Methane Production of Fresh Sainfoin, with or without PEG, and Fresh Alfalfa at Different Stages of Maturity is Similar but the Fermentation End Products Vary
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Design
2.2. Animal and Diets
2.2.1. Forages, Crop Management, and Harvest
2.2.2. Animals and Sampling of Ruminal Digesta
2.2.3. In Vitro Gas Production Technique and Sampling
2.3. Analytical Methods
2.3.1. Chemical Composition
2.3.2. Determination of Parameters of the In Vitro Gas Production Technique
2.4. Statistical Analyses
3. Results
3.1. Chemical Composition
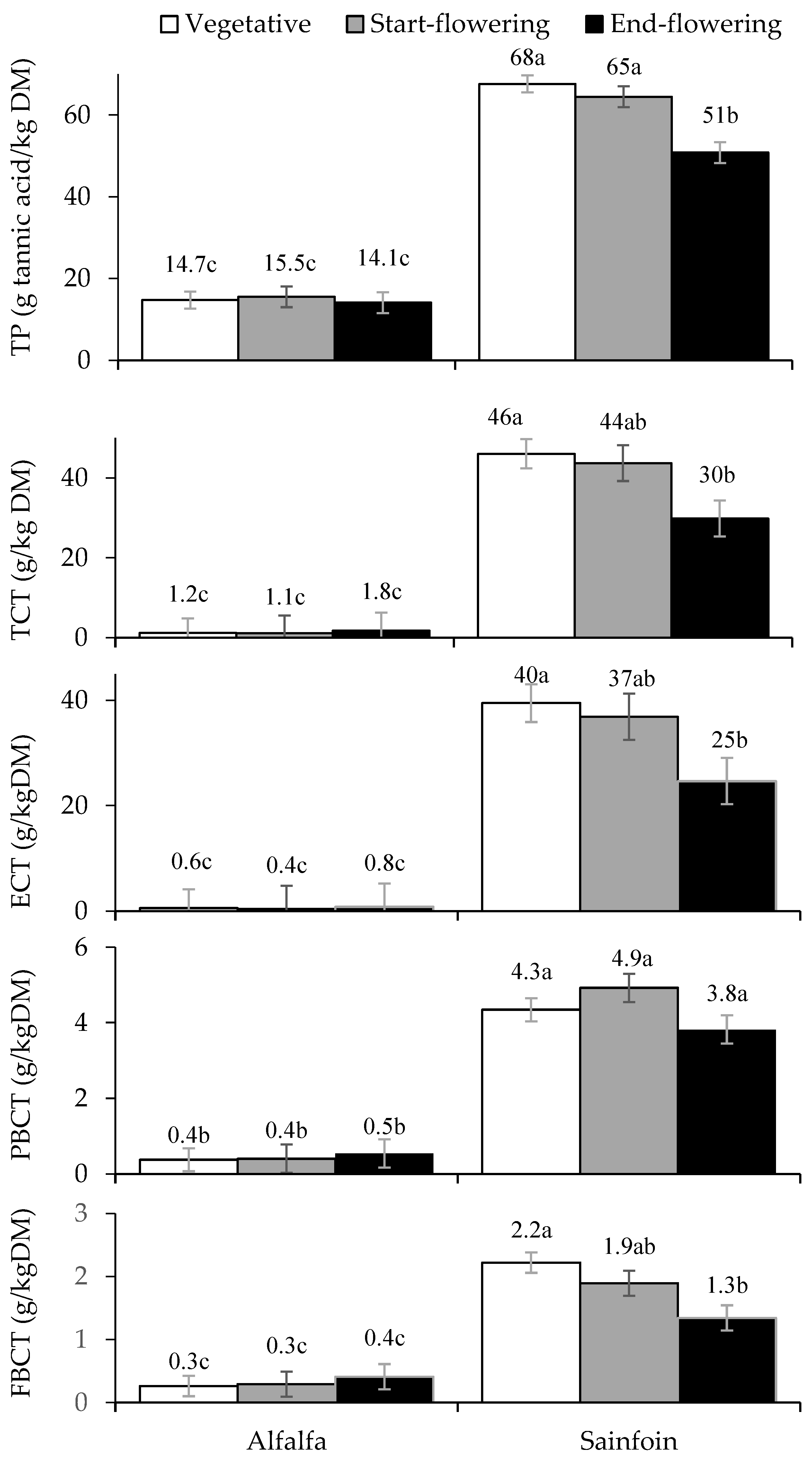
3.2. Contents of Total Polyphenols and Condensed Tannins
3.3. In Vitro Fermentation
4. Discussion
4.1. Effect of the Substrate
4.2. Effect of the Stage of Maturity
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Phelan, P.; Moloney, A.; McGeough, E.; Humphreys, J.; Bertilsson, J.; O’Riordan, E.; O’Kiely, P. Forage legumes for grazing and conserving in ruminant production systems. Crit. Rev. Plant Sci. 2015, 34, 281–326. [Google Scholar] [CrossRef]
- Delgado, I.; Muñoz, F.; Andueza, D. Evaluación comparativa de la alfalfa y la esparceta, en condiciones de secano y regadío de Aragón. Pastos Y PAC 2014, 2020, 304–310. [Google Scholar]
- Guglielmelli, A.; Calabrò, S.; Primi, R.; Carone, F.; Cutrignelli, M.I.; Tudisco, R.; Piccolo, G.; Ronchi, B.; Danieli, P.P. In vitro fermentation patterns and methane production of sainfoin (Onobrychis viciifolia Scop.) hay with different condensed tannin contents. Grass Forage Sci. 2011, 66, 488–500. [Google Scholar] [CrossRef] [Green Version]
- Toral, P.G.; Hervás, G.; Missaoui, H.; Andrés, S.; Giráldez, F.J.; Jellali, S.; Frutos, P. Effects of a tannin-rich legume (Onobrychis viciifolia) on in vitro ruminal biohydrogenation and fermentation. Span. J. Agric. Res. 2016, 14, 1–9. [Google Scholar] [CrossRef]
- Theodoridou, K.; Aufrère, J.; Andueza, D.; Le Morvan, A.; Picard, F.; Stringano, E.; Pourrat, J.; Mueller-Harvey, I.; Baumont, R. Effect of plant development during first and second growth cycle on chemical composition, condensed tannins and nutritive value of three sainfoin (Onobrychis viciifolia) varieties and lucerne. Grass Forage Sci. 2011, 66, 402–414. [Google Scholar] [CrossRef]
- Alvarez-Rodriguez, J.; Sanz, A.; Ripoll-Bosch, R.; Joy, M. Do alfalfa grazing and lactation length affect the digestive tract fill of light lambs? Small Rumin. Res. 2010, 94, 109–116. [Google Scholar] [CrossRef]
