Differential Expression of KCNJ12 Gene and Association Analysis of Its Missense Mutation with Growth Traits in Chinese Cattle
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. Animals and Data Collection
2.3. DNA Isolation and Genomic DNA Sequencing
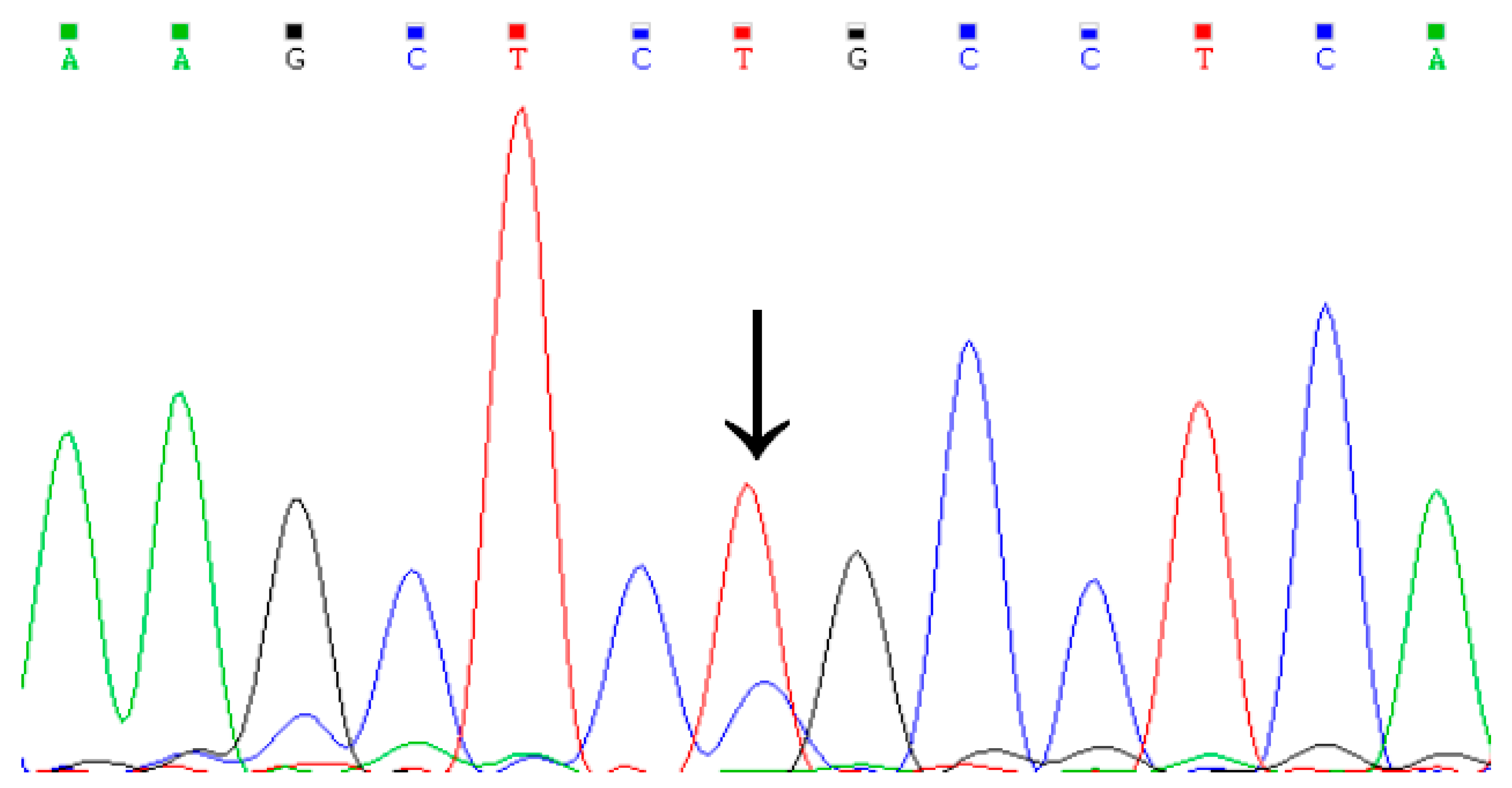
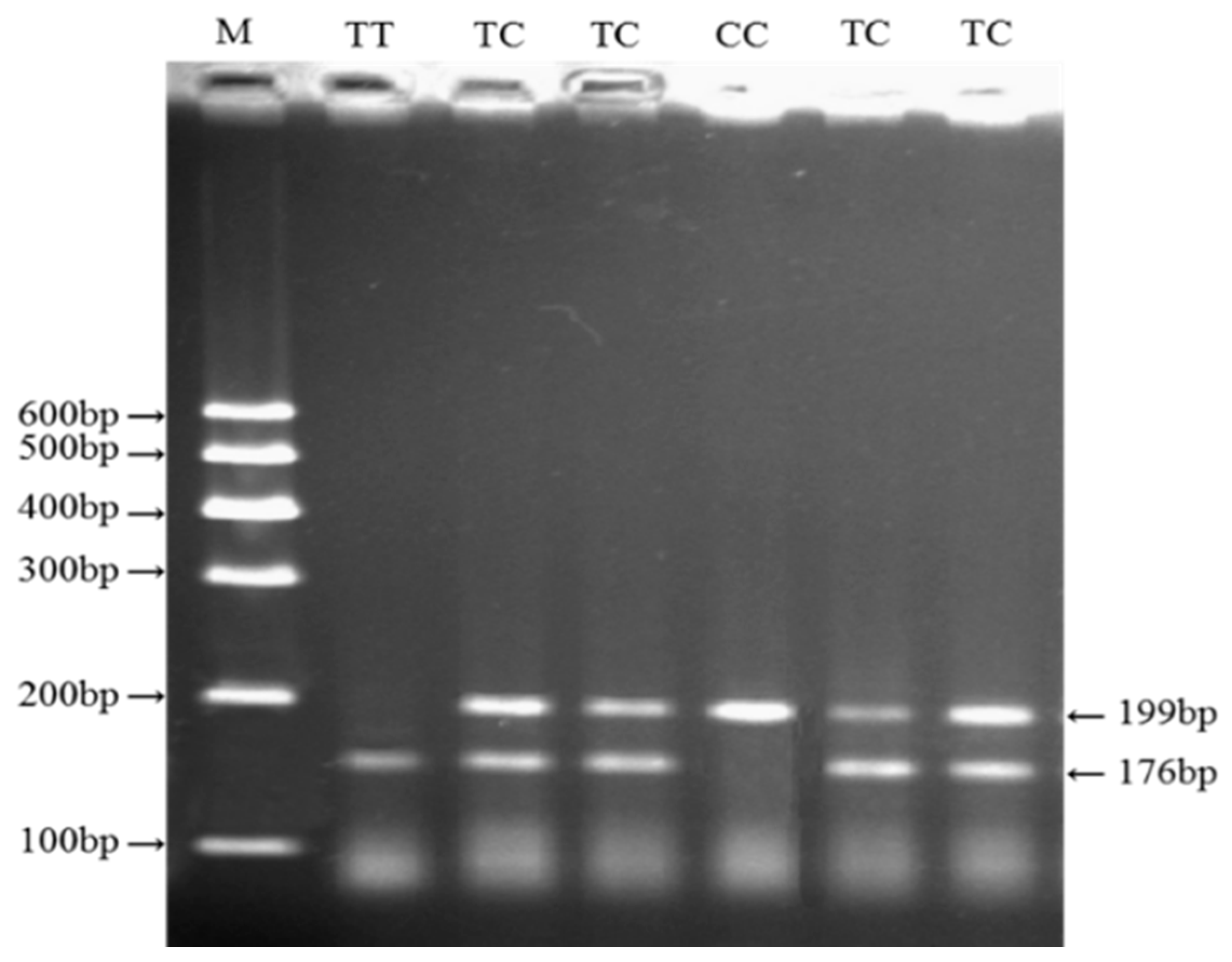
2.4. Genotyping of Four Variations within Cattle KCNJ12 Gene
2.5. Statistical Analysis
2.6. Using PolyPhen to Predict if the Missense Mutation Altered the Protein of KCNJ12
2.7. Cell Culture and Induction Differentiation
2.8. RNA Extraction, cDNA Synthesis, and Expression Analyses
3. Results
3.1. Identification of Genetic Variation in the Cattle KCNJ12 Gene
3.2. Genotypes, Allele Frequencies, and Genetic Diversity of the SNP in the KCNJ12 Gene
3.3. Association Study of g.35989944T>C with Cattle Stature
3.4. Using PolyPhen to Determine if the Missense Mutation May Alter the Protein
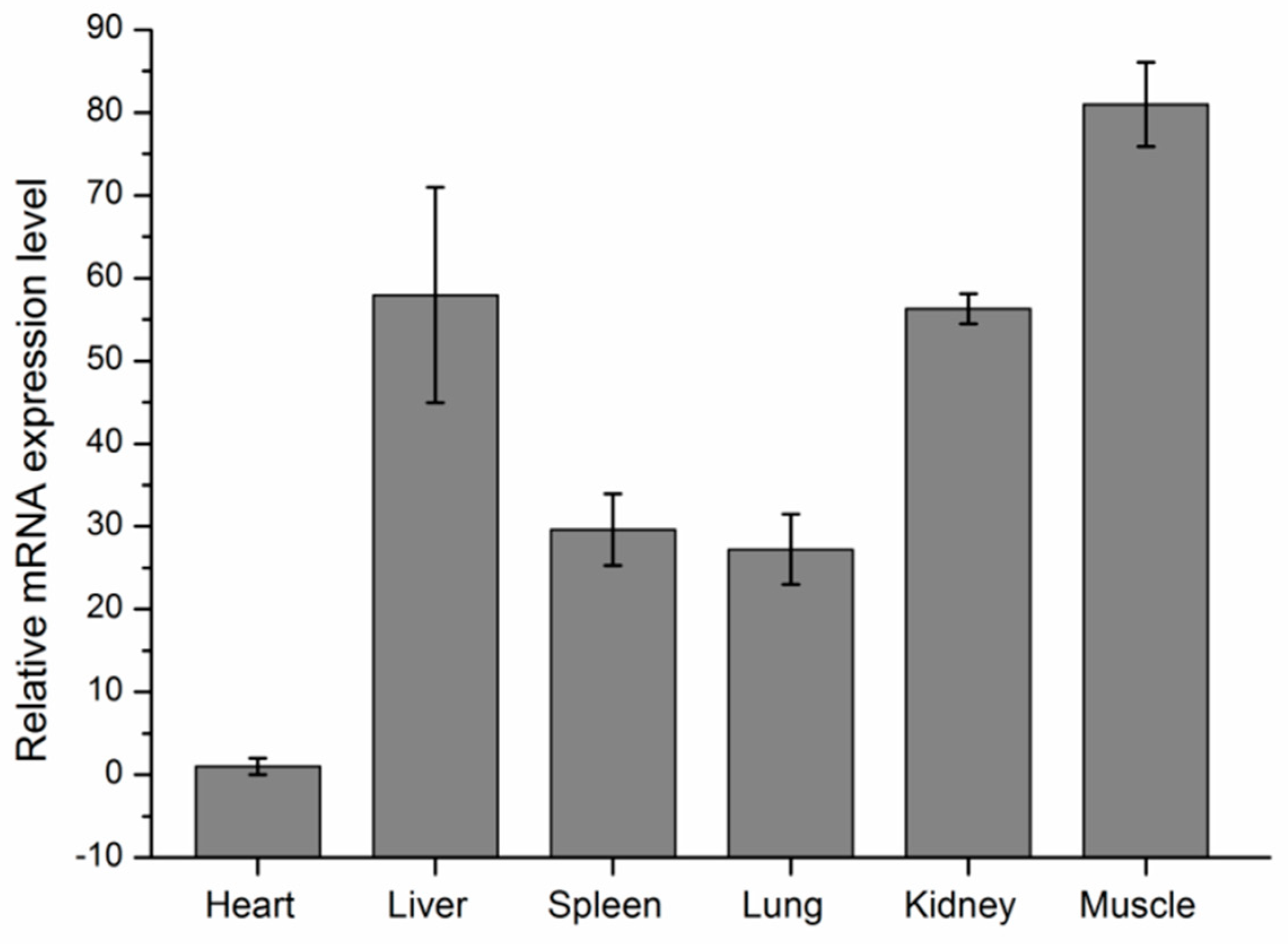
3.5. Expression Analyses of KCNJ12 in Cattle Tissues
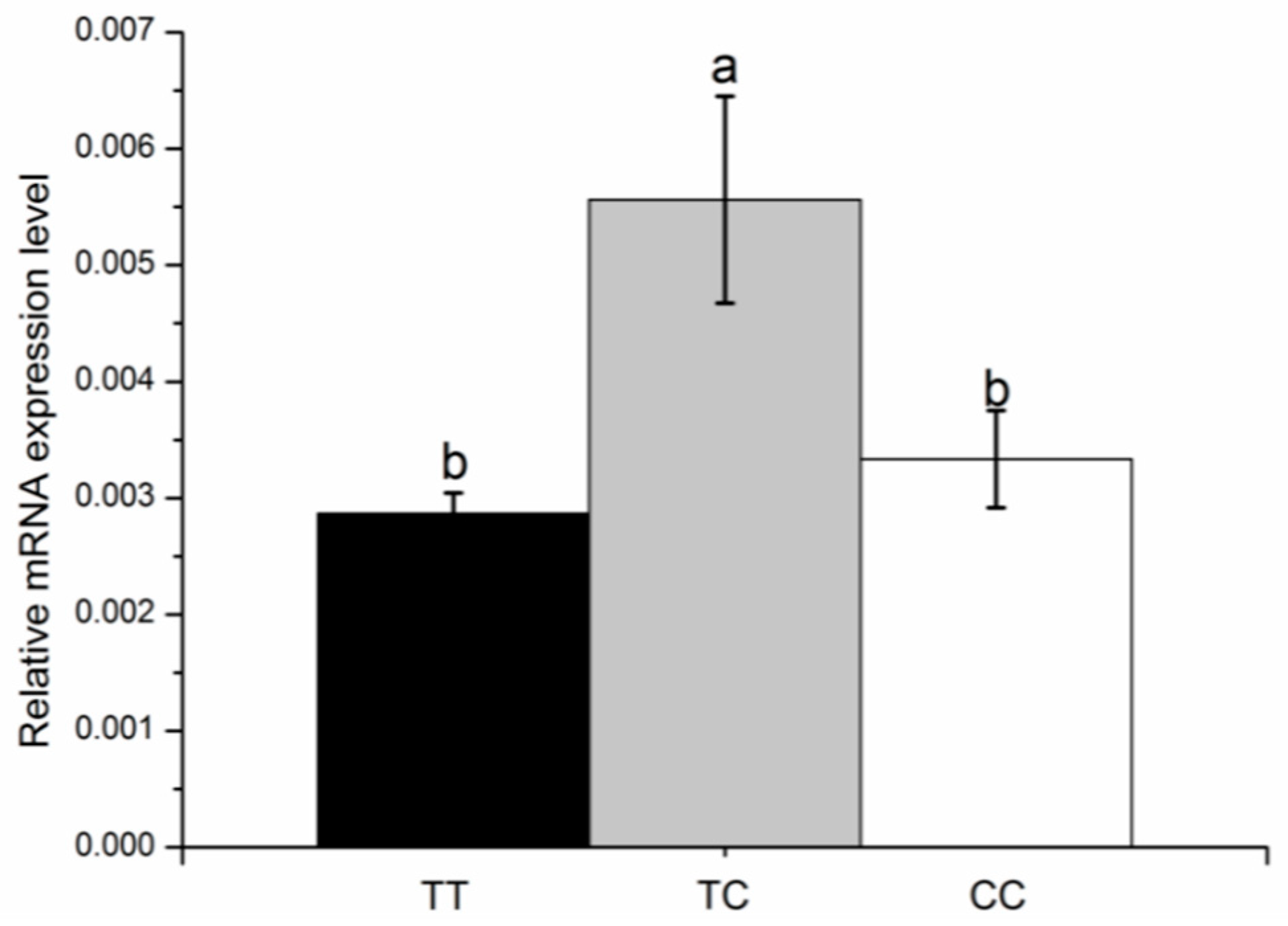
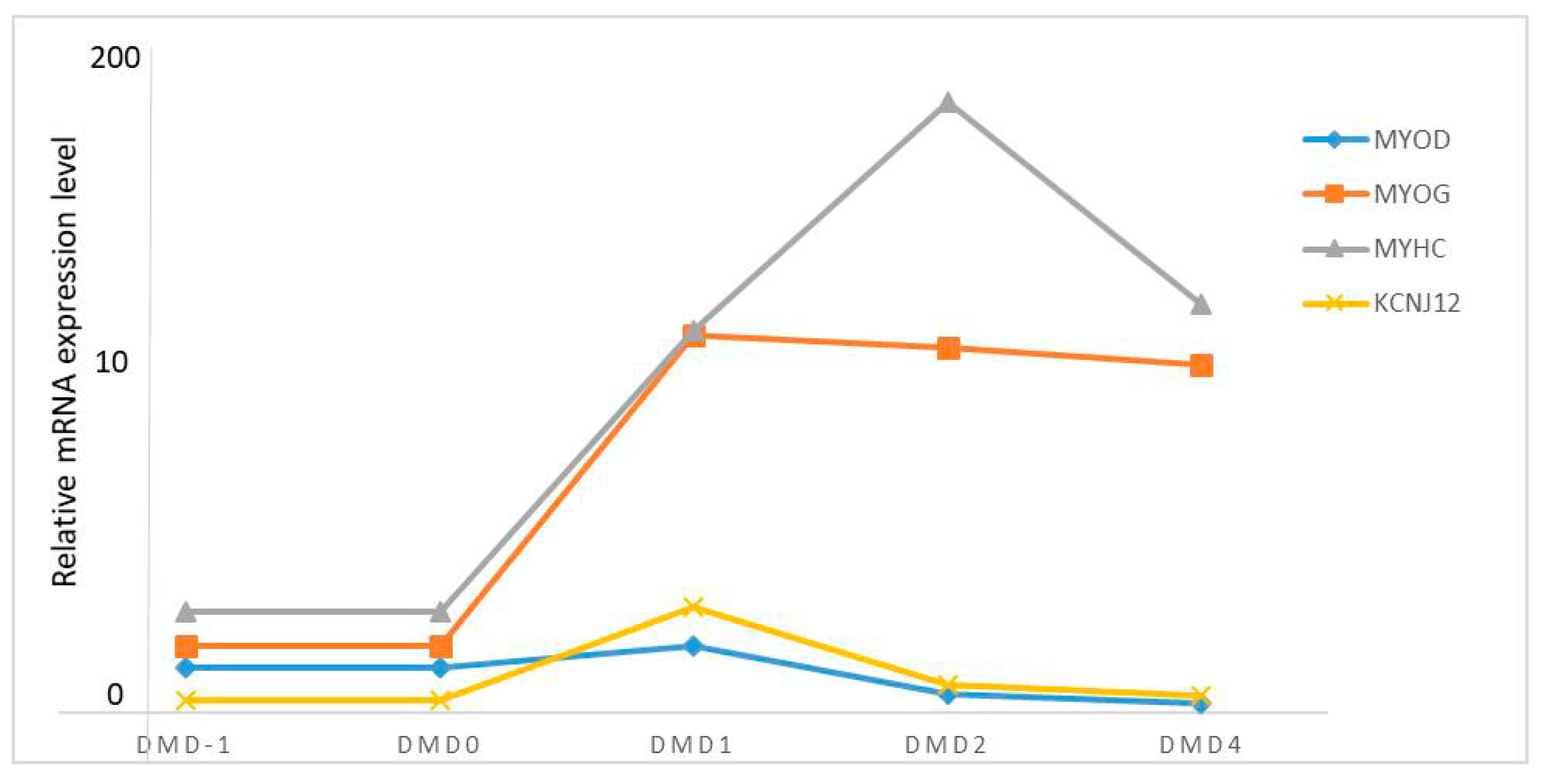
3.6. KCNJ12 Expression during Primary Bovine Skeletal Muscle Cell Differentiation
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
References
- Garrick, D.; Ruvinsky, A. The Genetics of Cattle; CABI: Oxfordshire, UK, 2014. [Google Scholar]
- Domenighetti, A.A.; Boixel, C.; Cefai, D.; Abriel, H.; Pedrazzini, T. Chronic angiotensin II stimulation in the heart produces an acquired long QT syndrome associated with IK1 potassium current downregulation. J. Mol. Cell. Cardiol. 2007, 42, 63–70. [Google Scholar] [CrossRef]
