Investigating Home Range, Movement Pattern, and Habitat Selection of Bar-headed Geese during Breeding Season at Qinghai Lake, China
Simple Summary
Abstract
1. Introduction
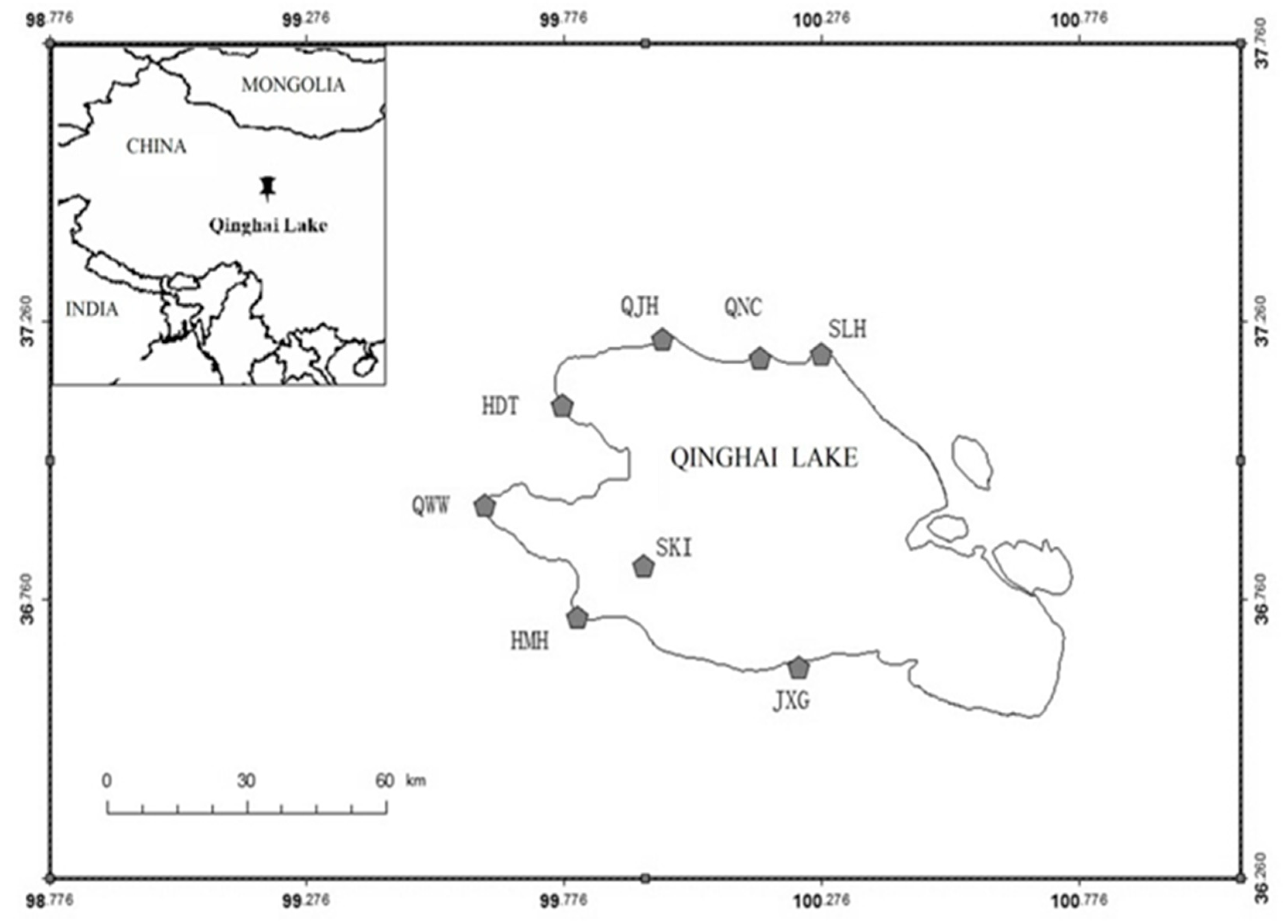
2. Study Area
3. Methods
3.1. Capture and Marking
3.2. Data Analysis
3.2.1. Home Range and Movement Pattern
3.2.2. Habitat Selection
4. Results
4.1. Marking and Telemetry
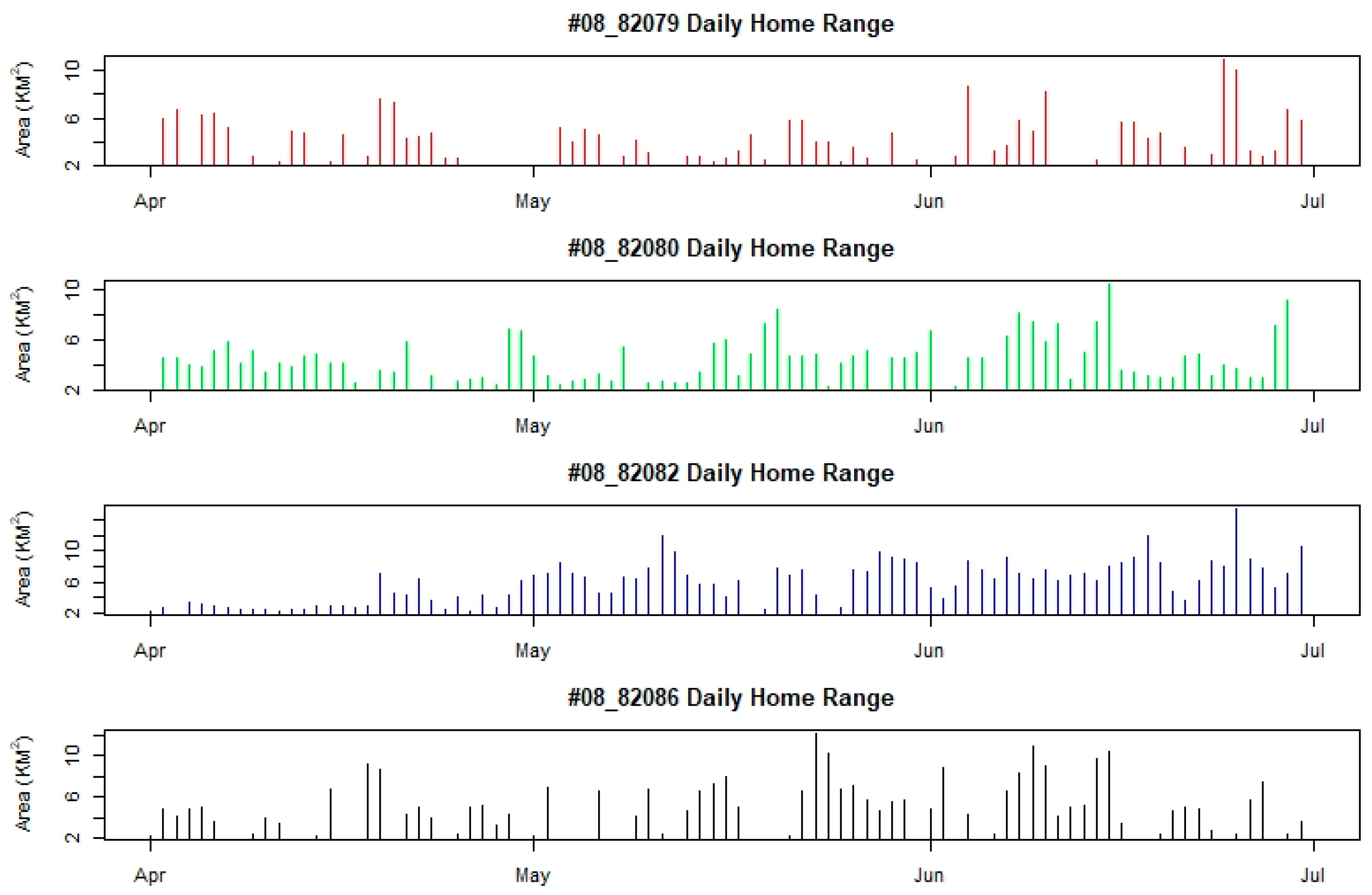
4.2. Home Range and Movement Pattern
4.3. Habitat Selection
5. Discussion
5.1. Home Range and Movement Pattern
5.2. Habitat Selection
5.3. Conservation Implications
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- BirdLife International. Species Factsheet: Anser indicus. 2016. Available online: http://www.birdlife.org/datazone/speciesfactsheet.php?id=379 (accessed on 12 September 2016).
