Effects of a Change from an Indoor-Based Total Mixed Ration to a Rotational Pasture System Combined with a Moderate Concentrate Feed Supply on the Health and Performance of Dairy Cows
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Design and Treatments
2.2. Pasture Management
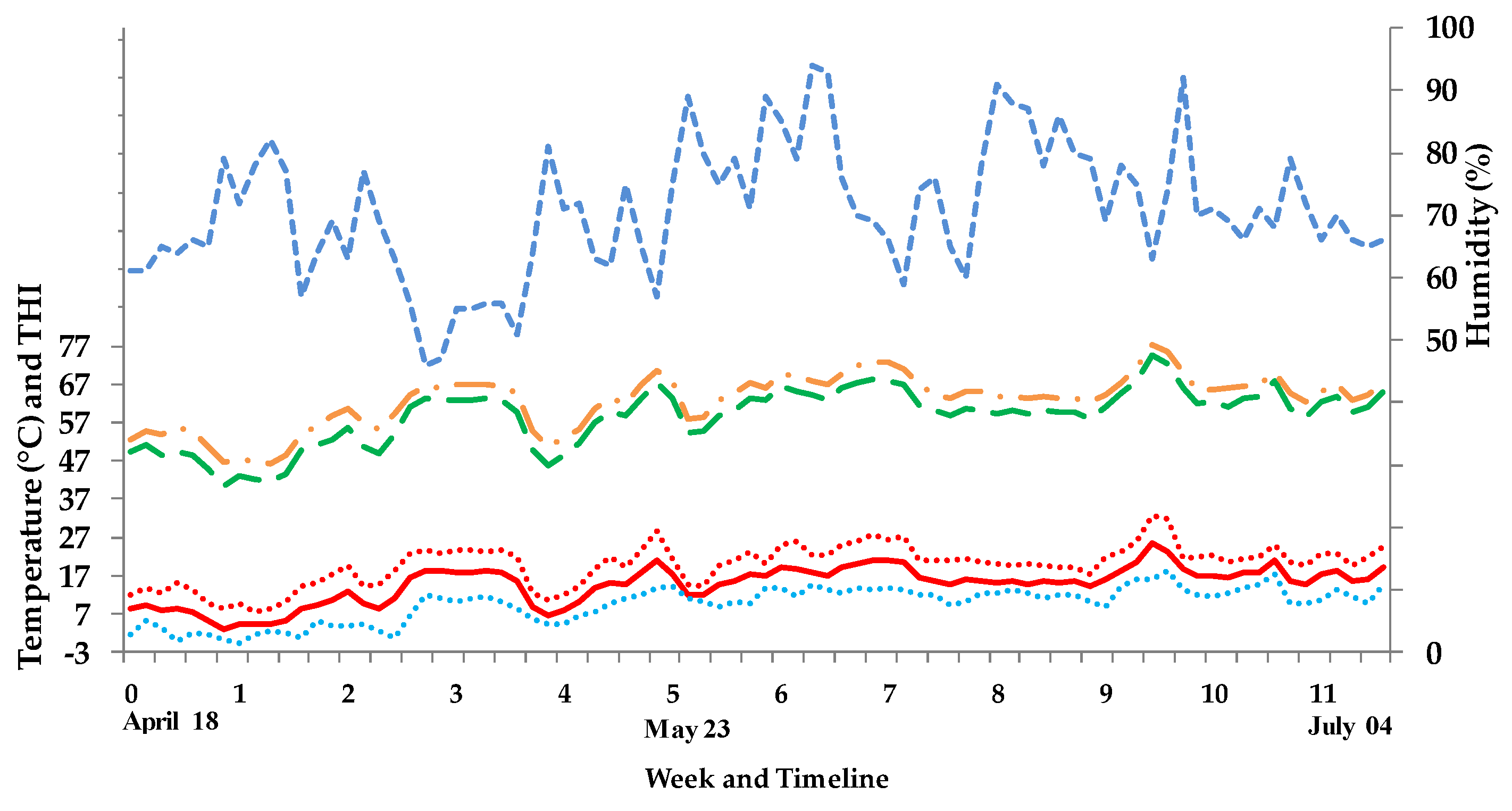
2.3. Weather and Barn Climate Conditions
2.4. Pasture and Feed Measurements
2.5. Animal Measurements
2.5.1. DMI
2.5.2. Milk, BCS and BW
2.5.3. Blood
2.5.4. Fat Depots
2.5.5. Liver Fat
2.5.6. Activity and Behavior Monitoring
2.6. Calculations
2.7. Statistical Analysis
3. Results
3.1. Weather and Barn Climate Conditions
3.2. Feed Composition
3.3. Paddock Characteristics
3.4. Animal Measurements
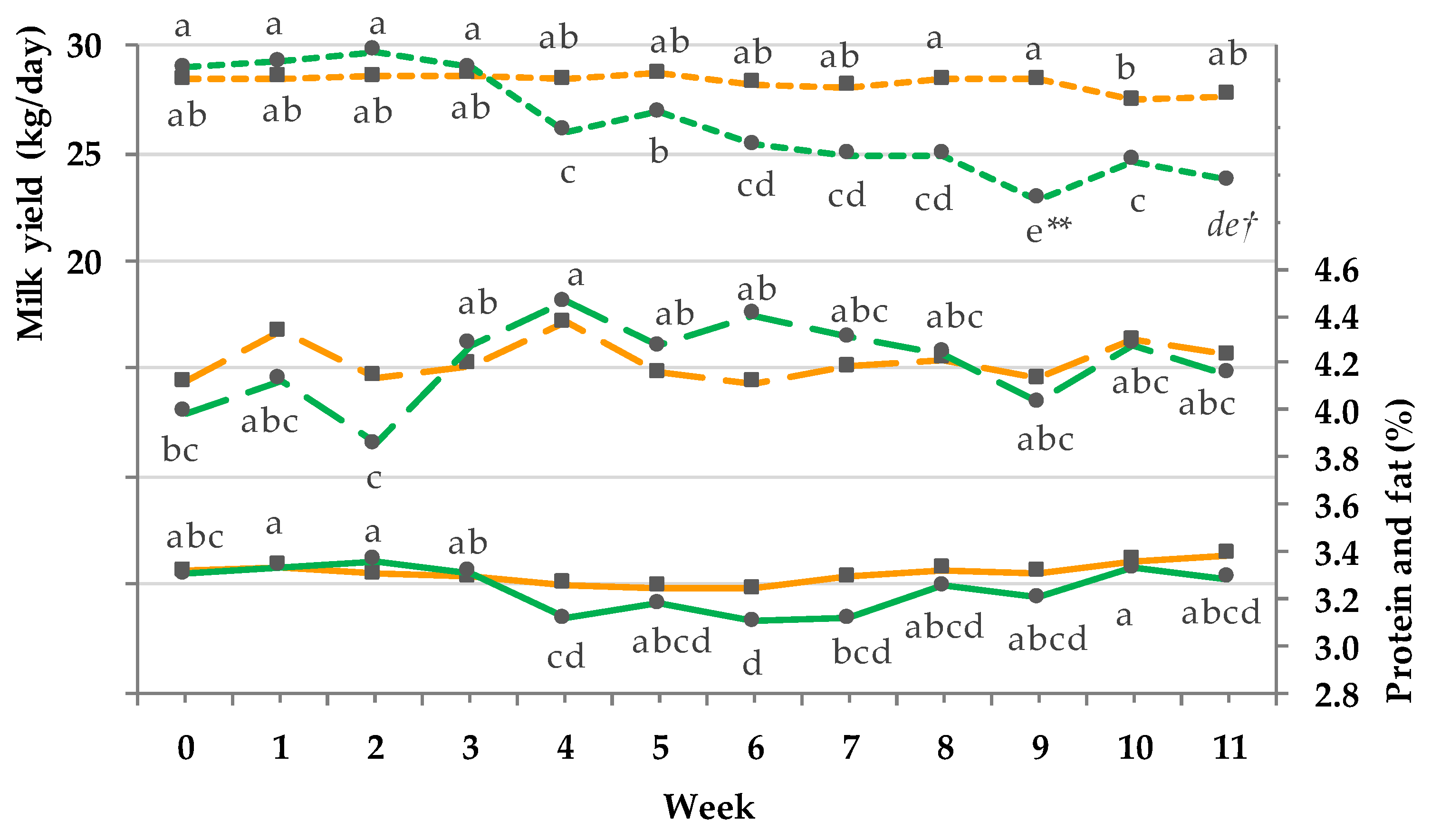
3.5. Milk Yield and Composition
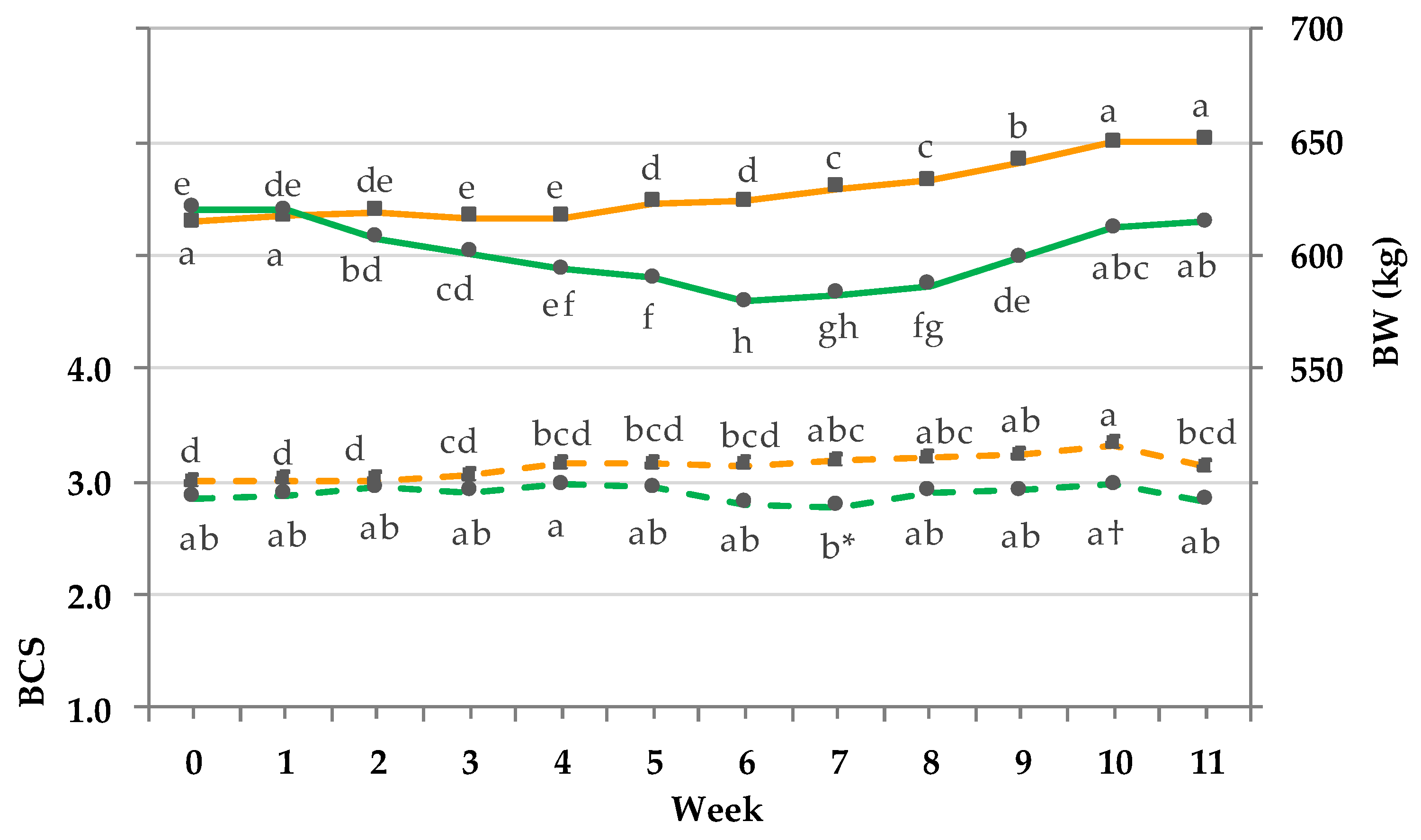
3.6. BW and BCS.
3.7. Hematology
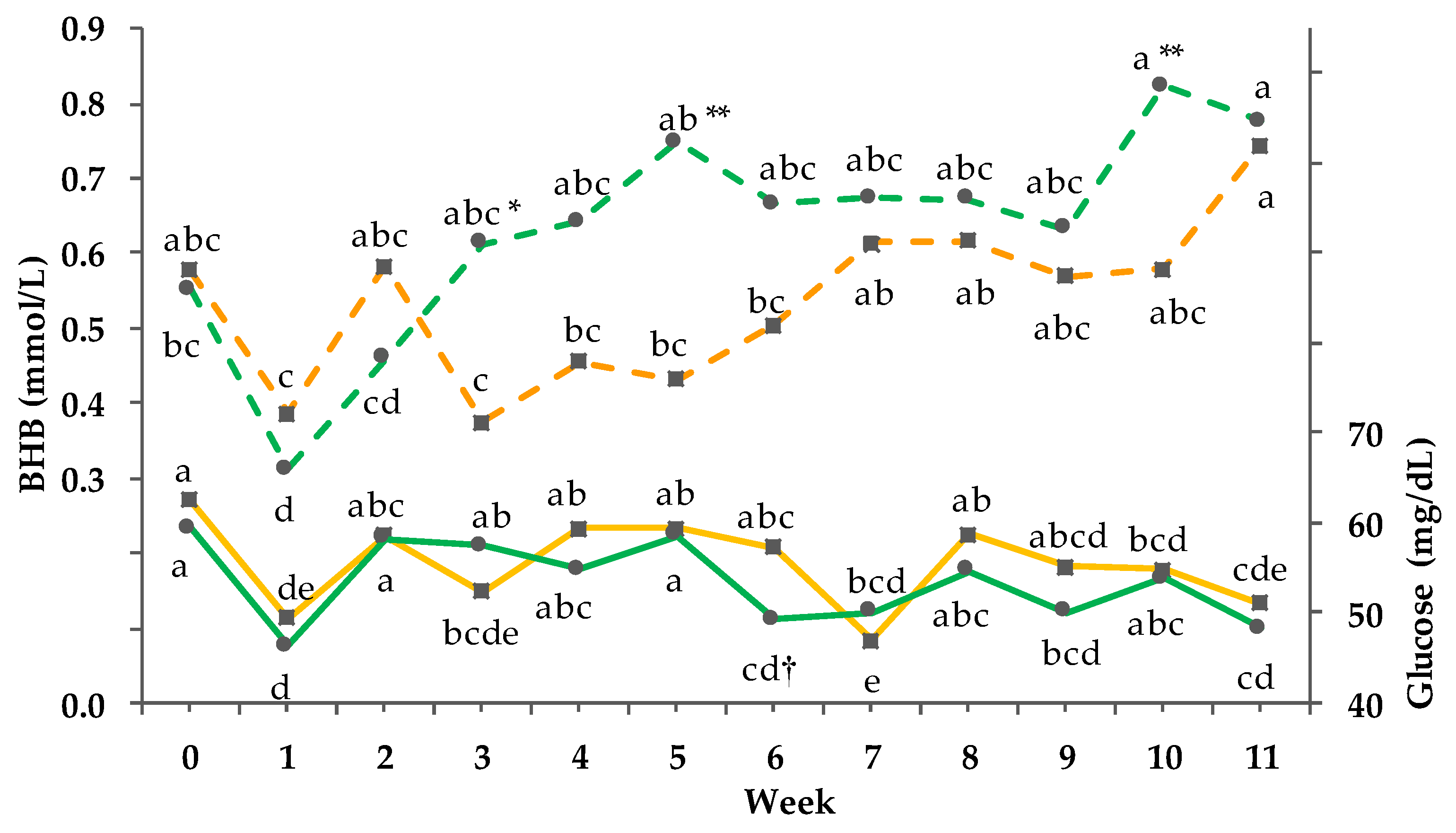
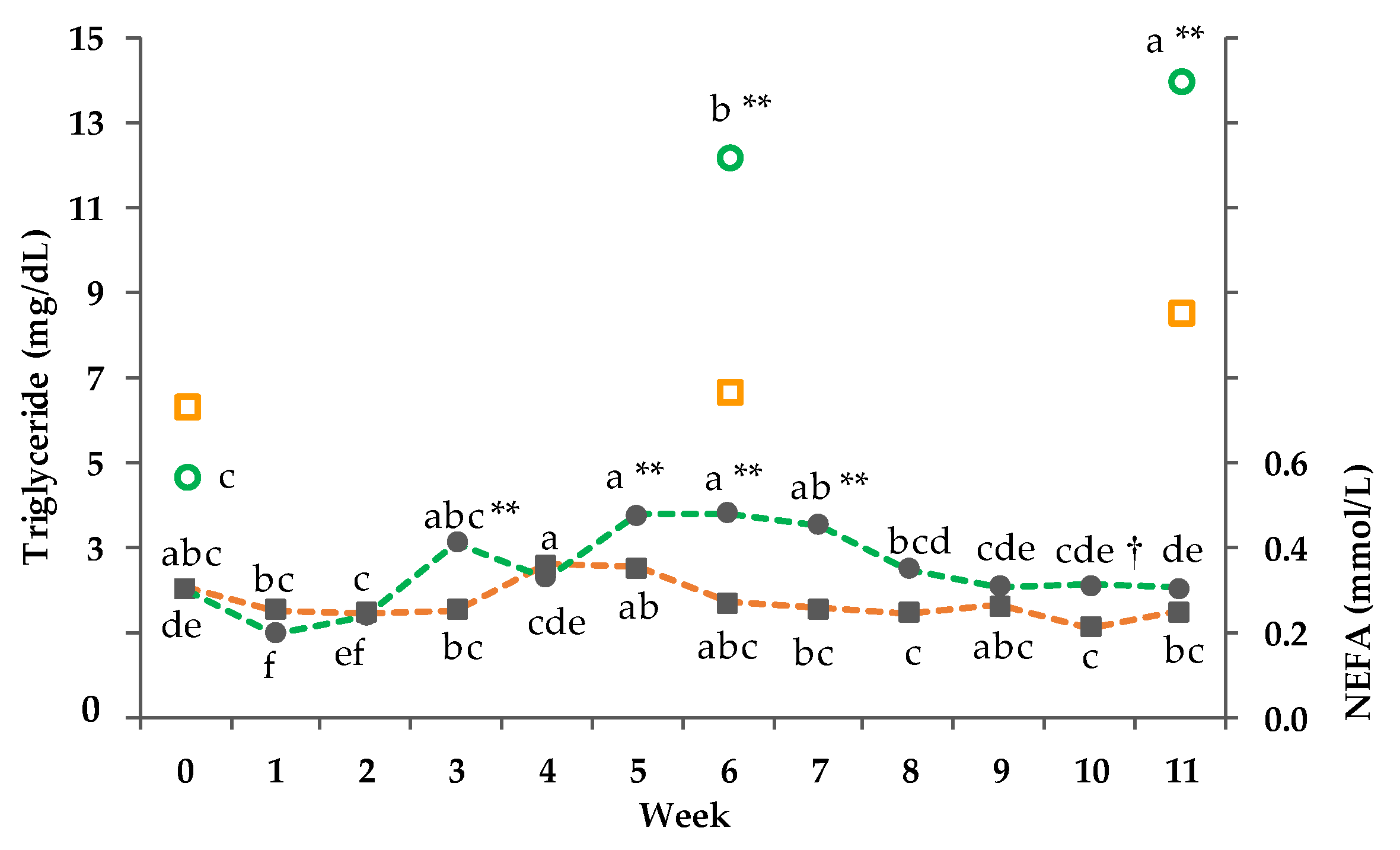
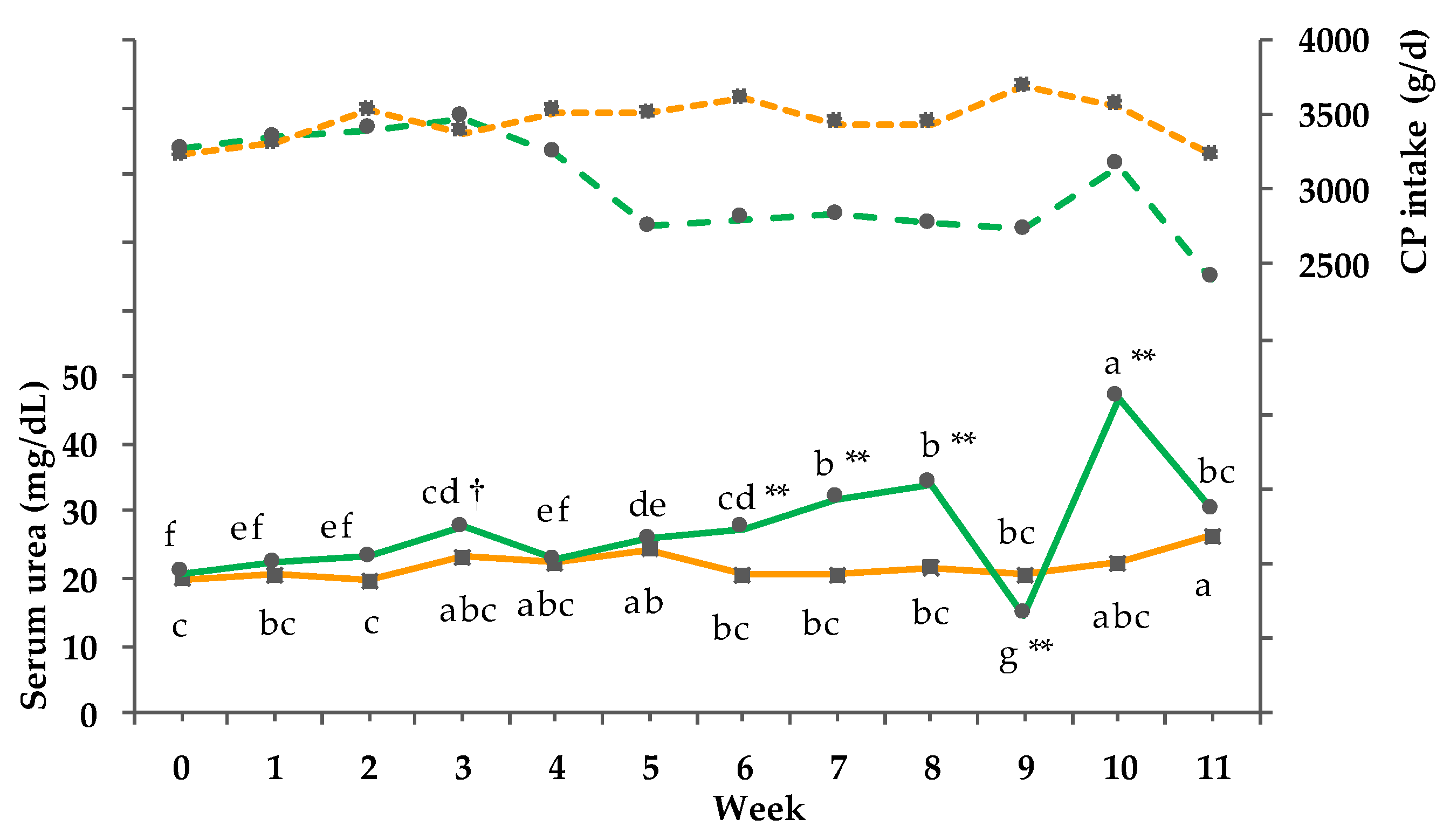
3.8. Clinical-Chemical Traits
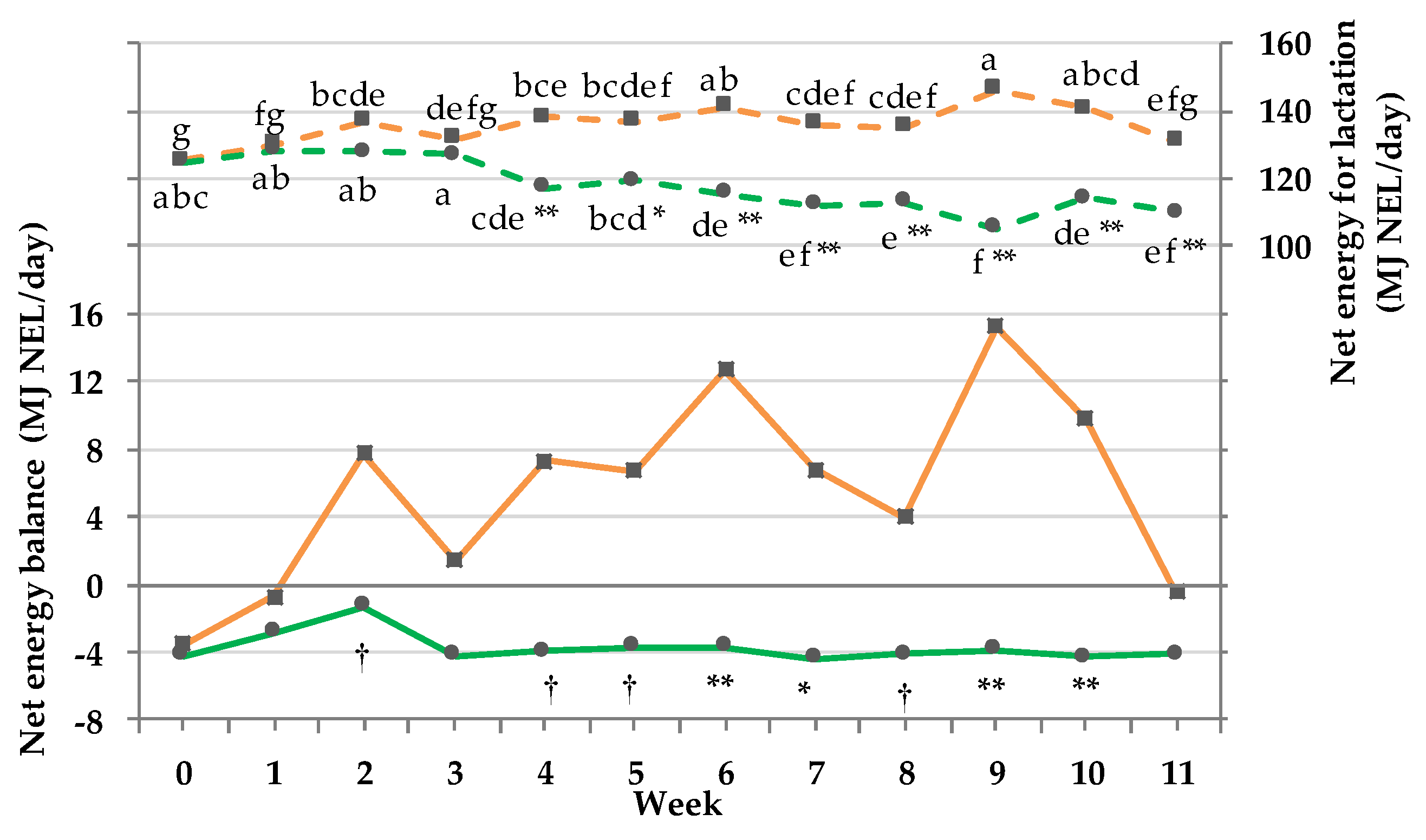
3.9. Net energy Intake and Balance
3.10. Fat Depots and Liver Fat
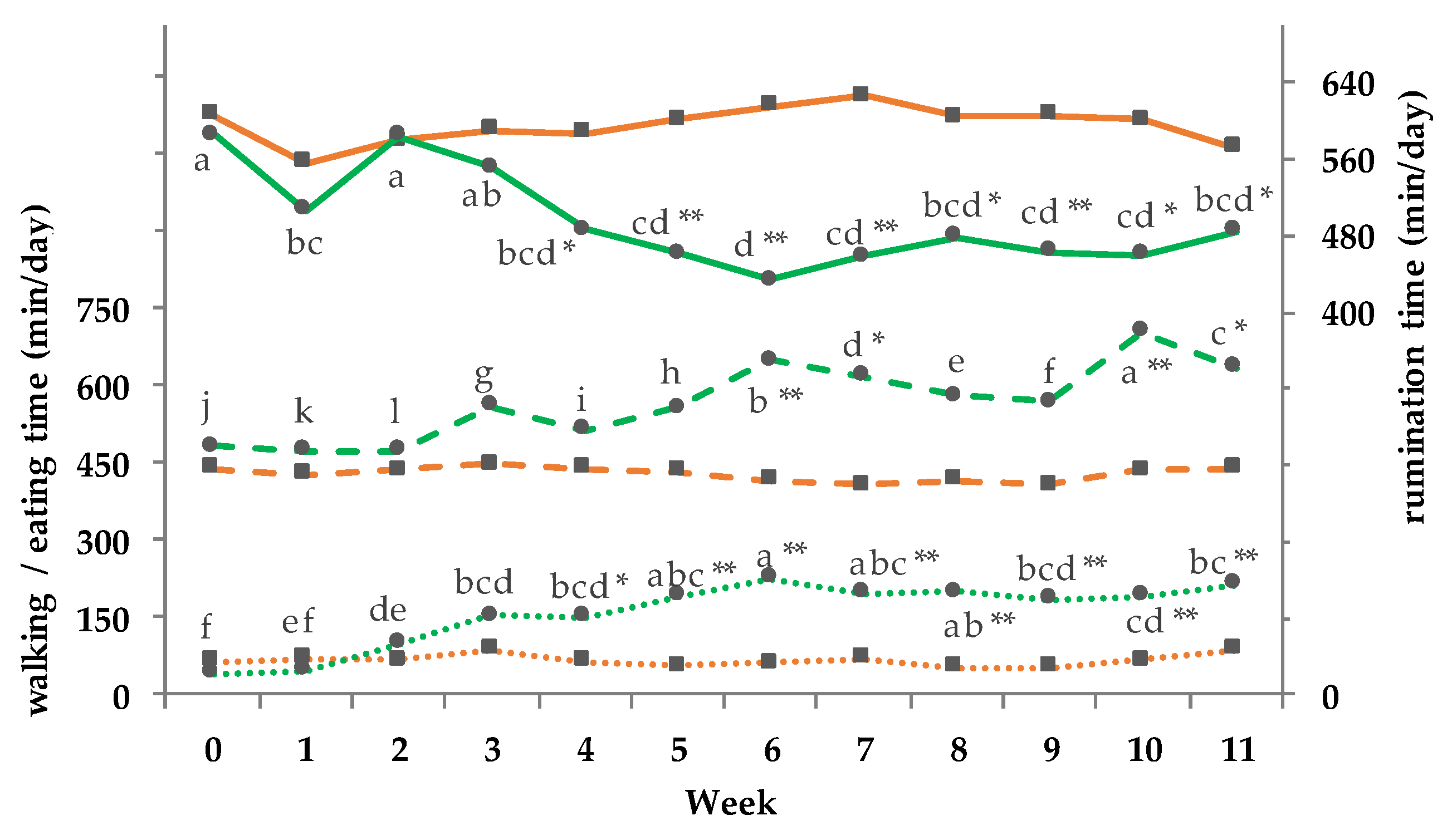
3.11. Activity and Behavior Monitoring
4. Discussion
4.1. Effects of Transition to Grazing on DMI
4.2. Changes of Grazing Behavior and Its Effect on DMI
4.3. Effects of DMI on Body Condition
4.4. Effect of Behavioral Adaption on Energy Metabolism
4.5. Effect of Transition on Milking Performance
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Plant species (%) | Paddock 1 * | Paddock 2 * | Paddock 3 * | Paddock 4 * |
|---|---|---|---|---|
| Grass | 80.8 ± 4.4 | 71.2 ± 32.6 | 81.3 ± 16.3 | 92 |
| Herbs | 5.8 ± 4.6 | 8.3 ± 6.7 | 11.3 ± 6.5 | 2.0 |
| Legumes | 1.8 ± 3.0 | 6.7 ± 8.3 | 5.0 ± 8.7 | 6.0 |
| Lolium perenne | 42.3 ± 34.5 | 27.2 ± 11.2 | 32.8 ± 18.8 | 40 |
| Phleum pretense | 32.3 ± 31.7 | 28.2 ± 10.6 | 22.5 ± 31.7 | 10 |
| Poa trivalis | 7.0 ± 0.0 | 2.5 ± 0.7 | ||
| Poa annua | 4.5 ± 2.5 | |||
| Holus Lanatus | 5.0 ± 0.0 | |||
| Dactylis glomerata | 2.0 ± 0.5 | 14.2 ± 10.8 | 5.0 ± 0.0 | |
| Poa pratensis | 5.0 ± 2.4 | 5.0 ± 0.0 | 6.5 ± 2.1 | 7.0 |
| Elymus repens | 0.7 ± 1.2 | |||
| Festuca pratensis | 5.0 ± 0.0 | 72.0 ± 0.0 | 35 | |
| Festuca rubra | 4.0 ± 0.0 | |||
| Stellaria media | 5.3 ± 4.6 | 3.3 ± 2.0 | 9.0 ± 5.2 | |
| Achillea millefolium | 5.0 ± 0.0 | |||
| Lamium Purpureum | 0.5 ± 0.0 | |||
| Capsella bursa-pastoris | 2.0 ± 1.0 | |||
| Taracum officinale | 4.0 ± 0.0 | 1.0 | ||
| Veronica chamaedrys | ||||
| Geranium dissectum | 10.0 ± 0.0 | |||
| Trifolium repens | 2.3 ± 4.0 | 7.3 ± 9 0 | 20.0 ± 0.0 | |
| Trifolium pratense | 6.0 |
References
- Bargo, F.; Muller, L.D.; Kolver, E.S.; Delahoy, J.E. Invited review: Production and digestion of supplemented dairy cows on pasture. J. Dairy Sci. 2003, 86, 1–42. [Google Scholar] [CrossRef]