- Magnusson, C.; Svensson, A.; Christerson, U.; Tågerud, S. Denervation. induced alterations in gene expression in mouse skeletal muscle. Eur. J. Neurosci. 2005, 21, 577–580. [Google Scholar] [CrossRef]
- Paninka, R.M.; Mazzotti, D.R.; Kizys, M.M.; Vidi, A.C.; Rodrigues, H.; Silva, S.P.; Kunii, I.S.; Furuzawa, G.K.; Arcisio-Miranda, M.; Dias-da-Silva, M.R. Whole genome and exome sequencing realignment supports the assignment of KCNJ12, KCNJ17, and KCNJ18 paralogous genes in thyrotoxic periodic paralysis locus: Functional characterization of two polymorphic Kir2.6 isoforms. Mol. Genet. Genom. 2016, 291, 1535–1544. [Google Scholar] [CrossRef] [PubMed]
- Sher, R.B.; Cox, G.A.; Mills, K.D.; Sundberg, J.P. Rhabdomyosarcomas in aging A/J mice. PLoS ONE 2011, 6, e23498. [Google Scholar] [CrossRef]
- Szuts, V.; Ménesi, D.; Varga-Orvos, Z.; Zvara, Á.; Houshmand, N.; Bitay, M.; Bogáts, G.; Virág, L.; Baczkó, I.; Szalontai, B.; et al. Altered expression of genes for Kir ion channels in dilated cardiomyopathy. Can. J. Physiol. Pharmacol. 2013, 91, 648–656. [Google Scholar] [CrossRef]
- Girirajan, S.; Williams, S.; Garbern, J.; Nowak, N.; Hatchwell, E.; Elsea, S.H. 17p11. 2p12 triplication and del (17) q11. 2q12 in a severely affected child with dup (17) p11. 2p12 syndrome. Clin. Genet. 2007, 72, 47–58. [Google Scholar] [CrossRef]
- Zhou, Y.; Utsunomiya, Y.T.; Xu, L.; Bickhart, D.M.; Alexandre, P.A.; Rosen, B.D.; Schroeder, S.G.; Carvalheiro, R.; de Rezende Neves, H.H.; Sonstegard, T.S.; et al. Genome-wide CNV analysis reveals variants associated with growth traits in Bos indicus. BMC Genom. 2016, 17, 419. [Google Scholar] [CrossRef]
