The Facilitating Effect of Tartary Buckwheat Flavonoids and Lactobacillus plantarum on the Growth Performance, Nutrient Digestibility, Antioxidant Capacity, and Fecal Microbiota of Weaned Piglets
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals, Diets and Management
2.2. Digestibility Trial and Chemical Analyses
2.3. Blood Profiles
2.4. DNA Extraction, pCR Amplification of 16S rRNA and Illumina Hiseq Sequencing
2.5. Processing of Sequencing Data
2.6. Statistical Analysis
3. Results
3.1. Growth Performance of Piglets
3.2. Nutrient Digestibility Analysis
3.3. Blood Biochemical Parameters
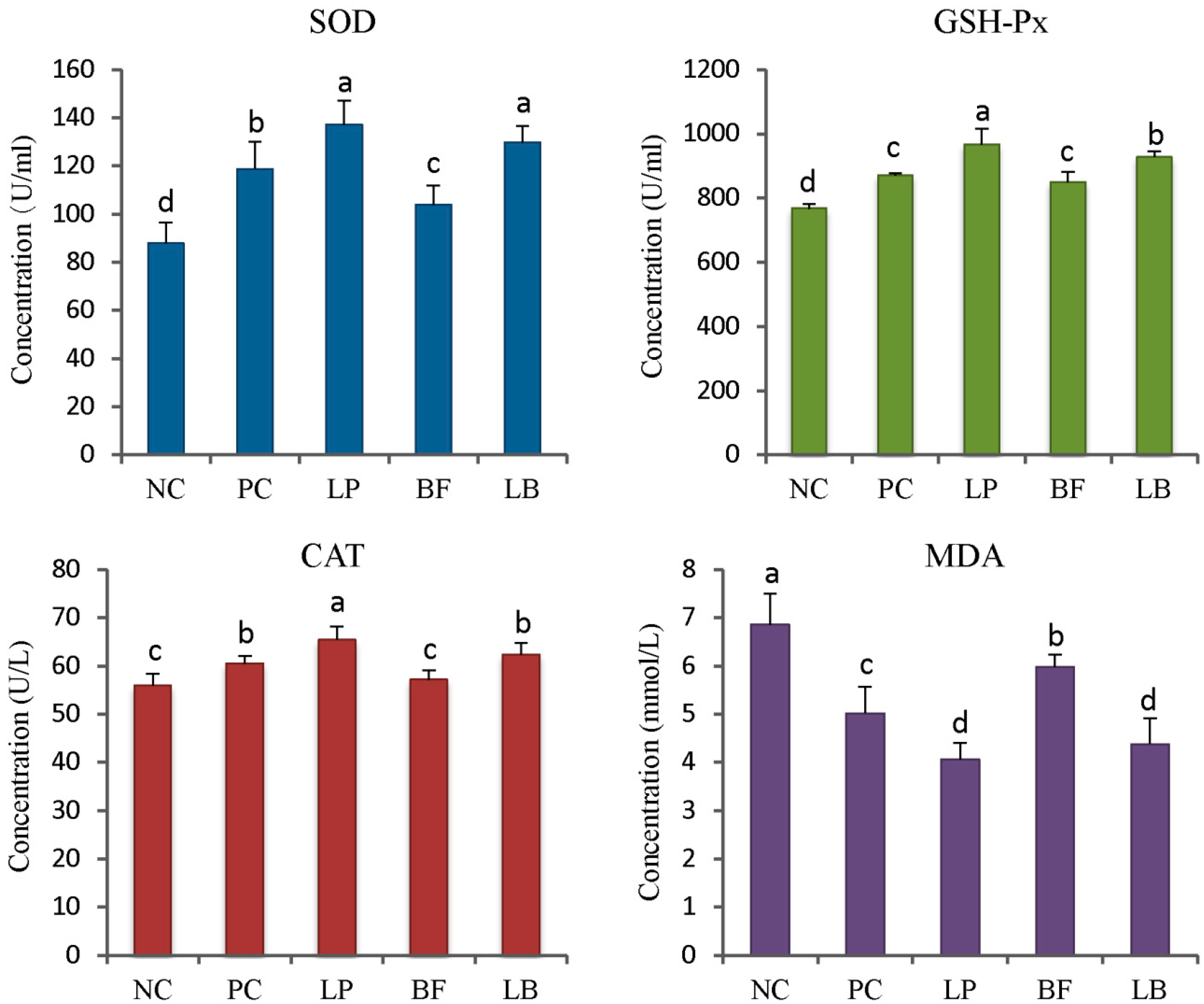
3.4. Antioxidant Enzyme Activities
3.5. Sequencing Depth and Index of Microbial Community
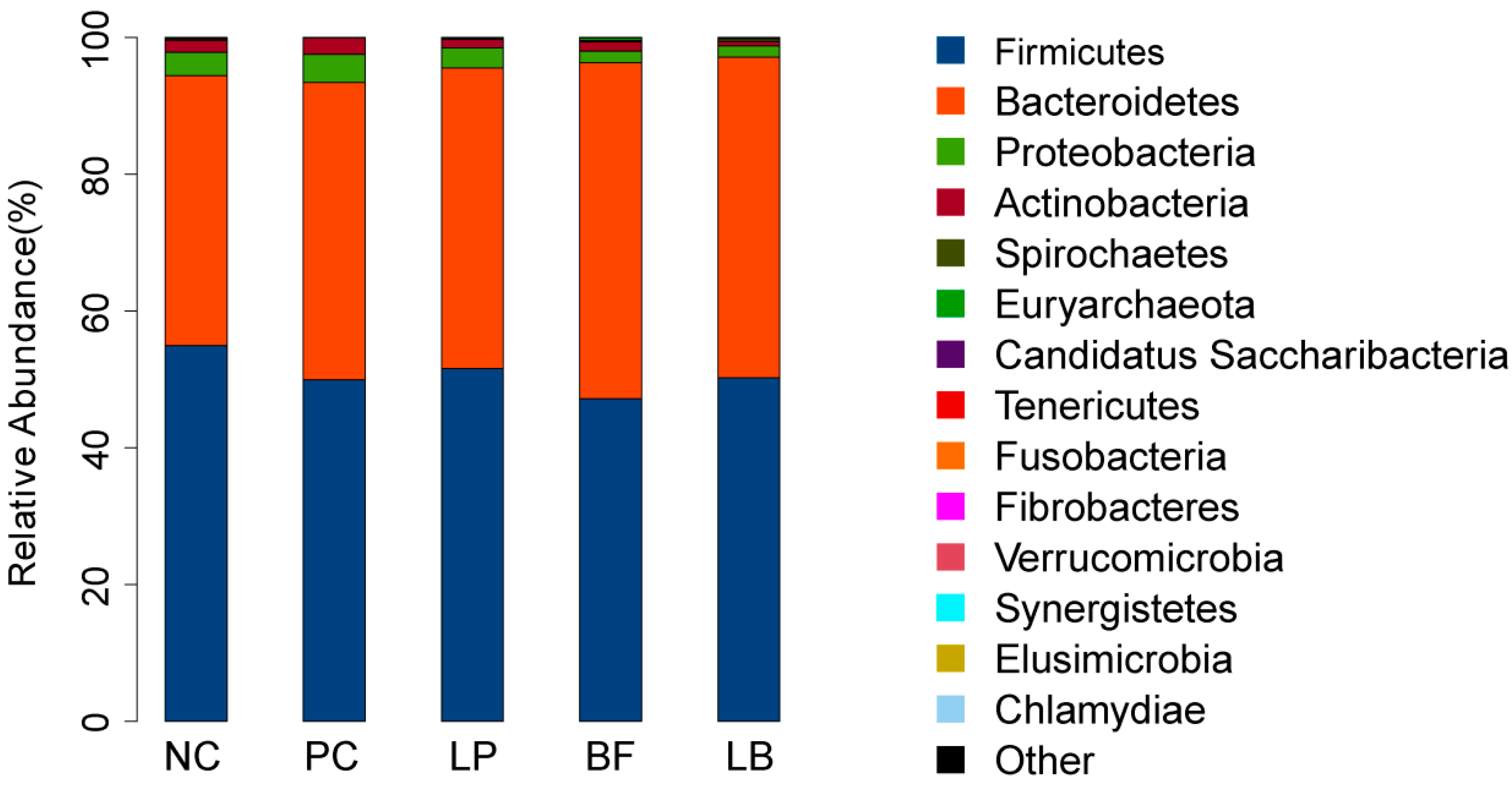
3.6. Relative Abundance and Diversity of Bacterial Communities
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Ray, P.D.; Huang, B.W.; Tsuji, Y. Reactive oxygen species (ROS) homeostasis and redox regulation in cellular signaling. Cell. Signal. 2012, 24, 981–990. [Google Scholar] [CrossRef] [PubMed]
- Weary, D.M.; Jasper, J.; Hötzel, M.J. Understanding weaning distress. Appl. Anim. Behav. Sci. 2008, 110, 24–41. [Google Scholar] [CrossRef]
- Campbell, J.M.; Crenshaw, J.D.; Polo, J. The biological stress of early weaned piglets. J. Anim. Sci. Biotechnol. 2013, 4, 19. [Google Scholar] [CrossRef] [PubMed]
- Gu, X.; Li, D.; She, R. Effect of weaning on small intestinal structure and function in the piglet. Arch. Tierernaehr. 2002, 56, 275–286. [Google Scholar] [CrossRef] [PubMed]
- Barton, M.D. Antibiotic use in animal feed and its impact on human healt. Nutr. Res. Rev. 2000, 13, 279–299. [Google Scholar] [CrossRef] [PubMed]
- Chattopadhyay, M.K. Use of antibiotics as feed additives: A burning question. Front. Microbiol. 2014, 5, 334. [Google Scholar] [CrossRef]
- Casewell, M.; Friis, C.; Marco, E.; McMullin, P.; Phillips, I. The European ban on growth-promoting antibiotics and emerging consequences for human and animal health. J. Antimicrob. Chemother. 2003, 52, 159–161. [Google Scholar] [CrossRef]
- Maciej, J.; Schäff, C.T.; Kanitz, E.; Tuchscherer, A.; Bruckmaier, R.M.; Wolffram, S.; Hammon, H.M. Short communication: Effects of oral flavonoid supplementation on the metabolic and antioxidative status of newborn dairy calves. J. Dairy Sci. 2016, 99, 805–811. [Google Scholar] [CrossRef]
