Phenotypic Stability of Sex and Expression of Sex Identification Markers in the Adult Yesso Scallop Mizuhopecten yessoensis throughout the Reproductive Cycle
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Tracking Experiment for the Analysis of Phenotypic Stability of Sex in the Yesso Scallop
2.2. Sample Preparation for mRNA Expression Analyses
2.3. Transcriptomic Survey and cDNA Cloning
2.4. Bioinformatic Analyses
2.5. Semi-Quantitative RT-PCR Assay
2.6. ISH Detection
2.7. qPCR Assay
3. Results
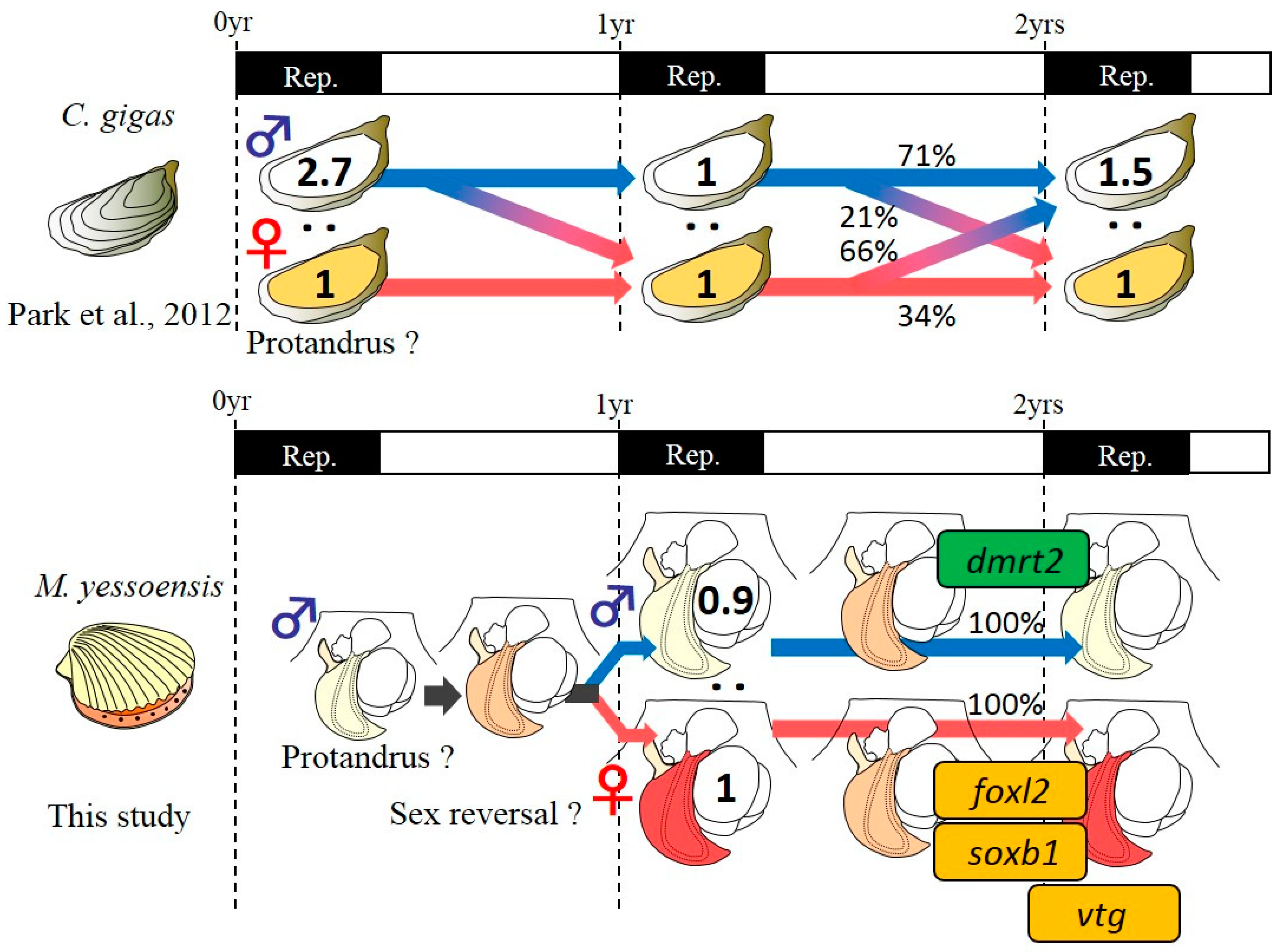
3.1. Tracking of the Sex Phenotype in the Yesso Scallops between One and Two Years of Age
3.2. Characterization of dmrts and foxl2 in the Yesso Scallops
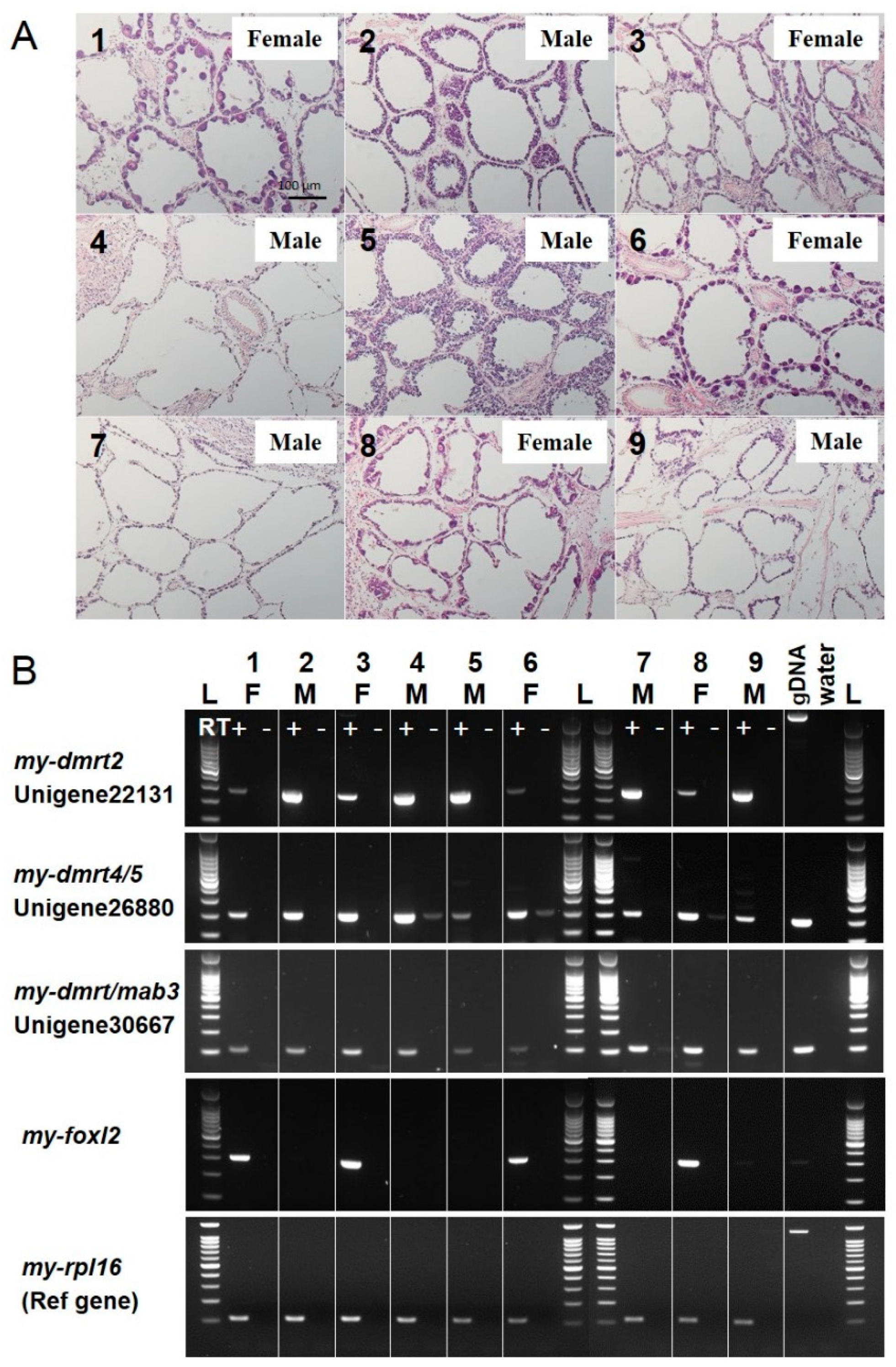
3.3. Validation of Molecular Markers of Sex Identification for Testis/Male or Ovary/Female in the Adult Yesso Scallop
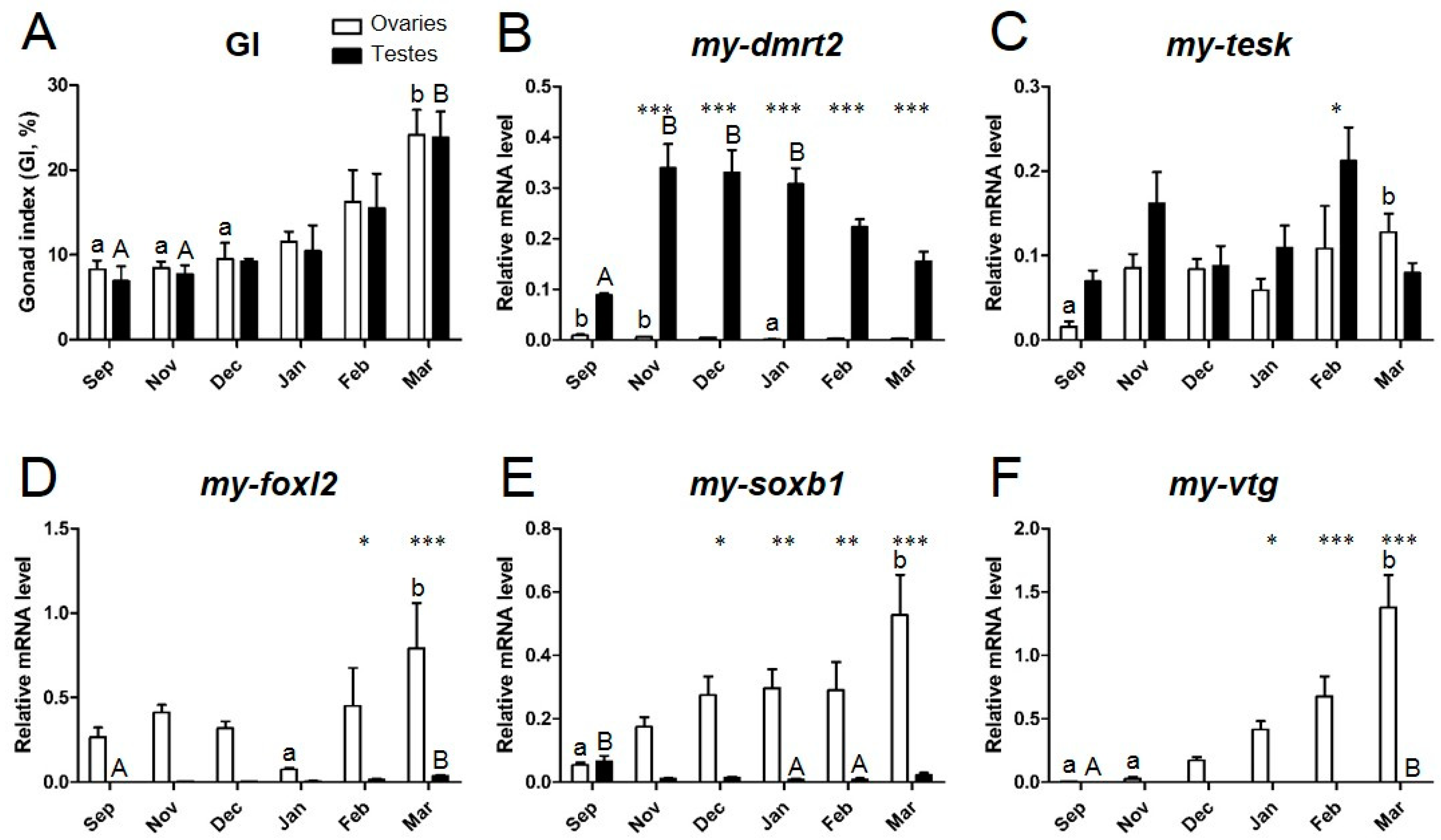
3.4. Expression Profiles of Sex Identification Markers in the Yesso Scallop Gonads during the Reproductive Cycle
4. Discussion
4.1. Phenotypic Stability of Sex in the Yesso Scallop
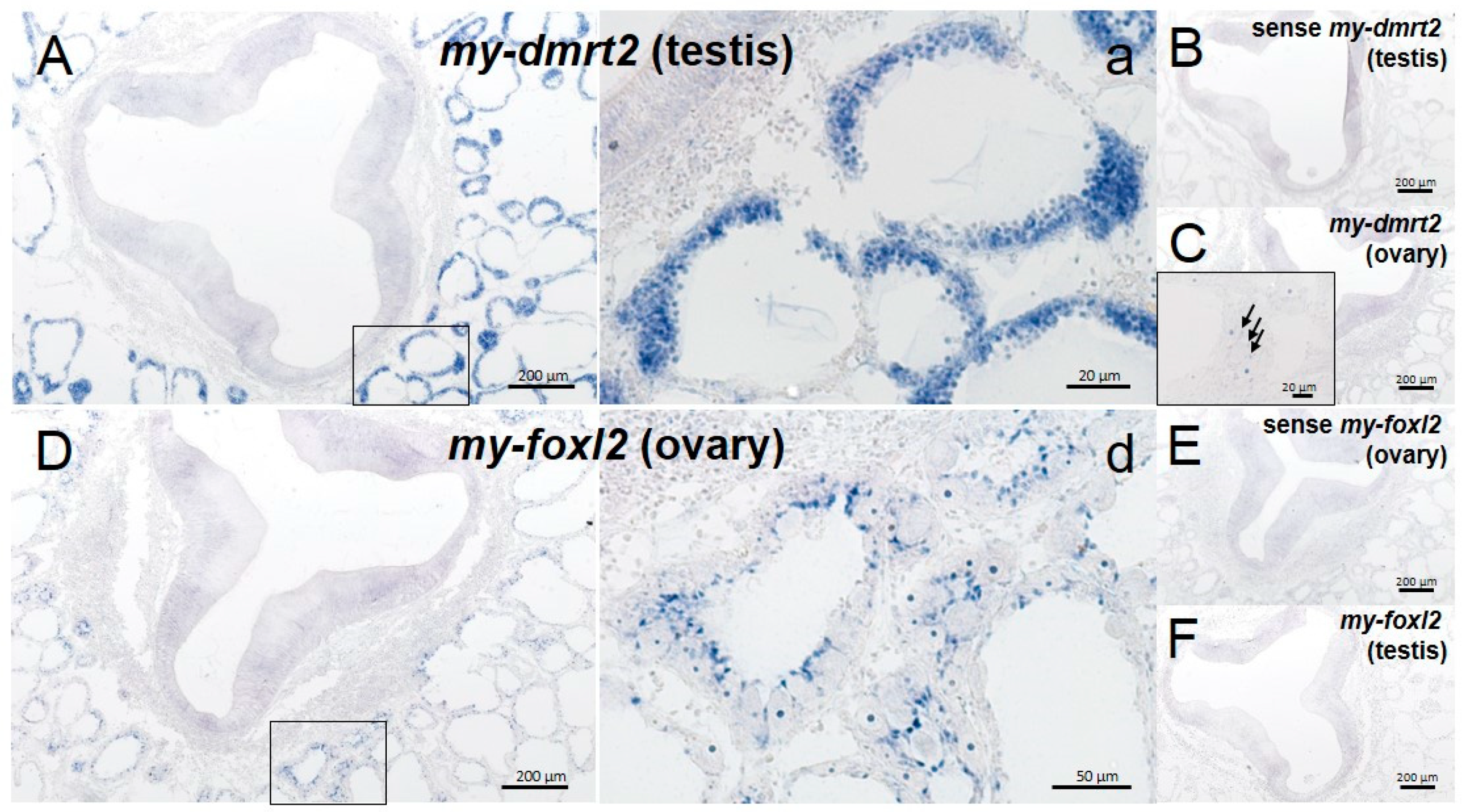
4.2. my-dmrt2 as a Testis Marker in the Yesso Scallop
4.3. my-foxl2 as an Ovary Marker in the Yesso Scallop
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Shumway, S.; Parsons, G.J. Scallops: Biology, Ecology, Aquaculture, and Fisheries. In Scallops: Biology, Ecology, Aquaculture, and Fisheries; Shumway, S.E., Parsons, G.J., Eds.; Elsevier: Amsterdam, The Netherlands, 2016; Volume 40. [Google Scholar]
- Wakui, T.; Obara, A. On the seasonal change of the gonads of scallop, Patinopecten yessoensis (JAY), in lake Saroma, Hokkaido. Bull. Hokkaido Reg. Fish. Res. Lab. 1967, 32, 15–22. [Google Scholar]
- Osanai, K.; Hirai, S.; Odashima, M.; Kyozuka, K. Sexual differentiation in the juveniles of the scallop, Patinopecten yessoensis. Bull. Mar. Biol. Stn. Asamushi Tohoku Univ. 1980, 16, 221–230. [Google Scholar]
- Mori, K.; Osanai, K.; Sato, R. Seasonal gonad changes in scallops under culture in Toni Bay, Iwate Prefecture. Bull. Jpn. Soc. Sci. Fish. 1977, 43, 1–8. [Google Scholar] [CrossRef]
