Vaccinations and Functional Feed Supplements as Alternatives to Coccidiostats for the Control of Coccidiosis in Raising Broiler Chickens
Simple Summary
Abstract
1. Introduction
2. Coccidiosis
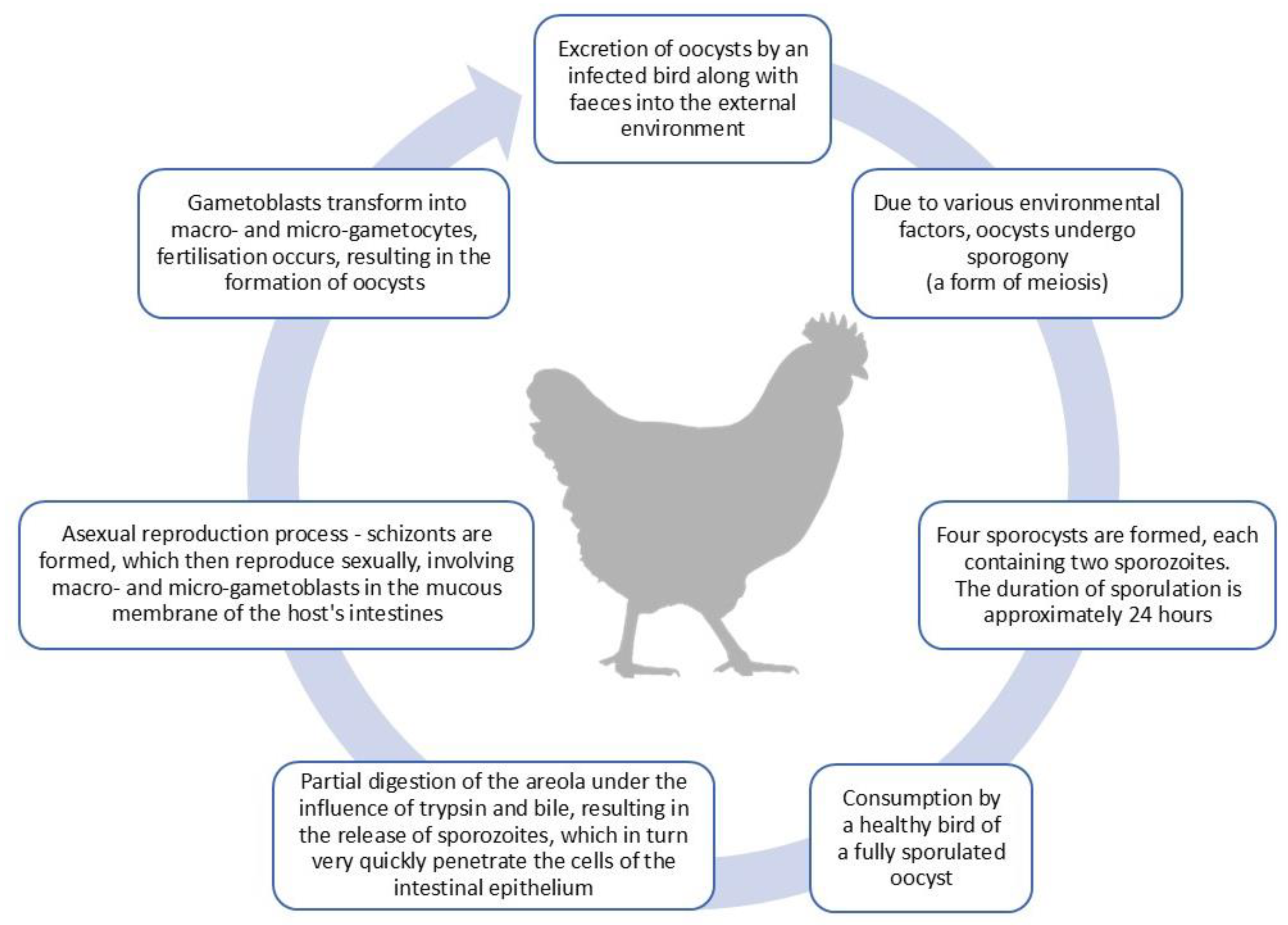
2.1. Life Cycle of Eimeria
2.2. Prevention of Coccidiosis
- isolating individual poultry houses by using separate staff, feeding, and tools;
- applying the principle “the whole poultry house full—the whole poultry house empty”;
- maintaining appropriate conditions in terms of temperature, access to light, humidity, and air exchange;
- protecting fodder against the access of wild birds and rodents;
- keeping a register of third parties entering the farm;
- using the right stocking in the poultry house;
- isolating poultry in one poultry house, keeping the herd at a uniform age;
- regular disinfestation, disinfection, and pest control, along with recording these activities;
- disinfection of the wheels of vehicles entering the farm;
- regular updating of the rodent protection plan [41].