- Kolver, E.S.; Muller, L.D. Performance and nutrient intake of high producing holstein cows consuming pasture or a total mixed ration. J. Dairy Sci. 1998, 81, 1403–1411. [Google Scholar] [CrossRef]
- Gibb, M.J.; Huckle, C.A.; Nuthall, R.; Rook, A.J. Effect of sward surface height on intake and grazing behaviour by lactating holstein friesian cows. Grass Forage Sci. 1997, 52, 309–321. [Google Scholar] [CrossRef]
- Dohme-Meier, F.; Kaufmann, L.D.; Görs, S.; Junghans, P.; Metges, C.C.; van Dorland, H.A.; Bruckmaier, R.M.; Münger, A. Comparison of energy expenditure, eating pattern and physical activity of grazing and zero-grazing dairy cows at different time points during lactation. Livest. Sci. 2014, 162, 86–96. [Google Scholar] [CrossRef]
- Schären, M.; Jostmeier, S.; Ruesink, S.; Hüther, L.; Frahm, J.; Bulang, M.; Meyer, U.; Rehage, J.; Isselstein, J.; Breves, G.; et al. The effects of a ration change from a total mixed ration to pasture on health and production of dairy cows. J. Dairy Sci. 2016, 99, 1183–1200. [Google Scholar] [CrossRef] [PubMed]
- Drong, C.; Meyer, U.; von Soosten, D.; Frahm, J.; Rehage, J.; Schirrmeier, H.; Beer, M.; Dänicke, S. Effects of monensin and essential oils on immunological, haematological and biochemical parameters of cows during the transition period. J. Anim. Physiol. Anim. Nutr. 2017, 101, 791–806. [Google Scholar] [CrossRef] [PubMed]
- Schäfers, S.; von Soosten, D.; Meyer, U.; Drong, C.; Frahm, J.; Kluess, J.; Raschka, C.; Rehage, J.; Tröscher, A.; Pelletier, W.; et al. Influence of conjugated linoleic acid and vitamin e on performance, energy metabolism, and change of fat depot mass in transitional dairy cows. J. Dairy Sci. 2017, 100, 3193–3208. [Google Scholar] [CrossRef] [PubMed]
- Hills, J.L.; Wales, W.J.; Dunshea, F.R.; Garcia, S.C.; Roche, J.R. Invited review: An evaluation of the likely effects of individualized feeding of concentrate supplements to pasture-based dairy cows. J. Dairy Sci. 2015, 98, 1363–1401. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Belenguer, S.; Palacio, J.; Gascon, M.; Acena, C.; Revilla, R.; Mormède, P. Differences in the biological stress responses of two cattle breeds to walking up to mountain pastures in the pyrenees. Vet. Res. 1996, 27, 515–526. [Google Scholar] [PubMed]
- Gibb, M.J. Chapter 8: Grassland management with emphasis on grazing behaviour. In Fresh Herbage for Dairy Cattle; AA Dordrecht, T.N., Ed.; Springer: North Wyke, Devon, UK, 2007; pp. 141–157. [Google Scholar]
- Hahn, G.L. Dynamic responses of cattle to thermal heat loads. J. Anim. Sci. 1999, 77 (Suppl. 2), 10–20. [Google Scholar] [CrossRef] [PubMed]
