Red Wolf (Canis rufus) Recovery: A Review with Suggestions for Future Research
Abstract
:Simple Summary
Abstract
1. Introduction
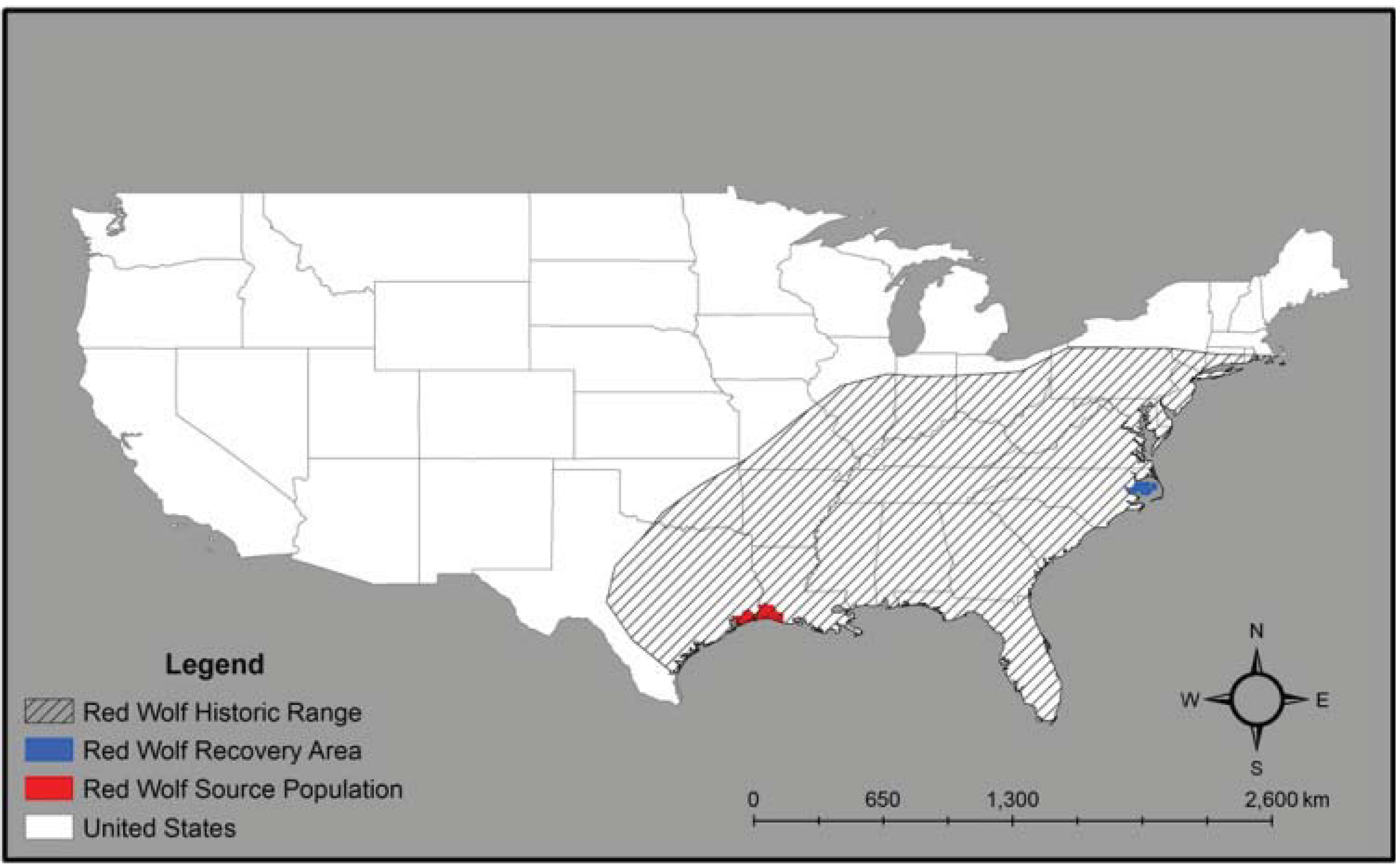
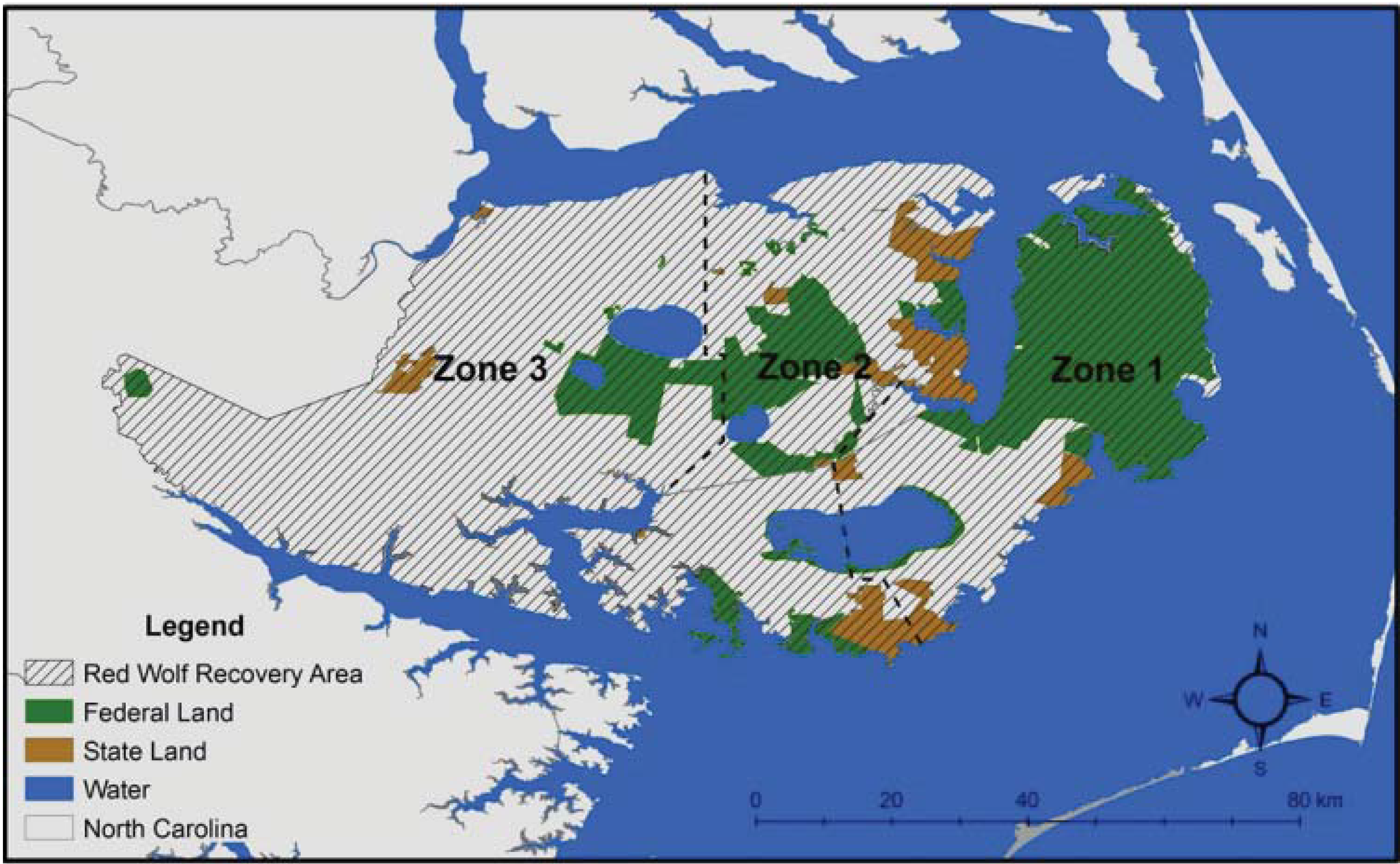
2. Red Wolf Taxonomy
3. Ecological Challenges
3.1. Red Wolf and Coyote Hybridization
3.2. Inbreeding Effects
3.3. Red Wolf Demographics
4. Conclusions
Acknowledgments
Conflict of Interest
References and Notes
- Nowak, R.M. The original status of wolves in eastern North America. Southeast. Nat. 2002, 1, 95–130. [Google Scholar] [CrossRef]
- Nowak, R.M. Wolf evolution and taxonomy. In Wolves: Behavior, Ecology, and Conservation; Mech, L.D., Boitani, L., Eds.; University of Chicago Press: Chicago, IL, USA, 2003; pp. 239–271. [Google Scholar]
- McCarley, H. The mammals of eastern Texas. J. Sci. 1959, 11, 385–426. [Google Scholar]
- McCarley, H. The taxonomic status of wild Canis (Canidae) in the south central United States. Southwest. Nat. 1962, 7, 227–235. [Google Scholar]
- Paradiso, J.L.; Nowak, R.M. Canis rufus. Mamm. Species 1972, 22, 1–4. [Google Scholar] [CrossRef]
- Carley, C.J. Activities and Findings of the Red Wolf Recovery Program from Late 1973 to July 1, 1975; U.S. Fish and Wildlife Service: Albuquerque, NM, USA, 1975; p. 215. [Google Scholar]
- Red Wolf Recovery/Species Survival Plan; U.S. Fish and Wildlife Service: Atlanta, GA, USA, 1989; p. 110.