- Wang, Y.; Gu, Q. Effect of probiotic on growth performance and digestive enzyme activity of Arbor Acres broilers. Res. Vet. Sci. 2010, 89, 163–167. [Google Scholar] [CrossRef]
- Yuan, X.X.; Zhang, B.; Li, L.L.; Xiao, C.W.; Fan, J.X.; Geng, M.M.; Yin, Y.L. Effects of soybean isoflavones on reproductive parameters in Chinese mini-pig boars. J. Anim. Sci. Biotechnol. 2012, 3, 31. [Google Scholar] [CrossRef]
- Zhu, C.; Wu, Y.; Jiang, Z.; Zheng, C.; Wang, L.; Yang, X.; Ma, X.; Gao, K.; Hu, Y. Dietary soy isoflavone attenuated growth performance and intestinal barrier functions in weaned piglets challenged with lipopolysaccharide. Int. Immunopharmacol. 2015, 28, 288–294. [Google Scholar] [CrossRef] [PubMed]
- Ma, T.; Chen, D.D.; Tu, Y.; Zhang, N.F.; Si, B.W.; Diao, Q.Y. Dietary supplementation with mulberry leaf flavonoids inhibits methanogenesis in sheep. Anim. Sci. J. 2017, 88, 72–78. [Google Scholar] [CrossRef] [PubMed]
- Wijngaard, H.H.; Arendt, E.K. Buckwheat. Cereal. Chem. 2006, 83, 391–401. [Google Scholar] [CrossRef]
- Zhu, F. Chemical composition and health effects of Tartary buckwheat. Food Chem. 2016, 203, 231–245. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.; Shin, Y.; Jung, S.; Kim, S.; Jo, Y.; Kim, C.; Yun, M.; Lee, S.; Sohn, J.; Yu, H.; et al. The Inhibitory Effect of Tartary Buckwheat Extracts on Adipogenesis and Inflammatory Response. Molecules 2017, 22, 1160. [Google Scholar] [CrossRef] [PubMed]
- Hosaka, T.; Nii, Y.; Tomotake, H.; Ito, T.; Tamanaha, A.; Yamasaka, Y.; Sasaga, S.; Edazawa, K.; Tsutsumi, R.; Shuto, E. Extracts of common buckwheat bran prevent sucrose digestion. J. Nutr. Sci. Vitaminol. 2011, 57, 441. [Google Scholar] [CrossRef] [PubMed]
- Merendino, N.; Molinari, R.; Costantini, L.; Mazzucato, A.; Pucci, A.; Bonafaccia, F.; Esti, M.; Ceccantoni, B.; Papeschi, C.; Bonafaccia, G. A new “functiona” pasta containing tartary buckwheat sprouts as an ingredient improves the oxidative status and normalizes some blood pressure parameters in spontaneously hypertensive rats. Food Funct. 2014, 5, 1017–1026. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.; Shen, S.; Lai, Y.; Wu, S. Rutin and quercetin, bioactive compounds from tartary buckwheat, prevent liver inflammatory injury. Food Funct. 2013, 4, 794. [Google Scholar] [CrossRef] [PubMed]
