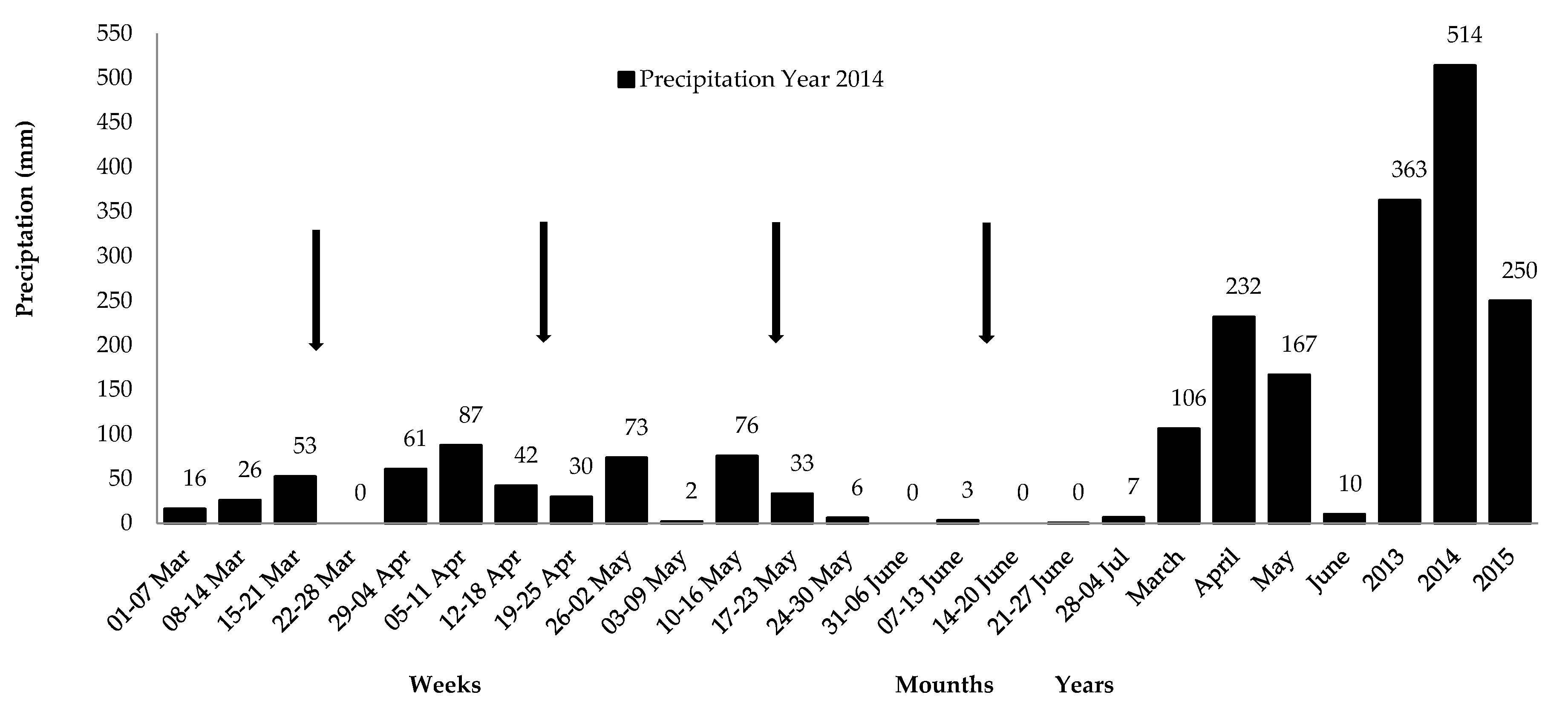
3. Results
The herbaceous stratum was composed of 28% of species from legumes and 72% of species from grasses, predominantly from
Euphorbiaceae, Poaceae, and
Fabaceae families. The pasture area at the beginning of the study showed availability of total DM from the herbaceous stratum of 1897 kg hectare
–1, favored by adequate rainfall average in the first months of the rainy season (
Table 1;
Figure 1).
The most frequent herbaceous species in the area were
Arachis dardani Krapov., & W.C. Greg.,
Oxalis corniculata L.,
Hyptis suaveolens,
Alternanthera tenella colla,
Acalypha communis,
Aspilia martii Baker,
Centrosema Pascuorum Mart. Ex Benth.,
Alternanthera brasiliana (L.) Kuntze),
Stylosanthes humilis,
Merremia aegyptia,
Commelina diffusa,
Digitaria Sanguinalis (L.). Scop,
Wissadula rostrata, in which CP levels of legumes ranged from 125 to 295 g kg
−1 of CP (
Table 2 and
Table 3).