- Carely, C.J. The Red Wolf (Canis rufus) Recovery Program: Things they didn’t tell me in school. In Reflections of a Naturalist: Papers Honoring Professor Eugene D. Fleharty; Choate, J.R., Ed.; Fort Hays State University: Hays, KS, USA, 2000; pp. 125–141. [Google Scholar]
- Phillips, M.K.; Henry, V.G.; Kelly, B.T. Restoration of the red wolf. In Wolves: Behavior, Ecology, and Conservation; Mech, L.D., Boitani, L., Eds.; University of Chicago Press: Chicago, IL, USA, 2003; pp. 272–288. [Google Scholar]
- A Proposal to Reintroduce the Red Wolf into the Great Smoky Mountains National Park; Red Wolf Management Series Technical Report No. 7; U.S. Fish and Wildlife Service: Atlanta, GA, USA, 1990; p. 33.
- Lucash, C.F.; Crawford, B.; Clark, J.D. Species repatriation: Red wolf. In Ecosystem Management for Sustainability; Piene, J.D., Ed.; Lewis Publishing: Boca Raton, FL, USA, 1998; pp. 225–246. [Google Scholar]
- Henry, V.G. Notice of termination of the red wolf reintroduction project in the Great Smoky Mountains National Park. Federal Register 1998, 63, 54152–54153. [Google Scholar]
- Red Wolf Recovery Program 3rd Quarter Report: April–June 2013; U.S. Fish and Wildlife Service: Atlanta, GA, USA, 2013; p. 9.
- Phillips, M.K.; Henry, V.G. Comments on red wolf taxonomy. Conserv. Biol. 1992, 6, 596–599. [Google Scholar]
- Nowak, R.M.; Federoff, N.E. Validity of the red wolf: Response to Roy et al. Conserv. Biol. 1998, 12, 722–725. [Google Scholar] [CrossRef]
- Population and Habitat Viability Assessment Workshop for the Red Wolf (Canis rufus); Kelly, B.T.; Miller, P.S.; Seal, U.S. (Eds.) Conservation Breeding Specialist Group (SSC/IUCN): Apple Valley, MN, USA, 1999; p. 88.
- Stoskopf, M.K.; Beck, K.; Fazio, B.B.; Fuller, T.K.; Gese, E.M.; Kelly, B.T.; Knowlton, F.F.; Murray, D.L.; Waddell, W.T.; Waits, L.P. Implementing recovery of the red wolf: Integrating research, scientists, and managers. Wildl. Soc. Bullet. 2005, 33, 1145–1152. [Google Scholar] [CrossRef]
- Red Wolf (Canis rufus) 5-Year Review: Summary and Evaluation; U.S. Fish and Wildlife Service: Atlanta, GA, USA, 2007; p. 58.
- Rabon, D.R.; Waddell, W. Effects of inbreeding on reproductive success, performance, litter size, and survival in captive red wolves (Canis rufus). Zoo Biol. 2010, 29, 36–49. [Google Scholar]
- Chambers, S.M.; Fain, S.R.; Fazio, B.; Amaral, M. An account of the taxonomy of North American wolves from morphological and genetic analyses. North Am. Fauna 2012, 77, 1–67. [Google Scholar] [CrossRef]
- Nowak, R.M. North American quaternary Canis. Monogr. Mus. Nat. Hist., Univ. Kans. 1979, 6, 1–154. [Google Scholar]
- Wayne, R.K.; Jenks, S.M. Mitochondrial DNA analysis implying extensive hybridization of the endangered red wolf Canis rufus. Nature 1991, 351, 565–568. [Google Scholar] [CrossRef]
- Wilson, P.J.; Grewal, S.; Lawford, I.D.; Heal, J.N.M.; Granacki, A.G.; Pennock, D.; Theberge, J.B.; Theberge, M.T.; Voight, D.R.; Waddell, W.; Chambers, R.E.; Paquet, P.C.; Goulet, G.; Cluff, D.; White, B.N. DNA profiles of the eastern Canadian wolf and the red wolf provide evidence for a common evolutionary history independent of the gray wolf. Can. J. Zool. 2000, 78, 2156–2166. [Google Scholar] [CrossRef]