- Yeo, S.; Park, H.; Ji, Y.; Park, S.; Yang, J.; Lee, J.; Mathara, J.M.; Shin, H.; Holzapfel, W. Influence of gastrointestinal stress on autoinducer-2 activity of two Lactobacillus species. Fems Microbiol. Ecol. 2015. [Google Scholar] [CrossRef]
- Yang, K.M.; Jiang, Z.Y.; Zheng, C.T.; Wang, L.; Yang, X.F. Effect of Lactobacillus plantarum on diarrhea and intestinal barrier function of young piglets challenged with enterotoxigenic Escherichia coli K88. J. Anim. Sci. 2014, 92, 1496–1503. [Google Scholar] [CrossRef]
- Palócz, O.; Pászti-Gere, E.; Gálfi, P.; Farkas, O. Chlorogenic Acid Combined with Lactobacillus plantarum 2142 Reduced LPS-Induced Intestinal Inflammation and Oxidative Stress in IPEC-J2 Cells. PLoS ONE 2016, 11, e166642. [Google Scholar] [CrossRef] [PubMed]
- Myers, W.D.; Ludden, P.A.; Nayigihugu, V.; Hess, B.W. Technical note: A procedure for the preparation and quantitative analysis of samples for titanium dioxide. J. Anim. Sci. 2004, 82, 179–183. [Google Scholar] [CrossRef] [PubMed]
- Lammers, P.J.; Kerr, B.J.; Weber, T.E.; Dozier, W.R.; Kidd, M.T.; Bregendahl, K.; Honeyman, M.S. Digestible and metabolizable energy of crude glycerol for growing pigs. J. Anim. Sci. 2008, 86, 602–608. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Li, K.; Su, R.; Liu, W.; Ren, Y.; Zhang, C.; Du, M.; Zhang, J. Effect of dietary Tartary buckwheat extract supplementation on growth performance, meat quality and antioxidant activity in ewe lambs. Meat Sci. 2017, 134, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Dabbou, S.; Gasco, L.; Rotolo, L.; Pozzo, L.; Tong, J.M.; Dong, X.F.; Rubiolo, P.; Schiavone, A.; Gai, F. Effects of dietary alfalfa flavonoids on the performance, meat quality and lipid oxidation of growing rabbits. Asian Australas. J. Anim. Sci. 2018, 31, 270–277. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Gong, X.; Li, G.; Lin, M.; Huo, Y.; Li, S.; Zhao, G. Effects of dietary alfalfa flavonoids extraction on growth performance, organ development and blood biochemical indexes of Yangzhou geese aged from 28 to 70 days. Anim. Nutr. 2016, 2, 318–322. [Google Scholar] [CrossRef] [PubMed]
- Qi, S.; Wang, T.; Chen, R.; Wang, C.; Ao, C. Effects of flavonoids from Allium mongolicum Regel on growth performance and growth-related hormones in meat sheep. Anim. Nutr. 2017, 3, 33–38. [Google Scholar]