- Blanco, M.; Joy, M.; Ripoll, G.; Sauerwein, H.; Casasús, I. Grazing lucerne as fattening management for young bulls: Technical and economic performance and diet authentication. Animal 2011, 5, 113–122. [Google Scholar] [CrossRef]
- Calabrò, S.; Guglielmelli, A.; Iannaccone, F.; Danieli, P.P.; Tudisco, R.; Ruggiero, C.; Piccolo, G.; Cutrignelli, M.I.; Infascelli, F. Fermentation kinetics of sainfoin hay with and without PEG. J. Anim. Physiol. Anim. Nutr. 2012, 96, 842–849. [Google Scholar] [CrossRef]
- McMahon, L.; Majak, W.; McAllister, T.; Hall, J.; Jones, G.; Popp, J.; Cheng, K.-J. Effect of sainfoin on in vitro digestion of fresh alfalfa and bloat in steers. Can. J. Anim. Sci. 1999, 79, 203–212. [Google Scholar] [CrossRef] [Green Version]
- Chung, Y.H.; Mc Geough, E.J.; Acharya, S.; McAllister, T.A.; McGinn, S.M.; Harstad, O.M.; Beauchemin, K.A. Enteric methane emission, diet digestibility, and nitrogen excretion from beef heifers fed sainfoin or alfalfa. J. Anim. Sci. 2013, 91, 4861–4874. [Google Scholar] [CrossRef]
- Hatew, B.; Stringano, E.; Mueller-Harvey, I.; Hendriks, W.H.; Carbonero, C.H.; Smith, L.M.J.; Pellikaan, W.F. Impact of variation in structure of condensed tannins from sainfoin (Onobrychis viciifolia) on in vitro ruminal methane production and fermentation characteristics. J. Anim. Physiol. Anim. Nutr. 2016, 100, 348–360. [Google Scholar] [CrossRef]
- Theodoridou, K.; Aufrère, J.; Niderkorn, V.; Andueza, D.; Le Morvan, A.; Picard, F.; Baumont, R. In vitro study of the effects of condensed tannins in sainfoin on the digestive process in the rumen at two vegetation cycles. Anim. Feed Sci. Technol. 2011, 170, 147–159. [Google Scholar] [CrossRef]
- Azuhnwi, B.; Boller, B.; Martens, M.; Dohme-Meier, F.; Ampuero, S.; Günter, S.; Kreuzer, M.; Hess, H. Morphology, tannin concentration and forage value of 15 Swiss accessions of sainfoin (Onobrychis viciifolia Scop.) as influenced by harvest time and cultivation site. Grass Forage Sci. 2011, 66, 474–487. [Google Scholar] [CrossRef]
- Hatew, B.; Hayot Carbonero, C.; Stringano, E.; Sales, L.F.; Smith, L.M.J.; Mueller-Harvey, I.; Hendriks, W.H.; Pellikaan, W.F. Diversity of condensed tannin structures affects rumen in vitro methane production in sainfoin (Onobrychis viciifolia) accessions. Grass Forage Sci. 2015, 70, 474–490. [Google Scholar] [CrossRef]