- VDLUFA. Handbuch der Landwirtschaftlichen Versuchs- und Untersuchungsmethodik (VDLUFA-Methodenbuch), Bd. III Die chemische Untersuchung von Futtermitteln; Verband Deutscher Landwirtschaftlicher Untersuchungs- und Forschunganstalten; VDLUFA-Verlag: Darmstadt, Germany, 2012. (In German) [Google Scholar]
- Heublein, C.; Dohme-Meier, F.; Sudekum, K.H.; Bruckmaier, R.M.; Thanner, S.; Schori, F. Impact of cow strain and concentrate supplementation on grazing behaviour, milk yield and metabolic state of dairy cows in an organic pasture-based feeding system. Anim. Int. J. Anim. Biosci. 2017, 11, 1163–1173. [Google Scholar] [CrossRef] [PubMed]
- Elwert, C.; Kluth, H.; Rodehutscord, M. Effect of variable intake of alfalfa and wheat on faecal alkane recoveries and estimates of roughage intake in sheep. J. Agric. Sci. 2004, 142, 213–223. [Google Scholar] [CrossRef]
- Taweel, H.Z.; Tas, B.M.; Smit, H.J.; Elgersma, A.; Dijkstra, J.; Tamminga, S. Grazing behaviour, intake, rumen function and milk production of dairy cows offered lolium perenne containing different levels of water-soluble carbohydrates. Livest. Sci. 2006, 102, 33–41. [Google Scholar] [CrossRef]
- Dove, H.; Mayes, R. The use of plant wax alkanes as marker substances in studies of the nutrition of herbivores: A review. Aust. J. Agric. Res. 1991, 42, 913–952. [Google Scholar] [CrossRef]
- Edmonson, A.J.; Lean, I.J.; Weaver, L.D.; Farver, T.; Webster, G. A body condition scoring chart for holstein dairy cows. J. Dairy Sci. 1989, 72, 68–78. [Google Scholar] [CrossRef]
- Raschka, C.; Ruda, L.; Wenning, P.; von Stemm, C.I.; Pfarrer, C.; Huber, K.; Meyer, U.; Danicke, S.; Rehage, J. In vivo determination of subcutaneous and abdominal adipose tissue depots in german holstein dairy cattle. J. Anim. Sci. 2016, 94, 2821–2834. [Google Scholar] [CrossRef] [PubMed]
- Starke, A.; Haudum, A.; Busche, R.; Beyerbach, M.; Danicke, S.; Rehage, J. Technical note: Analysis of total lipid and triacylglycerol content in small liver biopsy samples in cattle. J. Anim. Sci. 2010, 88, 2741–2750. [Google Scholar] [CrossRef] [PubMed]
- GfE. Empfehlungen zur Energie- und Nährstoffversorgung von Milchkühen und Aufzuchtrinder; DLG-Verlags GmbH: Frankfurt am Main, Germany, 2001. (In German) [Google Scholar]
- Collier, R.; Zimbelman, R.; Rhoads, R.; Rhoads, M.; Baumgard, L. A re-evaluation of the impact of temperature humidity index (thi) and black globe humidity index (bghi) on milk production in high producing dairy cows. In Proceedings of the 24th Annual Southwest Nutrition and Management Conference, Tempe, AZ, USA, 2009. [Google Scholar]
- Gorniak, T.; Meyer, U.; Sudekum, K.H.; Danicke, S. Impact of mild heat stress on dry matter intake, milk yield and milk composition in mid-lactation holstein dairy cows in a temperate climate. Arch. Anim. Nutr. 2014, 68, 358–369. [Google Scholar] [CrossRef] [PubMed]