To better characterize the pasture, the dietary nutrient composition of the ruminal extrusa samples were determined in different months of the experimental period (
Table 4). Variations were evidenced as to the composition of the ruminal extrusa sample over the months for CP (192–131 g kg
−1), DM digestibility (537–441 g kg
−1) and OM digestibility (468 and 359 g kg
−1) in the months of March and June, respectively (
Table 4).
There was no effect for treatments and treatment x period interaction as to OM intake (OMI), CP intake (CPI), and fibrous fractions NDF intake (NDFI), ADF intake (ADFI), Cellulose intake (CELI) in g day
−1 and g/kgLW
0.75 (
p > 0.05;
Table 5).
For OMI in the different periods, greater intakes were obtained at the beginning of the rainy season (March) compared to the other months. OMI was 23.9% greater in March compared to June. For CPI, there was a monthly reduction in intake, with a greater difference between the first and the last months of the studied period, being 54.5% lower in June compared to March (
p < 0.05;
Table 5).
The intake of fibrous fractions over the periods was greater in March (
p < 0.05;
Table 5). In June, intake was lower than in the initial period (March), with a reduction in NDFI (34.8%), ADFI (33.3%), and CELI (39.4%). At the same time, there was a pattern of intake reduction for the other periods (April and May) in relation to the initial period, which was, however, more evident in the month of June. A greater organic matter digestibility (OMD) was found for PG. Concerning ADFD, there were greater coefficients for CT and PG (
p < 0.05;
Table 6). Considering the digestibility coefficients in the periods, values were greater in March, at the same time, with a reduction in the digestibility of CP (57.0%) and fibrous fractions (NDF, 39.7%; ADF, 36.4%; CEL, 46.5%) in June (
p < 0.05;
Table 6).
The intake partition assessment took into account key species, recognized with greater contribution to sheep’s diet during the rainy season. The main key species established were subdivided into grasses:
Cyperus uniciualatus Schrad. ex Ness,
Cynodon sp.,
Digitaria Sanguinalis (L.). Scop; herbaceous legumes:
Alternanthera brasiliana (L.) Kuntze,
Alternathera tenella Colla,
Amaranthus blitum,
Arachis dardani,
Aspilia martii Baker,
Borreria verticillata,
Centrosema pascuorum Mart. Ex Benth.,
Commelina diffusa,
Delilia biflora (L.) Kuntze,
Oxalis corniculata L.,
Sesuvium portulacastrum,
Stylosanthes humilis,
Wissadula rostrata); and some arboreal legumes species:
Auxemma oncocalix,
Mimosa caesalpinifolia and Zizyphus joazeiro, totaling 19 species (
Table 7).
For intake partition, there was no effect of treatments (
p > 0.05) for species selection. Over the periods, there were no major changes in the proportion of species ingested during the experiment (
p < 0.05;
Table 7). Overall, the results indicate that the ingestion for each species was variable with the months, but between species the behavior was similar. Although floristic composition in each month was not determined, there seems to be, by comparison among species, little variation in intake by the animals. Greater intakes were observed in April compared to May, except for
Zizyphus joazeiro intake, which was greater in March (
p < 0.05;
Table 7).
4. Discussion
Evaluating goats supplemented in the finishing phase on
Caatinga-native pasture, [
35] observed correlations in DM and CP concentration during the rainy season, in May and June, and, at the end, in July, leading to a reduction in the levels of these fractions. The values obtained in the current research seem adequate to meet the protein requirements of animals raised on this type of pasture, emphasizing that one should observe how much of this protein is degradable in the rumen for utilization by microorganisms.
Pfister and Malechek [
3] reported a gradual decline in CP levels of diets selected by sheep and goats in the rainy season from 180 g kg
−1 of CP in May to 120 g kg
−1 of CP in December in
Caatinga pasture. Still, in said study, at the beginning of the rainy season, CP levels were close to 250 g kg
−1, then dropped (170 g kg
−1 CP) in the middle of the rainy season (April).
Free-grazing ruminants are selective as to what to eat and, in general, select the diet with better quality, i.e., greater digestibility and protein levels, and fewer secondary compounds than the average vegetal biomass supplied [
12]. Thus, even in the rainy season with a greater supply of forage mass constituted by varied amount of species, changes naturally occur in the proportion and quality of dietary constituents consumed by grazing sheep over this period.