- Miksicek, R.J. Commonly occurring plant flavonoids have estrogenic activity. Mol. Pharm. 1993, 44, 37–43. [Google Scholar]
- Wang, Z.; Guo, Y.; Yuan, J.; Zhang, B. Effect of dietary beta-1,3/1,6-glucan supplementation on growth performance, immune response and plasma prostaglandin E2, growth hormone and ghrelin in weanling piglets. Asian Australas. J. Anim. Sci. 2008, 21, 707–714. [Google Scholar] [CrossRef]
- Suo, C.; Yin, Y.; Wang, X.; Lou, X.; Song, D.; Wang, X.; Gu, Q. Effects of lactobacillus plantarum ZJ316 on pig growth and pork quality. BMC Vet. Res. 2012, 8, 89. [Google Scholar] [CrossRef]
- Le, M.H.A.; Galle, S.; Yang, Y.; Landero, J.L.; Beltranena, E.; Ganzle, M.G.; Zijlstra, R.T. Effects of feeding fermented wheat with Lactobacillus reuteri on gut morphology, intestinal fermentation, nutrient digestibility, and growth performance in weaned pigs. J. Anim. Sci. 2016, 94, 4677–4687. [Google Scholar] [CrossRef] [PubMed]
- Meng, Q.W.; Yan, L.; Ao, X.; Zhou, T.X.; Wang, J.P.; Lee, J.H.; Kim, I.H. Influence of probiotics in different energy and nutrient density diets on growth performance, nutrient digestibility, meat quality, and blood characteristics in growing-finishing pigs. J. Anim. Sci. 2010, 88, 3320–3326. [Google Scholar] [CrossRef] [PubMed]
- Lan, R.X.; Lee, S.I.; Kim, I.H. Effects of multistrain probiotics on growth performance, nutrient digestibility, blood profiles, faecal microbial shedding, faecal score and noxious gas emission in weaning pigs. J. Anim. Physiol. Anim. Nutr. (Berl.) 2016, 100, 1130–1138. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.H.; Qiao, S.Y.; Li, D.; Piao, X.S.; Ren, J. Effects of Lactobacilli on the performance, diarrhea incidence, VFA concentration and gastrointestinal microbial flora of weaning pigs. Asian Australas. J. Anim. Sci. 2004, 17, 401–409. [Google Scholar] [CrossRef]
- Murota, K.; Nakamura, Y.; Uehara, M. Flavonoid metabolism: The interaction of metabolites and gut microbiota. Biosci. Biotechnol. Biochem. 2018, 82, 600–610. [Google Scholar] [CrossRef] [Green Version]
- Phuoc, T.L.; Jamikorn, U. Effects of probiotic supplement (Bacillus subtilis and Lactobacillus acidophilus) on feed efficiency, growth performance, and microbial population of weaning rabbits. Asian Australas. J. Anim. Sci. 2017, 30, 198–205. [Google Scholar] [CrossRef] [Green Version]