- Bartram, W. Travels through North and South Carolina, Georgia, East and West Florida, the Cherokee Country, the Extensive Territories of the Muscogulges or Creek Confederacy, and the Country of the Chactaws. Containing an Account of the Soil and Natural Productions of Those Regions; Together with Observations on the Manners of the Indians; James & Johnson: Philadelphia, PA, USA, 1791. [Google Scholar]
- Harper, F. The name of the Florida wolf. J. Mammal. 1942, 23, 339. [Google Scholar]
- Nowak, R.M. The red wolf is not a hybrid. Conserv. Biol. 1992, 6, 593–595. [Google Scholar]
- Audubon, J.J.; Bachman, J. Viviparous Quadrupeds of North America; J.J. Audubon: New York, NY, USA, 1851; Volume 2. [Google Scholar]
- Bangs, O. The land mammals of peninsular Florida and the coast region of Georgia. Proc. Boston Soc. Nat. Hist. 1898, 28, 157–235. [Google Scholar]
- Baily, V. Biological survey of Texas. North Am. Fauna 1905, 25, 1–255. [Google Scholar] [CrossRef]
- Miller, G.S. The names of two North American wolves. Proc. Biol. Soc. Wash. 1912, 25, 95. [Google Scholar]
- Goldman, E.A. The wolves of North America. J. Mammal. 1937, 18, 37–45. [Google Scholar] [CrossRef]
- Goldman, E.A. Classification of wolves. In The Wolves of North America; Young, S.P., Goldman, E.A., Eds.; American Wildlife Institute: Washington, DC, USA, 1944; Volume 2, pp. 389–636. [Google Scholar]
- Paradiso, J.L. Recent records of red wolves from the Gulf Coast of Texas. Southwest. Nat. 1965, 10, 218–319. [Google Scholar] [CrossRef]
- Paradiso, J.L. Canids recently collected in east Texas, with comments on the taxonomy of the red wolf. Am. Midl. Nat. 1968, 80, 529–534. [Google Scholar] [CrossRef]
- Pimlott, D.H.; Joslin, P.W. The status and distribution of the red wolf. T. N. Am. Wildl. Nat. Res. 1968, 33, 373–389. [Google Scholar]
- Mech, L.D. The Wolf: The Ecology and Behavior of an Endangered Species; University of Minnesota Press: Minneapolis, MN, USA, 1970. [Google Scholar]
- Roy, M.S.; Geffen, E.; Ostrander, D.; Wayne, R.K. Patterns of differentiation and hybridization in North American wolflike canids, revealed by analysis of microsatellite loci. Mol. Biol. Evol. 1994, 11, 533–570. [Google Scholar]
- Roy, M.S.; Geffen, E.; Smith, E.; Wayne, R.K. Molecular genetics of pre-1940 red wolves. Conserv. Biol. 1996, 10, 1413–1424. [Google Scholar]
- Reich, D.E.; Wayne, R.K.; Goldstein, D.B. Genetic evidence for a recent origin by hybridization of red wolves. Mol. Ecol. 1999, 8, 139–144. [Google Scholar] [CrossRef]
- vonHoldt, B.M.; Pollinger, J.P.; Earl, D.A.; Knowles, J.C.; Boyko, A.R.; Parker, H.; Geffen, E.; Pilot, M.; Jedrzejewski, W.; Jedrzejewski, B.; Sidorovich, V.; Creco, C.; Ettore, R.; Musiani, M.; Kays, R.; Bustamante, C.D.; Ostrander, E.A.; Novembre, J.; Wayne, R.K. A genome-wide perspective on the evolutionary history of enigmatic wolf-like canids. Genome Res. 2011, 21, 1294–1305. [Google Scholar] [CrossRef]
- Nowak, R.M. Another look at wolf taxonomy. In Ecology and Conservation of Wolves in a Changing World: Proceedings of the Second North American Symposium on Wolves, Edmonton, Alberta, 25–27 August 1992; Carbyn, L.N., Fritts, S.H., Seip, D.R., Eds.; Canadian Circumpolar Institute, University of Alberta: Edmonton, AB, Canada, 1995; pp. 375–397. [Google Scholar]
- Nowak, R.M.; Federoff, N.E. Systematics of wolves in eastern North America. In Proceedings of Defenders of Wildlife’s Wolves of America Conference, Albany, NY, USA, 14–16 November 1996; pp. 187–203.