- Dave, K.N. Birds in Sanskrit Literature: With 107 Bird Illustrations; Motilal Banarsidass Publishers: New Delhi, India, 2005. [Google Scholar]
- Hawkes, L.; Balachandran, S.; Batbayar, N.; Butler, P.; Chua, B.; Douglas, D.; Frappell, P.; Hou, Y.; Milsom, W.; Newman, S.; et al. The paradox of extreme high-altitude migration in bar-headed geese Anser indicus. Proc Biol. Sci. 2013, 280, 20122114. [Google Scholar] [CrossRef] [PubMed]
- Hawkes, L.A.; Balachandran, S.; Batbayar, N.; Butler, P.J.; Frappell, P.B.; Milsom, W.K.; Tseveenmyadag, N.; Newman, S.H.; Scott, G.R.; Sathiyaselvam, P.; et al. The trans-Himalayan flights of bar-headed geese (Anser indicus). Proc. Natl. Acad. Sci. USA 2011, 108, 9516–9519. [Google Scholar] [CrossRef] [PubMed]
- Namgail, T.; Takekawa, J.Y.; Balachandran, S.; Palm, E.C.; Mundkur, T.; Vélez, V.M.; Prosser, D.J.; Newman, S.H. Himalayan Thoroughfare: Migratory Routes of Ducks over the Rooftop of the World; Cambridge University Press: Cambridge, MA, USA, 2017. [Google Scholar]
- Prosser, D.J.; Cui, P.; Takekawa, J.Y.; Tang, M.; Hou, Y.; Collins, B.M.; Yan, B.; Hill, N.J.; Li, T.; Li, Y.; et al. Wild bird migration across the Qinghai-Tibetan plateau: A transmission route for highly pathogenic h5n1. PLoS ONE 2011, 6, e17622. [Google Scholar] [CrossRef] [PubMed]
- Takekawa, J.Y.; Newman, S.H.; Xiao, X.; Prosser, D.J.; Spragens, K.A.; Palm, E.C.; Yan, B.; Li, T.; Lei, F.; Zhao, D.; et al. Migration of waterfowl in the east Asian flyway and spatial relationship to hpai h5n1 outbreaks. Avian Dis. 2010, 54, 466–476. [Google Scholar] [CrossRef] [PubMed]
- Takekawa, J.; Heath, S.R.; Douglas, D.C.; Perry, W.M.; Javed, S.; Newman, S.H.; Suwal, R.N.; Rahmani, A.R.; Choudhury, B.C.; Prosser, D.J.; et al. Geographic variation in bar-headed geese Anser indicus: Connectivity of wintering areas and breeding grounds across a broad front. Wildfowl 2013, 59, 100–123. [Google Scholar]
- Bishop, C.M.; Spivey, R.J.; Hawkes, L.A.; Batbayar, N.; Chua, B.; Frappell, P.B.; Milsom, W.K.; Natsagdorj, T.; Newman, S.H.; Scott, G.R.; et al. The roller coaster flight strategy of bar-headed geese conserves energy during Himalayan migrations. Science 2015, 347, 250–254. [Google Scholar] [CrossRef] [PubMed]