- Zheng, L.; Xu, J.W.; Li, J.C.; Wang, D.H.; An, Q.M.; Xu, L.N.; Ma, Y.L.; Wang, J.; Peng, S.J.; Lei, C.Z. Distribution and association study in copy number variation of KCNJ12 gene across four Chinese cattle populations. Gene 2019, 689, 90–96. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, R.; Bailey, D.; Shannon, N. Linear body measurements of cattle before and after 20 years of selection for postweaning gain when fed two different diets. J. Anim. Sci. 1993, 71, 1712–1720. [Google Scholar] [CrossRef]
- Sambrook, J.; Russell, D.W.; Russell, D.W. Molecular Cloning: A Laboratory Manual (3-Volume Set); Cold Spring Harbor Laboratory Press: Cold Spring Harbor, NY, USA, 2001; Volume 999. [Google Scholar]
- Cao, X.K.; Zhan, Z.Y.; Huang, Y.Z.; Lan, X.Y.; Lei, C.Z.; Qi, X.L.; Chen, H. Variants and haplotypes within MEF2C gene influence stature of chinese native cattle including body dimensions and weight. Livest. Sci. 2016, 185, 106–109. [Google Scholar] [CrossRef]
- See, L.M.; Hassan, R.; Tan, S.G.; Bhassu, S.; Ambak, M.A.; Bolong, A.M.; Ismail, P.; Tam, B.M.; Armstrong, A.; Gibbs, A.; et al. POPGENE, the user-friendly shareware for population genetic analysis. Biotechnology 2006, 7, 104–110. [Google Scholar]
- Adzhubei, I.A.; Schmidt, S.; Peshkin, L.; Ramensky, V.E.; Gerasimova, A.; Bork, P.; Kondrashov, A.S.; Sunyaev, S.R. A method and server for predicting damaging missense mutations. Nat. Methods 2010, 7, 248–249. [Google Scholar] [CrossRef] [PubMed]
- Miyake, M.; Takahashi, H.; Kitagawa, E.; Watanabe, H.; Sakurada, T.; Aso, H.; Yamaguchi, T. AMPK activation by AICAR inhibits myogenic differentiation and myostatin expression in Cattle. Cell Tissue Res. 2012, 349, 615–623. [Google Scholar] [CrossRef]
- Hu, J.; Shi, B.; Xie, J.; Zhou, H.; Wang, J.; Liu, X.; Li, S.; Zhao, Z.; Luo, Y. Tissue Expression and Variation of the DGAT2 Gene and Its Effect on Carcass and Meat Quality Traits in Yak. Animals 2019, 9, 61. [Google Scholar] [CrossRef]