- Kawamata, K. Gonadal development of cultured scallops, Patinopecten yessoensis (JAY), in Lake Saroma, Hokkaido. Sci. Rep. Hokkaido Fish. Exp. Stn. 1994, 45, 37–44. [Google Scholar]
- Maru, K. Studies on the reproduction of a scallop, Patinopecten yessoensis (Jay), 1: Reproductive cycle of the cultured scallop. Sci. Rep. Hokkaido Fish. Exp. Stn. 1976, 18, 9–26. [Google Scholar]
- Park, J.J.; Kim, H.; Kang, S.W.; An, C.M.; Lee, S.H.; Gye, M.C.; Lee, J.S. Sex ratio and sex reversal in two-year-old class of oyster, Crassostrea gigas (Bivalvia: Ostreidae). Dev. Reprod. 2012, 16, 385–388. [Google Scholar] [CrossRef] [PubMed]
- Breton, S.; Capt, C.; Guerra, D.; Stewart, D. Sex Determining mechanisms in bivalves. Preprints 2017, 2017, 2017060127. [Google Scholar] [CrossRef][Green Version]
- Lee, J.S.; Park, J.J.; Shin, Y.K.; Kim, H.; Jeon, M.A. Sex change and sequential hermaphroditism in Tegillarca granosa (Bivalvia: Arcidae). Invertebr. Reprod. Dev. 2014, 58, 314–318. [Google Scholar] [CrossRef]
- Coe, W.R. Development of reproductive system and variations in sexuality in Pecten and other pelecypod mollusks. Trans. Conn. Acad. Arts Sci. 1945, 36, 673–700. [Google Scholar]
- Osada, M.; Nakamura, S.; Kijima, A. Quantitative analysis of pattern of gonial proliferation during sexual maturation in Japanese scallop Patinopecten yessoensis. Fish. Sci. 2007, 73, 1318–1324. [Google Scholar] [CrossRef]
- Osanai, K. Seasonal gonad development and sex alteration in the scallop, Patinopecten yessoensis. Bull. Mar. Biol. Stn. Asamushi Tohoku Univ. 1975, 15, 81–88. [Google Scholar]
- Osada, M.; Treen, N. Molluscan GnRH associated with reproduction. Gen. Comp. Endocrinol. 2013, 181, 254–258. [Google Scholar] [CrossRef]
- Devlin, R.H.; Nagahama, Y. Sex determination and sex differentiation in fish: An overview of genetic, physiological, and environmental influences. Aquaculture 2002, 208, 191–364. [Google Scholar] [CrossRef]
- Ijiri, S.; Kaneko, H.; Kobayashi, T.; Wang, D.S.; Sakai, F.; Paul-Prasanth, B.; Nakamura, M.; Nagahama, Y. Sexual dimorphic expression of genes in gonads during early differentiation of a teleost fish, the Nile tilapia Oreochromis niloticus. Biol. Reprod. 2008, 78, 333–341. [Google Scholar] [CrossRef]