- Kellaway, R.C.; Colditz, P.J. The effect of heat stress on growth and nitrogen metabolism in friesian and f 1 brahman × friesian heifers. Aust. J. Agric. Res. 1975, 26. [Google Scholar] [CrossRef]
- Undi, M.; Wilson, C.; Ominski, K.H.; Wittenberg, K.M. Comparison of techniques for estimation offorage dry matter intake bygrazing beef cattle. Can. J. Anim. Sci. 2008, 88, 693–701. [Google Scholar] [CrossRef]
- Decruyenaere, V.; Buldgen, A.; Stilmant, D. Factors affecting intake by grazing ruminants and related quantification methods: A review. BASE 2009, 13, 559–573. [Google Scholar]
- Roca Fernandez, A.I.; Gonzalez Rodriguez, A. Sward factors influence on pasture dry matter intake of grazing dairy cows: A review. Iran. J. Appl. Anim. Sci. 2013, 3, 629–651. [Google Scholar]
- Oshita, T.; Sudo, K.; Nonaka, K.; Kume, S.; Ochiai, K. The effect of feed regimen on chewing time, digesta passage rate and particle size distribution in holstein non-lactating cows fed pasture ad libitum. Livest. Sci. 2008, 113, 243–250. [Google Scholar] [CrossRef]
- Parker, W. Pasture and grazing management made simple 6: Cow grazing behavior and intake. Landcare Res. 2002. Available online: http://www.milkproduction.com/Library/Scientific-articles/Nutrition/Pasture-and-grazing-6/ (accessed on 27 September 2018).
- Schirmann, K.; Chapinal, N.; Weary, D.M.; Heuwieser, W.; von Keyserlingk, M.A.G. Rumination and its relationship to feeding and lying behavior in holstein dairy cows. J. Dairy Sci. 2012, 95, 3212–3217. [Google Scholar] [CrossRef] [PubMed]
- Kaufmann, L.D.; Münger, A.; Rérat, M.; Junghans, P.; Görs, S.; Metges, C.C.; Dohme-Meier, F. Energy expenditure of grazing cows and cows fed grass indoors as determined by the 13c bicarbonate dilution technique using an automatic blood sampling system. J. Dairy Sci. 2011, 94, 1989–2000. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, J.; Zsédely, E. Nutrition of Ruminants; Várhegyi, Józsefné, University of West-Hungary: Sopron, Hungary, 2011; pp. 56–87. [Google Scholar]
- Schären, M.; Kiri, K.; Riede, S.; Gardener, M.; Meyer, U.; Hummel, J.; Urich, T.; Breves, G.; Dänicke, S. Alterations in the rumen liquid-, particle- and epithelium-associated microbiota of dairy cows during the transition from a silage- and concentrate-based ration to pasture in spring. Front. Microbiol. 2017, 100, 2765. [Google Scholar] [CrossRef] [PubMed]
- Schären, M.; Seyfang, G.M.; Steingass, H.; Dieho, K.; Dijkstra, J.; Hüther, L.; Frahm, J.; Beinke, A.; von Soosten, D.; Meyer, U.; et al. The effects of a ration change from a total mixed ration to pasture on rumen fermentation, volatile fatty acid absorption characteristics, and morphology of dairy cows. J. Dairy Sci. 2016, 99, 3549–3565. [Google Scholar] [CrossRef] [PubMed]