- Miyabayashi, Y.; Mundkur, T. Atlas of Key Sites for Anatidae in the East Asian Flyway; Wetlands International: Tokyo, Japan, 1999. [Google Scholar]
- Liu, X.; Chen, B. Climatic warming in the Tibetan plateau during recent decades. Int. J. Climatol. 2000, 20, 1729–1742. [Google Scholar] [CrossRef]
- Liu, D.; Zhang, G.; Li, F.; Ma, T.; Lu, J.; Qian, F. A revised species population estimate for the bar-headed goose (Anser indicus). Avian Res. 2017, 8. [Google Scholar] [CrossRef]
- Abraham, K.F.; Jefferies, R.L.; Alisauskas, R.T. The dynamics of landscape change and snow geese in mid-continent North America. Glob. Chang. Biol. 2005, 11, 841–855. [Google Scholar] [CrossRef]
- Cui, P.; Hou, Y.; Tang, M.; Zhang, H.; Zhou, Y.; Yin, Z.; Li, T.; Guo, S.; Xing, Z.; He, Y.; et al. Movement patterns of bar-headed geese Anser indicus during breeding and post-breeding periods at Qinghai Lake, China. J. Ornithol. 2011, 152, 83–92. [Google Scholar] [CrossRef]
- Liu, Y.; Zha, Y.; Gao, J.; Ni, S. Assessment of grassland degradation near Lake Qinghai, west China, using landsat tm and in situ reflectance spectra data. Int. J. Remote Sens. 2004, 25, 4177–4189. [Google Scholar] [CrossRef]
- Rhode, D.; Haizhou, M.; Madsen, D.B.; Brantingham, P.J.; Forman, S.L.; Olsen, J.W. Paleoenvironmental and archaeological investigations at Qinghai Lake, western China: Geomorphic and chronometric evidence of lake level history. Quat. Int. 2010, 218, 29–44. [Google Scholar] [CrossRef]
- Xu, H.; Hou, Z.; Ai, L.; Tan, L. Precipitation at Lake Qinghai, ne Qinghai–Tibet plateau, and its relation to Asian summer monsoons on decadal/interdecadal scales during the past 500 years. Palaeogeogr. Palaeoclim. Palaeoecol. 2007, 254, 541–549. [Google Scholar] [CrossRef]
- Li, L. Survey results and new records of waterfowl at Qinghai Lake. China Crane News 2001, 5, 36–37. [Google Scholar]
- Horne, J.S.; Garton, E.O.; Krone, S.M.; Lewis, J.S. Analyzing animal movements using Brownian bridges. Ecology 2007, 88, 2354–2363. [Google Scholar] [CrossRef] [PubMed]