- Zhang, N.; Xu, F.; Guo, X.M. Genomic analysis of the Pacific oyster (Crassostrea gigas) reveals possible conservation of vertebrate sex determination in a mollusc. G3 Genes Genomes Genet. 2014, 4, 2207–2217. [Google Scholar] [CrossRef]
- Shi, Y.; Wang, Q.; He, M.X. Molecular identification of dmrt2 and dmrt5 and effect of sex steroids on their expressions in Chlamys nobilis. Aquaculture 2014, 426, 21–30. [Google Scholar] [CrossRef]
- Galindo-Torres, P.; Garcia-Gasca, A.; Llera-Herrera, R.; Escobedo-Fregoso, C.; Abreu-Goodger, C.; Ibarra, A.M. Sex determination and differentiation genes in a functional hermaphrodite scallop, Nodipecten subnodosus. Mar. Genom. 2018, 37, 161–175. [Google Scholar] [CrossRef] [PubMed]
- Mulyana, J.S.; Iwai, T.; Takahashi, M.; Farajallah, A.; Wardiatno, Y.; Miura, C.; Miura, T. Sex-changing patterns of Akoya pearl oyster (Pinctada fucata). Zool. Lett. 2018, 4, 11. [Google Scholar] [CrossRef]
- Nagahama, Y. Molecular mechanisms of sex determination and gonadal sex differentiation in fish. Fish Physiol. Biochem. 2005, 31, 105–109. [Google Scholar] [CrossRef]
- Liu, X.L.; Zhang, Z.F.; Shao, M.Y.; Liu, J.G.; Muhammad, F. Sexually dimorphic expression of foxl2 during gametogenesis in scallop Chlamys farreri, conserved with vertebrates. Dev. Genes Evol. 2012, 222, 279–286. [Google Scholar] [CrossRef]
- Teaniniuraitemoana, V.; Huvet, A.; Levy, P.; Klopp, C.; Lhuillier, E.; Gaertner-Mazouni, N.; Gueguen, Y.; Le Moullac, G. Gonad transcriptome analysis of pearl oyster Pinctada margaritifera: Identification of potential sex differentiation and sex determining genes. BMC Genom. 2014, 15. [Google Scholar] [CrossRef]
- Li, R.; Zhang, L.; Li, W.; Zhang, Y.; Li, Y.; Zhang, M.; Zhao, L.; Hu, X.; Wang, S.; Bao, Z. FOXL2 and DMRT1L are yin and yang genes for determining timing of sex differentiation in the bivalve mollusk Patinopecten yessoensis. Front. Physiol. 2018, 9, 1166. [Google Scholar] [CrossRef] [PubMed]
