sheep in Australia by capillary electrophoresis. LWT 2006, 39, 63–69. [Google Scholar] [CrossRef]
- Folch, J.; Lees, M.; Stanley, G.H.S. A simple method for the isolation and purification of lipids from animal tissue. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar]
- Christie, W.W. A simple procedure for rapid transmethylation of glycerolipids and cholesteryl esters. J. Lipid Res. 1982, 23, 1072–1075. [Google Scholar]
- Valenti, B.; Luciano, G.; Pauselli, M.; Mattioli, S.; Biondi, L.; Priolo, A.; Natalello, A.; Morbidini, L.; Lanza, M. Dried tomato pomace supplementation to reduce lamb concentrate intake: Effects on growth performance and meat quality. Meat Sci. 2018, 145, 63–70. [Google Scholar] [CrossRef]
- Prior, R.L.; Hoang, H.; Gu, L.; Wu, X.; Bacchiocca, M.; Howard, L.; Hampsch-Woodill, M.; Huang, D.; Ou, B.; Jacob, R. Assays for hydrophilic and lipophilic antioxidant capacity (oxygen radical absorbance capacity (ORACFL)) of plasma and other biological and food samples. J. Agric. Food Chem. 2003, 51, 3273–3279. [Google Scholar] [CrossRef]
- Wu, X.; Beecher, G.; Holden, J.; Haytowitz, D.; Gebhardt, S.E.; Prior, R.L. Lipophilic and hydrophilic antioxidant capacities of common foods in the U.S. J. Agric. Food Chem. 2004, 52, 4026–4037. [Google Scholar] [CrossRef]
- Zulueta, A.; Maurizi, A.; Frigola, A.; Esteve, M.J.; Coli, R.; Burini, B. Antioxidant capacity of cow milk, whey and deproteinized milk. Int. Dairy J. 2009, 19, 380–385. [Google Scholar] [CrossRef]
- Ou, B.; Hampsch-Woodill, M.; Prior, R.L. Development and validation of an improved oxygen radical absorbance capacity assay using fluorescein as the fluorescent probe. J. Agric. Food Chem. 2001, 49, 4619–4626. [Google Scholar] [CrossRef]
- Eliyahu, D.; Shaani, Y.; Yosef, E.; Ben-Meir, Y.; Nikbachat, M.; Solomon, R.; Mabjeeshc, S.J.; Weinbergd, Z.G.; Mirona, J. Effect of ensiling pomegranate pulp with solid additives on chemical composition, intake and digestibility by sheep. Small Rum. Res. 2015, 131, 93–98. [Google Scholar] [CrossRef]
