Pumpkin Waste as Livestock Feed: Impact on Nutrition and Animal Health and on Quality of Meat, Milk, and Egg
Simple Summary
Abstract
1. Introduction
2. Human Population and Food Demand
3. Food of Animal Origin and Its Impact on Human Health
3.1. Meat
3.2. Milk
3.3. Egg
4. Plant Residues in Animal Feed
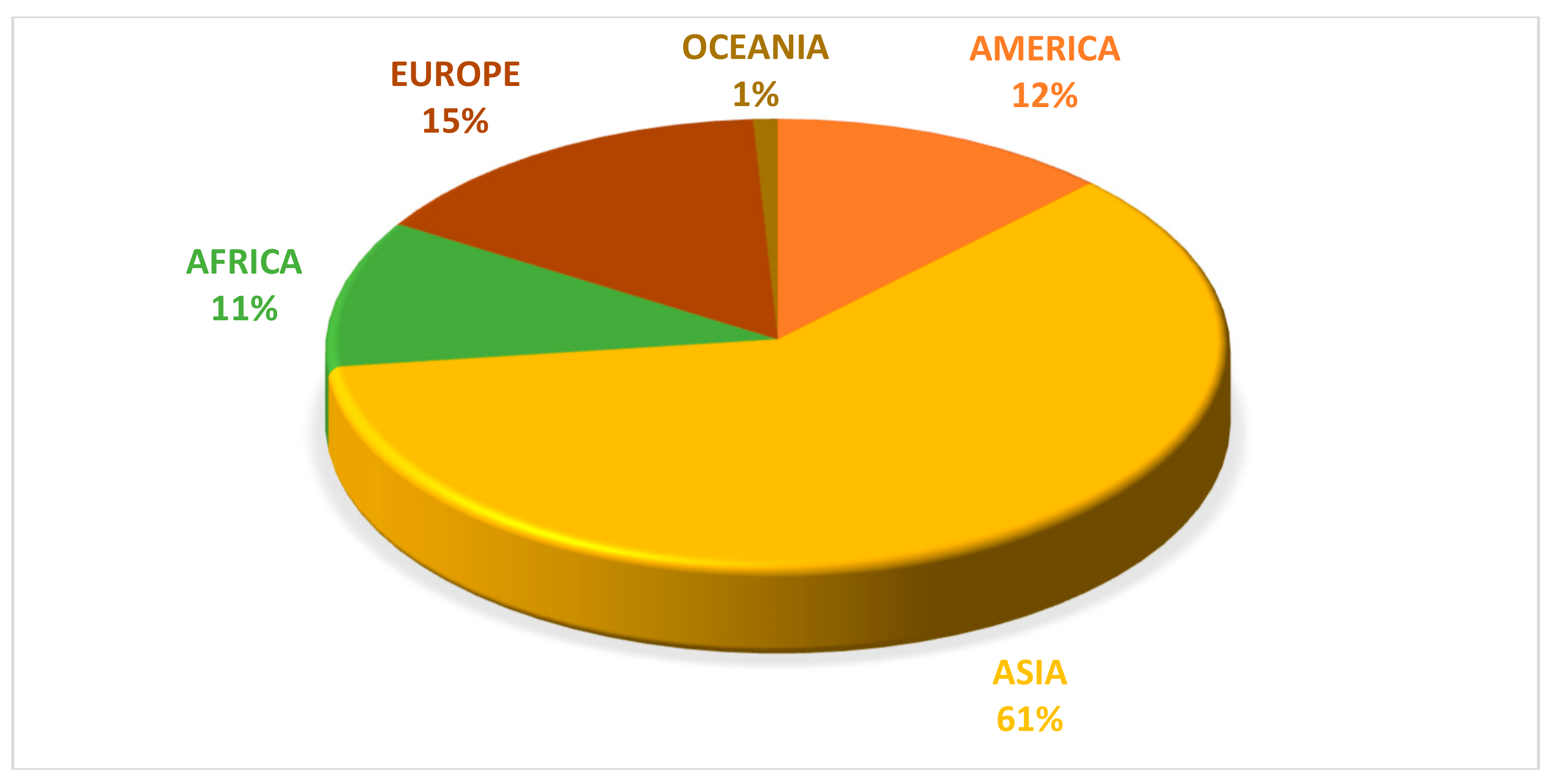
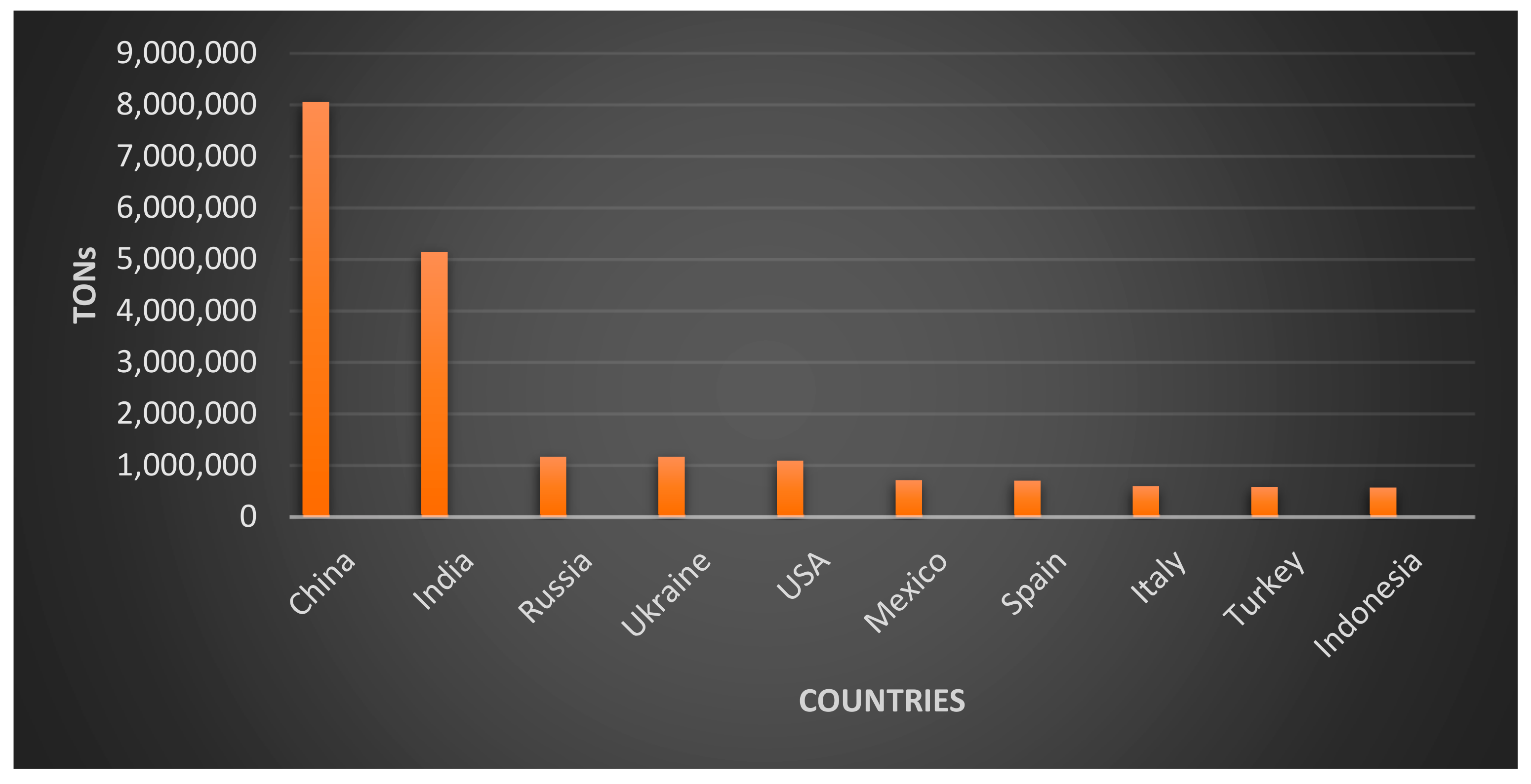
5. World Overview of Pumpkin Production
6. The Genus Cucurbita
7. Nutritional and Phytochemical Composition of Pumpkins
8. Impact on Livestock Nutrition and Productivity
9. Effect on the Composition of Meat, Milk, and Egg
10. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- ONU. Población. Naciones Unidas. 2019. Available online: https://www.un.org/es/sections/issues-depth/population/index.html (accessed on 13 August 2019).