- Hou, R.; Bao, Z.; Wang, S.; Su, H.; Li, Y.; Du, H.; Hu, J.; Hu, X. Transcriptome sequencing and de novo analysis for Yesso scallop (Patinopecten yessoensis) using 454 GS FLX. PLoS ONE 2011, 6, e21560. [Google Scholar] [CrossRef]
- Nagasawa, K.; Muroi, K.; Thitiphuree, T.; Minegishi, Y.; Itoh, N.; Osada, M. Cloning of invertebrate gonadotropin-releasing hormone receptor (GnRHR)-like gene in Yesso scallop, Patinopecten yessoensis. Agri Gene 2017, 3, 46–56. [Google Scholar] [CrossRef]
- Nagasawa, K.; Takeuchi, Y.; Miwa, M.; Higuchi, K.; Morita, T.; Mitsuboshi, T.; Miyaki, K.; Kadomura, K.; Yoshizaki, G. cDNA cloning and expression analysis of a vasa-like gene in Pacific bluefin tuna Thunnus orientalis. Fish. Sci. 2009, 75, 71–79. [Google Scholar] [CrossRef]
- Feng, L.; Yu, Q.; Li, X.; Ning, X.; Wang, J.; Zou, J.; Zhang, L.; Wang, S.; Hu, J.; Hu, X.; et al. Identification of reference genes for qRT-PCR analysis in Yesso scallop Patinopecten yessoensis. PLoS ONE 2013, 8, e75609. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Zhang, J.B.; Jiao, W.Q.; Li, J.; Xun, X.G.; Sun, Y.; Guo, X.M.; Huan, P.; Dong, B.; Zhang, L.L.; et al. Scallop genome provides insights into evolution of bilaterian karyotype and development. Nat. Ecol. Evol. 2017, 1. [Google Scholar] [CrossRef]
- Guo, X.; Hedgecock, D.; Hershberger, W.K.; Cooper, K.; Allen, S.K., Jr. Genetic determinants of protandric sex in the Pacific oyster, Crassostrea Gigas Thunberg. Evolution 1998, 52, 394–402. [Google Scholar] [CrossRef]
- Mori, K.; Muramatsu, T.; Nakamura, Y. Effect of steroid on oyster-III, sex reversal from male to female in Crassostrea gigas by estradiol-17beta. Nippon Suisan Gakkaishi 1969, 35, 1072–1076. [Google Scholar] [CrossRef]
- Chen, C.J.; Shikina, S.; Chen, W.J.; Chung, Y.J.; Chiu, Y.L.; Bertrand, J.A.; Lee, Y.H.; Chang, C.F. A novel female-specific and sexual reproduction-associated dmrt gene discovered in the stony coral, Euphyllia ancora. Biol. Reprod. 2016, 94, 40. [Google Scholar] [CrossRef][Green Version]