- Liu, W.; Devi, S.; Park, J.; Kim, I. Effects of complex probiotic supplementation in growing pig diets with and without palm kernel expellers on growth performance, nutrient digestibility, blood parameters, fecal microbial shedding and noxious gas emission. Anim. Sci. J. 2018, 89, 552. [Google Scholar] [CrossRef]
- Tao, H.; Guo, F.; Tu, Y.; Si, B.W.; Xing, Y.C.; Huang, D.J.; Diao, Q.Y. Effect of weaning age on growth performance, feed efficiency, nutrient digestibility and blood-biochemical parameters in Droughtmaster crossbred beef calves. Asian Australas. J. Anim. Sci. 2018, 31, 864–872. [Google Scholar] [CrossRef]
- Crozier, A.; Jaganath, I.B.; Clifford, M.N. Dietary phenolics: Chemistry, bioavailability and effects on health. Nat. Prod. Rep. 2009, 26, 1001–1043. [Google Scholar] [CrossRef]
- Peluso, I.; Miglio, C.; Morabito, G.; Ioannone, F.; Serafini, M. Flavonoids and immune function in human: A systematic review. Crit. Rev. Food Sci. Nutr. 2015, 55, 383–395. [Google Scholar] [CrossRef]
- Suzuki, Y.; Kosaka, M.; Shindo, K.; Kawasumi, T.; Kimoto-Nira, H.; Suzuki, C. Identification of antioxidants produced by Lactobacillus plantarum. Biosci. Biotechnol. Biochem. 2013, 77, 1299–1302. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Guo, Y.; Chen, H.; Wei, H.; Wan, C. Potential of Lactobacillus plantarum ZDY2013 and Bifidobacterium bifidum WBIN03 in relieving colitis by gut microbiota, immune, and anti-oxidative stress. Can. J. Microbiol. 2018, 64, 327–337. [Google Scholar] [CrossRef] [PubMed]
- Tang, W.; Xing, Z.; Li, C.; Wang, J.; Wang, Y. Molecular mechanisms and in vitro antioxidant effects of Lactobacillus plantarum MA2. Food Chem. 2017, 221, 1642–1649. [Google Scholar] [CrossRef] [PubMed]
- Lin, X.; Xia, Y.; Wang, G.; Xiong, Z.; Zhang, H.; Lai, F.; Ai, L. Lactobacillus plantarum AR501 Alleviates the Oxidative Stress of D-Galactose-Induced Aging Mice Liver by Upregulation of Nrf2-Mediated Antioxidant Enzyme Expression. J. Food Sci. 2018, 83, 1990–1998. [Google Scholar] [CrossRef] [PubMed]
- Cheng, N.; Wu, L.; Zheng, J.; Cao, W. Buckwheat Honey Attenuates Carbon Tetrachloride-Induced Liver and DNA Damage in Mice. Evid. Based Complement. Altern. Med. 2015, 2015, 987385. [Google Scholar] [CrossRef] [Green Version]
- Zhang, J.; Zhao, X.; Jiang, Y.; Zhao, W.; Guo, T.; Cao, Y.; Teng, J.; Hao, X.; Zhao, J.; Yang, Z. Antioxidant status and gut microbiota change in an aging mouse model as influenced by exopolysaccharide produced by Lactobacillus plantarum YW11 isolated from Tibetan kefir. J. Dairy Sci. 2017, 100, 6025–6041. [Google Scholar] [CrossRef]