- OCDE. OCDE-FAO Perspectivas Agrícolas 2018–2027; OCDE-Publishing: Paris, France, 2018. [Google Scholar] [CrossRef]
- Gerber, P.J.; Steinfeld, H.; Henderson, B.; Mottet, A.; Opio, C.; Dijkman, J.; Falcucci, A.; Tempio, G. Tackling Climate Change through Livestock—A Global Assessment of Emissions and Mitigation Opportunities; Food and Agriculture Organization of the United Nations (FAO): Rome, Italy, 2013; pp. 15–40. [Google Scholar]
- Tilman, D.; Clark, M. Global diets link environmental sustainability and human health. Nature 2014, 515, 518–522. [Google Scholar] [CrossRef] [PubMed]
- Williams, A.C.; Hill, L.J. Meat and nicotinamide: A causal role in human evolution, history, and demographics. Int. J. Tryptophan Res. 2017, 10, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Wadhwa, M.; Bakshi, M.P.S. Utilization of Fruit and Vegetable Wastes as Livestock Feed and as Substrates for Generation of Other Value-Added Products; Food and Agriculture Organization of the United Nations (FAO): Rome, Italy, 2013; pp. 1–46. [Google Scholar]
- Salami, S.; Luciano, G.; O’Grady, M.; Biondi, L.; Newbold, C.; Kerry, J.; Priolo, A. Sustainability of feeding plant by-products: A review of the implications for ruminant meat production. Anim. Feed. Sci. Technol. 2019, 251, 37–55. [Google Scholar] [CrossRef]
- Kim Young, M.; Kim Jin, E.; Kim, M.; Choi, C.; Lee, B. Comparison of the chemical compositions and nutritive values of various pumpkin (Cucurbitaceae) Species and Parts. Nutr. Res. Pract. 2012, 6, 21–27. [Google Scholar] [CrossRef]
- Perez Gutiérrez, R.M. Review of Cucurbita Pepo (Pumpkin) Its Phytochemistry and Pharmacology. Med. Chem. (Los Angeles) 2016, 6, 12–21. [Google Scholar] [CrossRef]
- Kulczyński, B.; Gramza-Michałowska, A. The profile of secondary metabolites and other bioactive compounds in Cucurbita pepo L. and Cucurbita moschata pumpkin cultivars. Molecules 2019, 24, 2945. [Google Scholar] [CrossRef]
- Yadav, M.; Jain, S.; Tomar, R.; Prasad, G.B.K.S.; Yadav, H. Medicinal and biological potential of pumpkin: An updated review. Nutr. Res. Rev. 2010, 23, 184–190. [Google Scholar] [CrossRef]
- Badr, S.E.A.; Shaaban, M.; Elkholy, Y.M.; Helal, M.H.; Hamza, A.S.; Masoud, M.S.; El Safty, M.M. Chemical composition and biological activity of ripe pumpkin fruits (Cucurbita pepo L.) cultivated in egyptian habitats. Nat. Prod. Res. 2011, 25, 1524–1539. [Google Scholar] [CrossRef]
- Achilonu, M.C.; Nwafor, I.C.; Umesiobi, D.O.; Sedibe, M.M. Biochemical Proximates of Pumpkin (Cucurbitaeae Spp.) and Their Beneficial Effects on the General Well-Being of Poultry Species. J. Anim. Physiol. Anim. Nutr. (Berlin) 2018, 102, 5–16. [Google Scholar] [CrossRef]
- FAO. El Futuro de La Alimentación y La Agricultura Tendencias y Desafíos; Food and Agriculture Organization of the United Nations (FAO): Rome, Italy, 2017; pp. 15–40. [Google Scholar]
- Spitsberg, V.L. Invited Review: Bovine milk fat globule membrane as a potential nutraceutical. J. Dairy Sci. 2005, 88, 2289–2294. [Google Scholar] [CrossRef]
- Pereira, P.C. Milk nutritional composition and its role in human health. Nutrition 2014, 30, 619–627. [Google Scholar] [CrossRef] [PubMed]
- Fricker, R.A.; Green, E.L.; Jenkins, S.I.; Griffin, S.M. The influence of nicotinamide on health and disease in the Central Nervous System. Int. J. Tryptophan Res. 2018, 11, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Kailasapathy, K. Chemical Composition, Physical, and Functional Properties of Milk and Milk Ingredients. In Dairy Processing and Quality Assurance, 2nd ed.; Chandan, R., Kilara, A., Shah, N., Eds.; John Wiley & Sons: Hoboken, NJ, USA, 2016; pp. 77–105. [Google Scholar]
- Milke-García, M.P. La importancia de la leche y los lácteos en la dieta. In El Libro Blanco de La Leche; CANILEC: Mexico City, Mexico, 2011; pp. 103–113. [Google Scholar]