- Calenge, C. The package adehabitat for the R software: A tool for the analysis of space and habitat use by animals. Ecol. Model. 2006, 197, 516–519. [Google Scholar] [CrossRef]
- Bullard, F. Estimating the Home Range of an Animal: A Brownian Bridge Approach; University of North Carolina: Chapel Hill, NC, USA, 1991. [Google Scholar]
- Keating, K.A.; Cherry, S. Modeling utilization distributions in space and time. Ecology 2009, 90, 1971–1980. [Google Scholar] [CrossRef] [PubMed]
- Noguchi, K.; Gel, Y.R.; Brunner, E.; Konietschke, F. nparLD: An r software package for the nonparametric analysis of longitudinal data in factorial experiments. J. Stat. Softw. 2012, 50. [Google Scholar] [CrossRef]
- Manly, B.; McDonald, L.; Thomas, D.L.; McDonald, T.L.; Erickson, W.P. Resource Selection by Animals: Statistical Design and Analysis for Field Studies; Springer Science & Business Media: London, UK, 2007. [Google Scholar]
- Lamprecht, J. Social dominance and reproductive success in a goose flock (Anser indicus). Behavior 1986, 97, 50–65. [Google Scholar] [CrossRef]
- Del Hoyo, J.; Elliott, A.; Sargatal, J. Handbook of the Birds of the World; Volume 1: Ostrich to Ducks; Lynx Edicions: Barcelona, Spain, 1992. [Google Scholar]
- Wan, G.; Yang, M.; Liu, Z.; Wang, X.; Liang, X. The precipitation variations in the Qinghai-Xizang (Tibetan) plateau during 1961–2015. Atmosphere 2017, 8, 80. [Google Scholar] [CrossRef]
- Kranstauber, B.; Kays, R.; LaPoint, S.D.; Wikelski, M.; Safi, K. A dynamic Brownian bridge movement model to estimate utilization distributions for heterogeneous animal movement. J. Anim. Ecol. 2012, 81, 738–746. [Google Scholar] [CrossRef] [PubMed]
- Barron, D.G.; Brawn, J.D.; Weatherhead, P.J. Meta-analysis of transmitter effects on avian behaviour and ecology. Methods Ecol. Evol. 2010, 1, 180–187. [Google Scholar] [CrossRef]
- De Vries, M. The Effect of Telemetric Devices on the Flight and Swimming Performance of Birds. Master’s Thesis, Utrecht University, Utrecht, The Netherlands, 2014. [Google Scholar]