- von Soosten, D.; Meyer, U.; Weber, E.M.; Rehage, J.; Flachowsky, G.; Dänicke, S. Effect of trans-10, cis-12 conjugated linoleic acid on performance, adipose depot weights, and liver weight in early-lactation dairy cows. J. Dairy Sci. 2011, 94, 2859–2870. [Google Scholar] [CrossRef] [PubMed]
- Rebuffe-Scrive, M.; Andersson, B.; Olbe, L.; Bjorntorp, P. Metabolism of adipose tissue in intraabdominal depots of nonobese men and women. Metab: Clin. Exp. 1989, 38, 453–458. [Google Scholar] [CrossRef]
- Brouwer, E. Report on sub-committee on constants and factors. Energy Metab. 1965, 441–443. [Google Scholar]
- Moe, P.W.; Tyrrell, H.F.; Flatt, W.P. Energetics of body tissue mobilization. J. Dairy Sci. 1971, 54, 548–553. [Google Scholar] [CrossRef]
- Abrahamse, P.A.; Dijkstra, J.; Vlaeminck, B.; Tamminga, S. Frequent allocation of rotationally grazed dairy cows changes grazing behavior and improves productivity. J Dairy Sci 2008, 91, 2033–2045. [Google Scholar] [CrossRef] [PubMed]
- Delaby, L.; Peyraud, J.-L.; Foucher, N.; Michel, G. The effect of two contrasting grazing managements and level of concentrate supplementation on the performance of grazing dairy cows. Anim. Res. 2003, 52, 437–460. [Google Scholar] [CrossRef]
- Morales-Almaráz, E.; Soldado, A.; González, A.; Martínez-Fernández, A.; Domínguez-Vara, I.; de la Roza-Delgado, B.; Vicente, F. Improving the fatty acid profile of dairy cow milk by combining grazing with feeding of total mixed ration. J. Dairy Res. 2010, 77, 225–230. [Google Scholar] [CrossRef] [PubMed]
- Rivero, M.J.; Anrique, R. Milk fat depression syndrome and the particular case of grazing cows: A review. Acta. Agric. Scand. A Anim. Sci. 2015, 65, 42–54. [Google Scholar] [CrossRef]
- Bargo, F.; Muller, L.D.; Delahoy, J.E.; Cassidy, T.W. Performance of high producing dairy cows with three different feeding systems combining pasture and total mixed rations. J. Dairy Sci. 2002, 85, 2948–2963. [Google Scholar] [CrossRef]
- Santana, A.; Cajarville, C.; Mendoza, A.; Repetto, J.L. Combination of legume-based herbage and total mixed ration (tmr) maintains intake and nutrient utilization of tmr and improves nitrogen utilization of herbage in heifers. Animal 2017, 11, 616–624. [Google Scholar] [CrossRef] [PubMed]
- Kohn, R. Use of milk or blood urea nitrogen to identify feed management inefficiencies and estimate nitrogen excretion by dairy cattle and other animals; Florida Ruminant Nutrition Symposium: Best Western Gateway Grand Gainesville, FL, USA, 30–31 January 2007. [Google Scholar]