- Abarghuei, M.J.; Rouzbehan, Y.; Salem, A.Z.M.; Zamiri, M.J. Nitrogen balance, blood metabolites and milk fatty acid composition of dairy cows fed pomegranate-peel extract. Livest. Sci. 2014, 164, 72–80. [Google Scholar] [CrossRef]
- Makkar, H.P.S. Effects and fate of tannins in ruminant animals, adaptation to tannins, and strategies to overcome detrimental effects of feeding tannin-rich feeds. Small Rum Res. 2003, 49, 241–256. [Google Scholar] [CrossRef]
- Valenti, B.; Natalello, A.; Vasta, V.; Campidonico, L.; Roscini, V.; Mattioli, S.; Pauselli, M.; Priolo, A.; Lanza, M.; Luciano, G. Effect of different dietary tannin extracts on lamb growth performances and meat oxidative stability: Comparison between mimosa, chestnut and tara. Animal 2019, 13, 435–443. [Google Scholar] [CrossRef]
- Buccioni, A.; Serra, A.; Minieri, S.; Mannelli, F.; Cappucci, A.; Benvenuti, D.; Rapaccini, S.; Conte, G.; Mele, M. Milk production, composition, and milk fatty acid profile from grazing sheep fed diets supplemented with chestnut tannin extract and extruded linseed. Small Rum Res. 2015, 130, 200–207. [Google Scholar] [CrossRef]
- Aguerre, M.J.; Capozzolo, M.C.; Lencioni, P.; Cabral, C.; Wattiaux, M.A. Effect of quebracho-chestnut tannin extracts at 2 dietary crude protein levels on performance, rumen fermentation, and nitrogen partitioning in dairy cows. J. Dairy Sci. 2016, 99, 4476–4486. [Google Scholar] [CrossRef]
- Jeronimo, E.; Pinheiro, C.; Lamy, E.; Dentinho, M.T.; Sales-Baptista, E.; Lopes, O.; Capela e Silva, F. Tannins in ruminant nutrition: Impact on animal performance and quality of edible products. In Tannins: Biochemistry, Food Sources and Nutritional Properties; Combs, C.A., Ed.; Nova Science Publisher Inc.: Hauppauge, NY, USA, 2016; pp. 121–168. [Google Scholar]
- Min, B.R.; Barry, T.N.; Attwood, G.T.; McNabb, W.C. The effect of condensed tannins on the nutrition and health of ruminants fed fresh temperate forages: A review. Anim. Feed Sci. Technol. 2003, 106, 3–19. [Google Scholar] [CrossRef]
- Waghorn, G. Beneficial and detrimental effects of dietary condensed tannins for sustainable sheep and goat production—Progress and challenges. Anim. Feed Sci. Technol. 2008, 147, 116–139. [Google Scholar] [CrossRef]