- Bellefroid, E.J.; Leclere, L.; Saulnier, A.; Keruzore, M.; Sirakov, M.; Vervoort, M.; De Clercq, S. Expanding roles for the evolutionarily conserved Dmrt sex transcriptional regulators during embryogenesis. Cell. Mol. Life Sci. 2013, 70, 3829–3845. [Google Scholar] [CrossRef]
- Ottolenghi, C.; Veitia, R.; Barbieri, M.; Fellous, M.; McElreavey, K. The human doublesex-related gene, DMRT2 is homologous to a gene involved in somitogenesis and encodes a potential bicistronic transcript. Genomics 2000, 64, 179–186. [Google Scholar] [CrossRef]
- Raymond, C.S.; Murphy, M.W.; O’Sullivan, M.G.; Bardwell, V.J.; Zarkower, D. Dmrt1, a gene related to worm and fly sexual regulators, is required for mammalian testis differentiation. Genes Dev. 2000, 14, 2587–2595. [Google Scholar] [CrossRef]
- Matsuda, M.; Nagahama, Y.; Shinomiya, A.; Sato, T.; Matsuda, C.; Kobayashi, T.; Morrey, C.E.; Shibata, N.; Asakawa, S.; Shimizu, N.; et al. DMY is a Y-specific DM-domain gene required for male development in the medaka fish. Nature 2002, 417, 559–563. [Google Scholar] [CrossRef]
- Yu, F.F.; Wang, M.F.; Zhou, L.; Gui, J.F.; Yu, X.Y. Molecular Cloning and expression characterization of Dmrt2 in Akoya pearl oysters, Pinctada Martensii. J. Shellfish Res. 2011, 30, 247–254. [Google Scholar] [CrossRef]
- Guo, Y.Q.; Cheng, H.H.; Huang, X.; Gao, S.; Yu, H.S.; Zhou, R.J. Gene structure, multiple alternative splicing, and expression in gonads of zebrafish Dmrt1. BBRC 2005, 330, 950–957. [Google Scholar] [CrossRef]
- Li, Q.; Zhou, X.; Guo, Y.Q.; Shang, X.; Chen, H.; Lu, H.; Cheng, H.H.; Zhou, R.J. Nuclear localization, DNA binding and restricted expression in neural and germ cells of zebrafish Dmrt3. Biol. Cell 2008, 100, 453–463. [Google Scholar] [CrossRef]
- Guo, Y.Q.; Li, Q.; Gao, S.; Zhou, X.; He, Y.; Shang, X.; Cheng, H.H.; Zhou, R.J. Molecular cloning, characterization, and expression in brain and gonad of Dmrt5 of zebrafish. BBRC 2004, 324, 569–575. [Google Scholar] [CrossRef]
- Kobayashi, T.; Kajiura-Kobayashi, H.; Guan, G.; Nagahama, Y. Sexual dimorphic expression of DMRT1 and Sox9a during gonadal differentiation and hormone-induced sex reversal in the teleost fish Nile tilapia (Oreochromis niloticus). Dev. Dyn. 2008, 237, 297–306. [Google Scholar] [CrossRef]