- Sun, Y.; Liu, K.; Huang, Y.; Lan, X.; Chen, H. Differential expression of FOXO1 during development and myoblast differentiation of Qinchuan cattle and its association analysis with growth traits. Sci. China Life Sci. 2018, 61, 826–835. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Zhu, B.; Wang, Z.; Xu, L.; Liu, Y.; Chen, Y.; Zhang, L.; Gao, X.; Gao, H.; Zhang, S.; et al. Evaluation of Linkage Disequilibrium, Effective Population Size and Haplotype Block Structure in Chinese Cattle. Animals 2019, 9, 83. [Google Scholar] [CrossRef]
- Hua, J.T.; Ahmed, M.; Guo, H.; Zhang, Y.; Chen, S.; Soares, F.; Lu, J.; Zhou, S.; Wang, M.; Li, H.; et al. Risk SNP-mediated promoter-enhancer switching drives prostate cancer through lncRNA PCAT19. Cell 2018, 174, 564–575.e18. [Google Scholar] [CrossRef] [PubMed]
- Sandestig, A.; Green, A.; Jonasson, J.; Vogt, H.; Wahlström, J.; Pepler, A.; Ellnebo, K.; Biskup, S.; Stefanova, M. Could Dissimilar Phenotypic Effects of ACTB Missense Mutations Reflect the Actin Conformational Change? Two Novel Mutations and Literature Review. Mol. Syndromol. 2019, 9, 259–265. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Xi, J.; Zhu, W.; Lin, J.; Luo, S.; Yue, D.; Cai, S.; Sun, C.; Zhao, C.; Mitsuhashi, S.; et al. GNE myopathy in Chinese population: Hotspot and novel mutations. J. Hum. Genet. 2019, 64, 11–16. [Google Scholar] [CrossRef]
- Huang, Y.; He, H.; Wang, J.; Li, Z.J.; Lan, X.Y.; Lei, C.Z.; Zhang, E.P.; Zhang, C.L.; Wang, J.Q.; Shen, Q.W.; et al. Sequence variants in the bovine nucleophosmin 1 gene, their linkage and their associations with body weight in native cattle breeds in China. Anim. Genet. 2011, 42, 556–559. [Google Scholar] [CrossRef]