- Bishop, M.A.; Yanling, S.; Zhouma, C.; Binyuan, G. Bar-headed geese Anser indicus wintering in south-central Tibet. Wildfowl 1997, 48, 118–126. [Google Scholar]
- Li, X.; Ma, Y.; Xu, H.; Wang, J.; Zhang, D. Impact of land use and land cover change on environmental degradation in lake Qinghai watershed, northeast Qinghai-Tibet Plateau. Land Degrad. Dev. 2009, 20, 69–83. [Google Scholar] [CrossRef]
- Ramanathan, V.; Feng, Y. Air pollution, greenhouse gases and climate change: Global and regional perspectives. Atmos. Environ. 2009, 43, 37–50. [Google Scholar] [CrossRef]
- Holmes, J.A.; Cook, E.R.; Yang, B. Climate change over the past 2000 years in western China. Quat. Int. 2009, 194, 91–107. [Google Scholar] [CrossRef]
- Xu, X.; Chen, H.; Levy, J.K. Spatiotemporal vegetation cover variations in the Qinghai-Tibet plateau under global climate change. Chin. Sci. Bull. 2008, 53, 915–922. [Google Scholar] [CrossRef]
- Brown, J.D.; Stallknecht, D.E.; Swayne, D.E. Experimental infection of swans and geese with highly pathogenic avian influenza virus (h5n1) of Asian lineage. Emerg. Infect. Dis. 2008, 14, 136–142. [Google Scholar] [CrossRef] [PubMed]
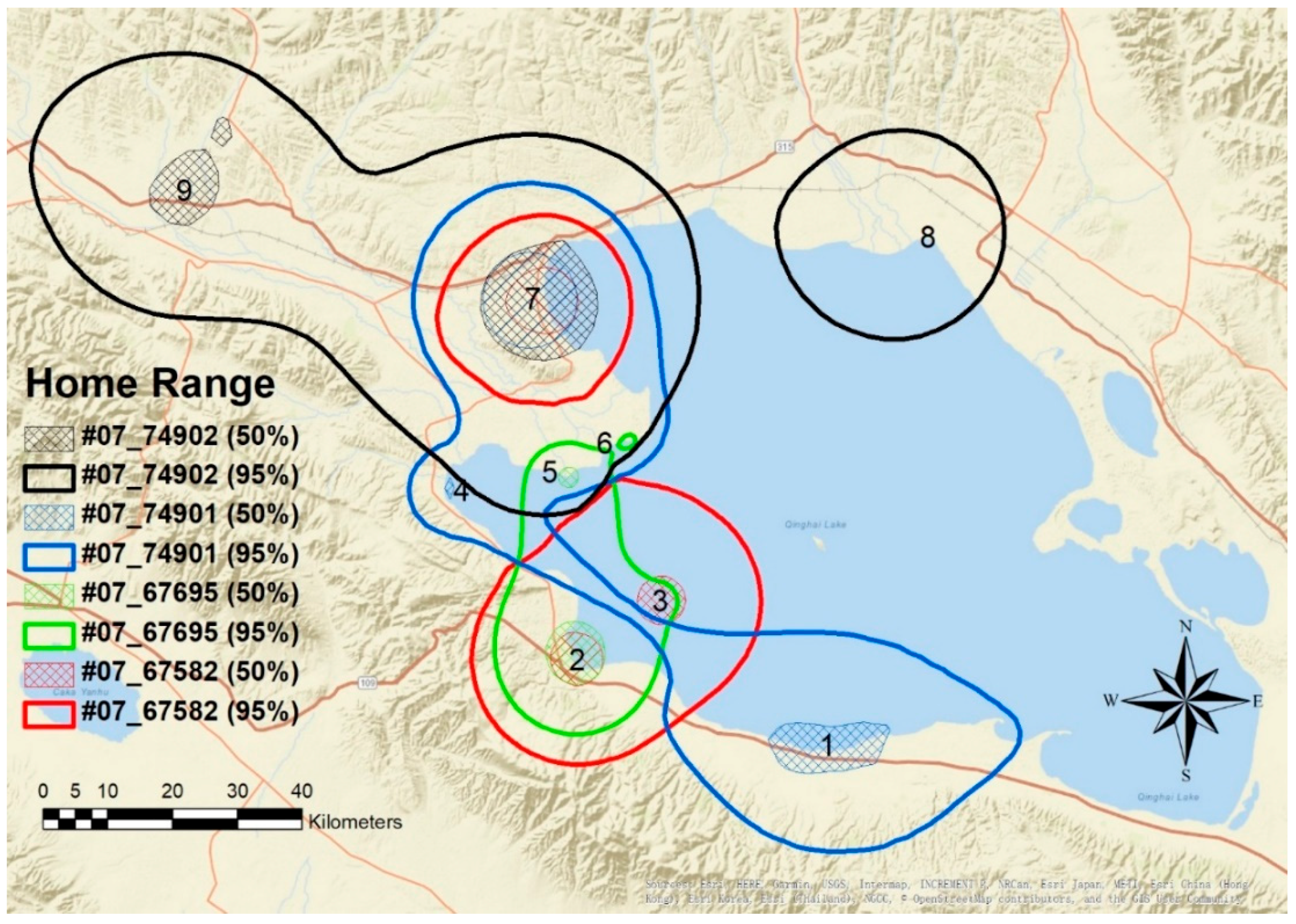
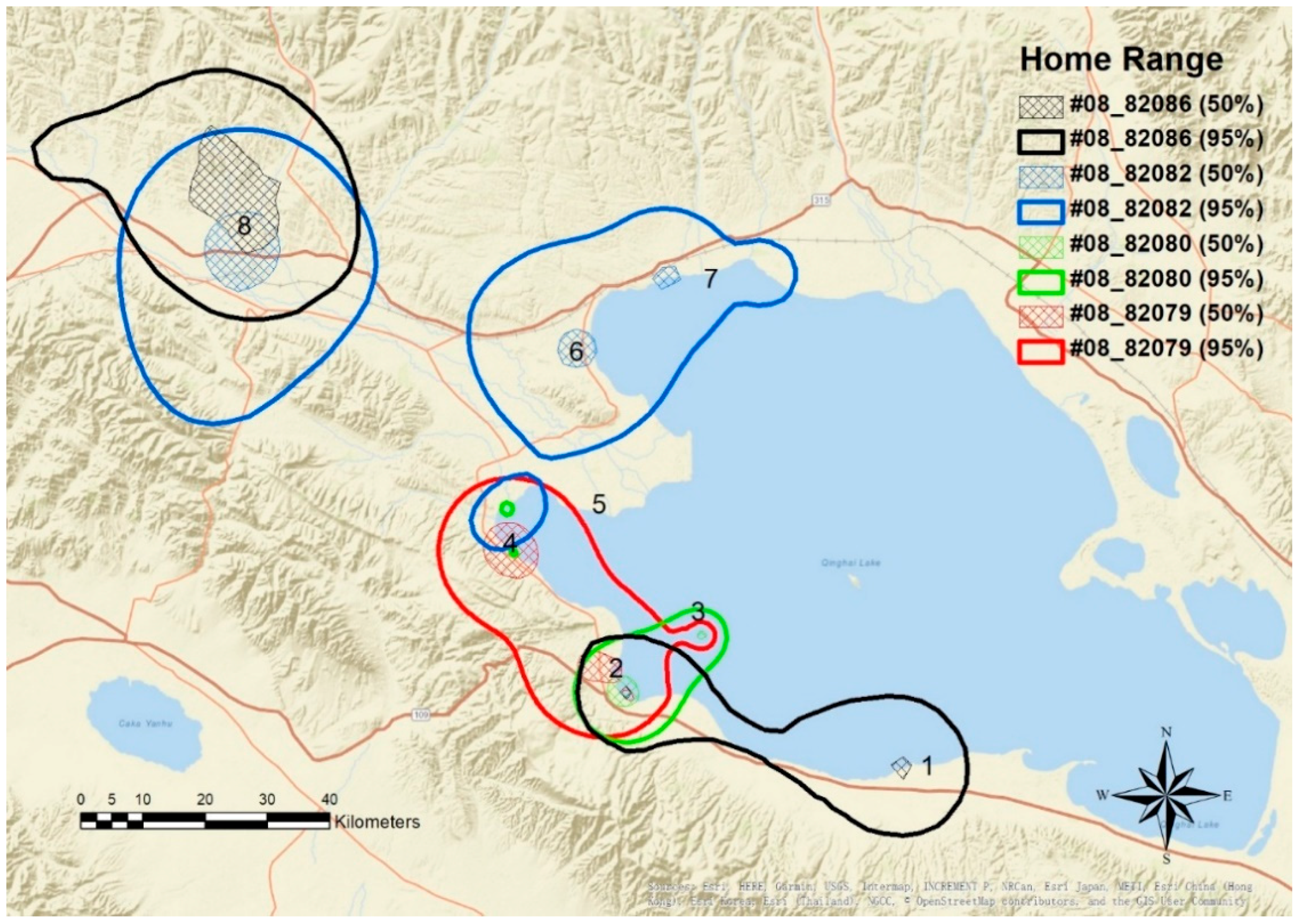
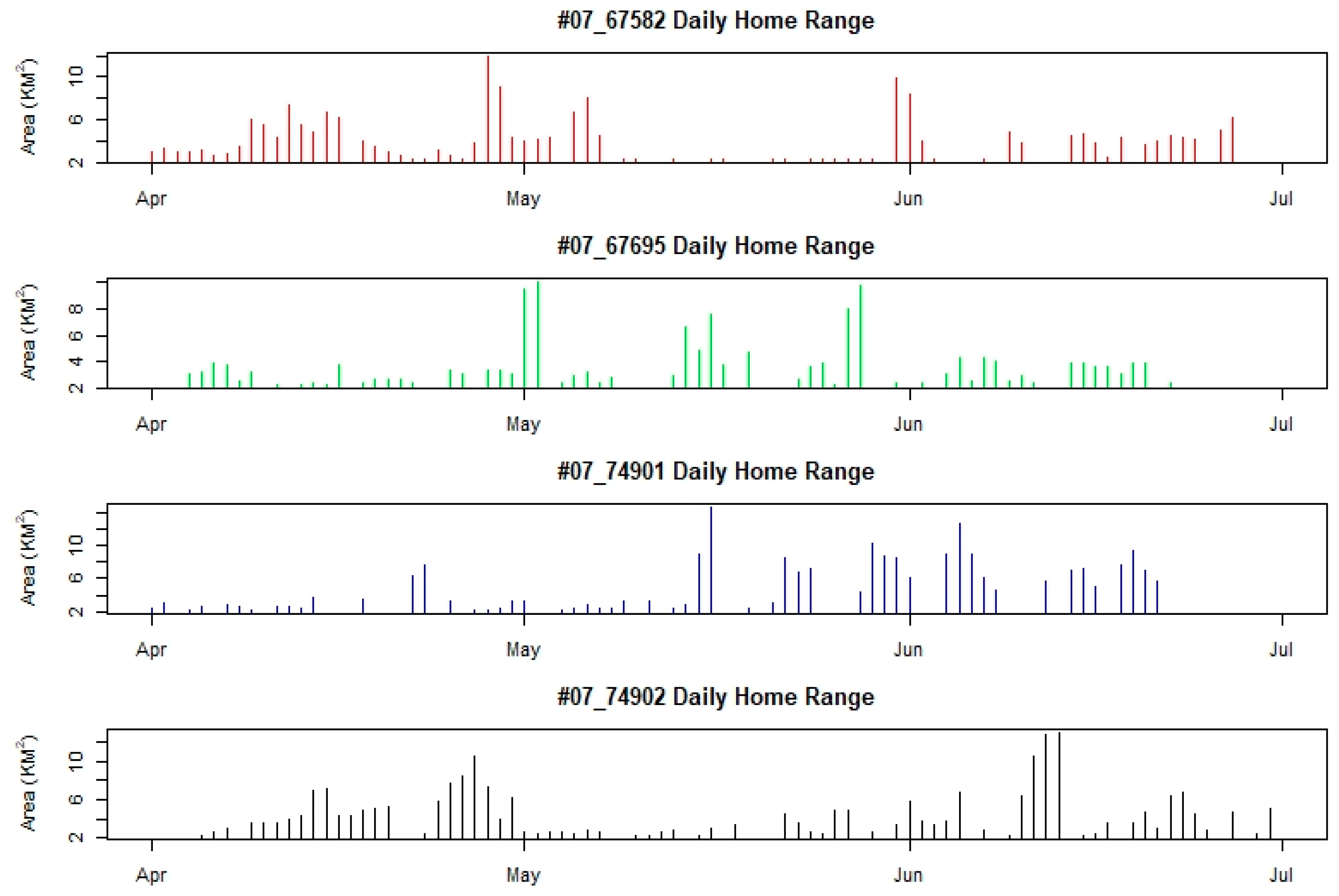
| ID | Sex | Capture Date | Departure Date | Number of GPS Fixes | Home Range Area (km2) | Core Area (km2) |
|---|---|---|---|---|---|---|
| 07_67582 | F | 03/25/07 | 07/02/07 | 327 | 1476.15 | 126.5 |
| 07_67695 | M | 03/29/07 | 06/22/07 | 252 | 543.89 | 49.22 |
| 07_74901 | M | 03/31/07 | 06/21/07 | 234 | 1369.48 | 187.62 |
| 07_74902 | F | 03/30/07 | 10/24/07 | 334 | 3910.36 | 242.03 |
| 08_82079 | M | 04/02/08 | 08/31/08 | 329 | 612.82 | 58.42 |
| 08_82080 | M | 04/02/08 | 06/29/08 | 561 | 211.57 | 13.57 |
| 08_82082 | F | 03/30/08 | 09/29/08 | 573 | 1788.28 | 103.63 |
| 08_82086 | F | 03/31/08 | 08/21/08 | 266 | 1409.34 | 120.37 |
| Year | n | Home Range (km2) (min–max) | Core Area (km2) (min–max) |
|---|---|---|---|
| Female | 4 | 2146.03 ± 1187.76 (1409.34–3910.36) | 245.34 ± 89.41 (137.84.86–321.61) |
| Male | 4 | 990.14 ± 778.22 (220.36–2011.66) | 84.93 ± 75.21 (16.23–191.49) |