- Bertorelle, G.; Excoffier, L. Inferring admixture proportions from molecular data. Mol. Biol. Evol. 1998, 15, 1298–1311. [Google Scholar] [CrossRef]
- Hedrick, P.W.; Lee, R.N.; Garrigan, D. Major histocompatibility complex variation in red wolves: Evidence for common ancestry with coyotes and balancing selection. Mol. Ecol. 2002, 11, 1905–1913. [Google Scholar] [CrossRef]
- Adams, J.R.; Kelly, B.T.; Waits, L.P. Using faecal DNA sampling and GIS to monitor hybridization between red wolves (Canis rufus) and coyote (Canis latrans). Mol. Ecol. 2003, 12, 2175–2186. [Google Scholar] [CrossRef]
- Wilson, P.J.; Grewal, S.; McFadden, T.; Chambers, R.C.; White, B.N. DNA extracted from eastern North American wolves killed in the 1800s is not of gray wolf origin. Can. J. Zool. 2003, 81, 936–940. [Google Scholar] [CrossRef]
- Hailer, F.; Leonard, J.A. Hybridization among three native North American Canis species in a region of natural sympatry. PLoS ONE 2008, 3. [Google Scholar] [CrossRef]
- Rutledge, L.Y.; Wilson, P.J.; Klütsch, F.C.; Patterson, B.R.; White, B.N. Conservation genomics in perspective: A holistic approach to understanding Canis evolution in North America. Biol. Conserv. 2012, 155, 186–192. [Google Scholar] [CrossRef]
- Kyle, C.J.; Johnson, A.R.; Patterson, B.R.; Wilson, P.J.; Shami, K.; Grewal, S.K.; White, B.N. Genetic nature of eastern wolves: Past, present and future. Conserv. Genet. 2006, 7, 273–287. [Google Scholar] [CrossRef]
- Wheeldon, T.; Patterson, B.R.; White, B.N. Sympatric wolf and coyote populations of the western Great Lakes region are reproductively isolated. Mol. Ecol. 2010, 19, 4428–4440. [Google Scholar] [CrossRef]
- Rabon, D.R.; Bartel, R.; Beyer, A. Red Wolf Adaptive Management Plan FY13-FY15; U.S. Fish and Wildlife Service: Manteo, NC, USA, 2013. [Google Scholar]
- Miller, C.; Adams, J.; Waits, L. Pedigree based assignment tests for reversing coyote (Canis latrans) introgression into the wild red wolf (Canis rufus) population. Mol. Ecol. 2003, 12, 3287–3301. [Google Scholar] [CrossRef]
- Adams, J.R. A multi-faceted molecular approach to red wolf (Canis rufus) conservation and management. Ph.D. Thesis, University of Idaho, Moscow, ID, USA, 2006. [Google Scholar]
- Red Wolf Recovery Program, U.S. Fish and Wildlife Service: Manteo, NC, USA, Unpublished data; 2013.
- Fredrickson, R.J.; Hedrick, P.W. Dynamics of hybridization and introgression in red wolves and coyotes. Conserv. Biol. 2006, 20, 1272–1283. [Google Scholar] [CrossRef]
- Bohling, J.H. Exploring patterns and mechanisms of red wolf (Canis rufus) hybridization in North Carolina. Ph.D. Thesis, University of Idaho, Moscow, ID, USA, 2011. [Google Scholar]
- Rabon, D.R. Factors affecting reproduction in the red wolf (Canis rufus). Ph.D. Thesis, North Carolina State University, Raleigh, NC, USA, 2009. [Google Scholar]
- Jordan, P.A.; Shelton, P.C.; Allen, D.L. Numbers, turnover, and social structure of the Isle Royale wolf population. Am. Zool. 1967, 7, 233–252. [Google Scholar]
- Mech, L.D. Alpha status, dominance, and division of labor in wolf packs. Can. J. Zool. 1999, 77, 1196–1203. [Google Scholar] [CrossRef]
- Hinton, J.W.; Chamberlain, M.J. Space and habitat use by a red wolf pack and their pups during pup-rearing. J. Wildl. Manag. 2010, 74, 55–58. [Google Scholar] [CrossRef]
- Sparkman, A.M.; Adams, J.; Beyer, A.; Steury, T.D.; Waits, L.; Murray, D.L. Helper effects on pup lifetime fitness in the cooperatively breeding red wolf (Canis rufus). Proc. Roy. Soc. B. 2011, 278, 1381–1389. [Google Scholar] [CrossRef]
- Gese, E.M.; Ruff, R.L.; Crabtree, R.L. Social and nutritional factors influencing dispersal of resident coyotes. Anim. Behav. 1996, 52, 1025–1043. [Google Scholar] [CrossRef]