- Lobón, S.; Blanco, M.; Sanz, A.; Ripoll, G.; Joy, M. Effects of feeding strategies during lactation and the inclusion of quebracho in the fattening on performance and carcass traits in light lambs. J. Sci. Food Agric. 2019, 99, 457–463. [Google Scholar] [CrossRef]
- Borreani, G.; Peiretti, P.G.; Tabacco, E. Evolution of yield and quality of sainfoin (Onobrychis viciifolia Scop.) in the spring growth cycle. Agronomie 2003, 23, 193–201. [Google Scholar] [CrossRef]
- Kalu, B.A.; Fick, G.W. Quantifying Morphological Development of Alfalfa for Studies of Herbage Quality. Crop Sci. 1981, 21, 267–271. [Google Scholar] [CrossRef]
- Rufino-Moya, P.J.; Blanco, M.; Bertolín, J.R.; Joy, M. Effect of the method of preservation on the chemical composition and in vitro fermentation characteristics in two legumes rich in condensed tannins. Anim. Feed Sci. Technol. 2019, 251, 12–20. [Google Scholar] [CrossRef]
- Menke, K.H.; Steingass, H. Estimation of the energetic feed value obtained from chemical analysis and in vitro gas production using rumen fluid. Anim. Res. Dev. 1988, 28, 7–55. [Google Scholar]
- AOAC. Official Methods of Analysis, 17th ed.; Association of Official Analytical Chemist: Arlington, VA, USA, 2000. [Google Scholar]
- Van Soest, P.J.; Robertson, J.B.; Lewis, B.A. Methods for dietary fiber, neutral detergent fiber, and nonstarch polysaccharides in relation to animal nutrition. J. Dairy Sci. 1991, 74, 3583–3597. [Google Scholar] [CrossRef]
- AOCS. Approved Procedure Am 5-04, Rapid Determination of Oil/Fat Utilizing High. Temperature Solvent Extraction; AOCS Press: Urbana, IL, USA, 2005. [Google Scholar]
- Makkar, H.P.S. Quantification of Tannins in Tree Foliage; FAO/IAEA Working Document; IAEA: Vienna, Austria, 2000. [Google Scholar]
- Julkunen-Tiitto, R. Phenolic Constituents in the Leaves of Northern Willows: Methods for the Analysis of Certain Phenolics. J. Agric. Food Chem. 1985, 33, 213–217. [Google Scholar] [CrossRef]
- Terrill, T.H.; Windham, W.R.; Hoveland, C.S.; Amos, H.E. Forage preservation method influences on tannin concentration, intake and digestibility of Sericea lespedeza by sheep. Agron. J. 1989, 81, 435–439. [Google Scholar] [CrossRef]
- Grabber, J.H.; Zeller, W.E.; Mueller-Harvey, I. Acetone enhances the direct analysis of procyanidin- and prodelphinidin-based condensed tannins in lotus species by the butanol-HCl-iron assay. J. Agric. Food Chem. 2013, 61, 2669–2678. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, R.M.; Terrill, T.H.; Muir, J.P. Drying method and origin of standard affect condensed tannin (CT) concentrations in perennial herbaceous legumes using simplified butanol-HCl CT analysis. J. Sci. Food Agric. 2008, 88, 1060–1067. [Google Scholar] [CrossRef]