- Orlandi, T.; Kozloski, G.V.; Alves, T.P.; Mesquita, F.R.; Ávila, S.C. Digestibility, ruminal fermentation and duodenal flux of amino acids in steers fed grass forage plus concentrate containing increasing levels of Acacia mearnsii tannin extract. Anim. Feed Sci. Technol. 2015, 210, 37–45. [Google Scholar] [CrossRef]
- López-Andrés, P.; Luciano, G.; Vasta, V.; Gibson, T.M.; Biondi, L.; Priolo, A.; Mueller-Harvey, I. Dietary quebracho tannins are not absorbed, but increase the antioxidant capacity of liver and plasma in sheep. Br. J. Nutr. 2013, 110, 632–639. [Google Scholar] [CrossRef] [PubMed]
- Clausen, M.R.; Skibsted, L.H.; Stagsted, J. Characterization of major radical scavenger species in bovine milk through size exclusion chromatography and functional assays. J. Agric. Food Chem. 2009, 57, 2912–2919. [Google Scholar] [CrossRef] [PubMed]
- Chilliard, Y.; Glasser, F.; Ferlay, A.; Bernard, L.; Rouel, J.; Doreau, M. Diet, rumen biohydrogenation and nutritional quality of cow and goat milk fat. Eur. J. Lipid Sci. Technol. 2007, 109, 828–855. [Google Scholar] [CrossRef]
- Verardo, V.; Garcia-Salas, P.; Baldi, E.; Segura-Carretero, A.; Fernandez-Gutierrez, A.; Caboni, M. Pomegranate seeds as a source of nutraceutical oil naturally rich in bioactive lipids. Food Res. Int. 2014, 65, 445–452. [Google Scholar] [CrossRef]
- Buccioni, A.; Decandia, M.; Minieri, S.; Molle, G.; Cabiddu, A. Lipid metabolism in the rumen: New insights on lipolysis and biohydrogenation with an emphasis on the role of endogenous plant factors. Anim. Feed Sci. Technol. 2012, 174, 1–25. [Google Scholar] [CrossRef]
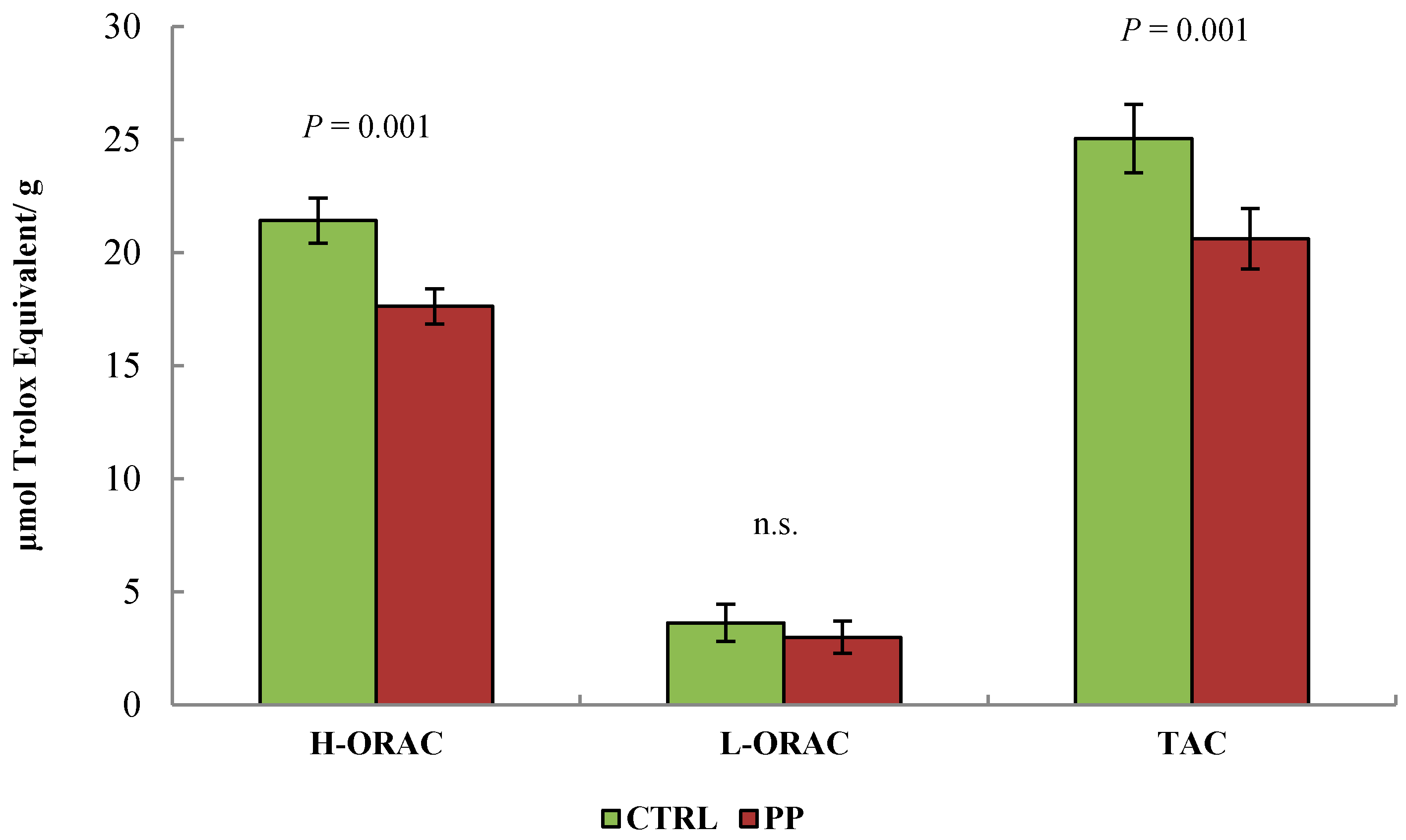
| Item | Pomegranate Pulp | Pasture | Hay | Barley | Concentrate 1 | |
|---|---|---|---|---|---|---|
| CTRL | PP | |||||
| Ingredients (g/kg dry matter) | ||||||
| Wheat bran | 205.6 | 71.4 | ||||
| Corn | 345.0 | 40.1 | ||||
| Barley | 336.0 | 32.7 | ||||
| Soybean meal | 35.5 | 130.4 | ||||
| Pomegranate pulp | - | 648.4 | ||||
| Molasses | 51.9 | 51.9 | ||||
| Mineral mix | 25.9 | 25.6 | ||||