- Cao, X.K.; Wang, J.; Lan, X.Y.; Lei, C.Z.; Zhang, C.L.; Qi, X.L.; Chen, H.; et al. Genetic variants in BMP8B gene are associated with growth traits in Chinese native cattle. Gene 2013, 532, 115–120. [Google Scholar] [CrossRef]
- Xu, Y.; Shi, T.; Zhou, Y.; Liu, M.; Klaus, S.; Lan, X.; Lei, C.; Chen, H. A novel PAX7 10-bp indel variant modulates promoter activity, gene expression and contributes to different phenotypes of Chinese cattle. Sci. Rep. 2018, 8, 1724. [Google Scholar] [CrossRef]
- Wang, X.; Yang, Q.; Wang, K.; Yan, H.; Pan, C.; Chen, H.; Liu, J.; Zhu, H.; Qu, L.; Lan, X. Two strongly linked single nucleotide polymorphisms (Q320P and V397I) in GDF9 gene are associated with litter size in cashmere goats. Theriogenology 2019, 125, 115–121. [Google Scholar] [CrossRef]
- Buckingham, M.; Bajard, L.; Chang, T.; Daubas, P.; Hadchouel, J.; Meilhac, S.; Montarras, D.; Rocancourt, D.; Relaix, F. The formation of skeletal muscle: From somite to limb. J. Anat. 2003, 202, 59–68. [Google Scholar] [CrossRef]
- Perry, R.L.; Rudnick, M.A. Molecular mechanisms regulating myogenic determination and differentiation. Front. Biosci. 2000, 5, D750–D767. [Google Scholar] [CrossRef]
- Hernández-Hernández, J.M.; García-González, E.G.; Brun, C.E.; Rudnicki, M.A. The myogenic regulatory factors, determinants of muscle development, cell identity and regeneration. Semin. Cell Dev. Biol. 2017, 72, 10–18. [Google Scholar] [CrossRef]
- Braun, T.; Gautel, M. Transcriptional mechanisms regulating skeletal muscle differentiation, growth and homeostasis. Nat. Rev. Mol. Cell Biol. 2011, 12, 349. [Google Scholar] [CrossRef]
