The Wily and Courageous Red Fox: Behavioural Analysis of a Mesopredator at Resource Points Shared by an Apex Predator
Abstract
Simple Summary
Abstract
1. Introduction
“Look at him. His coat is russet with sufficient gold in it to make him glow. He has just enough of a ruff to please a very young lion and enough tail to be the pleasure of any animal that grows a tail. His snout is a bit pinched-looking and would be mean if there wasn’t so much pride in his carriage—he walks the Australian earth as though it was a carpet especially laid for him”.(Rolls 1969)
2. Materials and Methods
2.1. Study Sites
2.2. Recording Fox Behaviour
2.3. Construction of a Fox Ethogram
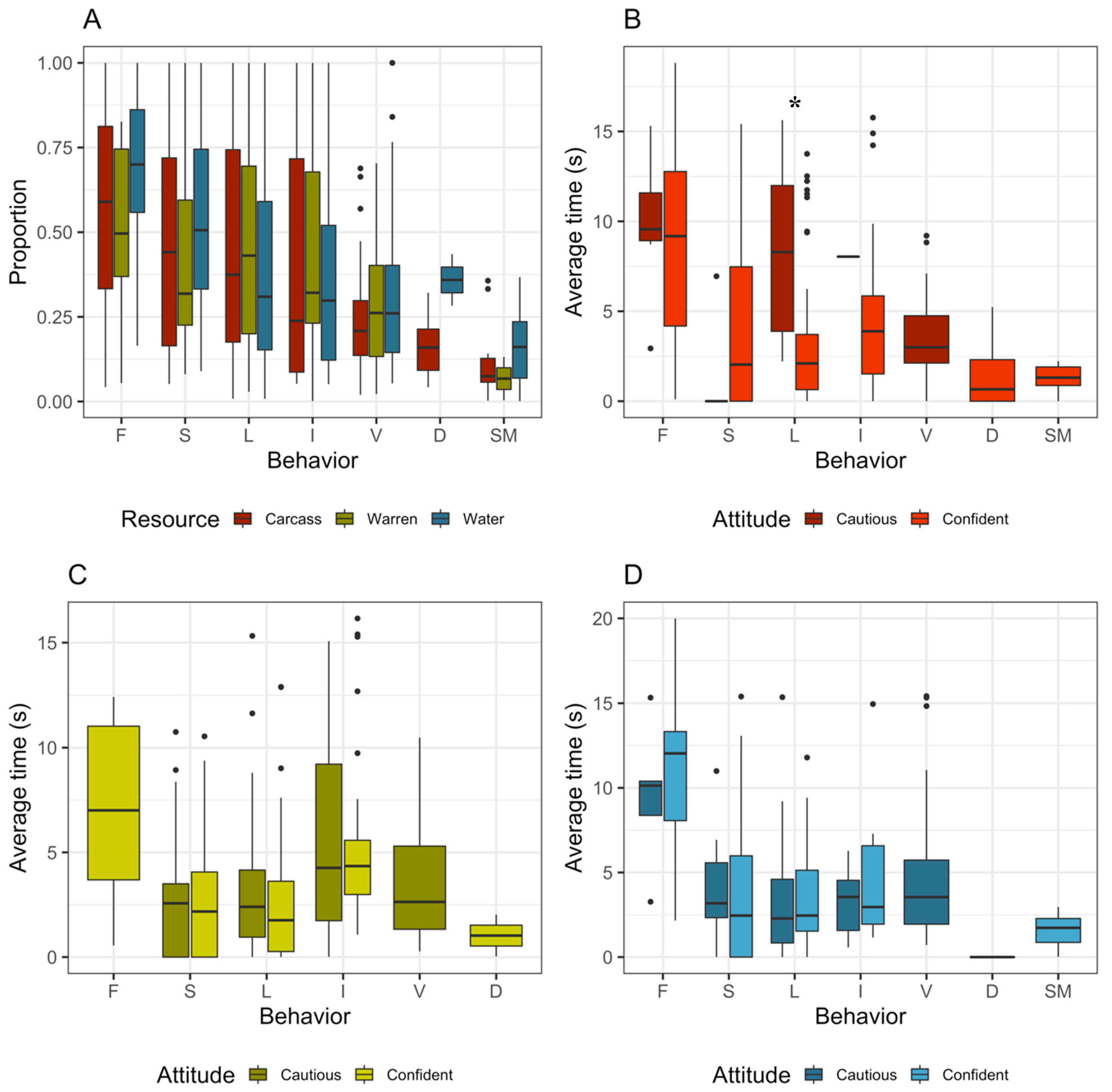
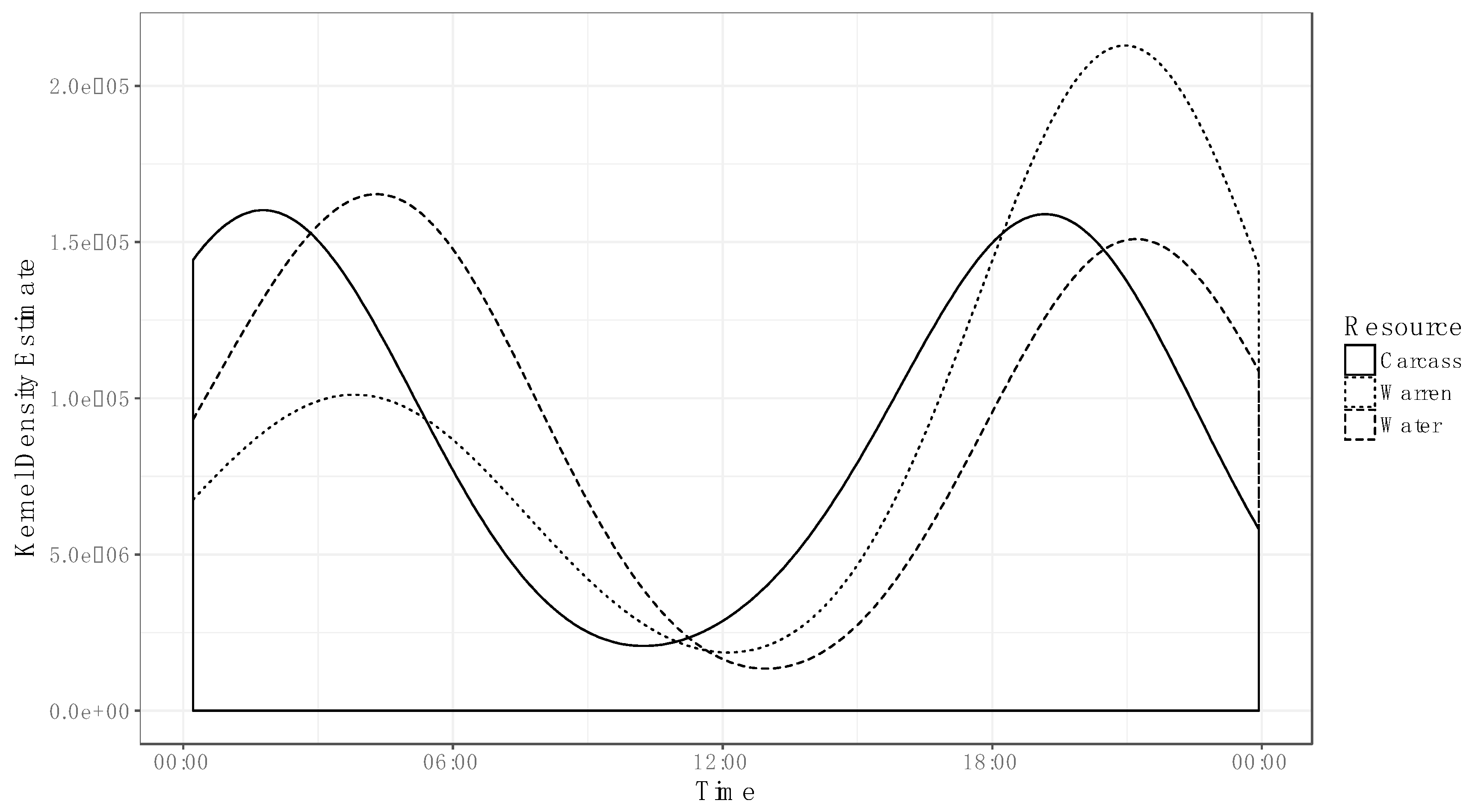
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Macdonald, D.W.; Reynolds, J. Red Fox (Vulpes vulpes). Canids: Foxes, Wolves, Jackals, and Dogs. In Status Survey and Conservation Action Plan; Sillero-Zubiri, C., Hoffmann, M., Macdonald, D.W., Eds.; IUCN: Gland, Switzerland; Cambridge, UK, 2004; pp. 129–135. [Google Scholar]
- Contesse, P.; Hegglin, D.; Gloor, S.; Bontadina, F.; Deplazes, P. The diet of urban foxes (Vulpes vulpes) and the availability of anthropogenic food in the city of Zurich, Switzerland. Mamm. Biol. 2004, 69, 81–95. [Google Scholar] [CrossRef]
- Prugh, L.R.; Stoner, C.J.; Epps, C.W.; Bean, W.T.; Ripple, W.J.; Laliberte, A.S.; Brashares, J.S. The rise of the mesopredator. Bioscience 2009, 59, 779–791. [Google Scholar] [CrossRef]
- Long, J.L. Introduced Mammals of the World: Their History, Distribution and Influence; CSIRO Publishing: Clayton, Australia, 2003; pp. 239–242. [Google Scholar]
- Dickman, C.R. Impact of exotic generalist predators on the native fauna of Australia. Wildl. Biol. 1996, 2, 185–195. [Google Scholar] [CrossRef]
- Wallach, A.D.; Johnson, C.N.; Ritchie, E.G.; O’Neill, A.J. Predator control promotes invasive dominated ecological states. Ecol. Lett. 2010, 13, 1008–1018. [Google Scholar] [CrossRef]
- Haswell, P.M.; Kusak, J.; Hayward, M.W. Large carnivore impacts are context-dependent. Food Webs 2017, 12, 3–13. [Google Scholar] [CrossRef]
- Cupples, J.B.; Crowther, M.S.; Story, G.; Letnic, M. Dietary overlap and prey selectivity among sympatric carnivores: Could dingoes suppress foxes through competition for prey? J. Mammal. 2011, 92, 590–600. [Google Scholar] [CrossRef]
- Mueller, M.A.; Drake, D.; Allen, A.J. Coexistence of coyotes (Canis latrans) and red foxes (Vulpes vulpes) in an urban landscape. PLoS ONE 2018, 13, e0190971. [Google Scholar] [CrossRef]
- Newsome, T.M.; Greenville, A.C.; Cirovic, D.; Dickman, C.R.; Johnson, C.N.; Krofel, M.; Letnic, M.; Ripple, W.J.; Ritchie, E.G.; Stoyanov, S.; et al. Top predators constrain mesopredator distributions. Nat. Commun. 2017, 8, 15469. [Google Scholar] [CrossRef]
