Reproductive Characteristics of Thawed Stallion Sperm
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Fresh and Frozen Stallion Sperm Samples
Thawing of Frozen Sperm
2.2. Sperm Examination
2.2.1. Sperm Survival Test
2.2.2. Sperm DNA Fragmentation Test
2.3. Artificial Insemination (AI) and Pregnancy Control
2.4. Statistical Analysis
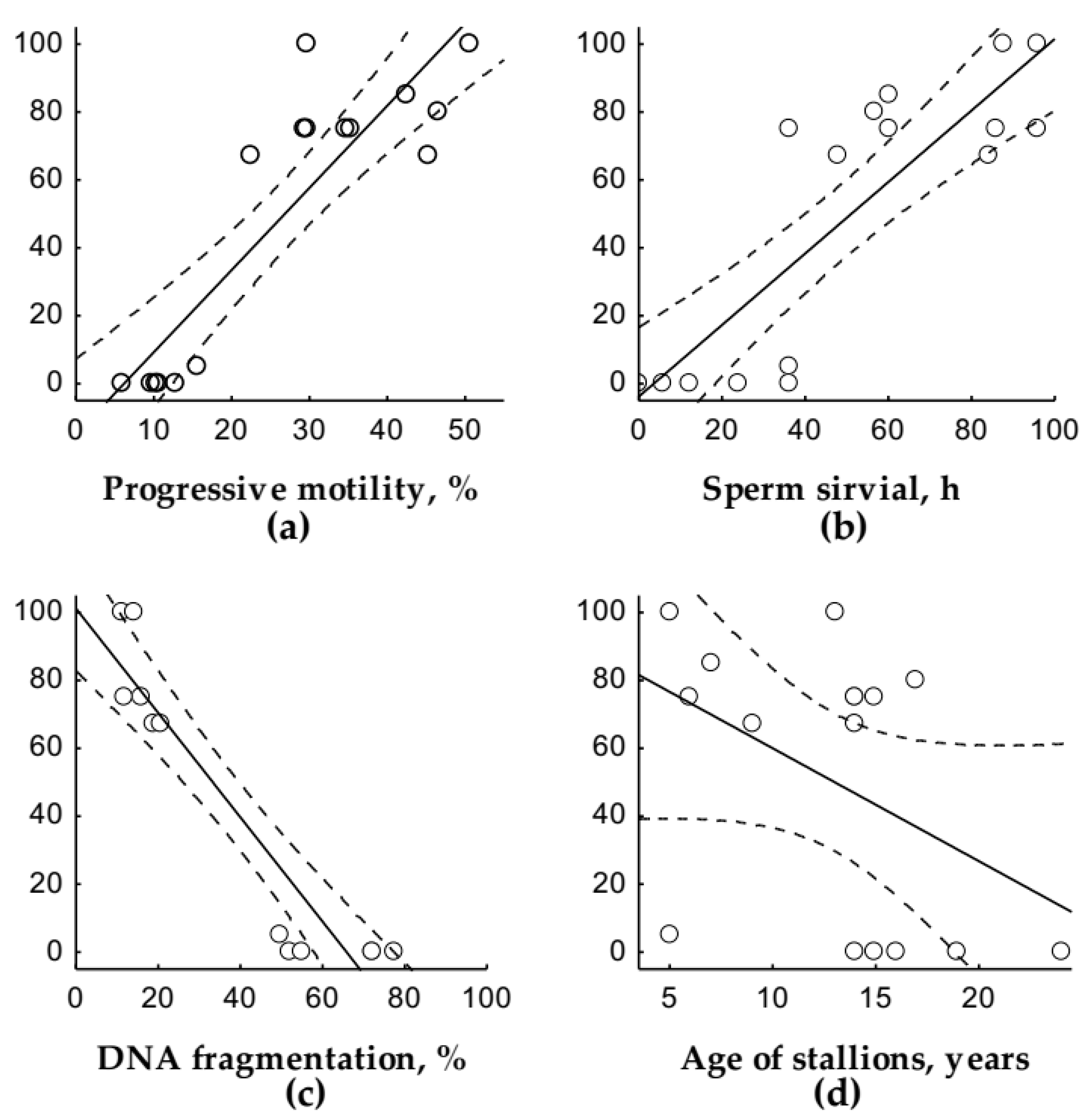
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Muriel, L.; Garrido, N.; Fernández, J.L.; Remohí, J.; Pellicer, A.; de los Santos, M.J.; Meseguer, M. Value of the sperm deoxyribonucleic acid fragmentation level, as measured by the sperm chromatin dispersion test, in the outcome of in vitro fertilization and intracytoplasmic sperm injection. Fertil. Steril. 2006, 85, 371–383. [Google Scholar] [CrossRef] [PubMed]
- Morrell, J.M.; Johannisson, A.; Dalin, A.-M.; Hammar, L.; Sandebert, T.; Rodriguez-Martinez, H. Sperm morphology and chromatin integrity in Swedish warmblood stallions and their relationship to pregnancy rates. Acta Vet. Scand. 2008, 50, 2. [Google Scholar] [CrossRef] [PubMed]
- Varner, D.D. Developments in stallion semen evaluation. Theriogenology 2008, 70, 448–462. [Google Scholar] [CrossRef] [PubMed]
- WHO Laboratory Manual for the Examination and Processing of Human Semen; World Health Organization: Geneva, Switzerland, 2010.
- Naumenkov, A.I.; Roman’kova, N.K. The method for stallion semen cryopreservation. In Theoretical and Practical Aspects of Horse Breeding: Scientific reports of Russian Institute of Horse Breeding; Russian Institute of Horse Breeding: Divovo, Russia, 1971; Volume XXV, pp. 128–132. [Google Scholar]
- Sestili, P.; Calcabrini, C.; Diaz, A.R.; Fimognari, C.; Stocchi, V. The Fast-Halo Assay for the Detection of DNA Damage. Methods Mol. Biol. 2017, 1644, 75–93. [Google Scholar] [CrossRef] [PubMed]
- Smetanina, N.M.; Pustovalova, M.V.; Osipov, A.N. Modified DNA-halo method for assessment of DNA damage induced by various genotoxic agents. Radiatsionnaia Biologiia Radioecologiia/Rossiiskaia Akademiia Nauk 2013, 53, 389–393. [Google Scholar]