| 2007 | 4 | 1987.93 ± 1419.39 (543.89–3910.36) | 148.13 ± 63.34 (103.63–242.03) |
| 2008 | 4 | 684.44 ± 489.14 (211.57–1369.48) | 77.21 ± 76.11 (13.57–187.62) |
| ID | Sex | Daily Home Range (km2) (min–max) | Daily Movement Distance (km) (min–max)-Median | Daily Movement Rate (Km/H) (Min–Max)-Median |
|---|---|---|---|---|
| 07_67582 | F | 4.08 ± 2.03 (2.30–11.95) | 4.07 ± 13.34 (0–96.23)-0.87 | 0.31 ± 0.78 (0–4.37)-0.09 |
| 07_67695 | M | 3.71 ± 1.84 (2.30–10.05) | 3.24 ± 7.42 (0–40.22)-0.71 | 0.25 ± 0.49 (0–2.51)-0.08 |
| 07_74901 | M | 5.04 ± 2.99 (2.20–14.62) | 5.34 ± 11.81 (0–73.70)-0.94 | 2.52 ± 0.94 (0–4.82)-0.13 |
| 07_74902 | F | 4.42±2.43 (2.25–12.99) | 4.27 ± 9.03 (0–51.67)-1.34 | 0.39 ± 0.99 (0–7.24)-0.12 |
| 08_82079 | M | 4.64 ± 1.95 (2.30–11.00) | 4.64 ± 7.92 (0–35.56)-1.88 | 0.51 ± 1.08 (0–6.98)-0.18 |
| 08_82080 | M | 4.52 ± 1.73 (2.32–10.49) | 5.89 ± 8.43 (0–37.32)-1.83 | 0.52 ± 0.86 (0–3.19)-0.12 |
| 08_82082 | F | 5.96 ± 2.68 (2.31–15.40) | 12.76 ± 23.58 (0–195.49)-4.28 | 0.87 ± 1.51 (0–12.22)-0.24 |
| 08_82086 | F | 5.37 ± 2.44 (2.17–12.24) | 7.99 ± 12.53 (0–78.34)-3.78 | 0.74 ± 1.12 (0–6.00)-0.33 |
| Characteristic | Category | Bonferroni CI | Selection | |||||
|---|---|---|---|---|---|---|---|---|
| Lower | Upper | |||||||
| Land cover | Cropland | 0.076 | 0.460 | 0.165 | 0.009 | 0.140 | 0.190 | - |
| Forest | 0.207 | 0.302 | 0.685 | 0.026 | 0.615 | 0.754 | - | |
| Shrubland | 0.006 | 0.000 | 26.28 | 26.75 | -44.31 | 96.87 | 0 | |
| Barrenland | 0.177 | 0.132 | 1.336 | 0.070 | 1.153 | 1.520 | + | |
| Wetland | 0.533 | 0.105 | 5.075 | 0.243 | 4.434 | 5.716 | + | |
| = 2700.529, = 4, p < 0.001 | ||||||||
| NDVI | −1–−0.5 | 0.0001 | 0.0001 | 1.000 | 1.000 | –1.576 | 3.576 | 0 |
| −0.5–0 | 0.339 | 0.185 | 1.836 | 0.071 | 1.652 | 2.020 | + | |
| 0–0.5 | 0.660 | 0.815 | 0.810 | 0.011 | 0.783 | 0.837 | - | |
| 0.5–1 | 0.000 | 0.000 | 1.000 | 1.414 | –2.643 | 4.643 | 0 | |
| = 324.91, = 3, p < 0.001 | ||||||||
| Distance to water (km) | 0–0.5 | 0.796 | 0.309 | 2.578 | 0.063 | 2.421 | 2.735 | + |
| 0.5–2 | 0.125 | 0.241 | 0.518 | 0.025 | 0.454 | 0.581 | - | |