- Letnic, M.; Greenville, A.C.; Denny, E.; Dickman, C.R.; Tischler, M.; Gordon, C.; Koch, F. Does a top predator suppress the abundance of an invasive mesopredator at a continental scale? Glob. Ecol. Biogeogr. 2011, 20, 343–353. [Google Scholar] [CrossRef]
- Scheinin, S.; Yom-Tov, Y.; Motro, U.; Geffen, E. Behavioural responses of red foxes to an increase in the presence of golden jackals: A field experiment. Anim. Behav. 2006, 71, 577–584. [Google Scholar] [CrossRef]
- Wikenros, C.; Jarnemo, A.; Frisen, M.; Kuijper, D.P.J.; Schmidt, K. Mesopredator behavioural response to olfactory signals of an apex predator. J. Ethol. 2017, 35, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Laundre, J.W.; Hernandez, L.; Ripple, W.J. The landscape of fear: Ecological implications of being afraid. Open Ecol. J. 2009, 3, 1–7. [Google Scholar] [CrossRef]
- Brawata, R.L.; Neeman, T. Is water the key? Dingo management, intraguild interactions and predator distribution around water points in arid Australia. Wildl. Res. 2011, 38, 426–436. [Google Scholar] [CrossRef]
- Haswell, P.M.; Jones, K.A.; Kusak, J.; Hayward, M.W. Fear, foraging and olfaction: How mesopredators avoid costly interactions with apex predators. Oecologia 2018, 187, 573–583. [Google Scholar] [CrossRef] [PubMed]
- Harding, E.; Doak, D.; Albertson, D. Evaluating the effectiveness of predator control - the non-native red fox as a case study. Conserv. Biol. 2001, 15, 114–1122. [Google Scholar] [CrossRef]
- Mahon, P.S. Targeted control of widespread exotic species for biodiversity conservation: The red fox (Vulpes vulpes) in New South Wales, Australia. Ecol. Manag. Restor. 2009, 10, 59–69. [Google Scholar] [CrossRef]
- Leo, V.; Reading, R.P.; Letnic, M. Interference competition: Odours of an apex predator and conspecifics influence resource acquisition by red foxes. Oecologia 2015, 179, 1033–1040. [Google Scholar] [CrossRef]
- Voigt, D.R.; Earle, B.D. Avoidance of coyotes by red fox families. J. Wildl. Manag. 1983, 47, 852–857. [Google Scholar] [CrossRef]
- Meyer, S.; Weber, J.M. Ontogeny of dominance in free-living red foxes. Ethology 1996, 102, 1008–1019. [Google Scholar] [CrossRef]
- Zabel, C.J.; Taggart, S.J. Shift in red fox (Vulpes vulpes) mating system associated with El Niño in the Bering Sea. Anim. Behav. 1989, 38, 830–838. [Google Scholar] [CrossRef]
- Baker, P.J.; Robertson, C.P.J.; Funk, S.M.; Harris, S. Potential fitness benefits of group living in the red fox (Vulpes vulpes). Anim. Behav. 1998, 56, 1411–1424. [Google Scholar] [CrossRef] [PubMed]
- Blizard, R.A.; Perry, G.C. Response of captive male red foxes (Vulpes vulpes) to some conspecific odors. J. Chem. Ecol. 1979, 5, 869–880. [Google Scholar] [CrossRef]
- Fox, M. Behaviour of Wolves Dogs and Related Canids; Dogwise Publishing: New York, NY, USA, 1971. [Google Scholar]
- Henry, J.D. The use of urine marking in the scavenging behaviour of the red fox (Vulpes vulpes). Behaviour 1977, 61, 82–105. [Google Scholar] [CrossRef]