- France, J.; Dijkstra, J.; Dhanoa, M.S.; Lopez, S.; Bannink, A. Estimating the extent of degradation of ruminant feeds from a description of their gas production profiles observed in vitro: Derivation of models and other mathematical considerations. Br. J. Nutr. 2000, 83, 143–150. [Google Scholar] [CrossRef] [PubMed]
- Chaney, A.L.; Marbach, E.P. Modified reagents for determination of urea and ammonia. Clin. Chem. 1962, 8, 130–132. [Google Scholar] [PubMed]
- Grosse-Brinkhaus, A.; Wyss, U.; Arrigo, Y.; Girard, M.; Bee, G.; Zeitz, J.O.; Kreuzer, M.; Dohme-Meier, F. In vitro ruminal fermentation characteristics and utilisable CP supply of sainfoin and birdsfoot trefoil silages and their mixtures with other legumes. Animal 2017, 11, 580–590. [Google Scholar] [CrossRef]
- Frutos, P.; Hervás, G.; Giráldez, F.J.; Mantecón, A.R. An in vitro study on the ability of polyethylene glycol to inhibit the effect of quebracho tannins and tannic acid on rumen fermentation in sheep, goats, cows, and deer. Aust. J. Agric. Res. 2004, 55, 1125–1132. [Google Scholar] [CrossRef] [Green Version]
- Martínez, T.F.; McAllister, T.A.; Wang, Y.; Reuter, T. Effects of tannic acid and quebracho tannins on in vitro ruminal fermentation of wheat and corn grain. J. Sci. Food Agric. 2006, 86, 1244–1256. [Google Scholar] [CrossRef]
- Scharenberg, A.; Arrigo, Y.; Gutzwiller, A.; Soliva, C.R.; Wyss, U.; Kreuzer, M.F.D. Palatability in sheep and in vitro nutritional value of dried and ensiled sainfoin (Onobrychis viciifolia) birdsfoot trefoil (Lotus corniculatus), and chicory (Cichorium intybus). Arch. Anim. Nutr. 2007, 61, 481–496. [Google Scholar] [CrossRef] [PubMed]
- Tavendale, M.H.; Meagher, L.P.; Pacheco, D.; Walker, N.; Attwood, G.T.; Sivakumaran, S. Methane production from in vitro rumen incubations with Lotus pedunculatus and Medicago sativa, and effects of extractable condensed tannin fractions on methanogenesis. Anim. Feed Sci. Technol. 2005, 123–124 Pt 1, 403–419. [Google Scholar] [CrossRef]
- Doane, P.H.; Pell, A.N.; Schofield, P. The Effect of Preservation Method on the Neutral Detergent Soluble Fraction of Forages. J. Anim. Sci. 1997, 75, 1140–1148. [Google Scholar] [CrossRef] [PubMed]