- Haug, A.; Høstmark, A.T.; Harstad, O.M. Bovine milk in human nutrition—A Review. Lipids Health Dis. 2007, 6, 25. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, T.C.; Wallace, R.J.; Moate, P.J.; Mosley, E.E. Board-Invited Review: Recent advances in biohydrogenation of unsaturated fatty acids within the rumen microbial ecosystem. J. Anim. Sci. 2008, 86, 397–412. [Google Scholar] [CrossRef] [PubMed]
- Kuang, H.; Yang, F.; Zhang, Y.; Wang, T.; Chen, G. The impact of egg nutrient composition and its consumption on cholesterol homeostasis. Cholesterol 2018, 2018, 1–22. [Google Scholar] [CrossRef]
- Fredriksson, S.; Elwinger, K.; Pickova, J. Fatty acid and carotenoid composition of egg yolk as an effect of microalgae addition to feed formula for laying hens. Food Chem 2006, 99, 530–537. [Google Scholar] [CrossRef]
- Cherian, G.; Holsonbake, T.B.; Goeger, M.P. Fatty acid composition and egg components of specialty eggs. Poult. Sci. 2002, 81, 30–33. [Google Scholar] [CrossRef]
- Grobas, S.; Mendez, J.; Lazaro, R.; de Blas, C.; Mateo, G.G. Influence of source and percentage of fat added to diet on performance and fatty acid composition of egg yolks of two strains of laying hens. Poult. Sci. 2001, 80, 1171–1179. [Google Scholar] [CrossRef]
- Leskanich, C.O.; Noble, R.C. Manipulation of the n -3 polyunsaturated fatty acid composition of avian eggs and meat. Worlds Poult. Sci. J. 1997, 53, 155–183. [Google Scholar] [CrossRef]
- Surai, P.; Sparks, N.H. Designer eggs: From improvement of egg composition to functional food. Trends Food Sci. Technol. 2001, 12, 7–16. [Google Scholar] [CrossRef]
- Kovacs-Nolan, J.; Phillips, M.; Mine, Y. Advances in the value of eggs and egg components for human health. J. Agric. Food Chem. 2005, 53, 8421–8431. [Google Scholar] [CrossRef] [PubMed]
- Hartikainen, H.; Mogensen, L.; Svanes, E.; Franke, U. Food waste quantification in primary—The Nordic Countries as a case study. Waste Manag. 2018, 71, 502–511. [Google Scholar] [CrossRef] [PubMed]
- Makkar, H.P.S. Animal nutrition in a 360-degree view and a framework for future R&D Work: Towards sustainable livestock production. Anim. Prod. Sci. 2016, 56, 1561. [Google Scholar] [CrossRef]
- Röös, E.; Bajželj, B.; Smith, P.; Patel, M.; Little, D.; Garnett, T. Greedy or needy? Land use and climate impacts of food in 2050 under different livestock futures. Glob. Environ. Chang. 2017, 47, 1–12. [Google Scholar] [CrossRef]
- Ulloa, J.B.; van Weerd, J.H.; Huisman, E.A.; Verreth, J.A.J. Tropical agricultural residues and their potential uses in fish feeds: The Costa Rican situation. Waste Manag. 2004, 24, 87–97. [Google Scholar] [CrossRef] [PubMed]
- Westendorf, M.L.; Zirkle Pas, E.W.; Gordon, R. Feeding food or table waste to livestock. Prof. Anim. Sci. 1996, 12, 129–137. [Google Scholar] [CrossRef]
- Angulo, J.; Mahecha, L.; Yepes, S.A.; Yepes, A.M.; Bustamante, G.; Jaramillo, H.; Valencia, E.; Villamil, T.; Gallo, J. Nutritional evaluation of fruit and vegetable waste as feedstuff for diets of lactating holstein cows. J. Environ. Manag. 2012, 95, S210–S214. [Google Scholar] [CrossRef]
- Esteban, M.B.; García, A.J.; Ramos, P.; Márquez, M.C. Evaluation of fruit-vegetable and fish wastes as alternative feedstuffs in pig diets. Waste Manag. 2007, 27, 193–200. [Google Scholar] [CrossRef]
- Cook, D.F.; Telfer, D.V.; Lindsey, J.B.; Deyl, R.A. Substrates across horticultural and livestock industries that support the development of stable fly, stomoxys calcitrans (Diptera: Muscidae). Austral Entomol. 2018, 57, 344–348. [Google Scholar] [CrossRef]
- Beausang, C.; Hall, C.; Toma, L. Food waste and losses in primary production: Qualitative insights from horticulture. Resour. Conserv. Recycl. 2017, 126, 177–185. [Google Scholar] [CrossRef]
- Gustavsson, J.; Cederberg, C.; Sonesson, U.; van Otterdijk, R.; Meybeck, A. Pérdidas y Desperdicios de Alimentos en el Mundo; Organización de las Naciones Unidas para la Alimentación y la Agricultura (FAO): Italy, Roma, 2012; pp. 1–17. [Google Scholar]
- Girotto, F.; Alibardi, L.; Cossu, R. Food waste generation and industrial uses: A Review. Waste Manag. 2015, 45, 32–41. [Google Scholar] [CrossRef] [PubMed]