- Wilson, J.R.; Deinum, B.; Engels, F.M. Temperature effects on anatomy and digestibility of leaf and stem of tropical and temperate forage species. Netherlands J. Agric. Sci. 1991, 39, 31–48. [Google Scholar]
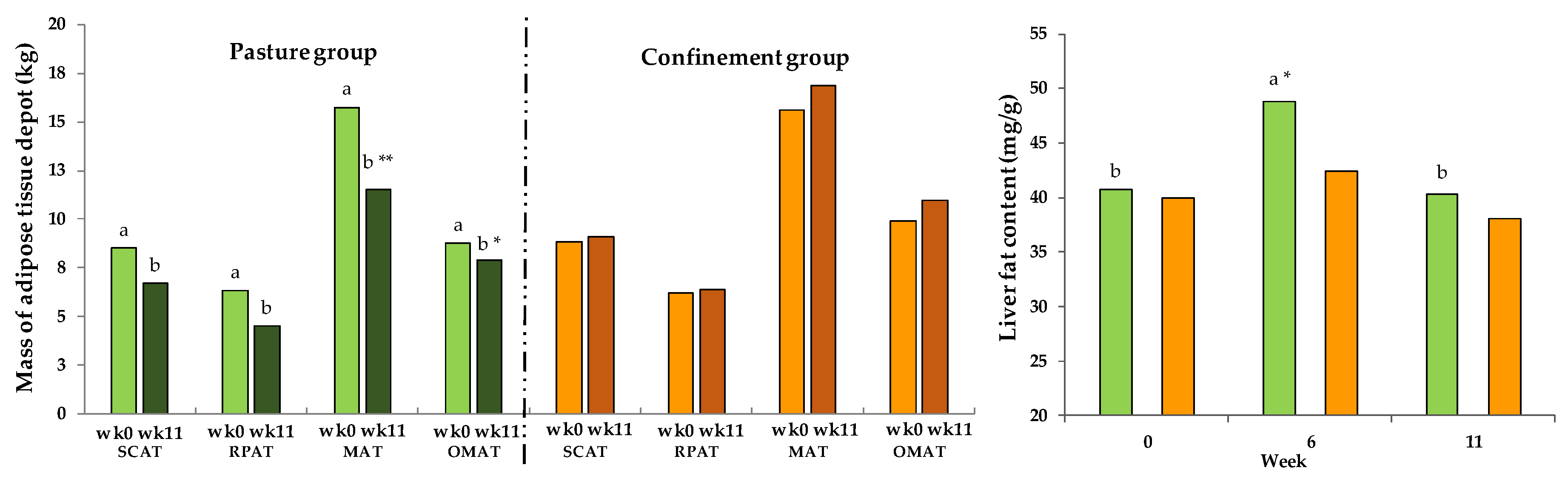
| Ingredient (% DM) | Concentrate TMR CG | Concentrate TMR PG | Concentrate PG |
|---|---|---|---|
| Soyabean meal | 20.7 | 20.7 | - |
| Rapeseed meal | 14.0 | 14.0 | - |
| Wheat | 18.2 | 18.1 | 29.6 |
| Corn | 18.2 | 18.1 | 29.6 |
| Barley | 18.2 | 18.1 | 29.6 |
| Dried sugar beet pulp | 5.0 | 5.0 | - |
| Feed lime | 1.9 | 1.9 | - |
| Soyabean oil | 1.0 | 1.0 | 1.0 |
| MgO | - | 0.3 | 1.2 |
| Urea | 1.0 | 1.0 | - |
| NaCl | 0.3 | 0.3 | - |
| Mineral feed 1 | 1.5 | 1.5 | 9.0 |
| Item (g/kg of DM) | TMR | TMR | Concentrate | Pasture (per week) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CG | PG | PG | |||||||||||
| (Week 0 to 11) | (Week 0 to 4) | (Week 5 to 11) | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | |
| DM (g/kg) | 465 | 477 | 899 | 196 | 217 | 198 | 164 | 147 | 183 | 145 | 147 | 167 | 172 |
| Ash | 66 | 6s6 | 108 | 80 | 82 | 85 | 90 | 105 | 90 | 105 | 102 | 93 | 105 |
| Cp | 157 | 159 | 93 | 213 | 199 | 188 | 178 | 168 | 201 | 168 | 198 | 209 | 155 |
| UCp | 152 | 151 | 148 | 154 | 153 | 142 | 143 | 131 | 150 | 126 | 142 | 143 | 133 |
| NEL (MJ/kg of DM) * | 6.8 | 6.8 | 7.6 | 6.9 | 6.8 | 6.4 | 6.3 | 5.7 | 6.5 | 5.6 | 6.1 | 6.1 | 5.9 |
| Sugar | 18 | 18 | 27 | 222 | 170 | 128 | 92 | 79 | 97 | 102 | 87 | 83 | 83 |
| Starch | 250 | 255 | 579 | - | - | - | - | - | - | - | - | - | - |
| RNB | 0.03 | −0.1 | −8.9 | 6.0 | 6.9 | 4.1 | 6.0 | 6.0 | 9.1 | 2.3 | 9.0 | 8.8 | 4.2 |
| CF | 184 | 184 | 31.5 | 157 | 189 | 211 | 254 | 262 | 238 | 249 | 242 | 236 | 271 |
| NDFOM | 363 | 363 | 124 | 377 | 433 | 472 | 509 | 514 | 462 | 479 | 472 | 468 | 521 |
| ADFOM | 206 | 203 | 42 | 174 | 208 | 233 | 278 | 288 | 255 | 277 | 270 | 261 | 300 |
| EE | 37 | 36 | 30 | 49 | 46 | 38 | 34 | 36 | 42 | 43 | 45 | 48 | 31 |
| Week | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | PSEM 3 | p-Value | |||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Week of Paddock Rotation * | 3–4 | 5–6.5 | 6.5–7.5 | 7.5–8.5 | 8.5–9.5 | 9.5–10.5 | 10.5–11 | Group (G) | Time (T) | GxT | |||||||
| CG/kg/day) | Water intake | 83 | 91 | 85 | 86 | 82 | 86 | 88 | 82 | 89 | 87 | 86 | 81 | 2.4 | <0.001 | <0.001 | <0.001 |
| PG (kg/day) | 80 | 76 | 67 | 46 | 31 | - | - | - | - | - | - | ||||||
| CG (kg/day DMI) | TMR | 20.5 | 21.0 | 22.5 | 21.5 | 22.4 | 22.4 | 23.0 | 21.9 | 21.9 | 23.5 | 22.7 | 20.6 | 0.5 | <0.001 | <0.001 | <0.001 |
| PG (kg/day DMI) | TMR | 20.6 | 21.1 | 17.6 | 13.0 | 11.0 | |||||||||||
| PI 1 Alkane | 14.6 | 12.8 | |||||||||||||||
| PI 1 Alkane + concentrate | 19.1 | 17.3 | |||||||||||||||