- Bodas, R.; Prieto, N.; García-González, R.; Andrés, S.; Giráldez, F.J.; López, S. Manipulation of rumen fermentation and methane production with plant secondary metabolites. Anim. Feed Sci. Technol. 2012, 176, 78–93. [Google Scholar] [CrossRef] [Green Version]
- Niderkorn, V.; Mueller-Harvey, I.; Le Morvan, A.; Aufrère, J. Synergistic effects of mixing cocksfoot and sainfoin on in vitro rumen fermentation. Role of condensed tannins. Anim. Feed Sci. Technol. 2012, 178, 48–56. [Google Scholar] [CrossRef]
- Huyen, N.; Fryganas, C.; Uittenbogaard, G.; Mueller-Harvey, I.; Verstegen, M.; Hendriks, W.; Pellikaan, W. Structural features of condensed tannins affect in vitro ruminal methane production and fermentation characteristics. J. Agric. Sci. 2016, 154, 1474–1487. [Google Scholar] [CrossRef]
- Lobón, S.; Molino, F.; Legua, A.; Eseverri, M.P.; Céspedes, M.A.; Joy, M. Efecto del Forraje y de La Inclusión de Concentrado En la Dieta Sobre La Producción de Gas y Metano En Ovino. 2015. Available online: http://www.uibcongres.org/imgdb/archivo_dpo19392.pdf (accessed on 1 March 2019).
- Mueller-Harvey, I. Unravelling the conundrum of tannins in animal nutrition and health. J. Sci. Food Agric. 2006, 86, 2010–2037. [Google Scholar] [CrossRef]
- Hassanat, F.; Benchaar, C. Assessment of the effect of condensed (acacia and quebracho) and hydrolysable (chestnut and valonea) tannins on rumen fermentation and methane production in vitro. J. Sci. Food Agric. 2013, 93, 332–339. [Google Scholar] [CrossRef]
- Theodoridou, K.; Aufrére, J.; Andueza, D.; Le Morvan, A.; Picard, F.; Pourrat, J.; Baumont, R. Effects of condensed tannins in wrapped silage bales of sainfoin (Onobrychis viciifolia) on in vivo and in situ digestion in sheep. Animal 2012, 6, 245–253. [Google Scholar] [CrossRef] [PubMed]
- Jin, L.; Wang, Y.; Iwaasa, A.; Xu, Z.; Schellenberg, M.; Zhang, Y.; Liu, X.; McAllister, T. Effect of condensed tannins on ruminal degradability of purple prairie clover (Dalea purpurea Vent.) harvested at two growth stages. Anim. Feed Sci. Technol. 2012, 176, 17–25. [Google Scholar] [CrossRef]
- Piluzza, G.; Bullitta, S. The dynamics of phenolic concentration in some pasture species and implications for animal husbandry. J. Sci. Food Agric. 2010, 90, 1452–1459. [Google Scholar] [CrossRef]
- Joseph, R.; Tanner, G.; Larkin, P. Proanthocyanidin synthesis in the forage legume Onobrychis viciifolia. A study of chalcone synthase, dihydroflavonol 4-reductase and leucoanthocyanidin 4-reductase in developing leaves. Funct. Plant Biol. 1998, 25, 271–278. [Google Scholar] [CrossRef]