- Sabourin, L.A.; Rudnicki, M.A. The molecular regulation of myogenesis. Clin. Genet. 2000, 57, 16–25. [Google Scholar] [CrossRef]
- Naya, F.J.; Olson, E. MEF2: A transcriptional target for signaling pathways controlling skeletal muscle growth and differentiation. Curr. Opin. Cell Biol. 1999, 11, 683–688. [Google Scholar] [CrossRef]
- Buckingham, M. Myogenic progenitor cells and skeletal myogenesis in vertebrates. Curr. Opin. Genet. Dev. 2006, 16, 525–532. [Google Scholar] [CrossRef]
- Kokabu, S.; Nakatomi, C.; Matsubara, T.; Ono, Y.; Addison, W.N.; Lowery, J.W.; Urata, M.; Hudnall, A.M.; Hitomi, S.; Nakatomi, M.; et al. The transcriptional co-repressor TLE3 regulates myogenic differentiation by repressing the activity of the MyoD transcription factor. J. Biol. Chem. 2017, 292, 12885–12894. [Google Scholar] [CrossRef]
| Name | Chr. Position | Primer Sequences (5′–3′) | Genotyping Method | Tm (°C) | Restriction Enzyme | Genotype Pattern (bp) |
|---|---|---|---|---|---|---|
| SNP | g.35989944 T>C | F: CGAGGAGTGCCCGGTGGCGGTGTTCAT | PCR-RFLP | 57 | Pst I, CTGCA↓G | 199 (176 + 23) |
| R: TAGGTTGCCCACGCGCCACATGCTGC |
| Gene Name | Primer Sequences (5′–3′) | Primer Efficiencies | Annealing Temp. (°C) |
|---|---|---|---|
| MyoD | F: ACGGCATGATGGACTACAGC | 97.2% | 60 |
| R: AGGCAGTCGAGGCTCGACA | |||
| MyoG | F: CAAATCCACTCCCTGAAA | 98.7% | 60 |
| R: GCATAGGAAGAGATGAACA | |||
| MyHC | F: TGCTCATCTCACCAAGTTCC | 98.3% | 60 |
| R: CACTCTTCACTCTCATGGACC | |||
| KCNJ12 | F: TGGGCAACCTACGCAAGAGC | 97.9% | 60 |
| R: GCAGGATGGTGATGGGAGACA | |||
| GAPDH | F: CGACTTCAACAGCGACACTCAC | 97.6% | 60 |
| R: CCCTGTTGCTGTAGCCAAATTC |
| Name | Breeds (Sizes) | Genotype Freq. | Allele Freq. | HWE-χ2 | p | Ho | He | Ne | PIC | |||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| TT | TC | CC | T | C | ||||||||