- Macdonald, D.W. Some observations and field experiments on the urine marking behaviour of the red fox (Vulpes vulpes). Z. Tierpsychol. 1979, 51, 1–22. [Google Scholar] [CrossRef]
- Monclús, R.; Arroyo, M.; Valencia, A.; de Miguel, F.J. Red foxes (Vulpes vulpes) use rabbit (Oryctolagus cuniculus) scent marks as territorial marking sites. J. Ethol. 2009, 27, 153–156. [Google Scholar] [CrossRef]
- Baker, P.J.; Dowding, C.V.; Molony, S.E.; White, P.C.L.; Harris, S. Activity patterns of urban red foxes (Vulpes vulpes) reduce the risk of traffic-induced mortality. Behav. Ecol. 2007, 18, 716–734. [Google Scholar] [CrossRef]
- Soulsbury, C.D.; Iossa, G.; Baker, P.J.; White, P.C.L.; Harris, S. Behavioural and spatial analysis of extraterritorial movements in red foxes (Vulpes vulpes). J. Mammal. 2011, 92, 190–199. [Google Scholar] [CrossRef]
- Tolhurst, B.A.; Grogan, H.; Hughes, H. Effects of temporary captivity on ranging behaviour in urban red foxes (Vulpes vulpes). Appl. Anim. Behav. Sci. 2016, 181, 182–190. [Google Scholar] [CrossRef]
- Iossa, G.; Soulsbury, C.D.; Baker, P.J.; Edwards, K.J.; Harris, S. Behavioural changes associated with a population density decline in the facultatively social red fox. Behav. Ecol. 2009, 20, 385–395. [Google Scholar] [CrossRef]
- Cagnacci, F.; Meriggi, A.; Lovari, S. Habitat selection by the red fox (Vulpes vulpes) in an Alpine area. Ethology, Ecol. Evol. 2004, 16, 103–116. [Google Scholar] [CrossRef]
- Cavallini, P.; Lovari, S. Environmental factors influencing the use of habitat in the red fox (Vulpes vulpes). J. Zool. 1991, 223, 323–339. [Google Scholar] [CrossRef]
- Lucherini, M.; Lovari, S.; Crema, G. Habitat use and ranging behaviour of the red fox (Vulpes vulpes) in a Mediterranean rural area: Is shelter availability a key factor? J. Zool. 1995, 237, 577–591. [Google Scholar] [CrossRef]
- Cavallini, P. Ranging behaviour of the red fox (Vulpes vulpes) in rural southern Japan. J. Mammal. 1992, 73, 321–325. [Google Scholar] [CrossRef]
- Kistler, C.; Hegglin, D.; Würbel, H.; König, B. Feeding enrichment in an opportunistic carnivore: The red fox. Appl. Anim. Behav. Sci. 2009, 116, 260–265. [Google Scholar] [CrossRef]
- Jarnemo, A. Predation processes: Behavioural interactions between red fox and roe deer during the fawning season. J. Ethol. 2004, 22. [Google Scholar] [CrossRef]
- Saunders, G.; Coman, B.; Kinnear, J.; Braysher, M. Managing Vertebrate Pests: Foxes; Australian Government Publishing Service: Canberra, Australia, 1995. [Google Scholar]
- Wallach, A.D.; Ramp, D.; O’Neill, A.J. Cattle mortality on a predator-friendly station in central Australia. J. Mammal. 2017, 98, 45–52. [Google Scholar] [CrossRef]
- Wallach, A.D.; Ritchie, E.G.; Read, J.; O’Neill, A.J. More than mere numbers: The impact of lethal control on the social stability of a top-order predator. PLoS ONE 2009, 4, e6861. [Google Scholar] [CrossRef]
- Moseby, K.E.; Stott, J.; Crisp, H. Movement patterns of feral predators in an arid environment–implications for control through poison baiting. Wildl. Res. 2009, 36, 422–435. [Google Scholar] [CrossRef]