- Bal, M.A.; Ozturk, D.; Aydin, R.; Erol, A.; Ozkan, C.O.; Ata, M.; Karakas, E.; Karabay, P. Nutritive value of sainfoin (Onobrychis viciaefolia) harvested at different maturity stages. Pak. J. Biol. Sci. 2006, 9, 205–209. [Google Scholar] [CrossRef]
- Navarro-Villa, A.; O’Brien, M.; López, S.; Boland, T.M.; O’Kiely, P. In vitro rumen methane output of red clover and perennial ryegrass assayed using the gas production technique (GPT). Anim. Feed Sci. Technol. 2011, 168, 152–164. [Google Scholar] [CrossRef]
| Alfalfa | Sainfoin | |||||
| Item | Vegetative | Start-Flowering | End-Flowering | Vegetative | Start-Flowering | End-Flowering |
| Chemical composition | ||||||
| Dry Matter (DM) (g/kg) | 249 | 261 | 333 | 241 | 262 | 224 |
| Ash (g/kg DM) | 103 | 111 | 98 | 83 | 113 | 82 |
| CP 1 (g/kg DM) | 227 | 207 | 166 | 204 | 201 | 181 |
| NDFom 2 (g/kg DM) | 336 | 352 | 405 | 313 | 324 | 394 |
| ADFom 3 (g/kg DM) | 201 | 230 | 276 | 199 | 213 | 264 |
| Lignin (sa), (g/kg DM) | 45 | 53 | 66 | 78 | 76 | 76 |
| Ether extract (g/kg DM) | 18 | 10 | 16 | 22 | 15 | 11 |
| NSC 4 (g/kg DM) | 317 | 320 | 316 | 379 | 347 | 332 |
| Plant components (%) | ||||||
| Leaves | 59.3 | 53.7 | 42.7 | 66.3 | 56.0 | 38.5 |
| Stems | 40.7 | 43.7 | 51.3 | 33.7 | 40.8 | 44.6 |
| Flowers | 0.0 | 2.5 | 5.9 | 0.0 | 3.2 | 16.8 |
| Alfalfa | Sainfoin | Sainfoin+PEG | p-Values | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Item | VEG | Start-F | End-F | VEG | Start-F | End-F | VEG | Start-F | End-F | r.s.d. 2 | S | SM | SxSM |
| pH | 6.42 a | 6.44 a | 6.41 a | 6.35 bx | 6.32 by | 6.33 by | 6.37 bx | 6.31 bz | 6.34 by | 0.032 | <0.001 | 0.002 | 0.01 |
| Gas production (mL/g dOM 3) | 179 | 184 | 188 | 183 | 163 | 181 | 180 | 199 | 173 | 26.5 | 0.43 | 0.97 | 0.12 |
| A (mL) | 68 x | 67 bxy | 62 y | 63 y | 74 bx | 70 x | 65 y | 78 ax | 67 y | 5.7 | 0.01 | <0.001 | <0.001 |
| c (h−1) | 0.2a | 0.22 a | 0.19 | 0.14 by | 0.16 bxy | 0.19 y | 0.15 bx | 0.15 bx | 0.19 y | 0.037 | <0.001 | 0.02 | 0.05 |
| CH4 production (mL/g dOM 3) | 37 | 38 | 39 | 37 | 33 | 38 | 37 | 39 | 36 | 5.0 | 0.29 | 0.73 | 0.17 |
| CH4: gas (mL/L) | 145 | 142 | 141 | 141xy | 134 y | 148 x | 145 | 144 | 144 | 8.8 | 0.35 | 0.13 | 0.11 |
| IVOMD (g/kg) | 851 bx | 837 bx | 770 by | 850 by | 926 ax | 856 ay | 868 ay | 936 ax | 864 ay | 16.9 | <0.001 | <0.001 | <0.001 |
| NH3-N (mg/L) | 240 a | 247 a | 210 | 206 b | 184 b | 201 | 242 a | 234 a | 215 | 31.7 | <0.001 | 0.06 | 0.20 |