- Bi, Y.; Yang, C.; Diao, Q.; Tu, Y. Effects of dietary supplementation with two alternatives to antibiotics on intestinal microbiota of preweaned calves challenged with Escherichia coli K99. Sci. Rep. 2017, 7, 5439. [Google Scholar] [CrossRef] [Green Version]
- Levy, S.B.; Marshall, B. Antibacterial resistance worldwide: Causes, challenges and responses. Nat. Med. 2004, 10 (Suppl. S12), S122–S129. [Google Scholar] [CrossRef]
- De Oliveira, M.N.; Jewell, K.A.; Freitas, F.S.; Benjamin, L.A.; Totola, M.R.; Borges, A.C.; Moraes, C.A.; Suen, G. Characterizing the microbiota across the gastrointestinal tract of a Brazilian Nelore steer. Vet. Microbiol. 2013, 164, 307–314. [Google Scholar] [CrossRef]
- Lanjekar, V.B.; Marathe, N.P.; Ramana, V.V.; Shouche, Y.S.; Ranade, D.R. Megasphaera indica sp. nov., an obligate anaerobic bacteria isolated from human faeces. Int. J. Syst. Evol. Microbiol. 2014, 64, 2250–2256. [Google Scholar] [CrossRef]
- Shetty, S.A.; Marathe, N.P.; Lanjekar, V.; Ranade, D.; Shouche, Y.S. Comparative genome analysis of Megasphaera sp. reveals niche specialization and its potential role in the human gut. PLoS ONE 2013, 8, e79353. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rath, C.B.; Schirmeister, F.; Figl, R.; Seeberger, P.H.; Schaffer, C.; Kolarich, D. Flagellin Glycoproteomics of the Periodontitis Associated Pathogen Selenomonas sputigena Reveals Previously Not Described O-glycans and Rhamnose Fragment Rearrangement Occurring on the Glycopeptides. Mol. Cell. Proteom. 2018, 17, 721–736. [Google Scholar] [CrossRef] [PubMed] [Green Version]


| Items | Content |
|---|---|
| Ingredients | |
| Corn | 59 |
| Soybean meal | 16.5 |
| Wheat bran | 5 |
| Rice polishings | 5 |
| DDGS | 5 |
| Wheat middling | 5 |
| Fat powder | 0.5 |
| premix a | 4 |
| Nutrient levels b | |
| DM | 88.03 |
| Cp | 18.95 |
| DE (MJ/kg) | 15.43 |
| EE | 2.90 |
| Ash | 5.41 |
| phosphorus | 0.58 |
| Calcium | 0.68 |
| Lys | 1.11 |
| Met | 0.32 |
| Cys | 0.36 |
| Thr | 0.70 |
| Items 1 | Treatments 2 | SEM | p-Value | ||||
|---|---|---|---|---|---|---|---|
| NC | pC | Lp | BF | LB | |||
| IBW, kg | 7.81 | 7.84 | 7.87 | 7.76 | 7.85 | 0.67 | 0.998 |