- Destefano, S.; Schmidt, S.L.; DeVos, C.J. Observations of predator activity at wildlife water developments in southern Arizona. J. Range Manag. 2000, 53, 255–258. [Google Scholar] [CrossRef]
- Mitchell, B.D.; Banks, P.B. Do wild dogs exclude foxes? Evidence for competition from dietary and spatial overlaps. Austral Ecol. 2005, 30, 581–591. [Google Scholar] [CrossRef]
- Coman, B.J. The diet of red foxes (Vulpes vulpes) in Victoria. Aust. J. Zool. 1973, 21, 391–401. [Google Scholar] [CrossRef]
- Glen, A.S.; Dickman, C.R.; Soulé, M.E.; Mackey, B.G. Evaluating the role of the dingo as a trophic regulator in Australian ecosystems. Austral Ecol. 2007, 32, 492–501. [Google Scholar] [CrossRef]
- Torretta, E.; Serafini, M.; Puopolo, F.; Schenone, L. Spatial and temporal adjustments allowing the coexistence among carnivores in Liguria (NW Italy). Acta Ethol. 2016, 19, 123–132. [Google Scholar] [CrossRef]
- Way, J.G.; Szumylo, D.L.M.; Strauss, E.G. An ethogram developed on captive eastern coyotes Canis Latrans. Can. Field-Nat. 2006, 120, 263–288. [Google Scholar] [CrossRef][Green Version]
- MacNulty, D.R.; Mech, L.D.; Smith, D.W. A proposed ethogram of large-carnivore predatory behaviour, exemplified by the wolf. J. Mammal. 2007, 88, 595–605. [Google Scholar] [CrossRef]
- Ghaskadbi, P.; Habib, B.; Qureshi, Q. A whistle in the woods: An ethogram and activity budget for the dhole in central India. J. Mammal. 2016, 97, 1745–1752. [Google Scholar] [CrossRef]
- Biro, P.A.; Stamps, J.A. Are animal personality traits linked to life-history productivity? Trends Ecol. Evol. 2008, 23, 361–368. [Google Scholar] [CrossRef]
- Mellor, D.J.; Beausoleil, N.J. Extending the ‘Five Domains’ model for animal welfare assessment to incorporate positive welfare states. Anim. Welf. 2015, 24, 241–253. [Google Scholar] [CrossRef]
- Friard, O.; Gamba, M. BORIS: A free, versatile open-source event-logging software for video/audio coding and live observations. Methods Ecol. Evol. 2016, 7, 1325–1330. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2010. [Google Scholar]
- Wikenros, C.; Ståhlberg, S.; Sand, H. Feeding under high risk of intraguild predation: Vigilance patterns of two medium-sized generalist predators. J. Mammal. 2014, 95, 862–870. [Google Scholar] [CrossRef]
- Wallach, A.D.; Ripple, W.J.; Carroll, S.P. Novel trophic cascades: Apex predators enable coexistence. Trends Ecol. Evol. 2015, 30, 146–153. [Google Scholar] [CrossRef] [PubMed]
- Swanson, A.; Arnold, T.; Kosmala, M.; Forester, J.; Packer, C. In the absence of a “landscape of fear”: How lions, hyenas, and cheetahs coexist. Ecol. Evol. 2016, 6, 8534–8545. [Google Scholar] [CrossRef] [PubMed]
- Corbett, L.K.; Newsome, A.E. The feeding ecology of the dingo. Oecologia 1987, 74, 215–227. [Google Scholar] [CrossRef] [PubMed]