| PI 1 according to equation (1) + (2) | 2.8 | 7.1 | 7.9 | 13.1 | 14.2 | 12.0 | 14.0 | 11.7 | 13.1 | 12.8 | 0.4 | <0.001 | <0.001 | <0.001 | |||
| PI 1 Heublein + TMR + concentrate 2 | 20.4 | 20.1 | 18.9 | 17.6 | 18.7 | 16.5 | 18.5 | 16.2 | 17.6 | 17.3 | 0.4 | <0.001 | <0.001 | <0.001 | |||
| PI1 exclosure cage * | 15.8 ± 3.4 | 21.9 ± 2.4 | 15.3 ± 1.8 | 19.4 ± 7.4 | 18.5 ± 1.7 | 25.6 ± 7.5 | 20.0 ± 4.9 | ||||||||||
| PI 1 exclosure cage + concentrate 2,* | 26.4 ± 2.4 | 19.8 ± 1.8 | 20.9 ± 7.4 | 22.9 ± 1.7 | 30.1 ± 7.5 | 24.5 ± 4.9 | |||||||||||
| Variale 1 | Group 2 | Week | PSEM 3 | p-Value | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | Group (G) | Time (T) | GxT | |||
| RBC | CG | 5.59 a | 5.41 ab | 5.41 ab | 5.45 ab | 5.50 ab | 5.44 ab | 5.45 ab | 5.42 ab | 5.42 ab | 5.38 ab | 5.26 b | 5.24 b | 0.1 | 0.114 | <0.001 | <0.001 |
| PG | 5.36 c | 5.36 c | 5.33 c | 5.54 bc | 5.73 ab | 5.71 ab | 5.87 a | 5.77 ab | 5.68 ab | 5.59 abc | 5.75 ab† | 5.54 bc | |||||
| HGB | CG | 8.59 | 8.36 | 8.33 | 8.23 | 8.62 | 8.66 | 8.62 | 8.43 | 8.41 | 8.29 | 8.22 | 8.28 | 0.16 | <0.1 | <0.001 | <0.001 |
| PG | 8.21 f | 8.28 ef | 8.45 def | 8.54 def | 9.20 abc | 9.30 ab | 9.23 abc | 9.15 abc | 8.83 bcd | 8.77 cde | 9.05 abc† | 9.37 a** | |||||
| HCT | CG | 25.0 | 24.2 | 24.2 | 24.3 | 24.6 | 24.4 | 24.4 | 24.3 | 24.5 | 24.3 | 23.7 | 23.8 | 0.5 | <0.05 | <0.001 | <0.001 |
| PG | 24.2 d | 24.3 cd | 24.2 d | 25.2 bcd | 26.2 ab | 26.3 ab | 26.7 a | 26.1 ab | 25.8 abc | 25.5 abcd | 26.0 ab† | 25.3 abcd | |||||
| MCV | CG | 44.7 | 44.8 | 44.8 | 44.8 | 44.9 | 44.8 | 44.9 | 44.8 | 45.2 | 45.2 | 45.1 | 45.4 | 0.7 | 0.534 | <0.05 | <0.1 |
| PG | 45.4 b | 45.5 ab | 45.6 ab | 45.7 ab | 45.9 ab | 46.2 a | 45.6 ab | 45.5 ab | 45.6 ab | 45.8 ab | 45.4 ab | 45.8 ab | |||||
| MCH | CG | 15.4 cd | 15.5 bcd | 15.4 cd | 15.2 def | 15.7 abc | 15.9 a | 15.8 ab | 15.6 abc | 15.5 bcd | 15.4 cde | 15.6 abc | 15.8 ab | 0.3 | 0.432 | <0.001 | <0.001 |
| PG | 15.4 e | 15.5 ef | 15.9 cd | 15.5 e | 16.1 bc | 16.3 b | 15.8 cde | 15.9 cd | 15.6 de | 15.7 de | 15.8 cde | 17.0 a | |||||
| MCHC | CG | 34.4 def | 34.6 cde | 34.5 cde | 33.8 f | 35.0 abc | 35.6 a | 35.3 ab | 34.7 bcd | 34.4 def | 34.1 ef | 34.7 cde | 34.3 def | 0.1 | <0.05 | <0.001 | <0.001 |
| PG | 33.9 f | 34.1 ef | 34.9 bc | 33.9 f | 35.2 bc | 35.4 b | 34.6 cde† | 35.0 bc | 34.3 def | 34.3 def | 34.8 cde | 36.9 a** | |||||
| RDW | CG | 16.0 e | 16.1 ef | 16.1 def | 16.3 cdef | 16.3 bcdef | 16.5 abcdef | 16.7 abcdef | 16.8 abcde | 16.8 abc | 16.8 abcd | 17.0 ab | 17.1 a | 0.2 | 0.986 | <0.001 | 0.188 |
| PG | 16.2 b | 16.2 ab | 16.5 ab | 16.6 ab | 16.6 ab | 16.8 a | 16.7 ab | 16.7 ab | 16.8 ab | 16.6 ab | 16.7 ab | 16.7 ab | |||||
| PDW | CG | 15.6 d | 15.6 d | 15.7 d | 15.6 def | 15.9 d | 15.9 d | 16.0 cde | 16.6 bcd | 18.5 a | 18.1 ab | 17.9 abc | 19.3 a | 0.4 | <0.05 | <0.001 | <0.01 |