- Gese, E.M.; Ruff, R.L.; Crabtree, R.L. Foraging ecology of coyotes (Canis latrans): the influence of extrinsic factors and a dominance hierarchy. Can. J. Zool. 1996, 74, 769–783. [Google Scholar] [CrossRef]
- Bekoff, M.; Gese, E.M. Coyote (Canis latrans). In Wild Mammals of North America, 2nd ed.; Feldhamer, G.A., Thompson, B.C., Chapman, J.A., Eds.; John Hopkins University Press: Baltimore, MD, USA, 2003; pp. 467–481. [Google Scholar]
- Gittleman, J.L. Carnivore group living: Comparative trends. In Carnivore Behavior, Ecology, and Evolution; Gittleman, J.L., Ed.; Cornell University Press: Ithaca, NY, USA, 1989; Volume 1, pp. 183–207. [Google Scholar]
- Geffen, E.; Gompper, M.E.; Gittleman, J.L.; Hang-Kwang, L.; Macdonald, D.W.; Wayne, R.K. Size, life-history traits and social organization in the Canidae: A reevaluation. Am. Nat. 1996, 147, 140–160. [Google Scholar]
- Hinton, J.H.; Chamberlain, M.J. Morphometrical discrimination of red wolves (Canis rufus), coyotes (Canis latrans), and their hybrids in North Carolina. J. Mammal. 2013. in review. [Google Scholar]
- Rosenzweig, M.L. Community structure in sympatric Carnivora. J. Mammal. 1966, 47, 602–612. [Google Scholar] [CrossRef]
- Rosenzweig, M.L. The strategy of body size in mammalian carnivores. Am. Midl. Nat. 1968, 80, 299–315. [Google Scholar] [CrossRef]
- Gittleman, J.L. Carnivore body size: Ecological and taxonomic correlates. Oecologia 1985, 67, 540–554. [Google Scholar] [CrossRef]
- Palomares, F.; Caro, T.M. Interspecific killing among mammalian carnivores. Am. Nat. 1999, 153, 492–508. [Google Scholar] [CrossRef]
- Huxley, J.S.; Tessier, G. Terminology of relative growth. Nature 1936, 137, 780–781. [Google Scholar] [CrossRef]
- McNab, B.K. Bioenergetics and the determination of home range size. Am. Nat. 1963, 97, 133–140. [Google Scholar]
- Brown, J.H.; Nicoletto, P.F. Spatial scaling of species composition: body masses of North American land mammals. Am. Nat. 1991, 138, 1478–1512. [Google Scholar]
- Brown, J.H.; Gillooly, J.F.; Allen, A.P.; Savage, V.M.; West, G.B. Toward a metabolic theory of ecology. Ecology 2004, 85, 1771–1789. [Google Scholar] [CrossRef]
- White, E.P.; Ernest, S.K.M.; Kerkhoff, A.J.; Enquist, B.J. Relationships between body size and abundance in ecology. Trends Ecol. Evol. 2007, 22, 323–330. [Google Scholar] [CrossRef]
- Mayr, E.W. Systematics and the Origin of Species from the Viewpoint of a Zoologist; Columbia University Press: New York, NY, USA, 1941. [Google Scholar]
- Charlesworth, D.; Charlesworth, B. Inbreeding depression and its evolutionary consequences. Annu. Rev. Ecol. Syst. 1987, 18, 237–268. [Google Scholar]
- Crnokrak, P.; Roff, D.A. Inbreeding depression in the wild. Heredity 1999, 83, 260–270. [Google Scholar] [CrossRef]
- Hedrick, P.W.; Kalinowski, S.T. Inbreeding depression in conservation biology. Annu. Rev. Ecol. Syst. 2000, 31, 139–162. [Google Scholar] [CrossRef]
- Charlesworth, D.; Willis, J.H. The genetics of inbreeding depression. Nat. Rev. Genet. 2009, 10, 783–789. [Google Scholar] [CrossRef]
- Lande, R. Genetics and demography in biological conservation. Science 1988, 241, 1455–1460. [Google Scholar]
- Allendorf, F.W.; Hohenlohe, P.A.; Luikart, G. Genomics and the future of conservation genetics. Nat. Rev. Genet. 2010, 11, 697–709. [Google Scholar] [CrossRef]
- Frankham, R. Where are we in conservation genetics and where do we need to go? Conserv. Genet. 2010, 11, 661–663. [Google Scholar] [CrossRef]
- Waddell, W.; Long, S. Population Analysis and Breeding and Transfer Plan—Red Wolf (Canis rufus gregoryi) Species Survival Plan® Program. Association of Zoos & Aquariums. Available online: http://redwolfssp.org/web/ssp_recommendations/red_wolf_recommendations_2010.pdf (accessed on 15 June 2013).