- Acosta-Martínez, A.; Avendaño-Ruiz, B.; Astorga-Ceja, J. Terminal market price relationships of fruits and vegetables from Mexico and US. Rev. Mexicana Cienc. Agric. 2015, 6, 1429–1442. [Google Scholar]
- Valbuena, D.; Tui, S.H.K.; Teufel, N.; Duncan, A.; Abdoulaye, T.; Swain, B.; Mekonnen, K.; Germaine, I. Identifying determinants, pressures and trade-offs of crop residue use in mixed smallholder farms in Sub-Saharan Africa and South Asia. Agric. Syst. 2015, 134, 107–118. [Google Scholar] [CrossRef]
- Hossain, M.E.; Sultana, S.A.; Karim, M.H.; Ahmed, M.I. Vegetable peels: A promising feed resource for livestock. J. Anim. Feed Res. 2015, 5, 33–39. [Google Scholar]
- Salinas-Chavira, J. Potential use of nonconventional silages in ruminant feeding for tropical and subtropical areas. In Advances in Silage Production and Utilization; InTech: Philadelphia, PA, USA, 2016. [Google Scholar] [CrossRef]
- Diaz-Monroy, B.L.; Iglesias, A.E.; Valiño-Cabrera, E.C. Evaluation of bioensilage of broccoli (Brassica oleracea L.) and oats (Avena sativa L.) as supplements for dairy cows. Arch. Latinoam. Prod. Anim. 2014, 22, 21–29. [Google Scholar]
- Caicedo, W.; Vargas, J.C.; Uvidia, H.; Samaniego, E.; Valle, S.; Flores, L.; Moyano, J.; Aguiar, S. Physicochemical, biological and organoleptic indicators in banana silage (Musa sapientum) for pig feeding. Cuba. J. Agric. Sci. 2017, 51, 85–92. [Google Scholar]
- Zivkov-Balos, M.; Jakšic, S.; Jovicin, M. The Determination of the Quality in Cereal Silages Supplemented with Pumpkin and Carrot. In Proceedings of the First International Symposium of Veterinary Medicine, Vrdnik, Serbia, 21–23 May 2015; Petrović, T., Ed.; Scientific Veterinary Institute “Novi Sad”: Novi Sad, Serbia, 2015; pp. 115–123. [Google Scholar]
- Martínez-Fernández, A.; Argamentería, A.; De-La Rosa, B. Manejo de Forrajes Para Ensilar; Servicio Regional de Investigación y Desarrollo Agroalimentaria (SERIDA) del Principado de Asturias: Asturias, España, 2014; pp. 215–247. [Google Scholar]
- Ferreira, M.S.L.; Santos, M.C.P.; Moro, T.M.A.; Basto, G.J.; Andrade, R.M.S.; Gonçalves, É.C.B.A. Formulation and characterization of functional foods based on fruit and vegetable residue flour. J. Food Sci. Technol. 2015, 52, 822–830. [Google Scholar] [CrossRef] [PubMed]
- Angulo, J.; Mahecha, L.; Yepes, S.A.; Yepes, A.M.; Bustamante, G.; Jaramillo, H.; Valencia, E.; Villamil, T.; Gallo, J. Quantitative and nutritional characterization of fruit and vegetable waste from marketplace: A potential use as bovine feedstuff? J. Environ. Manag. 2012, 95, S203–S209. [Google Scholar] [CrossRef] [PubMed]
- Bakshi, M.; Wadhwa, M.; Ps Makkar, H. Waste to worth: Vegetable wastes as animal feed. CAB Reviews 2016, 11. [Google Scholar] [CrossRef]
- FAOSTAT. Cultivos: Calabaza, zapallos, calabaza confitera. Available online: http://www.fao.org/faostat/es/#data/QC/visualize (accessed on 24 June 2019).
- Lira, R.; Caballero, J. Ethnobotany of the Wild Mexican Cucurbitaceae. Econ. Bot. 2002, 56, 380–398. [Google Scholar] [CrossRef]
- OECD. Squashes, pumkins, zucchinis and gourds (Cucurbita species). In Safety Assessment of Transgenic Organisms in the Environment; OECD Consensus Documents, OECD Publishing: Paris, France, 2016; Volume 5, pp. 83–149. [Google Scholar] [CrossRef]
- Ferriol, M.; Picó, B. Pumpkin and winter squash. In Vegetables I Asteraceae, Brassicaceae, Chenopodicaceae, and Cucurbitaceae; Prohens, J., Nuez, F., Eds.; Springer: New York, NY, USA, 2008; pp. 317–350. [Google Scholar]
- Paris, H.S.; Daunay, M.C.; Pitrat, M.; Janick, J. First known image of Cucurbita in Europe, 1503–1508. Ann. Bot. 2006, 98, 41–47. [Google Scholar] [CrossRef] [PubMed]
- Integrated Taxonomy Information System. ITIS Report Cucurbita, L. Available online: https://www.itis.gov/servlet/SingleRpt/SingleRpt?search_topic=TSN&search_value=22365#null (accessed on 13 August 2019).