| Chemical composition (g/kg dry matter) | ||||||
| Dry matter | 911.6 | 22.3 | 87.6 | 88.9 | 85.6 | 85.3 |
| Crude protein | 68.8 | 135.4 | 156.2 | 118.6 | 140.6 | 139.8 |
| Crude fat | 44.3 | 26.0 | 15.9 | 34.4 | 30.5 | 38.9 |
| Neutral detergent fiber | 314.0 | 485.1 | 443.3 | 269.3 | 221.0 | 237.1 |
| Acid detergent fiber | 227.8 | 318.8 | 348.9 | 102.5 | 100.9 | 159.6 |
| Acid detergent lignin | 68.8 | 35.9 | 70.7 | 24.4 | 20.3 | 39.4 |
| Ash | 36.4 | 93.3 | 98.7 | 26.8 | 61.2 | 67.4 |
| ME 2 | 10.7 | 10.4 | 9.52 | 13.47 | 10.9 | 12.2 |
| Total phenols 3 | 95.3 | 2.01 | 8.22 | 0.59 | 1.25 | 61.4 |
| Total tannins 3 | 93.4 | - | 2.48 | 0.03 | - | 60.9 |
| Fatty acids (g/100 g total fatty acids) | ||||||
| 14:0 | 0.06 | 0.42 | 0.78 | 0.33 | 0.14 | 0.07 |
| 16:0 | 3.54 | 13.6 | 20.7 | 20.2 | 14.2 | 5.80 |
| 18:0 | 1.92 | 1.43 | 3.90 | 2.49 | 2.05 | 2.05 |
| cis-9 18:1 | 5.43 | 3.25 | 4.85 | 14.1 | 22.0 | 7.84 |
| cis-9 cis-12 18:2 | 5.87 | 15.8 | 16.6 | 55.0 | 50.2 | 14.1 |
| cis-9 cis-12 cis-15 18:3 | 0.29 | 56.9 | 35.9 | 4.17 | 2.73 | 1.11 |
| cis-9 trans-11 cis-13 18:3 | 71.2 | - | - | - | - | 57.7 |
| Item | Concentrate 1 (Diet) | SEM 2 | p Value | |||
|---|---|---|---|---|---|---|
| CTRL | PP | Diet | Day | Diet × Day | ||
| Milk yield (g/day) | 332.2 | 339.7 | 19.9 | 0.910 | 0.252 | 0.025 |
| Fat (%) | 8.29 | 8.30 | 0.110 | 0.961 | 0.630 | 0.366 |
| Protein (%) | 6.53 | 6.45 | 0.069 | 0.521 | 0.064 | 0.764 |
| Lactose (%) | 4.29 | 4.21 | 0.048 | 0.382 | 0.725 | 0.604 |
| Urea (mg/mL) | 55.0 | 54.7 | 1.35 | 0.931 | 0.071 | 0.644 |
| Linear score | 3.40 | 4.30 | 0.227 | 0.031 | 0.609 | 0.406 |
| Protein fractions (% total protein) | ||||||
| α-lactalbumin | 2.19 | 2.30 | 0.076 | 0.441 | 0.031 | 0.447 |
| β-lactoglobulin | 14.2 | 14.6 | 0.167 | 0.277 | 0.063 | 0.142 |
| αs2-casein | 6.49 | 6.71 | 0.177 | 0.467 | 0.118 | 0.557 |
| αs1-casein | 33.9 | 35.3 | 0.368 | 0.056 | 0.590 | 0.542 |
| κ-casein | 6.02 | 5.92 | 0.179 | 0.801 | 0.469 | 0.280 |
| β-casein | 33.6 | 31.3 | 0.592 | 0.048 | 0.209 | 0.477 |
| Whey protein | 16.4 | 16.9 | 0.204 | 0.234 | 0.030 | 0.119 |