- Lebedeva, L.F.; Atroshchenko, M.M.; Burmistrova, S.A. Main factors affecting mare insemination with cryopreserved domestic and foreign sperm. Agric. Biol. 2015, 50, 476–485. [Google Scholar] [CrossRef]
- Anzar, M.; He, L.; Buhr, M.M.; Kroetsch, T.G.; Pauls, K.P. Sperm Apoptosis in Fresh and Cryopreserved Bull Semen Detected by Flow Cytometry and Its Relationship with Fertility1. Biol. Reprod. 2002, 66, 354–360. [Google Scholar] [CrossRef] [PubMed]
- Evenson, D.; Jost, L. Sperm chromatin structure assay is useful for fertility assessment. Methods Cell Sci. 2000, 22, 169–189. [Google Scholar] [CrossRef] [PubMed]
- Love, C.C. The sperm chromatin structure assay: A review of clinical applications. Anim. Reprod. Sci. 2005, 89, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Zini, A.; Fischer, M.A.; Sharir, S.; Shayegan, B.; Phang, D.; Jarvi, K. Prevalence of abnormal sperm DNA denaturation in fertile and infertile men. Urology 2002, 60, 1069–1072. [Google Scholar] [CrossRef] [PubMed]
- López-Fernández, C.; Crespo, F.; Arroyo, F.; Fernández, J.L.; Arana, P.; Johnston, S.D.; Gosálvez, J. Dynamics of sperm DNA fragmentation in domestic animals. Theriogenology 2007, 68, 1240–1250. [Google Scholar] [CrossRef] [PubMed]
- Krakowski, L.; Obara, J.; Wąchocka, A.; Piech, T.; Bartoszek, P.; Kostro, K.; Tatara, M.R. Assessment of Extent of Apoptosis and DNA Defragmentation in Chilled Semen of Stallions During the Breeding Season. Reprod. Domest. Anim. 2013, 48, 826–832. [Google Scholar] [CrossRef] [PubMed]
- Alessandra, G.; Lacalandra, G.M.; Filannino, A.; Pizzi, F.; Nicassio, M.; Dell’Aquila, M.E. Assessment of viability, chromatin structure stability, mitochondrial function and motility of stallion fresh sperm by using objective methodologies. J. Cell Anim. Biol. 2010, 4, 034–041. [Google Scholar]