- Allen, M.L.; Gunther, M.S.; Wilmers, C.C. The scent of your enemy is my friend? The acquisition of large carnivore scent by a smaller carnivore. J. Ethol. 2016, 35, 13–19. [Google Scholar] [CrossRef]
- McIntosh, D. Reproduction and growth of the fox in the Canberra district. Wildl. Res. 1963, 8, 132–141. [Google Scholar] [CrossRef]

| Behavioural Study | Country | Wild/Captive | Foxes Killed | Apex Predators | References |
|---|---|---|---|---|---|
| Anti-predator behaviour | Australia | Wild | Yes | Dingo (Canis dingo) | [19] |
| Canada | Wild | Unstated | Coyote (C.latrans) | [20] | |
| Croatia | Wild | No | Wolf (C.lupus) | [16] | |
| Israel | Wild | No | Golden jackal (C.aureus) | [12] | |
| North America | Wild | Unstated | Coyote | [9] | |
| Poland | Wild | Unstated | Lynx (Lynx lynx) | [13] | |
| Fox kit ethogram | Switzerland | Wild | Unstated | Unstated | [21] |
| Reproduction | North America | Wild | Yes | Unstated | [22] |
| Sociality | England | Wild | Yes | No | [23] |
| England | Captive | N/A | N/A | [24] | |
| North America | Captive | N/A | N/A | [25] | |
| Scent marking | Canada | Wild | Unstated | Unstated | [26] |
| Israel & North America | Both | Unstated | Unstated | [27] | |
| Spain | Wild | Unstated | Unstated | [28] | |
| Spatiotemporal patterns | England | Wild | Yes | No | [29] |
| England | Wild | Yes | No | [30] | |
| England | Wild | Yes | No | [31] | |
| England | Wild | Yes | No | [32] | |
| Italy | Wild | Yes | No | [33] | |
| Italy | Wild | Yes | Unstated | [34] | |
| Italy | Wild | Yes | Unstated | [35] | |
| Japan | Wild | Yes | Unstated | [36] | |
| Feeding behaviours | Switzerland | Captive | N/A | N/A | [37] |
| Sweden | Wild | Unstated | Unstated | [38] |
| Behaviour State | Modifiers | Definition |
|---|---|---|
| Locomotion (S) | A) Walk/Run/Jump/Perch | All spatial movements |
| B) Cautious/Confident | ||
| Sniffing (S) | Cautious/Confident | Exploring area of interest leading with the |
| nose, the head moves up and down with neck | ||
| extended | ||
| Digging (S) | Cautious/Confident | Investigation of ground utilising the front two |
| paws to remove a layer of soil | ||
| Vigilance (S) | Low/High | Examination of the surrounding environment |
| in a state of alert or heightened awareness, | ||
| the head moves directionally, head is moving | ||
| rapidly or focused on an object or location. | ||
| Individuals are positioned low to the ground | ||
| with legs splayed (see Figure 1D) | ||
| Foraging (S) | A) Scavenging/Drinking/Hunting | The act of feeding on carrion, ingesting water |
| from a natural or anthropogenic water source | ||
| or hunting for prey | ||
| B) Cautious/Confident | ||
| C) Alone/Social | ||
| Scent marking (S) | A) Defecation/Raking/Rubbing | The raising of a hind leg or leaning into a |
| squat position in order to deposit urine or | ||
| scats onto a point of interest, using a paw to | ||
| rake the ground or the act of rubbing face, | ||
| paw or tail glands on an object | ||
| B) Alone/Social | ||