| Total casein | 80.3 | 79.0 | 0.321 | 0.002 | 0.002 | 0.032 |
| Item | Concentrate 1 (Diet) | SEM 2 | p Value | |||
|---|---|---|---|---|---|---|
| CTRL | PP | Diet | Day | Diet × Day | ||
| 4:0 | 2.08 | 2.19 | 0.066 | 0.328 | <0.001 | 0.604 |
| 6:0 | 1.74 | 1.83 | 0.048 | 0.252 | 0.001 | 0.742 |
| 8:0 | 1.66 | 1.77 | 0.047 | 0.190 | 0.050 | 0.953 |
| 10:0 | 5.34 | 5.53 | 0.168 | 0.581 | 0.272 | 0.993 |
| 12:0 | 3.07 | 3.09 | 0.098 | 0.925 | 0.171 | 0.925 |
| cis-9 12:1 | 0.15 | 0.14 | 0.005 | 0.542 | 0.037 | 0.970 |
| 14:0 | 9.97 | 9.22 | 0.201 | 0.056 | 0.163 | 0.866 |
| cis-9 14:1 | 0.26 | 0.21 | 0.011 | 0.027 | 0.396 | 0.985 |
| iso 15:0 | 0.38 | 0.37 | 0.006 | 0.474 | 0.531 | 0.317 |
| anteiso 15:0 | 0.66 | 0.63 | 0.012 | 0.274 | 0.521 | 0.404 |
| 15:0 | 1.43 | 1.44 | 0.015 | 0.911 | 0.000 | 0.759 |
| iso 16:0 | 0.36 | 0.37 | 0.008 | 0.938 | 0.504 | 0.711 |
| 16:0 | 28.8 | 26.4 | 0.383 | 0.001 | 0.132 | 0.627 |
| cis-9 16:1 | 1.26 | 1.04 | 0.037 | 0.003 | 0.618 | 0.859 |
| iso 17:0 | 0.59 | 0.62 | 0.011 | 0.193 | 0.470 | 0.589 |
| anteiso 17:0 | 0.64 | 0.67 | 0.016 | 0.276 | 0.097 | 0.237 |
| 17:0 | 0.87 | 0.92 | 0.016 | 0.090 | 0.025 | 0.964 |
| 18:0 | 8.86 | 9.20 | 0.213 | 0.427 | 0.104 | 0.991 |
| trans-6+7+8 18:1 | 0.14 | 0.17 | 0.007 | 0.106 | 0.984 | 0.835 |
| trans-9 18:1 | 0.25 | 0.32 | 0.007 | <0.001 | 0.592 | 0.480 |
| trans-10 18:1 | 0.22 | 0.23 | 0.007 | 0.509 | 0.678 | 0.756 |
| trans-11 18:1 | 1.04 | 1.45 | 0.006 | <0.001 | 0.974 | 0.987 |
| cis-6 18:1 | 0.71 | 0.85 | 0.029 | 0.014 | 0.115 | 0.788 |
| cis-9 18:1 | 18.8 | 18.8 | 0.361 | 0.976 | 0.013 | 0.921 |
| cis-11 18:1 | 0.38 | 0.41 | 0.014 | 0.342 | 0.021 | 0.770 |
| cis-12 18:1 | 0.22 | 0.25 | 0.010 | 0.118 | 0.521 | 0.999 |
| cis-9 trans-11 18:2 | 0.69 | 1.28 | 0.051 | <0.001 | 0.999 | 0.779 |
| cis-9 cis-12 18:2 | 2.49 | 2.70 | 0.060 | 0.078 | 0.432 | 0.732 |
| cis-6 cis-9 cis-12 18:3 | 0.48 | 0.42 | 0.002 | 0.218 | 0.457 | 0.876 |
| cis-9 cis-12 cis-15 18:3 | 1.41 | 1.60 | 0.044 | 0.024 | 0.132 | 0.711 |
| cis-9 trans-11 cis-13 18:3 | n.d. | 0.19 | 0.018 | − | − | − |