| PG | 15.6 c | 15.9 c | 15.6 c | 15.6 cdef | 15.9 c | 16.2 c | 16.0 cde | 18.4 b | 19.6 ab | 20.4 a* | 18.4 b | 19.4 ab | |||||
| Variable 1 | Group 2 | Week | PSEM 3 | p-Value | ||||
|---|---|---|---|---|---|---|---|---|
| 0 | 6 | 11 | Group (G) | Time (T) | GxT | |||
| Albumin (g/L) | CG | 36.6 | 36.6 | 36.6 | 0.6 | 0.172 | 0.785 | 0.697 |
| PG | 35.4 | 35.9 | 35.9 | |||||
| Total protein (g/L) | CG | 68.3 | 70.1 | 70.1 | 1.3 | <0.05 | <0.05 | 0.553 |
| PG | 68.1 | 71.0 | 72.5 | |||||
| Cholesterol (mg/dL) | CG | 222.8 | 211.5 | 201.0 | 7.6 | <0.05 | <0.05 | 0.133 |
| PG | 212.2 | 214.7 | 206.1 | |||||
| AST (IU/L) | CG | 82.8 | 77.3 | 74.8 | 5.3 | 0.381 | 0.355 | 0.257 |
| PG | 84.5 | 81.4 | 86.3 | |||||
| γ-GT (IU/L) | CG | 42.2 | 39.6 | 39.4 | 3.7 | 0.285 | 0.285 | 0.494 |
| PG | 43.9 | 36.4 | 44.0 | |||||
| Bilirubin (mg/dL) | CG | 0.37 | 0.36 | 0.35 | 0.01 | <0.01 | <0.01 | <0.001 |
| PG | 0.37 b | 0.42 a** | 0.36 b | |||||
| GLDH (IU/L) | CG | 35.8 | 31.7 | 30.6 | 3.4 | 0.139 | 0.139 | 0.500 |
| PG | 32.9 | 28.1 | 37.0 | |||||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hartwiger, J.; Schären, M.; Gerhards, U.; Hüther, L.; Frahm, J.; Von Soosten, D.; Klüß, J.; Bachmann, M.; Zeyner, A.; Meyer, U.; et al. Effects of a Change from an Indoor-Based Total Mixed Ration to a Rotational Pasture System Combined with a Moderate Concentrate Feed Supply on the Health and Performance of Dairy Cows. Animals 2018, 8, 169. https://doi.org/10.3390/ani8100169
Hartwiger J, Schären M, Gerhards U, Hüther L, Frahm J, Von Soosten D, Klüß J, Bachmann M, Zeyner A, Meyer U, et al. Effects of a Change from an Indoor-Based Total Mixed Ration to a Rotational Pasture System Combined with a Moderate Concentrate Feed Supply on the Health and Performance of Dairy Cows. Animals. 2018; 8(10):169. https://doi.org/10.3390/ani8100169
Chicago/Turabian StyleHartwiger, Julia, Melanie Schären, Ursula Gerhards, Liane Hüther, Jana Frahm, Dirk Von Soosten, Jeanette Klüß, Martin Bachmann, Annette Zeyner, Ulrich Meyer, and et al. 2018. "Effects of a Change from an Indoor-Based Total Mixed Ration to a Rotational Pasture System Combined with a Moderate Concentrate Feed Supply on the Health and Performance of Dairy Cows" Animals 8, no. 10: 169. https://doi.org/10.3390/ani8100169
APA StyleHartwiger, J., Schären, M., Gerhards, U., Hüther, L., Frahm, J., Von Soosten, D., Klüß, J., Bachmann, M., Zeyner, A., Meyer, U., Isselstein, J., Breves, G., & Dänicke, S. (2018). Effects of a Change from an Indoor-Based Total Mixed Ration to a Rotational Pasture System Combined with a Moderate Concentrate Feed Supply on the Health and Performance of Dairy Cows. Animals, 8(10), 169. https://doi.org/10.3390/ani8100169