| Flight (P) | Startled Jump/Startled Flee | Dramatic and exaggerated responses to |
| environmental or camera born stimuli. Body | ||
| movements are rapid, legs, torso and head | ||
| perform sudden and reckless movements to | ||
| jump or flee away from the location where | ||
| they were startled | ||
| Investigating (S) | Cautious/Confident | The act of surveying the environment. |
| Head moves directionally, can be performed | ||
| stationary or during locomotion | ||
| Frustration (P) | An outburst of frustration manifested by | |
| biting or gnawing on an object in the | ||
| environment | ||
| Head shake (P) | A rapid shaking of the head in an attempt to | |
| remove or dislodge an item of irritation | ||
| Salivating (P) | A display of hunger at the anticipation of | |
| food involving the licking of the outside of | ||
| an individual’s mouth | ||
| Resting (S) | An absence of discernible activity. An | |
| individual laying on the ground with all four | ||
| legs relaxed or sitting down on back end with | ||
| front paws fully extended, supporting the | ||
| individual. Individual is motionless. Head | ||
| may be focused on the ground, sky or on | ||
| nothing in particular but is motionless | ||
| Greeting (S) | Facial/Posterior/Denial | The act of sniffing a conspecific to identify and |
| communicate with the individual | ||
| Play (S) | Jump/Chase | One individual actively solicits a non-agonistic interaction, with random and exaggerated movements |
| Behaviour | Modifiers | Description |
|---|---|---|
| Locomotion | A) Type | Walk: Slow quadrupedal movement Run: Fast quadrupedal movement Jump: Vertical or horizontal jump Perch: The lifting of two paws onto an object in order to investigate a resource or object of interest |
| B) Attitudinal | Confident: Head not focused on anything in particular, head movements are relaxed, ears are relaxed and kept vertical (unless sound is heard, if so, ears will move directionally), little concern over movement. Tail held high, parallel to the ground, level with the back, may have a kink towards the end pointing upwards Cautious: Head moves erratically, ears pricked forward, cautious paw placement with back feet placed firmly with movement only occurring in front feet, stands with legs close together and bent. Tail positioned closer to the back legs, lower than level with the back, with no kink, shoulders are raised | |
| Sniffing | Attitudinal | Confident: Sniffs are long and pronounced, little concern shown for anything apart from the object being sniffed. Head not focused on anything in particular, head movements are relaxed. Ears relaxed and kept vertical (unless sound is heard, if so, ears will move directionally), little concern over movement. Tail held high, parallel to the ground, level with the back, may have a kink towards the end pointing upwards Cautious: Sniffs are short. Head moves erratically, ears pricked forward, cautious paw placement with back feet placed firmly with movement only occurring in front feet stands with legs close together and bent. Tail positioned closer to the back legs, lower than level with the back, with no kink, shoulders are raised |