| 20:0 | 0.36 | 0.36 | 0.008 | 0.917 | 0.123 | 0.902 |
| cis-11 20:1 | 0.58 | 0.64 | 0.002 | 0.108 | 0.803 | 0.849 |
| 20:3 n-6 | 0.03 | 0.03 | 0.002 | 0.622 | 0.698 | 0.809 |
| 20:4 n-6 | 0.23 | 0.22 | 0.006 | 0.695 | 0.371 | 0.842 |
| 20:5 n-3 | 0.10 | 0.10 | 0.003 | 0.561 | 0.121 | 0.689 |
| 22:2 | 0.03 | 0.04 | 0.002 | 0.224 | 0.004 | 0.924 |
| 22:4 n-6 | 0.04 | 0.04 | 0.003 | 0.253 | 0.457 | 0.705 |
| 22:5 n-6 | 0.10 | 0.09 | 0.003 | 0.277 | 0.235 | 0.821 |
| 22:6 n-3 | 0.19 | 0.20 | 0.005 | 0.288 | 0.107 | 0.789 |
| 24:0 | 0.12 | 0.11 | 0.011 | 0.818 | 0.675 | 0.524 |
| Σ SFA 3 | 62.1 | 59.7 | 0.563 | 0.029 | 0.206 | 0.757 |
| Σ MUFA 4 | 23.5 | 23.9 | 0.375 | 0.566 | 0.023 | 0.885 |
| Σ PUFA 5 | 5.40 | 6.48 | 0.159 | <0.001 | 0.595 | 0.783 |
| Σ PUFA n-3 | 1.70 | 1.90 | 0.047 | 0.025 | 0.240 | 0.729 |
| Σ PUFA n-6 | 2.96 | 3.16 | 0.067 | 0.151 | 0.463 | 0.815 |
| Σ OBCFA 6 | 4.94 | 5.02 | 0.061 | 0.503 | 0.264 | 0.553 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Valenti, B.; Luciano, G.; Morbidini, L.; Rossetti, U.; Codini, M.; Avondo, M.; Priolo, A.; Bella, M.; Natalello, A.; Pauselli, M. Dietary Pomegranate Pulp: Effect on Ewe Milk Quality during Late Lactation. Animals 2019, 9, 283. https://doi.org/10.3390/ani9050283
Valenti B, Luciano G, Morbidini L, Rossetti U, Codini M, Avondo M, Priolo A, Bella M, Natalello A, Pauselli M. Dietary Pomegranate Pulp: Effect on Ewe Milk Quality during Late Lactation. Animals. 2019; 9(5):283. https://doi.org/10.3390/ani9050283
Chicago/Turabian StyleValenti, Bernardo, Giuseppe Luciano, Luciano Morbidini, Umberto Rossetti, Michela Codini, Marcella Avondo, Alessandro Priolo, Marco Bella, Antonio Natalello, and Mariano Pauselli. 2019. "Dietary Pomegranate Pulp: Effect on Ewe Milk Quality during Late Lactation" Animals 9, no. 5: 283. https://doi.org/10.3390/ani9050283
APA StyleValenti, B., Luciano, G., Morbidini, L., Rossetti, U., Codini, M., Avondo, M., Priolo, A., Bella, M., Natalello, A., & Pauselli, M. (2019). Dietary Pomegranate Pulp: Effect on Ewe Milk Quality during Late Lactation. Animals, 9(5), 283. https://doi.org/10.3390/ani9050283