| FBW, kg | 13.70 b | 14.55 ab | 14.37 b | 16.76 a | 15.90 ab | 0.36 | 0.047 |
| ADFI, g | 461.12 | 554.04 | 466.11 | 525.04 | 537.64 | - | - |
| ADG, g | 210.39 b | 239.56 b | 232.14 b | 321.34 a | 287.43 ab | 12.32 | 0.029 |
| F/G | 2.19 ab | 2.31 a | 2.01 abc | 1.63 c | 1.87 bc | 0.11 | 0.082 |
| Items 1 | Treatments 2 | SEM | p-Value | ||||
|---|---|---|---|---|---|---|---|
| NC | pC | Lp | BF | LB | |||
| DM (%) | 82.19 c | 84.03 b | 84.31 b | 84.26 b | 86.28 a | 0.264 | <0.001 |
| OM (%) | 85.25 c | 87.07 b | 87.21 b | 87.19 b | 88.70 a | 0.227 | <0.001 |
| GE (%) | 81.11 c | 83.34 b | 83.48 b | 83.88 b | 85.19 a | 0.277 | <0.001 |
| Cp (%) | 76.50 b | 80.50 a | 80.79 a | 81.69 a | 81.68 a | 0.423 | <0.001 |
| EE (%) | 33.32 b | 58.37 a | 61.24 a | 60.45 a | 62.59 a | 2.242 | <0.001 |
| Ca (%) | 35.55 c | 41.92 b | 42.88 b | 44.44 ab | 49.46 a | 1.132 | <0.001 |
| p (%) | 50.36 c | 54.92 b | 55.48 b | 55.27 b | 59.95 a | 0.752 | <0.001 |
| Items 1 | Treatments 2 | SEM | p-Value | ||||
|---|---|---|---|---|---|---|---|
| NC | pC | Lp | BF | LB | |||
| BUN, mmol/L | 6.68 c | 7.87 ab | 7.17 bc | 8.48 a | 7.49 bc | 0.17 | 0.003 |
| GLU, mmol/L | 2.54 c | 3.67 ab | 2.84 bc | 4.17 a | 3.94 a | 0.17 | 0.003 |
| IgG, g/L | 7.54 c | 8.29 ab | 7.65 bc | 8.78 a | 8.15 abc | 0.13 | 0.005 |
| IgA, g/L | 0.89 b | 0.86 b | 0.85 b | 1.16 a | 0.99 b | 0.03 | 0.004 |
| IgM, g/L | 0.69 c | 0.75 bc | 0.71 bc | 0.87 a | 0.80 ab | 0.02 | 0.003 |
| Items | Treatments a | SEM | p-Value | ||||
|---|---|---|---|---|---|---|---|
| NC | pC | Lp | BF | LB | |||
| Firmicutes | 47.21 | 50.25 | 51.62 | 54.93 | 49.96 | 4.51 | 0.7636 |
| Bacteroidetes | 49.10 | 46.86 | 43.90 | 39.49 | 43.44 | 4.35 | 0.519 |
| Proteobacteria | 1.70 | 1.68 | 2.97 | 3.42 | 4.13 | 1.04 | 0.4251 |
| Actinobacteria | 1.37 | 0.63 | 1.23 | 1.76 | 2.41 | 0.53 | 0.2762 |
| Phylum | Genus | Relative Abundance (%) 1 | SEM | p-Value | ||||
|---|---|---|---|---|---|---|---|---|
| NC | pC | Lp | BF | LB | ||||
| Firmicutes | Phascolarctobacterium | 6.54 | 6.35 | 7.14 | 9.38 | 6.61 | 1.07 | 0.2422 |
| Lactobacillus | 1.32 | 5.93 | 0.94 | 1.91 | 2.20 | 1.23 | 0.1686 | |
| Clostridium sensu stricto | 0.94 | 0.44 | 0.71 | 1.59 | 1.46 | 0.52 | 0.5263 | |
| Clostridium XlVa | 3.12 | 1.88 | 7.19 | 7.17 | 2.85 | 1.70 | 0.1103 | |
| Lachnospiracea_incertae_sedis | 1.74 | 0.57 | 3.29 | 2.22 | 3.07 | 0.58 | 0.0514 | |