The NRC (2007) [
12] establishes for live-weight lambs similar to those of the current study an intake of 64.5 g of DM/kgLW
0.75. Considering the average OM value (810 g kg
−1) of the selected diet in the different periods, and that, from this total, the OM requirement would be 52.2 g/kgLW
0.75, a deficit was verified in this intake only at the end of the rainy season (June). For CPI, the recommendation is 11.7 g CP/kgLW
0.75. Not considering any supplementation, and as verified by the CPI and CP percent in ruminal extrusa samples observed in the current study, CP requirements were only met in March.
Lower ingestion in the final periods is related to changes in grazing behavior, affected by the greater rainfall concentration in April and May and to the pasture’s lower nutritional availability and quality in June, notably IVOMD (
Figure 1;
Table 4). In addition, changes in intake are caused by grazing pressure, since the animals remained in the area during the entire finishing phase, but also by a lower availability of species that the animals prefer, and the poor quality of the diet ingested [
26], according to our study in June compared to March.
In a study evaluating the effect of grain supplementation on intake and digestibility of pasture and diets by goats in
Caatinga pasture, in the rainy season, total OMI was observed for non-supplemented and supplemented animals (0.6% LW, value close to that of our study) of 325 and 377 g day
−1, and 50.5 and 54.4 g/kgLW
0.75, respectively [
2].
Studies have indicated that CP intake by sheep in
Caatinga-native pasture during the rainy season was not a limiting factor to meet the animals’ demand [
24,
25]. However, this aspect only considers the pasture’s crude protein levels, and it is important to quantify how much of this fraction is available for utilization by microorganisms. This fact indicates that the utilization of protein fractions correlates with the availability, or even with factors associated with these proteins, such as secondary compounds, such as total tannins and lignin, which affect the degradation of protein constituents in the rumen [
34]. This condition, if not taken into account, impairs the indication of an adequate utilization of
Caatinga pasture, showing the need for strategic supplementations to adjust nutrient intake and maximize performance.
Some of the species that were collected in the first two months of the current study had adequate CP levels, with
Digitaria Sanguinalis (L.). Scop standing out in the grass group, and
Alternathera tenella Colla,
Commelina difusa,
Borreria verticillata,
Stylosanthes humilis,
Wissadula rostrata, and
Oxalis corniculata L. standing out among the legumes, but also with elevated lignin levels (
Table 4). During the rainy season months there are changes in the chemical composition of plants, with increased formation of CP-lignin bonds, which may lead to inefficient utilization of the diet’s CP by the animal (in the rumen) due to reduced digestibility over these months (
Table 4).
Changes in the botanical composition of the diet in this period derive from a lower availability and quality of herbaceous fractions, as well as edible portions of shrubs and trees. However, part of the diet at the end of the rainy season can be composed of leaves of deciduous species, due to a decline in available biomass [
3]. Although usually not considered as part of forage in traditional plant inventories for grazing use, litterfall, made up by leaves of deciduous trees (such as
Auxemma oncocalix,
Mimosa caesalpinifolia,
Zizyphus joazeiro), can be an important dietary component of grazing sheep, mainly at the end of the rainy season, extending to the dry season [
5]. Probably, these conditions and the greater presence of total tannins (14.8 g kg
−1) at the end of the rainy season, in June (
Table 4), favored reductions in OMI and CPI, and their consequent digestibility, as well as in fibrous fractions.
This research found no major changes in NDF levels obtained from the pasture during the periods, with a mean value of 637 g kg
−1, but there was a reduction in IVOMD by 23.3% when the rain began, compared to April, May, and June, resulting in lower intake during these periods. Intake is inversely related to NDF levels in diets with protein values of 60–80 g kg
−1 and NDF greater than 600 g kg
−1 [
36,
37], since there is correlation between NDF and volume and/or energy density of foods. On the other hand, intake by grazing animals is also influenced by the digestibility of the diet ingested [
36]. The supply of zinc sulfate and propylene glycol did not increase the intake and the efficiency of use of dietary nutrients in pasture. Greater OM and ADF digestibility for PG when compared to Zn may be favored by the low energy input from PG to microorganisms, providing conditions to improve fiber utilization in the rumen.
Based in our findings, the greatest intakes of dietary nutrients in the rainy season in the native pasture of the semi-arid region are a consequence of the greater amount and quality of pastures in the beginning and middle of the rainy season. Due to the ephemerality of some species, there are changes in the constitution of pasture canopies and succession by other species. Native pasture areas with adequate stocking rates allow for intake selectivity, making it possible to meet the requirements of sheep, especially as to CP, with emphasis on the availability aspects of this fraction.