- Zhabin, S.G.; Treshchenkov, E.A.; Artifeksov, S.B.; Pavlenko, I.I.; Polukarov, A.N. Comparative evaluation of sperm DNA fragmentation and other sperm parameters. Problemy Reproduktsii 2015, 21, 121–124. [Google Scholar] [CrossRef]
- Benchaib, M.; Braun, V.; Lornage, J.; Hadj, S.; Salle, B.; Lejeune, H.; Guérin, J.F. Sperm DNA fragmentation decreases the pregnancy rate in an assisted reproductive technique. Hum. Reprod. 2003, 18, 1023–1028. [Google Scholar] [CrossRef] [PubMed]
| Age of Stallions, Years | Sperm Volume, mL | Semen Concentration, 106 per mL | Spermatozoa of Normal Morphology, % | Progressive Motility, % | Sperm Survival, hours | Spermatozoa with Damaged DNA, % |
|---|---|---|---|---|---|---|
| Fresh sperm (n = 21) | ||||||
| 11.2 ± 1.1 | 42.3 ± 5.0 | 210.7 ± 22.1 | 74.6 ± 2.5 | 50.7 ± 1.7 | 117.7 ± 9.2 | 28.7 ± 2.1 |
| Thawed sperm (n = 17) | ||||||
| 12.9 ± 1.3 | — | — | 76.0 ± 2.2 | 26.9 ± 3.6 * | 48.5 ± 8.1 * | 39.3 ± 7.6 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Atroshchenko, M.M.; Arkhangelskaya, E.; Isaev, D.A.; Stavitsky, S.B.; Zaitsev, A.M.; Kalaschnikov, V.V.; Leonov, S.; Osipov, A.N. Reproductive Characteristics of Thawed Stallion Sperm. Animals 2019, 9, 1099. https://doi.org/10.3390/ani9121099
Atroshchenko MM, Arkhangelskaya E, Isaev DA, Stavitsky SB, Zaitsev AM, Kalaschnikov VV, Leonov S, Osipov AN. Reproductive Characteristics of Thawed Stallion Sperm. Animals. 2019; 9(12):1099. https://doi.org/10.3390/ani9121099
Chicago/Turabian StyleAtroshchenko, Mikhail M., Ekaterina Arkhangelskaya, Dmitry A. Isaev, Sergey B. Stavitsky, Alexander M. Zaitsev, Valery V. Kalaschnikov, Sergey Leonov, and Andreyan N. Osipov. 2019. "Reproductive Characteristics of Thawed Stallion Sperm" Animals 9, no. 12: 1099. https://doi.org/10.3390/ani9121099
APA StyleAtroshchenko, M. M., Arkhangelskaya, E., Isaev, D. A., Stavitsky, S. B., Zaitsev, A. M., Kalaschnikov, V. V., Leonov, S., & Osipov, A. N. (2019). Reproductive Characteristics of Thawed Stallion Sperm. Animals, 9(12), 1099. https://doi.org/10.3390/ani9121099