| Roseburia | 8.02 | 2.62 | 7.42 | 5.20 | 4.15 | 1.96 | 0.3769 | |
| Faecalibacterium | 1.60 | 3.58 | 1.87 | 1.58 | 0.88 | 0.73 | 0.2635 | |
| Oscillibacter | 1.63 | 0.57 | 0.94 | 2.49 | 1.34 | 0.61 | 0.2664 | |
| Acidaminococcus | 1.37 | 0.82 | 0.05 | 0.15 | 0.97 | 0.39 | 0.1065 | |
| Anaerovibrio | 1.64 | 0.94 | 2.85 | 2.53 | 1.57 | 0.91 | 0.6526 | |
| Dialister | 2.04 | 0.72 | 0.02 | 0.85 | 1.29 | 0.70 | 0.574 | |
| Megasphaera | 2.62 ab | 6.23 a | 0.08 b | 0.40 b | 6.08 a | 1.38 | 0.0094 | |
| Mitsuokella | 0.83 a | 0.79 a | 0.30 a | 0.63 a | 1.94 b | 0.36 | 0.0286 | |
| Selenomonas | 4.02 b | 0.30 b | 11.2 a | 0.47 b | 2.35 b | 1.70 | 0.0019 | |
| Bacteroidetes | Prevotella | 43.22 | 43.56 | 43.56 | 44.62 | 46.34 | 3.66 | 0.8588 |
| Barnesiella | 1.86 | 2.54 | 1.20 | 1.27 | 3.48 | 1.12 | 0.5283 | |
| Alloprevotella | 6.08 | 4.51 | 1.70 | 6.57 | 5.17 | 1.28 | 0.0944 | |
| Proteobacteria | Succinivibrio | 3.56 | 4.12 | 0.31 | 1.11 | 0.39 | 1.18 | 0.1252 |
| Actinobacteria | Olsenella | 1.53 | 2.48 | 0.63 | 1.05 | 0.37 | 0.60 | 0.2371 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cui, K.; Wang, Q.; Wang, S.; Diao, Q.; Zhang, N. The Facilitating Effect of Tartary Buckwheat Flavonoids and Lactobacillus plantarum on the Growth Performance, Nutrient Digestibility, Antioxidant Capacity, and Fecal Microbiota of Weaned Piglets. Animals 2019, 9, 986. https://doi.org/10.3390/ani9110986
Cui K, Wang Q, Wang S, Diao Q, Zhang N. The Facilitating Effect of Tartary Buckwheat Flavonoids and Lactobacillus plantarum on the Growth Performance, Nutrient Digestibility, Antioxidant Capacity, and Fecal Microbiota of Weaned Piglets. Animals. 2019; 9(11):986. https://doi.org/10.3390/ani9110986
Chicago/Turabian StyleCui, Kai, Qiang Wang, Shiqin Wang, Qiyu Diao, and Naifeng Zhang. 2019. "The Facilitating Effect of Tartary Buckwheat Flavonoids and Lactobacillus plantarum on the Growth Performance, Nutrient Digestibility, Antioxidant Capacity, and Fecal Microbiota of Weaned Piglets" Animals 9, no. 11: 986. https://doi.org/10.3390/ani9110986
APA StyleCui, K., Wang, Q., Wang, S., Diao, Q., & Zhang, N. (2019). The Facilitating Effect of Tartary Buckwheat Flavonoids and Lactobacillus plantarum on the Growth Performance, Nutrient Digestibility, Antioxidant Capacity, and Fecal Microbiota of Weaned Piglets. Animals, 9(11), 986. https://doi.org/10.3390/ani9110986