| Total VFAs (mmol/L) | 102 xy | 105 x | 97 y | 96 y | 103 x | 101 xy | 99 y | 105 x | 102 xy | 7.1 | 0.61 | 0.02 | 0.35 |
| Acetic acid (C2) (mol/100 mol) | 63.1 cy | 63.9 bx | 64.0 bx | 65.4 ay | 66.5 ax | 66.3 ax | 63.9 by | 64.6 bx | 64.5 bx | 0.76 | <0.001 | <0.001 | 0.95 |
| Propionic acid (C3) (mol/100 mol) | 15.5 a | 15.3 a | 15.3 a | 15 bx | 14.5 cy | 14.5 by | 15.3 ax | 14.9 by | 15.2 ax | 0.28 | <0.001 | <0.001 | 0.09 |
| Butyric acid (mol/100 mol) | 13.0 a | 12.6 a | 12.6 a | 12.1 c | 12.2 b | 12 b | 12.5 b | 12.8 a | 12.4 a | 0.45 | <0.001 | 0.20 | 0.16 |
| Iso-butyric acid (mol/100 mol) | 1.97 ax | 1.91 axy | 1.87 ay | 1.82 bx | 1.68 by | 1.72 by | 1.97 ax | 1.83 ay | 1.84 ay | 0.100 | <0.001 | <0.001 | 0.63 |
| Valeric acid (mol/100 mol) | 2.14 axy | 2.09 ay | 2.22 ax | 1.87 bx | 1.7 cy | 1.83 cx | 2.12 ay | 1.95 bx | 2.04 by | 0.107 | <0.001 | <0.001 | 0.11 |
| Iso-valeric acid (mol/100 mol) | 4.27 ax | 4.09 axy | 4.04 ay | 3.87 bx | 3.5 by | 3.65 by | 4.19 ax | 3.91 ay | 3.93 ay | 0.223 | <0.001 | <0.001 | 0.69 |
| C2:C3 (mol/mol) | 4.1 cy | 4.18 cxy | 4.2 bx | 4.39 ay | 4.62 ax | 4.59 ax | 4.18 by | 4.36 bx | 4.25 by | 0.109 | <0.001 | <0.001 | 0.13 |
| CH4:VFA (mL/mol) | 2.41 | 2.24 b | 2.35 | 2.53 | 2.22 b | 2.48 | 2.62 | 2.73 a | 2.4 | 0.313 | 0.01 | 0.26 | 0.10 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rufino-Moya, P.J.; Blanco, M.; Bertolín, J.R.; Joy, M. Methane Production of Fresh Sainfoin, with or without PEG, and Fresh Alfalfa at Different Stages of Maturity is Similar but the Fermentation End Products Vary. Animals 2019, 9, 197. https://doi.org/10.3390/ani9050197
Rufino-Moya PJ, Blanco M, Bertolín JR, Joy M. Methane Production of Fresh Sainfoin, with or without PEG, and Fresh Alfalfa at Different Stages of Maturity is Similar but the Fermentation End Products Vary. Animals. 2019; 9(5):197. https://doi.org/10.3390/ani9050197
Chicago/Turabian StyleRufino-Moya, Pablo José, Mireia Blanco, Juan Ramón Bertolín, and Margalida Joy. 2019. "Methane Production of Fresh Sainfoin, with or without PEG, and Fresh Alfalfa at Different Stages of Maturity is Similar but the Fermentation End Products Vary" Animals 9, no. 5: 197. https://doi.org/10.3390/ani9050197
APA StyleRufino-Moya, P. J., Blanco, M., Bertolín, J. R., & Joy, M. (2019). Methane Production of Fresh Sainfoin, with or without PEG, and Fresh Alfalfa at Different Stages of Maturity is Similar but the Fermentation End Products Vary. Animals, 9(5), 197. https://doi.org/10.3390/ani9050197