3. Coccidiostats
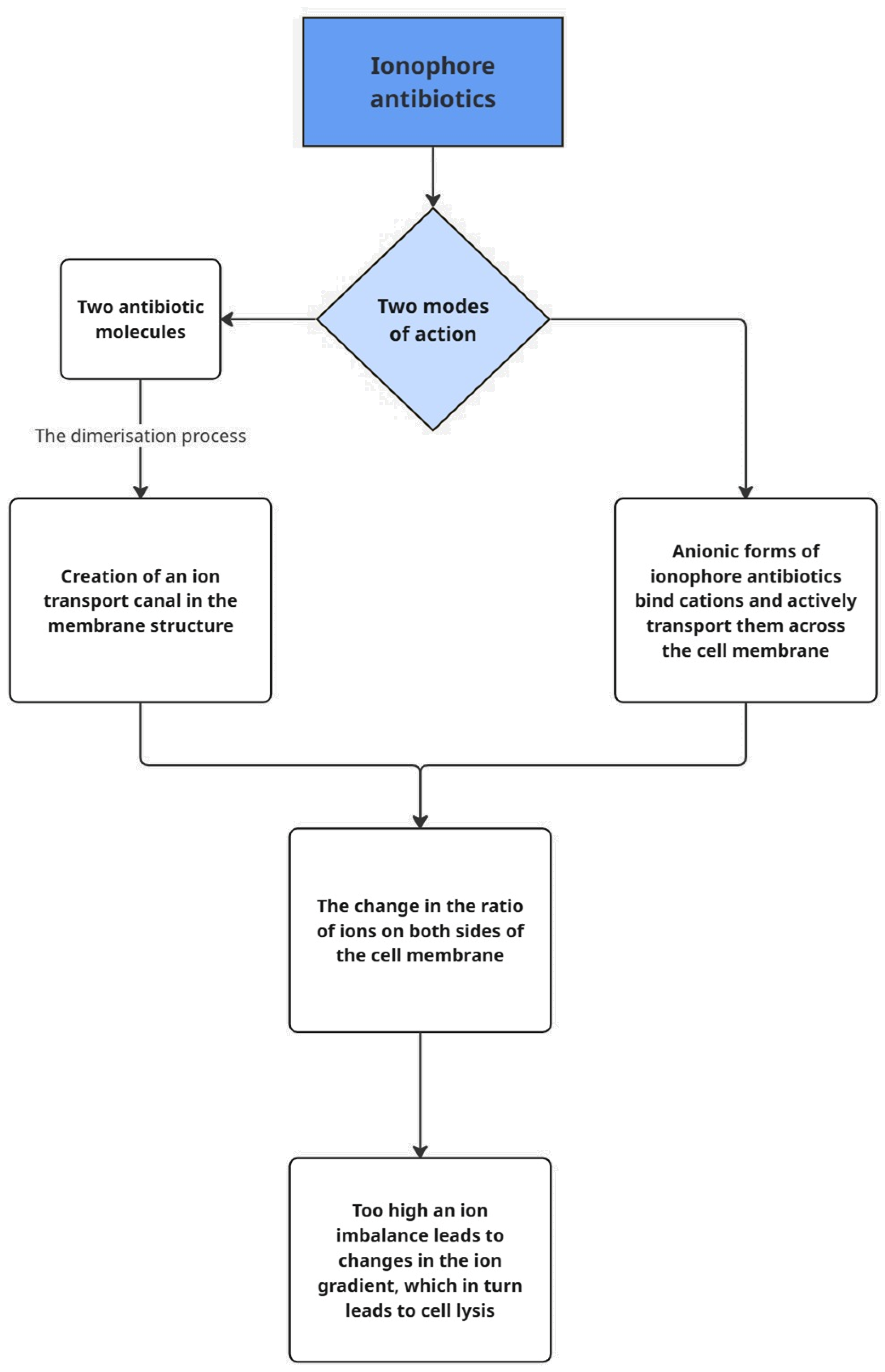
3.1. Modes of Action
3.2. Toxic Effect
3.3. Causes of Coccidiostats in Food
3.4. Resistance to Coccidiostats in Coccidia
- single-drug, where the coccidiostat is a single drug administered throughout the life of the entire flock,
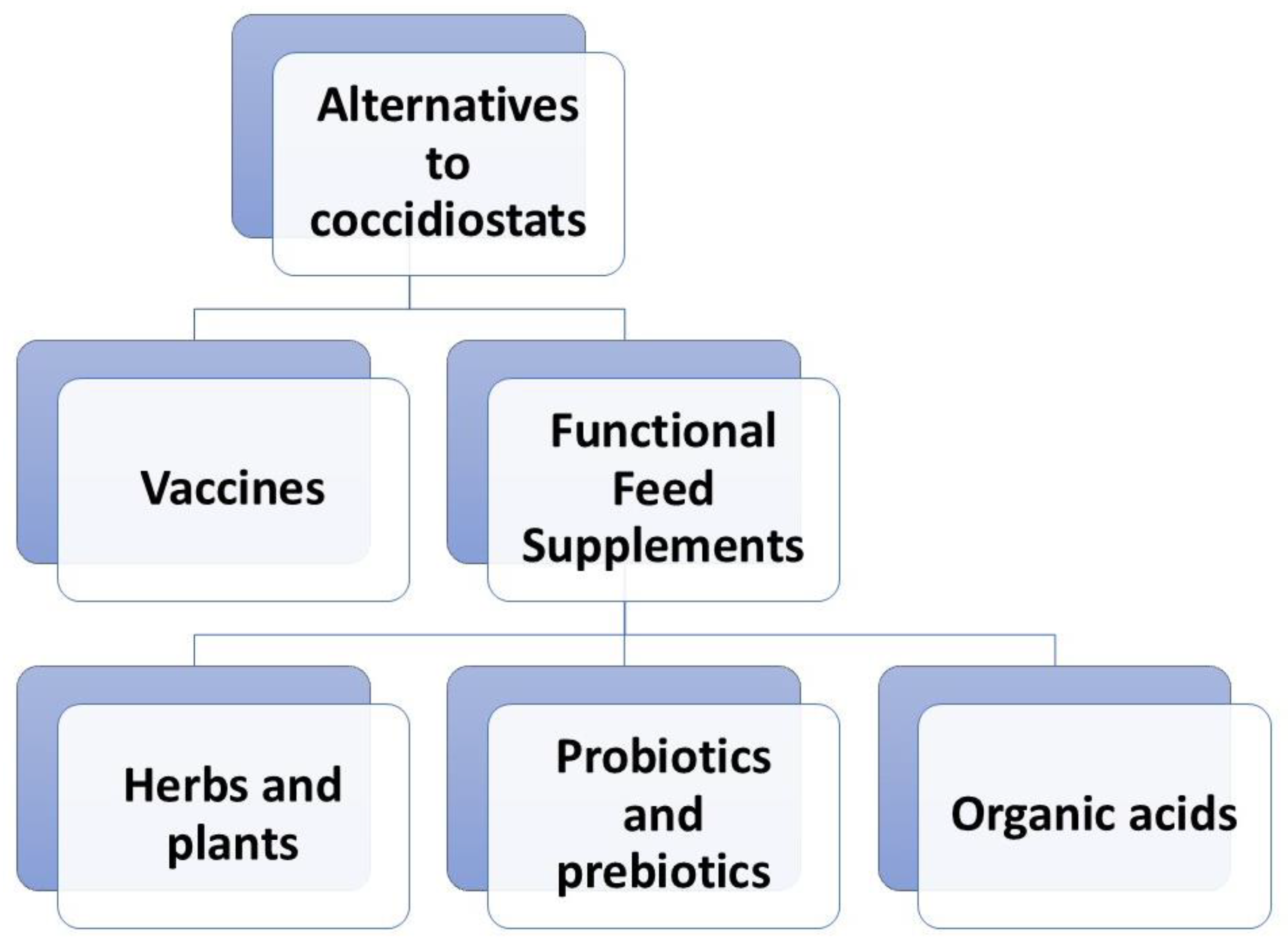