| SNP | XN/243 | 0.082 | 0.506 | 0.412 | 0.335 | 0.665 | 4.455 | <0.05 | 0.554 | 0.446 | 1.804 | 0.346 |
| g.35989944 | JN/205 | 0.063 | 0.293 | 0.644 | 0.210 | 0.790 | 2.813 | >0.05 | 0.668 | 0.332 | 1.496 | 0.277 |
| T>C | PN/372 | 0.167 | 0.602 | 0.231 | 0.468 | 0.532 | 16.301 | <0.05 | 0.502 | 0.498 | 1.992 | 0.374 |
| Locus | Genotypes | Body Trait (cm, Mean ± SE) | ||||
|---|---|---|---|---|---|---|
| Withers Height | Hip Width | Body Oblique Length | Chest Girth | Rump Length | ||
| g.35989944 T>C | CC | 129.61 A ± 0.55 | 132.02 A ± 0.65 | 153.26 ± 0.95 | 186.74 ± 1.13 | 48.63 ± 0.34 |
| TC | 127.00 B ± 0.73 | 129.50 B ± 0.92 | 151.41 ± 1.51 | 181.82 ± 2.25 | 47.34 ± 0.80 | |
| TT | 124.36 B ± 1.69 | 125.36 B ± 2.02 | 146.09 ± 3.16 | 176.64 ± 4.36 | 46.18 ± 1.43 | |
| p | 0.001 | 0.002 | 0.074 | 0.013 | 0.061 | |
| Locus | Genotypes | Body Trait (Mean ± SE) | |||
| Withers Height (cm) | Hip Width (cm) | Body Oblique Length (cm) | Chest Girth (cm) | ||
| g.35989944T>C | CC | 129.07 ± 0.73 | 137.86 ± 0.63 | 151.31 ± 1.53 | 189.32 a ± 1.45 |
| TC | 129.26 ± 0.92 | 135.58 ± 0.83 | 148.52 ± 4.61 | 185.16 a ± 2.42 | |
| TT | 127.00 ± 1.02 | 135.63 ± 1.60 | 148.45 ± 0.22 | 176.55 b ± 2.54 | |
| p | 0.383 | 0.246 | 0.751 | 0.003 | |
| Locus | Genotypes | Body Trait (Mean ± SE) | |||
| Paunch Girth (cm) | Cannon Bone CRCM (cm) | Body Weight (kg) | |||
| g.35989944T>C | CC | 220.98 A ± 2.06 | 18.11 ± 0.28 | 471.90 A ± 10.42 | |
| TC | 217.60 A ± 1.54 | 18.51 ± 0.18 | 455.55 A ± 7.24 | ||
| TT | 205.64 B ± 3.22 | 18.00 ± 0.39 | 385.45 B ± 12.52 | ||
| p | 0.001 | 0.467 | <0.001 | ||
| Locus | Genotypes | Body Trait (cm, Mean ± SE) | ||