- Kates, H.R. Pumpkins, Squashes, and Gourds (Cucurbita L.) of North America. In North American Crop Wild Relatives; Greene, L.S., Williams, K.A., Khoury, K.C., Kantar, B.M., Marek, F.L., Eds.; Springer: New York, NY, USA, 2019; Volume 2, pp. 195–224. [Google Scholar] [CrossRef]
- Dorantes-Jiménez, J.; Flota-Bañuelos, C.; Candelaria-Martínez, B.; Ramírez-Mella, M.; Crosby-Galván, M.M. Calabaza chihua (Cucurbita argyrosperma Huber), alternativa para alimentación animal en el trópico. Agroproductividad 2016, 9, 33–37. [Google Scholar]
- Zhang, Y.; Chen, P.; Zhang, Y.; Jin, H.; Zhu, L.; Li, J.; Yao, H. Effects of polysaccharide from pumpkin on biochemical indicator and pancreatic tissue of the diabetic rabbits. Int. J. Biol. Macromol. 2013, 62, 574–581. [Google Scholar] [CrossRef] [PubMed]
- Azevedo-Meleiro, C.H.; Rodriguez-Amaya, D.B. Qualitative and quantitative differences in carotenoid composition among Cucurbita moschata, Cucurbita maxima, and Cucurbita pepo. J. Agric. Food Chem. 2007, 55, 4027–4033. [Google Scholar] [CrossRef] [PubMed]
- Elinge, C.; Muhammad, A.; Atiku, F.; Itodo, A.U.; Peni, I.J.; Sanni, O.M.; Mbongo, A.N. Proximate, mineral and anti-nutrient composition of pumpkin (Cucurbita pepo L) seeds extract. Int. J. Plant Res. 2012, 2, 146–150. [Google Scholar] [CrossRef]
- Zhou, C.L.; Mi, L.; Hu, X.Y.; Zhu, B.H. Evaluation of three pumpkin species: Correlation with physicochemical, antioxidant properties and classification using SPME-GC–MS and E-Nose methods. J. Food Sci. Technol. 2017, 54, 3118. [Google Scholar] [CrossRef] [PubMed]
- Dhiman, A.K.; Sharma, K.D.; Attri, S. Functional constituents and processing of pumpkin: A review. J. Food Sci. Technol. 2009, 46, 411–417. [Google Scholar]
- Zdunić, G.M.; Menković, N.R.; Jadranin, M.B.; Novaković, M.M.; Šavikin, K.P.; Živković, J. Phenolic compounds and carotenoids in pumpkin fruit and related traditional products. Hem. Ind. 2016, 70, 429–433. [Google Scholar] [CrossRef]
- Andjelkovic, M.; Van Camp, J.; Trawka, A.; Verhé, R. Phenolic compounds and some quality parameters of pumpkin seed oil. Eur. J. Lipid Sci. Technol. 2010, 112, 208–217. [Google Scholar] [CrossRef]
- Montesano, D.; Blasi, F.; Simonetti, M.S.; Santini, A.; Cossignani, L. Chemical and nutritional characterization of seed oil from Cucurbita Maxima, L. (Var. Berrettina) pumpkin. Foods (Basel, Switzerland) 2018, 7. [Google Scholar] [CrossRef]
- Gry, J.; Søborg, I.; Andersson, H.C. Cucurbitacins in Plant Food; Nordic Council of Minister: Copenhagen, Denmark, 2006; pp. 15–38. [Google Scholar]
- Dhiman, K.; Gupta, A.; Sharma, D.K.; Gill, N.S.; Goyal, A. A review on the medicinally important plants of the family Cucurbitaceae. Asian J. Clin. Nutr. 2012, 4, 16–26. [Google Scholar] [CrossRef]
- Lans, C.; Turner, N.; Khan, T.; Brauer, G.; Boepple, W. Ethnoveterinary medicines used for ruminants in British Columbia, Canada. J. Ethnobiol. Ethnomed. 2007, 3, 11. [Google Scholar] [CrossRef] [PubMed]
- Lans, C.; Turner, N.; Brauer, G.; Lourenco, G.; Georges, K. Ethnoveterinary medicines used for horses in Trinidad and in British Columbia, Canada. J. Ethnobiol. Ethnomed. 2006, 2, 31. [Google Scholar] [CrossRef] [PubMed]
- Crosby-Galván, M.M.; Espinoza-Velasco, B.; Ramírez-Mella, M. Effect of chihua pumpkin residue (Cucurbita argyrosperma) in ruminal gas production and digestibility in vitro. Pak. J. Zool. 2018, 50, 1–3. [Google Scholar] [CrossRef]
- Halik, G.; Lozicki, A.; Koziorzębska, A.; Dymnicka, M.; Arkuszewska, E. Effect of ensiling pumpkin Cucurbita maxima with the addition of inoculant or without it on chemical composition and quality of silages. Ann. Warsaw Univ. of Life Sci.–SGGW Anim. Sci. 2014, 53, 103–110. [Google Scholar]
- Lozicki, A.; Koziorzebska, A.; Halik, G.; Dymnicka, M.; Arkuszewska, E.; Niemiec, T.; Bogdan, J. Effect of Ensiling Pumpkin (Cucurbita Maxima, D.) with Dried Sugar Beet Pulp on the Content of Bioactive Compounds in Silage and Its Antioxidant Potential. Anim. Feed Sci. Technol. 2015, 206, 108–113. [Google Scholar] [CrossRef]