4. Alternative Methods: Vaccination and Functional Feed Supplements
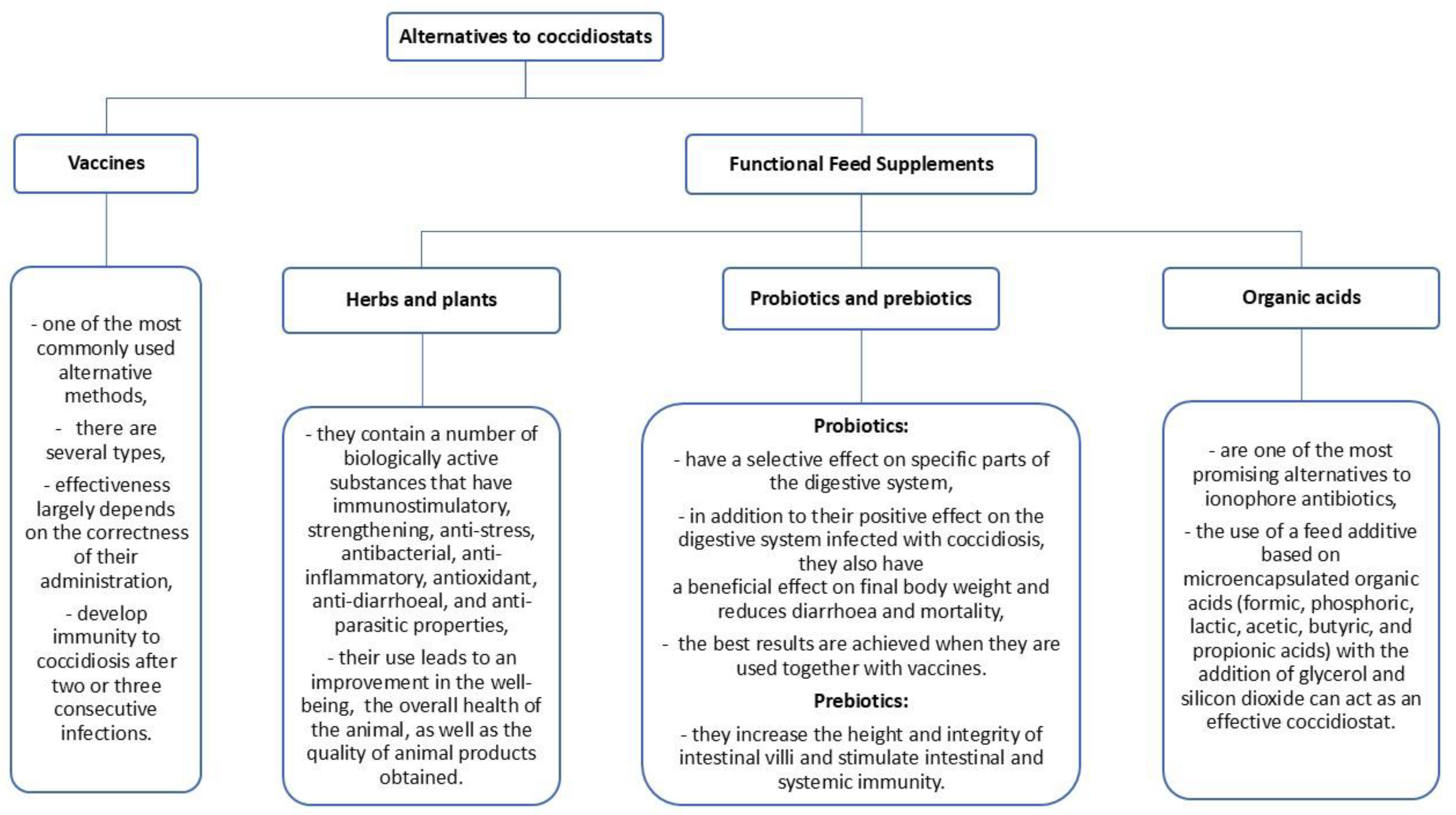
4.1. Vaccine
- Efficacy in inducing immunity against economically important Eimeria species.
- Safety for animals, other species and people.
- No negative impact on the environment.
- The content of low-virulence strains that displace more virulent strains.
- Storage stability, even under sub-optimal conditions.
- Protection against wild strains of coccidia from different geographical regions.
- Accurate, effective and easy dosage capability.
- No side effects.
- Compatibility with other poultry vaccines.
- No pollution (viruses, bacteria, fungi, chemical pollution).