| Vigilance | Intensity | Low: Head is most commonly focused on a single location, can be represented by low to moderate speed head movements, neck is extended, stands with legs close together shoulders are raised. Can be performed standing or sitting quadrupedally. Tail is position is lower than the level the back High: Head raised and moves erratically and quickly, regularly change focal point, neck is heavily extended, ears are pricked forward, stands with legs close together and shoulders are raised. Can be performed standing or sitting quadrupedally. Tail positioned closer to the back legs, lower than level with the back, with no kink |
| Foraging | A) Type | Scavenging: The investigation of carrion resulting in an individual attempting to or successfully feeding Drinking: The act of utilising either an anthropogenic or natural water resource. Hunting: The act of actively searching for and/or consuming live prey |
| B) Attitudinal | Confident: Individual attempting to consume resource makes slow movements, does not jump back after consuming the resource, consumes resource atop or very nearby resource. Ears perched vertical. Tail held high, parallel to the ground, level with the back, may have a kink towards the end pointing upwards Cautious: Individual attempting to consume resource is extremely jumpy, making erratic movements, ears perched forward, neck as elongated as possible to keep the majority of the body as far from resource as possible. Tail positioned closer to the back legs, lower than level with the back, with no kink, shoulders are raised. If possible, fox may take resource away from the resource to consume (most common during scavenging) | |
| C) Social | Social: The act of foraging with one or more conspecifics | |
| Scent marking | Type | Defecation: The act of squatting or raising a hind leg in order to spray urine or deposit faeces in the environment Raking: The act of dragging or clawing the dirt with paws in order to transfer scent Rubbing: The act of rubbing facial or tail scent glands on objects of interest to transfer scent Social: Scent marking in a group with more than one conspecific |
| Flight | Type | Startled jump: Quick jump backwards, erratic and quick movement. Limbs move in unison, back is arched during the jump, fox will land behind the point it jumped from Startled flee: Commonly initiated through a quick turn in opposite direction the fox was previously facing, then engaging in very fast running away from a specific location. Foxes head and tail will move erratically during the behaviour |