- Kalinowski, S.T.; Hedrick, P.W.; Miller, P.S. No evidence for inbreeding depression in Mexican and red wolf captive breeding programs. Conserv. Biol. 1999, 13, 1371–1377. [Google Scholar] [CrossRef]
- Hedrick, P.W.; Fredrickson, J. Captive breeding and the reintroduction of Mexican and red wolves. Mol. Ecol. 2008, 17, 344–350. [Google Scholar] [CrossRef]
- Smith, D.; Meier, T.; Geffen, E.; Mech, L.D.; Burch, J.W.; Adams, L.G.; Wayne, R.K. Is incest common in gray wolf packs? Behav. Ecol. 1997, 8, 384–391. [Google Scholar] [CrossRef]
- Geffen, E.; Kam, M.; Hefner, R.; Hersteinsson, P.; Angerbjörn, A.; Dalèn, L.; Fuglei, E.; Norèn, K.; Adams, J.R.; Vucetich, J.; Meier, T.J.; Mech, L.D.; vonHoldt, B.M.; Stahler, D.R.; Wayne, R.K. Kin encounter rate and inbreeding avoidance in canids. Mol. Ecol. 2011, 20, 5348–5358. [Google Scholar] [CrossRef]
- Jankovic, I.; vonHoldt, B.M.; Rosenberg, N.A. Heterozygosity of the Yellowstone wolves. Mol. Ecol. 2010, 19, 3246–3249. [Google Scholar] [CrossRef]
- Sparkman, A.M.; Adams, J.M.; Steury, T.D.; Waits, L.P.; Murray, D.L. Pack social dynamics and inbreeding avoidance in the cooperatively breeding red wolf. Behav. Ecol. 2012, 23, 1186–1194. [Google Scholar]
- Beyer, A.; Lucash, C. Personal communication. U.S. Fish and Wildlife Service: Manteo, NC, USA, 2013. [Google Scholar]
- Snell, G.D.; Higgins, G.F. Alleles at the histocompatibility-2 locus in the mouse as determined by tumor transplantation. Genetics 1951, 36, 306–310. [Google Scholar]
- Apanius, V.; Penn, D.; Slev, P.; Ruff, L.R.; Potts, W.K. The nature of selection on the major histocompatibility complex. Crit. Rev. Immunol. 1997, 17, 179–224. [Google Scholar] [CrossRef]
- Sommer, S. The importance of immune gene variability (MHC) in evolutionary ecology and conservation. Front. Zool. 2005, 2, 1–16. [Google Scholar] [CrossRef] [Green Version]
- Milinski, M. The major histocompatibility complex, sexual selection, and mate choice. Annu. Rev. Ecol. Evol. Syst. 2006, 37, 159–186. [Google Scholar] [CrossRef]
- Huchard, E.; Knapp, L.A.; Wand, J.; Raymond, M.; Cowlishaw, G. MHC, mate choice and heterozygote advantage in a wild social primate. Mol. Ecol. 2010, 19, 2545–2561. [Google Scholar]
- Piertney, S.B.; Oliver, M.K. The evolutionary ecology of the major histocompatibility complex. Heredity 2006, 96, 7–21. [Google Scholar]
- Leslie, P.H. On the use of matrices in certain population mathematics. Biometrika 1945, 33, 183–212. [Google Scholar] [CrossRef]
- Ginzburg, L.R. The theory of population dynamics: I. back to first principles. J. Theor. Biol. 1986, 122, 385–399. [Google Scholar] [CrossRef]
- Gotelli, N.J. A Primer of Ecology, 3rd ed.; Oxford University Press: New York, NY, USA, 2001. [Google Scholar]
- Watts, H.E.; Holekamp, K.E. Ecological determinants of survival and reproduction in the spotted hyena. J. Mammal. 2009, 90, 461–471. [Google Scholar] [CrossRef]
- Stahler, D.R.; MacNulty, D.R.; Wayne, R.K.; vonHoldt, B.; Smith, D.W. The adaptive value of morphological, behavioral and life-history traits in reproductive female wolves. J. Anim. Ecol. 2013, 82, 222–234. [Google Scholar] [CrossRef]
- Willams, B.K.; Nichols, J.D.; Conroy, M.J. Analysis and Management of Animal Populations; Academic Press: San Diego, CA, USA, 2002. [Google Scholar]
- Norris, K. Managing threatened species: The ecological toolbox, evolutionary theory and declining-population paradigm. J. Appl. Ecol. 2004, 41, 413–426. [Google Scholar] [CrossRef]
- Akçakaya, H.R. Population viability analyses with demographically and spatially structured models. Ecol. Bullet. 2000, 48, 23–38. [Google Scholar]
- Akçakaya, H.R. Conservation and management for multiple species: Integrating field research and modeling into management decisions. Environ. Manage. 2000, 26, S75–S83. [Google Scholar]
- Packer, C.; Kosmala, M.; Cooley, H.S.; Brink, H.; Pintea, L.; Garshelis, D. Sport hunting, predator control and conservation of large carnivores. PLoS ONE 2009, 4. [Google Scholar] [CrossRef]
- Rutledge, L.Y.; White, B.N.; Row, J.R.; Patterson, B.R. Intense harvesting of eastern wolves facilitated hybridization with coyotes. Ecol. Evol. 2012, 2, 19–33. [Google Scholar] [CrossRef]
- Burnham, K.P.; White, G.C.; Anderson, D.R. Model selection strategy in the analysis of capture-recapture data. Biometrics 1995, 51, 888–898. [Google Scholar] [CrossRef]
- Nichols, J.D.; Hines, J.E.; Pollock, K.H.; Hinz, R.L.; Link, W.A. Estimating breeding proportions and testing hypotheses about costs of reproduction with capture-recapture data. Ecology 1994, 75, 2052–2065. [Google Scholar] [CrossRef]
- Ivan, J.; White, G.C.; Shenk, T.M. Using auxiliary telemetry information to estimate animal density from capture-recapture data. Ecology 2013, 94, 809–816. [Google Scholar] [CrossRef]
- Gittleman, J.L.; Pimm, S.L. Crying wolf in North America. Nature 1991, 351, 524–525. [Google Scholar] [CrossRef]
- Wayne, R.K.; Gittleman, J.L. The problematic red wolf. Sci. Am. 1995, 273, 36–39. [Google Scholar] [CrossRef]
- Murray, D.L.; Waits, L.P. Taxonomic status and conservation strategy of the endangered red wolf: A response to Kyle et al. (2006). Conserv. Genet. 2007, 8, 1483–1485. [Google Scholar] [CrossRef]
- Kyle, C.J.; Johnson, A.R.; Patterson, B.R.; Wilson, P.J.; White, B.N. The conspecific nature of eastern and red wolves: conservation and management implications. Conserv. Genet. 2008, 9, 699–701. [Google Scholar] [CrossRef]
- North Carolina Wildlife Resources Commission. Fox and Coyote Population Study Final Report. Available online: http://www.ncwildlife.org/Portals/0/Learning/documents/Species/Fox_CoyotePopulationsReport.pdf (accessed on 10 June 2013).