- Economic competitiveness compared to other methods of coccidiosis control.
- Presence of drug-sensitive strains that may displace resistant strains [60].
- (1)
- fully virulent (non-attenuated),
- (2)
- fully virulent attenuated,
- (3)
- resistant to ionophore coccidiostats,
- (4)
- for in ovo administration and subunit vaccines with completely different principles of action than live vaccines [63].
4.2. Herbs and Plants
4.2.1. Herbs and Phytogenic Feed Supplements in the Diet of Poultry
4.2.2. Effect of Herbs and Plant Feed Supplements in Feed on the Welfare and Conditions of Poultry Farming
- single herbs or herbal mixtures, both fresh and dried,
- licks, infusions, decoctions, decoctions essential oils and mixtures of natural volatile compounds,
- herbal preparations containing a variety of plant substances in the form of aqueous, alcoholic or hydroalcoholic extracts, often standardized for the content of specific active ingredients [83].
4.2.3. Plants, Herbs and Substances Contained in Them as Agents with Coccidiostatic Properties
4.3. Probiotics and Prebiotics
- inhibiting the colonization of the bird’s intestine by pathogenic bacteria,
- adsorbing microorganisms and their toxins on its surface,
- prolonging the passage time of food contents,
- delaying gastric emptying—‘feeling of satiety’,
- stimulating the secretion of intestinal peptide hormones,
- reducing glucose absorption,
- lowering the pH of the gastrointestinal tract,
4.4. Organic Acids
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ATP | Adenosine Triphosphate |
| FCR | Feed Conversion Ratio |
| EC | European Commission |
| EU | European Union |
| EFSA | European Food Safety Authority |
| GALT | Gut-Associated Lymphoid Tissue |
| MoA | Modes of Action |
| MRLs | Maximum residue Limits |
| PLN | Polski Zloty |
| USD | United States Dollar |
References
- Bryan Salvage Global Meatconsumption to Rise 73 Percent by 2050:FAO12/14/2011. 2011. Available online: https://www.meatpoultry.com/articles/4395-global-meat-consumption-to-rise-73-percent-by-2050-fao (accessed on 17 August 2025).
- Yitbarek, M. Livestock and Livestock Product Trends by 2050: Review. Int. J. Anim. Res. 2020, 4, 30. [Google Scholar]
- John Kruse Estimating Demand for Agricultural Commodities to 2050. Global Harvest Initiative Sustainable Meeting the World Growing Need. 2010; pp. 1–26. Available online: https://pdf4pro.com/cdn/estimating-demand-for-agricultural-commodities-21712d.pdf (accessed on 17 August 2025).
- Michalczuk, M.; Łukasiewicz, M.; Wnuk, A.; Damaziak, K.; Niemiec, J. Wpływ Dostępu Do Wybiegów Na Wyniki Produkcyjne Oraz Wartość Rzeźną Kurcząt Wolno Rosnących Hubbard JA 957. Roczniki Naukowe Polskiego Towarzystwa Zootechnicznego 2013, 2, 23–31. [Google Scholar]
- Urban, J.; Michalczuk, M.; Batorska, M.; Marzec, A.; Jaroszek, A.; Bień, D. Effect of Crude Fibre Additives ARBOCEL and VITACEL on the Physicochemical Properties of Granulated Feed Mixtures for Broiler Chickens. Anim. Biosci. 2024, 37, 274–283. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.; Lu, M.; Lillehoj, H.S. Coccidiosis: Recent Progress in Host Immunity and Alternatives to Antibiotic Strategies. Vaccines 2022, 10, 215. [Google Scholar] [CrossRef] [PubMed]
- Lillehoj, H.S.; Lillehoj, E.P. Avian Coccidiosis. A Review of Acquired Intestinal Immunity and Vaccination Strategies. Avian Dis. 2000, 44, 408. [Google Scholar] [CrossRef]