- Halik, G.; Lozicki, A.; Wilczak, J.; Arkuszewska, E.; Makarski, M. Pumpkin (Cucurbita Maxima D.) Silage as a Feed That Improves Nutritional Properties of Cow’s Milk. J. Agric. Sci. Technol. 2018, 20, 1383–1394. [Google Scholar]
- Razzaghzadeh, S.; Amini-Jabalkandi, J.; Hashemi, A. Effects of different levels of pumpkin (Cucurbita pepo) residue silage replacement with forage part of ration on male buffalo calves fattening performance. Ital. J. Anim. Sci. 2007, 6, 575–577. [Google Scholar] [CrossRef]
- Klir, Ž.; Castro-Montoya, J.M.; Novoselec, J.; Molkentin, J.; Domacinovic, M.; Mioc, B.; Dickhoefer, U.; Antunovic, Z. Influence of pumpkin seed cake and extruded linseed on milk production and milk fatty acid profile in Alpine goats. Animal 2017, 11, 1772–1778. [Google Scholar] [CrossRef]
- Antunovic, Z.; Klir, Ž.; Šperanda, M.; Sičja, V.; Čolović, D.; Mioč, B.; Novoselec, J. Partial replacement of soybean meal with pumpkin seed cake in lamb diets: Effects on carcass traits, haemato-chemical parameters and fatty acids in meat. S. Afr. J. Anim. Sci. 2018, 48, 695–704. [Google Scholar] [CrossRef]
- Martínez, Y.; Yero, O.; Navarro, M.; Hurtado, C.; López, J.; Mejía, L. Effect of squash seed meal (Cucurbita moschata) on broiler performance, sensory meat quality, and blood lipid profile. Rev. Bras. Ciência Avícola 2011, 13, 219–226. [Google Scholar] [CrossRef]
- Zinabu, M.; Meseret, G.; Negassi, A.; Tesfaheywet, Z. Effects of neem (Azadirachta Indica) and pumpkin (Cucurbita maxima) seeds and their combination as feed additive on growth and carcass characteristics of broilers. Livest. Res. Rural Dev. 2019, 31. [Google Scholar]
- Hajati, H.; Hasanabadi, A.; Waldroup, P.W. Effects of dietary supplementation with pumpkin oil (Cucurbita pepo) on performance and blood fat of broiler chickens during finisher period. Am. J. Anim. Vet. Sci. 2011, 6, 40–44. [Google Scholar] [CrossRef]
- Martínez, Y.; Valdivié, M.; Solano, G.; Estarrón, M.; Martínez, O.; Córdova, J. Effect of pumpkin (Cucurbita maxima) seed meal on total cholesterol and fatty acids of laying hen eggs. Cuba. J. Agric. Sci. 2012, 46, 73–78. [Google Scholar]
- Martínez, Y.; Valdivié, M.; Estarrón, M.; Solano, G.; Córdova, J. Serum lipid profile of laying hens fed pumpkin (Cucurbita maxima) seed levels. Cuba. J. Agric. Sci. 2010, 44, 393–399. [Google Scholar]
- Machebe, N.; Ugwu, S.; Mbunwen, N. Intake of some biological seeds and root extracts of plants improves fertility and hatchability of turkey eggs. J. Basic Appl. Sci. 2013, 9. [Google Scholar] [CrossRef]
- Medina-González, R.; Ortiz-Milán, A.; Elias-Iglesias, A.; Álvarez-Villar, V.M.; Brea-Maure, O. Efecto de la calabaza fermentada (Cucurbita pepo) en los parámetros productivos y de salud en cerdos en preceba. Ciencia y Agricultura 2019, 16, 79–91. [Google Scholar] [CrossRef]
- Grabensteiner, E.; Liebhart, D.; Arshad, N.; Hess, M. Antiprotozoal activities determined in vitro and in vivo of certain plant extracts against Histomonas meleagridis, Tetratrichomonas gallinarum and Blastocystis Sp. Parasitol. Res. 2008, 103, 1257–1264. [Google Scholar] [CrossRef]
- Bahramsoltani, R.; Farzaei, M.H.; Abdolghaffari, A.H.; Rahimi, R.; Samadi, N.; Heidari, M.; Esfandyari, M.; Baeeri, M.; Hassanzadeh, G.; Abdollahi, M.; et al. Evaluation of phytochemicals, antioxidant and burn wound healing activities of Cucurbita moschata Duchesne Fruit Peel. Iran. J. Basic Med. Sci. 2017, 20, 798–805. [Google Scholar] [CrossRef]
- Decker, E.A.; Park, Y. Healthier meat products as functional foods. Meat Sci. 2010, 86, 49–55. [Google Scholar] [CrossRef]
- Daley, C.A.; Abbott, A.; Doyle, P.S.; Nader, G.A.; Larson, S. A Review of fatty acid profiles and antioxidant content in grass-fed and grain-fed beef. Nutr. J. 2010, 9, 10. [Google Scholar] [CrossRef] [PubMed]