- Davidson, W.R.; Appel, M.J.; Doster, G.L.; Baker, O.E.; Brown, J.F. Disease and parasites of red foxes, gray foxes, and coyotes from commercial sources selling to fox-chasing enclosures. J. Wildl. Dis. 1992, 28, 581–589. [Google Scholar]
- Baker, O.E. Status of fox and coyote hunting enclosures in the southeastern United States. Proc. Annu. Conf. Southeast. Assoc. Fish. Wildl. Agenc. 1993, 52, 367–376. [Google Scholar]
- Lee, G.W.; Lee, K.A.; Davidson, W.R. Evaluation of fox-chasing enclosures at sites of potential introduction and establishment of Echinococcus multilocularis. J. Wildl. Dis. 1993, 29, 498–501. [Google Scholar]
- LaBarbera, M. Analyzing body size as a factor in ecology and evolution. Annu. Rev. Ecol. Syst. 1989, 20, 97–117. [Google Scholar]
- Brown, J.H.; Marquet, P.A.; Taper, M.L. Evolution of body size: Consequences of an energetic definition of fitness. Am. Nat. 1993, 142, 573–584. [Google Scholar]
- Capellini, I.; Venditti, C.; Barton, R.A. Phylogeny and metabolic scaling in mammals. Ecology 2010, 91, 2783–2793. [Google Scholar] [CrossRef]
- Agrawal, A.A. Phenotypic plasticity in the interactions and evolution of species. Science 2001, 294, 321–326. [Google Scholar] [CrossRef]
- Crispo, E.; DiBattista, J.D.; Correa, C.; Thibert-Plante, X.; McKellar, A.E.; Schwartz, A.K; Berner, D.; De León, L.F.; Hendry, A.P. The evolution of phenotypic plasticity in response to anthropogenic disturbance. Evol. Ecol. Res. 2010, 12, 47–66. [Google Scholar]
- Schluter, D. Ecology and the origin of species. Trends Ecol. Evol. 2001, 16, 327–380. [Google Scholar]
- Price, T.D.; Qvarnström, A.; Irwin, D.E. The role of phenotypic plasticity in driving genetic evolution. Proc. Roy. Soc. Lond. B. 2003, 270, 1433–1440. [Google Scholar] [CrossRef]
- Kingsolver, J.G.; Pfennig, D.W. Patterns and power of phenotypic selection in nature. Bioscience 2008, 57, 561–572. [Google Scholar] [CrossRef]
- Siepielski, A.M.; DiBattista, J.D.; Carlson, S.M. It’s about time: The temporal dynamics of phenotypic selection in the wild. Ecol. Lett. 2009, 12, 1261–1276. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hinton, J.W.; Chamberlain, M.J.; Rabon, D.R., Jr. Red Wolf (Canis rufus) Recovery: A Review with Suggestions for Future Research. Animals 2013, 3, 722-744. https://doi.org/10.3390/ani3030722
Hinton JW, Chamberlain MJ, Rabon DR Jr. Red Wolf (Canis rufus) Recovery: A Review with Suggestions for Future Research. Animals. 2013; 3(3):722-744. https://doi.org/10.3390/ani3030722
Chicago/Turabian StyleHinton, Joseph W., Michael J. Chamberlain, and David R. Rabon, Jr. 2013. "Red Wolf (Canis rufus) Recovery: A Review with Suggestions for Future Research" Animals 3, no. 3: 722-744. https://doi.org/10.3390/ani3030722
APA StyleHinton, J. W., Chamberlain, M. J., & Rabon, D. R., Jr. (2013). Red Wolf (Canis rufus) Recovery: A Review with Suggestions for Future Research. Animals, 3(3), 722-744. https://doi.org/10.3390/ani3030722