- Kobayashi, T.; Matsuda, M.; Kajiura-Kobayashi, H.; Suzuki, A.; Saito, N.; Nakamoto, M.; Shibata, N.; Nagahama, Y. Two DM domain genes, DMY and DMRT1, involved in testicular differentiation and development in the medaka, Oryzias latipes. Dev. Dyn. 2004, 231, 518–526. [Google Scholar] [CrossRef]
- Sun, W.; Cai, H.; Zhang, G.; Zhang, H.Y.; Bao, H.S.; Wang, L.; Ye, J.; Qian, G.Y.; Ge, C.T. Dmrt1 is required for primary male sexual differentiation in Chinese soft-shelled turtle Pelodiscus sinensis. Sci. Rep. 2017, 7. [Google Scholar] [CrossRef]
- Otani, A.; Nakajima, T.; Okumura, T.; Fujii, S.; Tomooka, Y. Sex reversal and analyses of possible involvement of sex steroids in scallop gonadal development in newly established organ-culture systems. Zool. Sci. 2017, 34, 86–92. [Google Scholar] [CrossRef]
- Naimi, A.; Martinez, A.S.; Specq, M.L.; Diss, B.; Mathieu, M.; Sourdaine, P. Molecular cloning and gene expression of Cg-Foxl2 during the development and the adult gametogenetic cycle in the oyster Crassostrea gigas. Comp. Biochem. Phys. B 2009, 154, 134–142. [Google Scholar] [CrossRef]
- Osada, M.; Harata, M.; Kishida, M.; Kijima, A. Molecular cloning and expression analysis of vitellogenin in scallop, Patinopecten yessoensis (Bivalvia, Mollusca). Mol. Reprod. Dev. 2004, 67, 273–281. [Google Scholar] [CrossRef]
- Jiang, T.; Hou, C.C.; She, Z.Y.; Yang, W.X. The SOX gene family: Function and regulation in testis determination and male fertility maintenance. Mol. Biol. Rep. 2013, 40, 2187–2194. [Google Scholar] [CrossRef]
- Yu, J.; Zhang, L.; Li, Y.; Li, R.; Zhang, M.; Li, W.; Xie, X.; Wang, S.; Hu, X.; Bao, Z. Genome-wide identification and expression profiling of the SOX gene family in a bivalve mollusc Patinopecten yessoensis. Gene 2017, 627, 530–537. [Google Scholar] [CrossRef]
- Wei, L.; Yang, C.; Tao, W.J.; Wang, D.H. Genome-wide identification and transcriptome-based expression profiling of the sox gene family in the Nile Tilapia (Oreochromis niloticus). Int. J. Mol. Sci. 2016, 17. [Google Scholar] [CrossRef]