The interaction of some factors may explain differences in intake and digestibility among months in the rainy season. First, there was probably greater selective activity by the animals, with most of the dietary composition containing a blend of herbaceous legumes and fragments of shrub leaves, mainly from May to June, resulting in lower IVOMD. The digestibility of these constituents is moderately low [
3]. Second, the secondary polyphenolic compounds in these species, such as tannins, favored reductions in IVOMD and CPD and ADFD [
3].
Moreover, during the collection periods, even without botanical quantification, a greater participation of fractions of shrub and tree leaves, and of dry
Auxemma oncocalix and
Mimosa caesalpinifolia leaves were observed as of May. These species had greater lignification with levels at 234 g kg
−1 (
Auxemma oncocalix) and 212 g kg
−1 (
Mimosa caesalpinifolia) of lignin, favoring a decline in protein and digestibility levels. This reduction is attributed to the greater participation of stem and leaves of wood plants rich in secondary compounds, which, in turn, increases with the greater participation of shrubs in the diet at the end of the rainy season [
24].
The large contribution of annual plants such as herbaceous legumes occurs due to the greater distribution of the species in the area and, at the same time, they present a longer phenological cycle compared to grasses, which are more ephemeral. Grasses and herbaceous legumes account for about 70% of ruminants’ diet in the rainy season, and, specifically, in sheep’ diet, the participation of grasses and broadleaf herbs constitutes 85.9% [
1]. Considering the participation of each plant in the diet composition regarding intake partition by sheep in the four periods, and relating it to chemical composition (e.g., CP and NDF), herbaceous legume species contributed, on average, 73.4% of CPI and 61.7% of total NDFI in the ingested diet. This aspect indicates that when there is availability of herbaceous stratum composed of a larger fraction of legumes, resulting in a greater condition to meet requirements, e.g., CP, due to the high levels of this nutrient that are contained in these species (125–295 g kg
−1 of CP). As for concepts of availability and quality in native pasture for pastoral purposes, protein does not seem to be the limiting nutrient, especially in the rainy season, if these aspects are met.
In the current study, even with the adequate protein concentration in the species, CPI levels stood below 11.7 g/kgLW
0.75 during the months of April, May, and June [
12]. In addition, the low digestibility values during these months also affected CPI. Probably the increase of total tannins content in the selected diet during these months may have contributed for these reduction. In semiarid regions native pastures CP does not seem to be a limiting factor, but rather, the energy concentration, which negatively affects nutrient intake and utilization [
38].
The composition of the diet obtained was influenced by the month of collection, since there is variation in dietary composition, which is directly related to forage availability throughout the year and regulated by rainfall, which leads to the full development of plants in different seasons, being greater in the rainy season [
6]. In this period, sheep and goats selected diets containing herbaceous legumes, sprouts and leaves of trees and shrubs [
3], a situation similar to our study. Araújo (2015) [
19] determined intake partition by sheep during the rainy season (April) on
Caatinga-native pasture through micro-histological analyses of feces, and, according to intake level, for preferred and desirable species, the following species were observed:
Amaranthus blitum,
Alternanthera tenella Colla,
Alternanthera brasiliana (L.) Kuntze,
Auxemma oncoalyx,
Mimosa caesalpiniaefolia,
Libidibia ferrea,
Ipomoea sp.,
Sesuvium portulacastrum,
Arachis sp.,
Centrosema sp.,
Stylosanthes humilis,
Cyperus uniciualatus Schrad. ex Ness,
Cynodon dactilon,
Herissantia tiubae, K.Schum. Brizicky,
Melochia corchorifolia L.,
Melochia pyramidata L.,
Wissadula rostrata, and
Mimosa tenuiflora, accounting for 52.6% of the key species that represent the sheep’s diet in this research.
The proportion of species in the diet selected by the animals has different proportions of pasture composition [
6]. Variations in the dietary botanical composition of sheep are related to the eating strategies in native pasture areas, ability to select the diet in a heterogeneous pasture area, and, at the same time, with a large number of species, combined with the ability to select in a condition of adequate forage availability [
26].
The intake obtained from the sum of the intake partition was on average 552 g DM, with adequate partition estimation verified by using LIPE
® and KL. However, it is worth highlighting that results were adequate and in line with other research findings when key species were used, adjusting them to the model through the double marker [
18,
27].