- Jacobo-Valenzuela, N.; Maróstica-Junior, M.R.; Zazueta-Morales, J.D.J.; Gallegos-Infante, J.A. Physicochemical, technological properties, and health-benefits of Cucurbita moschata Duchense vs. Cehualca: A Review. Food Res. Int. 2011, 44, 2587–2593. [Google Scholar] [CrossRef]
- Jenness, R. Composition of Milk. In Fundamentals of Dairy Chemistry; Springer: Boston, MA, USA, 1988; pp. 1–38. [Google Scholar] [CrossRef]
- Khan, N.A.; Yu, P.; Ali, M.; Cone, J.W.; Hendriks, W.H. Nutritive value of maize silage in relation to dairy cow performance and milk quality. J. Sci. Food Agric. 2015, 95, 238–252. [Google Scholar] [CrossRef] [PubMed]
- Nys, Y. Dietary carotenoids and egg yolk coloration—A review. Arch. Geflügelk 2000, 45–54. [Google Scholar]
- Heying, E.K.; Tanumihardjo, J.P.; Vasic, V.; Cook, M.; Palacios-Rojas, N.; Tanumihardjo, S.A. Biofortified orange maize enhances β-cryptoxanthin concentrations in egg yolks of laying hens better than tangerine peel fortificant. J. Agric. Food Chem. 2014, 62, 11892–11900. [Google Scholar] [CrossRef]
- Liu, C.; Bronson, R.T.; Russell, R.M.; Wang, X.D. β-Cryptoxanthin supplementation prevents cigarette smoke-induced lung inflammation, oxidative damage, and squamous metaplasia in ferrets. Cancer Prev. Res. (Phila.) 2011, 4, 1255–1266. [Google Scholar] [CrossRef]
- Dunne, P.G.; Monahan, F.J.; O’Mara, F.P.; Moloney, A.P. Colour of bovine subcutaneous adipose tissue: A review of contributory factors, associations with carcass and meat quality and its potential utility in authentication of dietary history. Meat Sci. 2009, 81, 28–45. [Google Scholar] [CrossRef]
- Martínez-Aguilar, Y.; Córdova-López, J.; Santana-Pérez, A.; Martínez-Yero, O.; Valdivié-Navarro, M.; Betancur-Hurtado, C. Productivity and egg quality in laying hens fed increasing levels of pumpkin (Cucurbita maxima) seed meal. Tec. Pecu. en Mex. 2012, 3, 65–75. [Google Scholar]
| Kingdom | Plantae |
|---|---|
| Division | Tracheophyta |
| Class | Magnoliopsida |
| Order | Cucurbitales |
| Family | Cucurbitaceae |
| Genus | Cucurbita L. |
| Species | argyrosperma digitata ficifolia foetidissima máxima moschata okeechobeensis palmata pepo |
| Species | ||||
|---|---|---|---|---|
| Chemical Composition | Part | Cucurbita pepo | Cucurbita moschata | Cucurbita maxima |
| Carbohydrates | Peel | 43.76 | 96.29 | 206.78 |
| Pulp | 26.23 | 43.39 | 133.53 | |
| Seed | 122.20 | 140.19 | 129.08 | |
| Protein | Peel | 9.25 | 11.30 | 16.54 |
| Pulp | 2.08 | 3.05 | 11.31 | |
| Seed | 308.83 | 298.11 | 274.85 | |
| Fat | Peel | 4.71 | 6.59 | 8.59 |
| Pulp | 0.55 | 0.89 | 4.20 | |
| Seed | 439.88 | 456.76 | 524.34 | |
| Fiber | Peel | 12.28 | 34.28 | 22.35 |
| Pulp | 3.72 | 7.41 | 10.88 | |
| Seed | 148.42 | 108.51 | 161.54 | |
| Humidity | Peel | 935.98 | 871.86 | 756.79 |
| Pulp | 967.70 | 942.31 | 840.43 | |
| Seed | 74.06 | 51.79 | 27.51 | |
| Calcium | 9.78 |
|---|---|
| Magnesium | 67.41 |
| Sodium | 170.35 |
| Potassium | 237.24 |
| Phosphorus | 47.68 |
| Iron | 3.75 |
| Zinc | 14.14 |
| Magnesium | 0.06 |
| Cobalt | 2.17 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Valdez-Arjona, L.P.; Ramírez-Mella, M. Pumpkin Waste as Livestock Feed: Impact on Nutrition and Animal Health and on Quality of Meat, Milk, and Egg. Animals 2019, 9, 769. https://doi.org/10.3390/ani9100769
Valdez-Arjona LP, Ramírez-Mella M. Pumpkin Waste as Livestock Feed: Impact on Nutrition and Animal Health and on Quality of Meat, Milk, and Egg. Animals. 2019; 9(10):769. https://doi.org/10.3390/ani9100769
Chicago/Turabian StyleValdez-Arjona, Laura Patricia, and Mónica Ramírez-Mella. 2019. "Pumpkin Waste as Livestock Feed: Impact on Nutrition and Animal Health and on Quality of Meat, Milk, and Egg" Animals 9, no. 10: 769. https://doi.org/10.3390/ani9100769
APA StyleValdez-Arjona, L. P., & Ramírez-Mella, M. (2019). Pumpkin Waste as Livestock Feed: Impact on Nutrition and Animal Health and on Quality of Meat, Milk, and Egg. Animals, 9(10), 769. https://doi.org/10.3390/ani9100769