- Nagasawa, K.; Oouchi, H.; Itoh, N.; Takahashi, K.G.; Osada, M. In vivo administration of scallop GnRH-like peptide influences on gonad development in the Yesso scallop, Patinopecten yessoensis. PLoS ONE 2015, 10, e0129571. [Google Scholar] [CrossRef]
- Nagasawa, K.; Osugi, T.; Suzuki, I.; Itoh, N.; Takahashi, K.G.; Satake, H.; Osada, M. Characterization of GnRH-like peptides from the nerve ganglia of Yesso scallop, Patinopecten yessoensis. Peptides 2015, 71, 202–210. [Google Scholar] [CrossRef]
- Thitiphuree, T.; Nagasawa, K.; Osada, M. Molecular identification of steroidogenesis-related genes in scallops and their potential roles in gametogenesis. J. Steroid Biochem. Mol. Biol. 2019, 186, 22–33. [Google Scholar] [CrossRef]
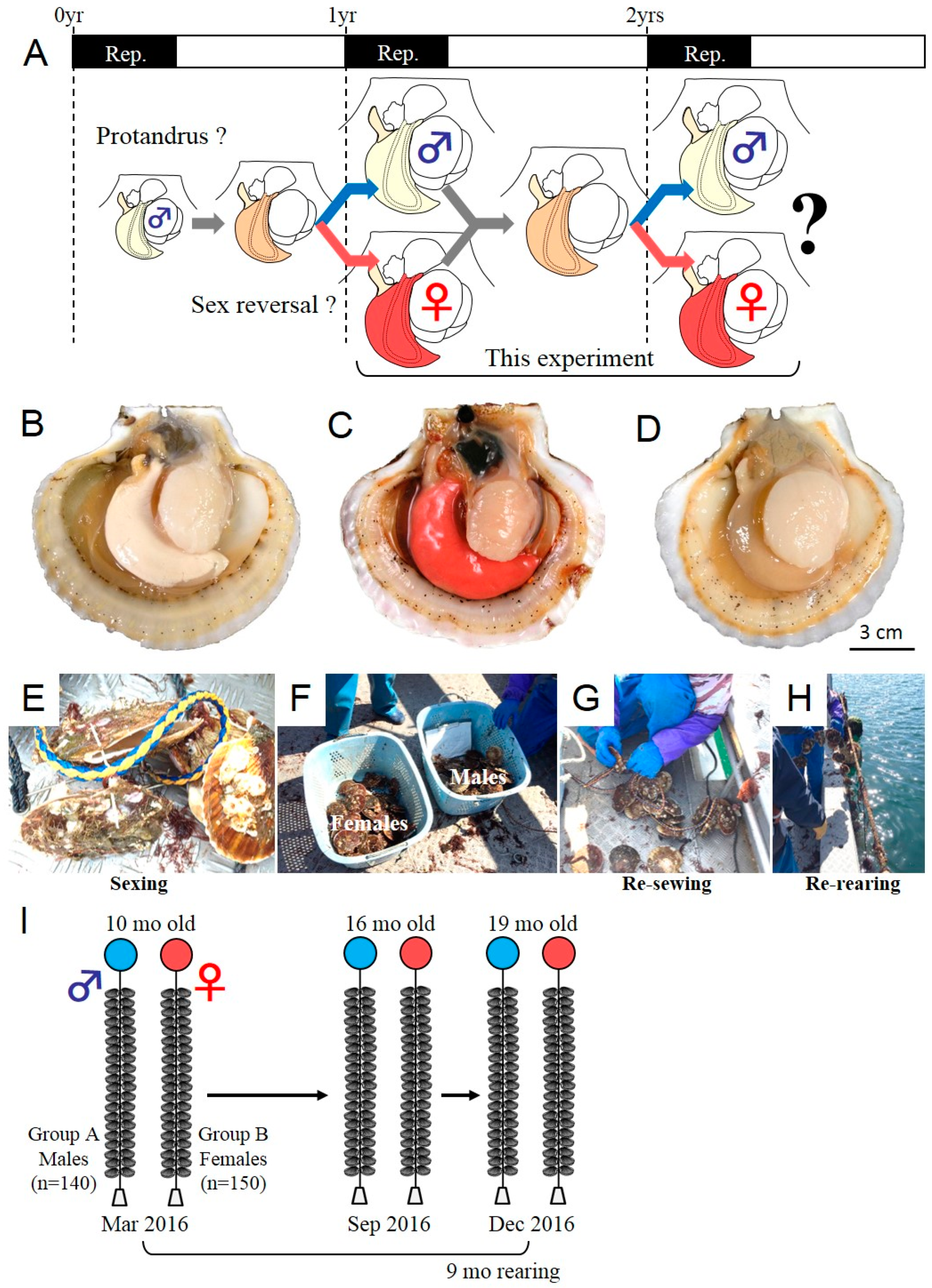
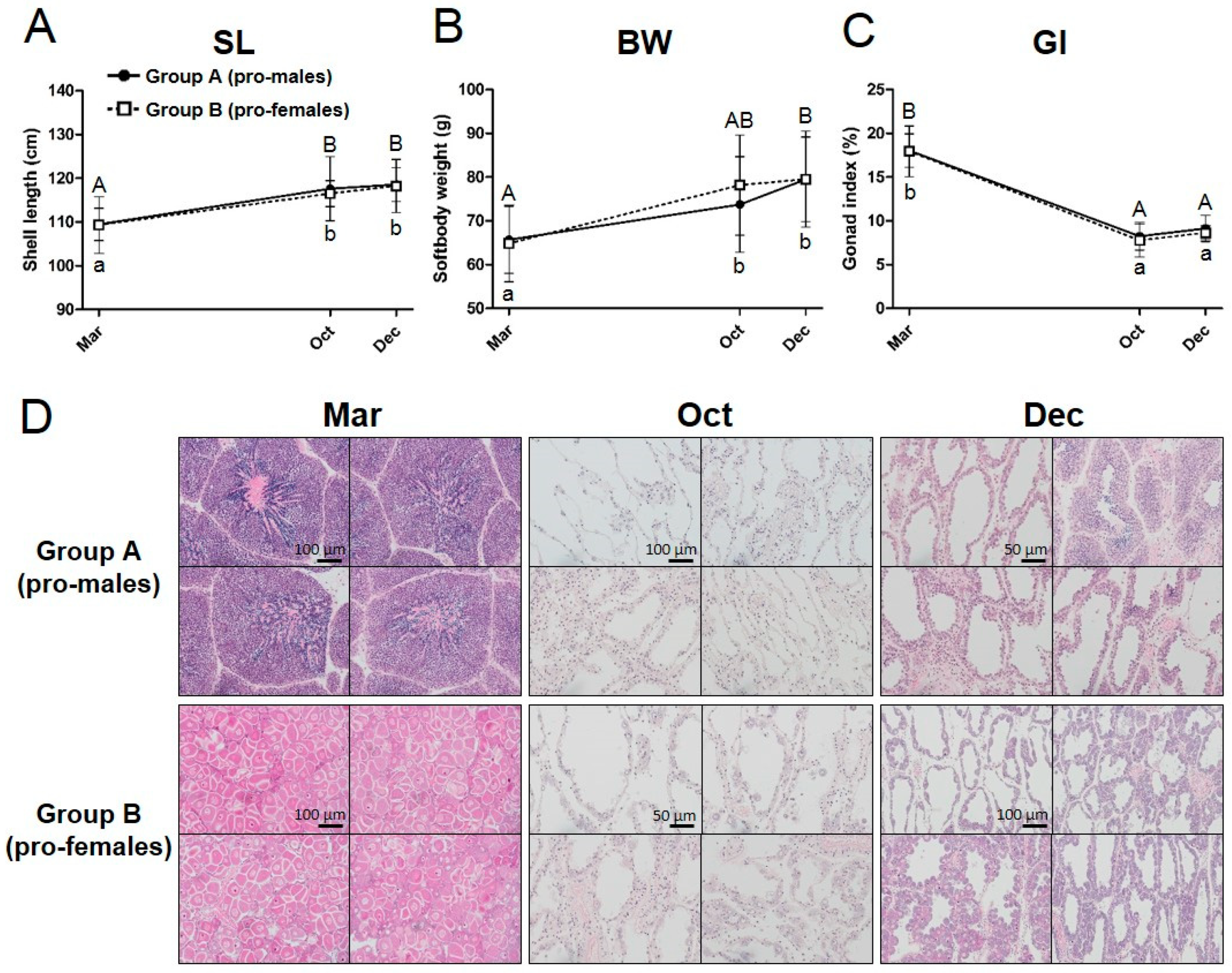
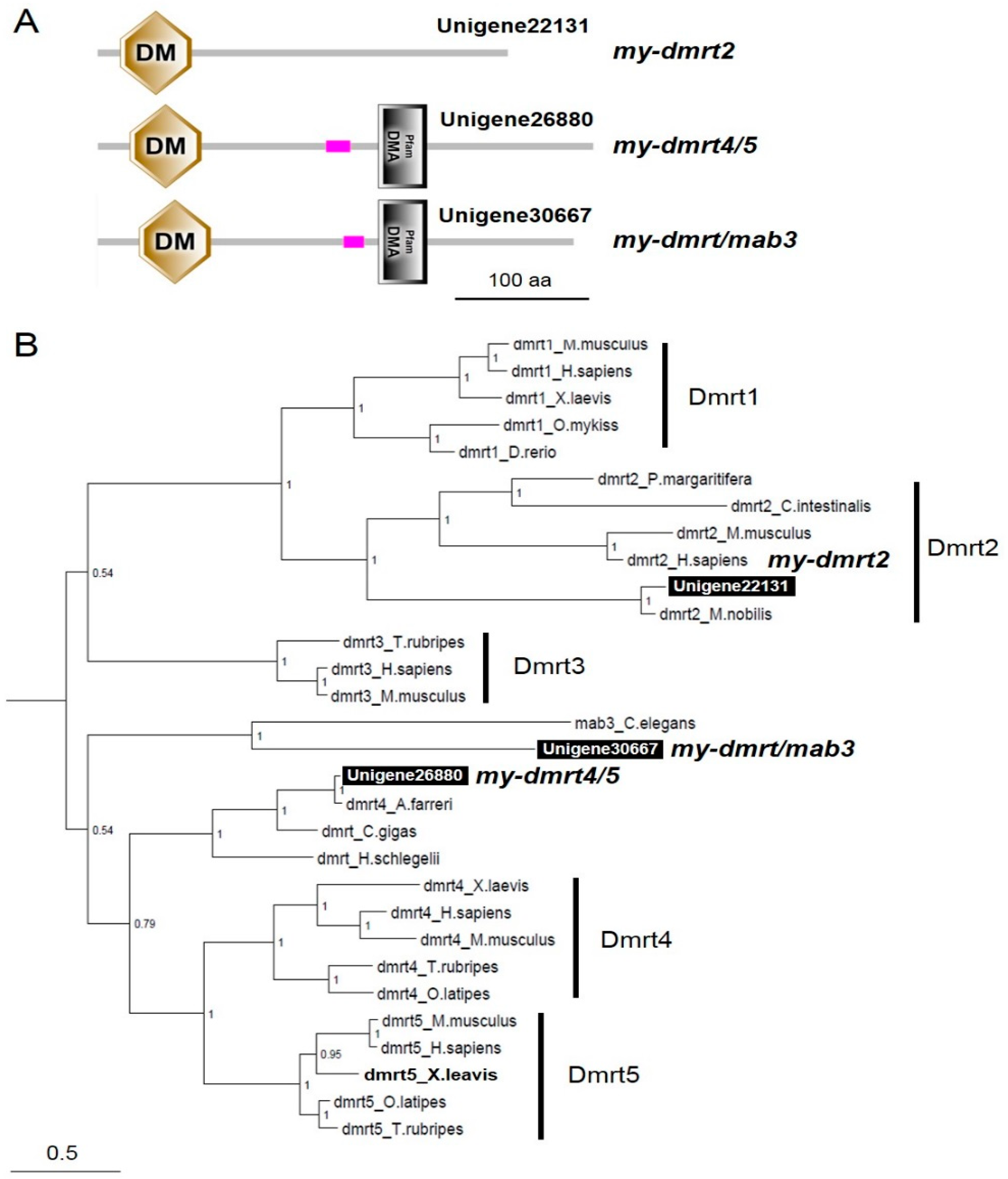
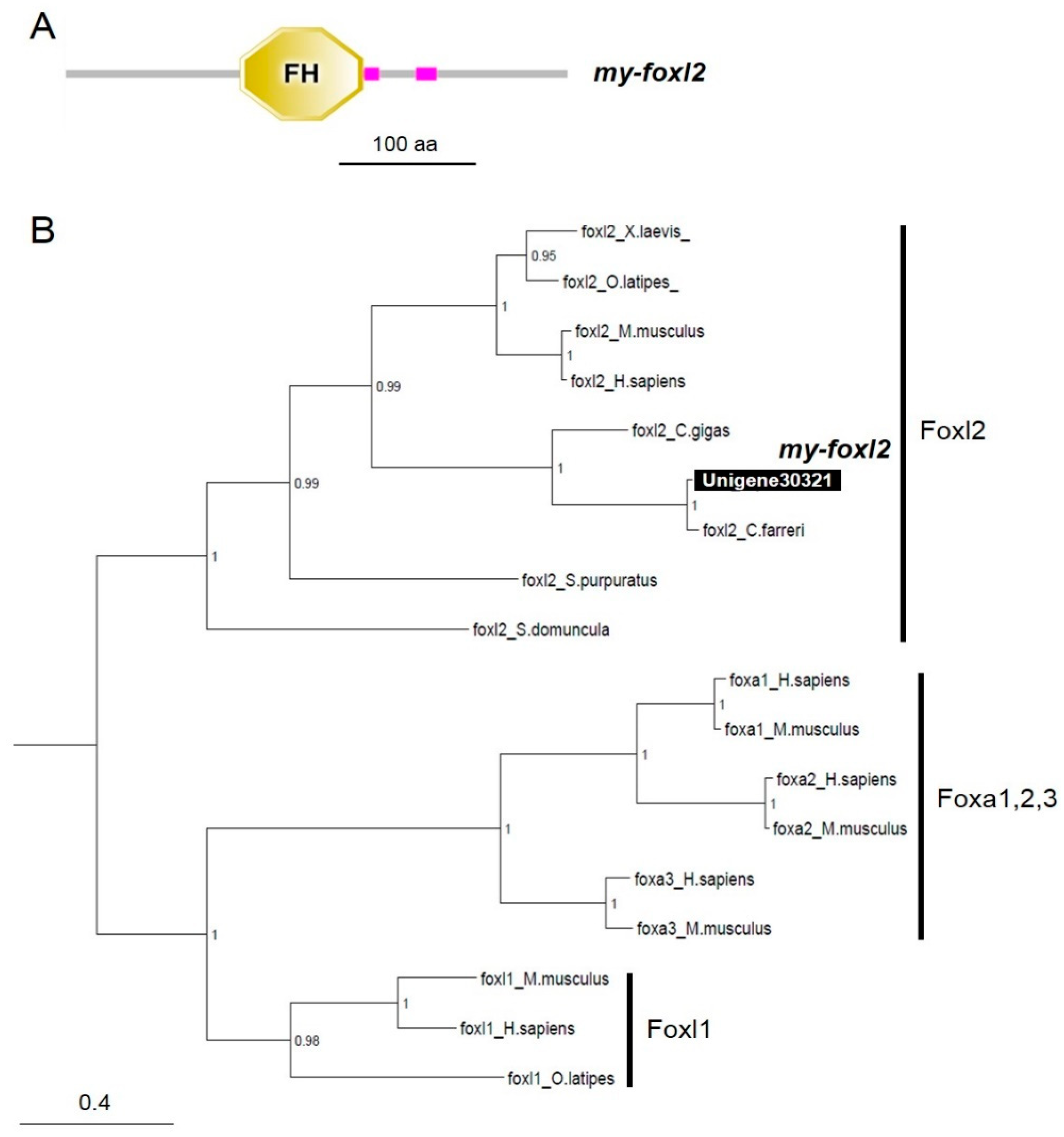
| Sampling Period (Age) | Mar 2016 (10 Months) | Oct 2016 (16 Months) | Dec 2016 (19 Months) | ||||
|---|---|---|---|---|---|---|---|
| Group | Intact | Survivors | Analyzed | Female:Male | Survivors | Analyzed | Female:Male |
| Group A (pro-males) | 140 | 45 | 10 | 0:10 | 22 | 22 | 0:22 |
| Group B (pro-females) | 150 | 51 | 10 | 10:0 | 21 | 21 | 21:0 |
| Gene | PCR Type | Sequence (5′–3′) | Amplicon (bp) | Intron Insertion | Genbank Acc. No. | r2 | E (%) | Reference |
|---|---|---|---|---|---|---|---|---|
| my-dmrt2 (Unigene22131) | RT-PCR, qPCR | Fw: TGAGCAATAACAAGGAGCTGTTAG | 264 | yes | XM_021498039 | 0.99 | 78.18 | This study |
| Rv: CTCAGGGCGATCTGTTCCTTTGA | ||||||||
| ISH | Fw: TGAGCAATAACAAGGAGCTGTTAG | 1026 | - | |||||
| Rv: TGTCATTCCTCAAGTAACCTTCAT | ||||||||
| my-dmrt4/5 (Unigene26880) | RT-PCR | Fw: TGCCTACAGTGGACTCACGACGGAC | 201 | no | XM_021521599 | This study | ||
| Rv: TCTCAGGCTCCGTCCTTACAGGCG | ||||||||
| my-dmrt/mab3 (Unigene30667) | RT-PCR | Fw: CTCAGAAGTCTGCAGCAACACAC | 174 | no | XM_021513113 | This study | ||
| Rv: CTCATGTTCGTACATTGCAAGCT | ||||||||
| my-foxl2 (Unigene30321) | RT-PCR, qPCR | Fw: TCAGAGTCACTCGATAACCTTACT | 304 | no | XM_021497746 | 0.97 | 90.84 | This study |
| Rv: CCTGGCTGCTACACCGTACGCCACT | ||||||||