| Investigating | Attitudinal | Confident: Body is relaxed, head movements are slow, ears are vertical, shoulders are lower, tail held high, parallel to the ground, level with the back, may have a kink towards the end pointing upwards Cautious: Head movements are slow, individual is not focused on a single point. Ears move directionally, shoulders are raised, tail is positioned towards the back legs |
| Greeting | Type | Facial: The act of sniffing the face and/or glands of the face of a conspecific Posterior: The act of sniffing the anus, anal glands or genitals of a conspecific Denial: The movement or jumping away from a conspecific after an attempted greeting |
| Play | Type | Jump: Leaping towards or away from a conspecific in a non-agonistic manner, with random and exaggerated movements Chase: The running or walking after or away from a conspecific in a non-agonistic manner, with random and exaggerated movements |
| Cautious | Confident | ||||||
|---|---|---|---|---|---|---|---|
| Resource | Behaviour | No. | Total Time | Average Time | No. | Total Time | Average Time |
| Carcass | Digging | 0 | N/A | 7 | 10.5 | 1.5 | |
| Foraging | 8 | 80.3 | 10.0 | 44 | 386.6 | 8.8 | |
| Investigating | 1 | 8.0 | 8.0 | 23 | 112.7 | 4.9 | |
| Locomotion | 8 | 66.4 | 8.3 | 60 | 191.8 | 3.2 | |
| Scent marking | 0 | N/A | 13 | 16.2 | 1.2 | ||
| Sniffing | 6 | 7.0 | 1.2 | 51 | 218.0 | 4.3 | |
| Vigilance | 30 | 108.3 | 3.6 | 0 | N/A | ||
| Warren | Foraging | 0 | N/A | 9 | 65.2 | 7.2 | |
| Investigating | 18 | 98.2 | 5.4 | 34 | 183.7 | 5.4 | |
| Locomotion | 26 | 89.3 | 3.4 | 67 | 160.2 | 2.4 | |
| Scent marking | 0 | N/A | 2 | 2.1 | 1.0 | ||
| Sniffing | 18 | 54.7 | 3.0 | 32 | 93.2 | 2.9 | |
| Vigilance | 28 | 101.7 | 3.6 | 0 | N/A | ||
| Water | Digging | 0 | N/A | 2 | 0 | 0 | |
| Foraging | 5 | 47.5 | 9.5 | 33 | 362.3 | 11.0 | |
| Investigating | 15 | 48.6 | 3.2 | 10 | 46.5 | 4.7 | |
| Locomotion | 42 | 131.1 | 3.1 | 99 | 316.0 | 3.2 | |
| Scent marking | 0 | N/A | 16 | 25.0 | 1.6 | ||
| Sniffing | 15 | 59.3 | 4.0 | 41 | 145.0 | 3.5 | |
| Vigilance | 55 | 236.7 | 4.3 | 0 | N/A | ||
| Total | 275 | 1137.1 | 4.1 | 543 | 2335.1 | 4.3 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wooster, E.; Wallach, A.D.; Ramp, D. The Wily and Courageous Red Fox: Behavioural Analysis of a Mesopredator at Resource Points Shared by an Apex Predator. Animals 2019, 9, 907. https://doi.org/10.3390/ani9110907
Wooster E, Wallach AD, Ramp D. The Wily and Courageous Red Fox: Behavioural Analysis of a Mesopredator at Resource Points Shared by an Apex Predator. Animals. 2019; 9(11):907. https://doi.org/10.3390/ani9110907
Chicago/Turabian StyleWooster, Eamonn, Arian D. Wallach, and Daniel Ramp. 2019. "The Wily and Courageous Red Fox: Behavioural Analysis of a Mesopredator at Resource Points Shared by an Apex Predator" Animals 9, no. 11: 907. https://doi.org/10.3390/ani9110907
APA StyleWooster, E., Wallach, A. D., & Ramp, D. (2019). The Wily and Courageous Red Fox: Behavioural Analysis of a Mesopredator at Resource Points Shared by an Apex Predator. Animals, 9(11), 907. https://doi.org/10.3390/ani9110907