- Gaweł, A.; Bobusia, K.; Bobrek, K. Identyfikacja Gatunków Eimeria Spp. Występujących u Kur i Kurcząt Brojlerów Na Terenie Polski. Med. Weter. 2015, 71, 382–384. [Google Scholar]
- Tewari, A.K.; Maharana, B.R. Control of Poultry Coccidiosis: Changing Trends. J. Parasit. Dis. 2011, 35, 10–17. [Google Scholar] [CrossRef]
- Lillehoj, H.S. Role of T Lymphocytes and Cytokines in Coccidiosis. Int. J. Parasitol. 1998, 28, 1071–1081. [Google Scholar] [CrossRef]
- El-Shall, N.A.; Abd El-Hack, M.E.; Albaqami, N.M.; Khafaga, A.F.; Taha, A.E.; Swelum, A.A.; El-Saadony, M.T.; Salem, H.M.; El-Tahan, A.M.; AbuQamar, S.F.; et al. Phytochemical Control of Poultry Coccidiosis: A Review. Poult. Sci. 2022, 101, 101542. [Google Scholar] [CrossRef]
- Gardiner, J.L. The Severity of Cecal Coccidiosis Infection in Chickens as Related to the Age of the Host and the Number of Oocysts Ingested. Poult. Sci. 1955, 34, 415–420. [Google Scholar] [CrossRef]
- Al-Natour, M.Q.; Suleiman, M.M.; Abo-Shehada, M.N. Flock-Level Prevalence of Eimeria Species among Broiler Chicks in Northern Jordan. Prev. Vet. Med. 2002, 53, 305–310. [Google Scholar] [CrossRef] [PubMed]
- Reza Razmi, G.; Ali Kalideri, G. Prevalence of Subclinical Coccidiosis in Broiler-Chicken Farms in the Municipality of Mashhad, Khorasan, Iran. Prev. Vet. Med. 2000, 44, 247–253. [Google Scholar] [CrossRef] [PubMed]
- Doner, S.; Szeleszczuk, P.; Żbikowski, A. Kokcydioza Kur. Życie Weterynaryjne 2019, 7, 494–502. [Google Scholar]
- Mazurkiewicz, M. Choroby Drobiu; Wydawnictwo Akademii Rolniczej we Wrocławiu: Wrocław, Poland, 2011. [Google Scholar]
- Meireles, M.; Roberto, L.; Riera, R. Identification of Eimeria mitis and Eimeria praecox in Broiler Feces Using Polymerase Chain Reaction. Rev. Bras. Cienc. Avic. 2004, 6, 249–252. [Google Scholar] [CrossRef]
- Jorgensen, W.; Stewart, N.; Jeston, P.; Molloy, J.; Blight, G.; Dalgliesh, R. Isolation and Pathogenicity of Australian Strains of Eimeria praecox and Eimeria mitis. Aust. Vet. J. 1997, 75, 592–595. [Google Scholar] [CrossRef]
- Blake, D.P.; Clark, E.L.; Macdonald, S.E.; Thenmozhi, V.; Kundu, K.; Garg, R.; Jatau, I.D.; Ayoade, S.; Kawahara, F.; Moftah, A.; et al. Population, Genetic, and Antigenic Diversity of the Apicomplexan Eimeria tenella and Their Relevance to Vaccine Development. Proc. Natl. Acad. Sci. USA 2015, 112, E5343–E5350. [Google Scholar] [CrossRef]
- Shirley, M.W.; Smith, A.L.; Tomley, F.M. The Biology of Avian Eimeria with an Emphasis on Their Control by Vaccination. Adv. Parasitol. 2005, 60, 285–330. [Google Scholar]
- Dalloul, R.A.; Lillehoj, H.S. Poultry Coccidiosis: Recent Advancements in Control Measures and Vaccine Development. Expert Rev. Vaccines 2006, 5, 143–163. [Google Scholar] [CrossRef]
- Szeleszczuk, P. Chemioprofilaktyka Kokcydiozy Drobiu. Magazyn Weterynaryjny 2005, 5, 59–64. [Google Scholar]
- Szeleszczuk, P.; Doner, S.; Nerc, J. Wstępna Próba Oceny Strat Finansowych Spowodowanych Kokcydiozą w Produkcji Kurcząt Brojlerów. In Proceedings of the I Międzynarodowa Konf. Tech. EIMERIANA AVIA. Kokcydioza Drobiu—Aktualne Wyzwania AD 2016, Wrocław, Poland, 26–27 February 2016; pp. 89–98. [Google Scholar]
- De Gussem, M. Coccidiosis in Poultry: Review on Diagnosis, Control, Prevention and Interaction with Overall Gut Health. Available online: https://www.researchgate.net/publication/242089929_Coccidiosis_in_poultry_Review_on_diagnosis_control_prevention_and_interaction_with_overall_gut_health (accessed on 4 April 2025).