| Withers Height | Body Oblique Length | Hip Width | ||
| g.35989944T>C | CC | 126.70 ± 0.75 | 151.41 A ± 1.24 | 133.37 A ± 0.74 |
| TC | 124.59 ± 0.41 | 147.42 B ± 0.72 | 131.41 B ± 0.40 | |
| TT | 122.59 ± 0.70 | 143.98 C ± 1.32 | 129.46 C ± 0.69 | |
| p | 0.04 | <0.001 | 0.001 | |
| Locus | Genotypes | Body Trait (cm, Mean ± SE) | ||
| Chest Girth | Thurl Width | Rump Length | ||
| g.35989944T>C | CC | 178.39 A ± 1.38 | 47.05 A ± 0.38 | 49.35 a ± 0.40 |
| TC | 172.91 B ± 0.88 | 45.43 B ± 0.28 | 48.28 b ± 0.26 | |
| TT | 169.22 B ± 1.72 | 44.57 B ± 0.52 | 47.31 b ± 0.49 | |
| p | <0.001 | 0.001 | 0.008 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cheng, J.; Peng, W.; Cao, X.; Huang, Y.; Lan, X.; Lei, C.; Chen, H. Differential Expression of KCNJ12 Gene and Association Analysis of Its Missense Mutation with Growth Traits in Chinese Cattle. Animals 2019, 9, 273. https://doi.org/10.3390/ani9050273
Cheng J, Peng W, Cao X, Huang Y, Lan X, Lei C, Chen H. Differential Expression of KCNJ12 Gene and Association Analysis of Its Missense Mutation with Growth Traits in Chinese Cattle. Animals. 2019; 9(5):273. https://doi.org/10.3390/ani9050273
Chicago/Turabian StyleCheng, Jie, Wenwen Peng, Xiukai Cao, Yongzhen Huang, Xianyong Lan, Chuzhao Lei, and Hong Chen. 2019. "Differential Expression of KCNJ12 Gene and Association Analysis of Its Missense Mutation with Growth Traits in Chinese Cattle" Animals 9, no. 5: 273. https://doi.org/10.3390/ani9050273
APA StyleCheng, J., Peng, W., Cao, X., Huang, Y., Lan, X., Lei, C., & Chen, H. (2019). Differential Expression of KCNJ12 Gene and Association Analysis of Its Missense Mutation with Growth Traits in Chinese Cattle. Animals, 9(5), 273. https://doi.org/10.3390/ani9050273