| 2–4 | 0.076 | 0.178 | 0.428 | 0.027 | 0.361 | 0.496 | - | |
| >4 | 0.003 | 0.272 | 0.010 | 0.003 | 0.002 | 0.017 | - | |
| = 2751.1, = 3, p < 0.001 | ||||||||
| Distance to road (km) | 0–0.2 | 0.013 | 0.029 | 0.444 | 0.071 | 0.266 | 0.621 | - |
| 0.2–0.5 | 0.022 | 0.030 | 0.740 | 0.099 | 0.492 | 0.988 | - | |
| 0.5–2 | 0.531 | 0.169 | 3.138 | 0.116 | 2.847 | 3.428 | + | |
| >2 | 0.434 | 0.771 | 0.563 | 0.011 | 0.535 | 0.590 | - | |
| = 1260.51, = 3, p < 0.001 | ||||||||
| Elevation (km) | 2.8–3.2 | 0.544 | 0.214 | 2.538 | 0.085 | 2.319 | 2.758 | + |
| 3.2–3.3 | 0.289 | 0.255 | 1.130 | 0.041 | 1.025 | 1.236 | + | |
| 3.3–3.4 | 0.146 | 0.176 | 0.828 | 0.042 | 0.720 | 0.936 | - | |
| 3.4–3.6 | 0.021 | 0.184 | 0.112 | 0.012 | 0.080 | 0.144 | - | |
| >3.6 | 0.001 | 0.170 | 0.006 | 0.003 | -0.002 | 0.013 | - | |
| = 2167.94, = 4, p < 0.001 | ||||||||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zheng, R.; Smith, L.M.; Prosser, D.J.; Takekawa, J.Y.; Newman, S.H.; Sullivan, J.D.; Luo, Z.; Yan, B. Investigating Home Range, Movement Pattern, and Habitat Selection of Bar-headed Geese during Breeding Season at Qinghai Lake, China. Animals 2018, 8, 182. https://doi.org/10.3390/ani8100182
Zheng R, Smith LM, Prosser DJ, Takekawa JY, Newman SH, Sullivan JD, Luo Z, Yan B. Investigating Home Range, Movement Pattern, and Habitat Selection of Bar-headed Geese during Breeding Season at Qinghai Lake, China. Animals. 2018; 8(10):182. https://doi.org/10.3390/ani8100182
Chicago/Turabian StyleZheng, Ruobing, Lacy M. Smith, Diann J. Prosser, John Y. Takekawa, Scott H. Newman, Jeffery D. Sullivan, Ze Luo, and Baoping Yan. 2018. "Investigating Home Range, Movement Pattern, and Habitat Selection of Bar-headed Geese during Breeding Season at Qinghai Lake, China" Animals 8, no. 10: 182. https://doi.org/10.3390/ani8100182
APA StyleZheng, R., Smith, L. M., Prosser, D. J., Takekawa, J. Y., Newman, S. H., Sullivan, J. D., Luo, Z., & Yan, B. (2018). Investigating Home Range, Movement Pattern, and Habitat Selection of Bar-headed Geese during Breeding Season at Qinghai Lake, China. Animals, 8(10), 182. https://doi.org/10.3390/ani8100182