| ISH | Fw: TCAGAGTCACTCGATAACCTTACT | 828 | - | |||||
| Rv: TAGGGACCGCAGTGGTTGTCAGCA | ||||||||
| my-soxb1 | qPCR | Fw: ACAAACTCTCGCAGGGGTAG | 309 | NT | KY523527 | 0.99 | 84.4 | Otani et al., 2017 |
| Rv: GCACCGAGTCGCTTACTGAT | Yu et al., 2017 | |||||||
| my-tesk | qPCR | Fw: CACGCCAAGGAATCTGAGG | 472 | NT | NA | 0.99 | 111.14 | Otani et al., 2017 |
| Rv: AGGATGTGAAGGGTCGTTG | ||||||||
| my-vtg | qPCR | Fw: CCTCTATGCCGGACATTTGC | 100 | NT | AB055960 | 0.97 | 83.03 | Osada et al., 2004 |
| Rv: CAAAGCCACGCTGCTATCTTT | ||||||||
| my-heli | qPCR | Fw: CCAGGAGCAGAGGGAGTTCG | 186 | yes | NA | 0.99 | 77.02 | Feng et al., 2013 |
| Rv: GTCTTACCAGCCCGTCCAGTTC | ||||||||
| my-rpl16 | RT-PCR, qPCR | Fw: CTGCCAGACAGACTGAATGATGCC | 117 | yes | NA | 0.99 | 95.49 | Feng et al., 2013 |
| Rv: ACGCTCGTCACTGACTTGATAAACCT | ||||||||
| my-ubq | qPCR | Fw: TCGCTGTAGTCTCCAGGATTGC | 184 | yes | NA | 0.99 | 80.05 | Feng et al., 2013 |
| Rv: TCGCCACATACCCTCCCAC |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nagasawa, K.; Thitiphuree, T.; Osada, M. Phenotypic Stability of Sex and Expression of Sex Identification Markers in the Adult Yesso Scallop Mizuhopecten yessoensis throughout the Reproductive Cycle. Animals 2019, 9, 277. https://doi.org/10.3390/ani9050277
Nagasawa K, Thitiphuree T, Osada M. Phenotypic Stability of Sex and Expression of Sex Identification Markers in the Adult Yesso Scallop Mizuhopecten yessoensis throughout the Reproductive Cycle. Animals. 2019; 9(5):277. https://doi.org/10.3390/ani9050277
Chicago/Turabian StyleNagasawa, Kazue, Tongchai Thitiphuree, and Makoto Osada. 2019. "Phenotypic Stability of Sex and Expression of Sex Identification Markers in the Adult Yesso Scallop Mizuhopecten yessoensis throughout the Reproductive Cycle" Animals 9, no. 5: 277. https://doi.org/10.3390/ani9050277
APA StyleNagasawa, K., Thitiphuree, T., & Osada, M. (2019). Phenotypic Stability of Sex and Expression of Sex Identification Markers in the Adult Yesso Scallop Mizuhopecten yessoensis throughout the Reproductive Cycle. Animals, 9(5), 277. https://doi.org/10.3390/ani9050277