- Haug, A.; Gjevre, A.-G.; Skjerve, E.; Kaldhusdal, M. A Survey of the Economic Impact of Subclinical Eimeria Infections in Broiler Chickens in Norway. Avian Pathol. 2008, 37, 333–341. [Google Scholar] [CrossRef]
- Williams, R.B. A Compartmentalised Model for the Estimation of the Cost of Coccidiosis to the World’s Chicken Production Industry. Int. J. Parasitol. 1999, 29, 1209–1229. [Google Scholar] [CrossRef]
- Dardi, M.; Bosh, M.P.; Benitez, E.M.; Rubio, J. Występowanie Eimeria Praecox w Europie—Wstępne Wyniki Badań Przeprowadzonych w Stadach Brojlerów Kurzych w Belgii, Hiszpanii i We Włoszech. Mag. Weterynaryjny. Chor. Drobiu—Monografia. Mat. Konf. Firmy Hipra Polanica Zdrój 2011, 10–14. [Google Scholar]
- Trout, J.M.; Lillehoj, H.S. Eimeria Acervulina Infection: Evidence for the Involvement of CD8+ T Lymphocytes in Sporozoite Transport and Host Protection. Poult. Sci. 1995, 74, 1117–1125. [Google Scholar] [CrossRef] [PubMed]
- Nesse, L.L.; Bakke, A.M.; Eggen, T.; Hoel, K.; Kaldhusdal, M.; Ringø, E.; Yazdankhah, S.P.; Lock, E.-J.; Olsen, R.E.; Ørnsrud, R.; et al. The Risk of Development of Antimicrobial Resistance with the Use of Coccidiostats in Poultry Diets. Eur. J. Nutr. Food Saf. 2019, 11, 40–43. [Google Scholar] [CrossRef]
- Urban, J.; Kareem, K.Y.; Matuszewski, A.; Bień, D.; Ciborowska, P.; Lutostański, K.; Michalczuk, M. Enhancing Broiler Chicken Health and Performance: The Impact of Phytobiotics on Growth, Gut Microbiota, Antioxidants, and Immunity. Phytochem. Rev. 2024, 24, 2131–2145. [Google Scholar] [CrossRef]
- Fantham, H.B. The Morphology and Life-History of Eimeria (Coccidium) avium: A Sporozoö Causing a Fatal Disease among Young Grouse. Proc. Zool. Soc. Lond. 1910, 80, 672–691. [Google Scholar] [CrossRef]
- Allen, P.C.; Fetterer, R.H. Recent Advances in Biology and Immunobiology of Eimeria Species and in Diagnosis and Control of Infection with These Coccidian Parasites of Poultry. Clin. Microbiol. Rev. 2002, 15, 58–65. [Google Scholar] [CrossRef]
- Jeurissen, S.H.M.; Janse, E.M.; Vermeulen, A.N.; Vervelde, L. Eimeria Tenella Infections in Chickens: Aspects of Host-Parasite: Interaction. Vet. Immunol. Immunopathol. 1996, 54, 231–238. [Google Scholar] [CrossRef]
- Levine, P.P. The Effects of Sulfanilamide on the Course of Experimental Avian Coccidiosis. Cornell Vet. 1936, 29, 309–322. [Google Scholar]
- Mehlorn, H. Encyclopedia of Parasitology, 3rd ed.; Springer: Berlin/Heilderberg, Germany, 2008. [Google Scholar]
- Markiewicz, W.; Barski, D.; Tomaszewska, E.; Burmańczuk, A. Toxicity of Salinomycin and Narasin in Turkeys. J. Elem. 2014, 19, 903–914. [Google Scholar] [CrossRef]
- Tykałowski, B.; Śmiałek, M.; Łoś, A. Kokcydioza u Kur—Opis Przypadku Klinicznego; Warmińsko-Mazurski Portal Weterynaryjny: Olsztyn, Poland, 2015. [Google Scholar]
- McManus, E.C.; Campbell, W.C.; Cuckler, A.C. Development of Resistance to Quinoline Coccidiostats under Field and Laboratory Conditions. J. Parasitol. 1968, 54, 1190–1193. [Google Scholar] [CrossRef]
- Fabia, K.; Wolski, D.; Bieńko, M.; Radzki, R.P.; Szymańczyk, S. The Problem of Coccidiosis in Broiler Chickens and Laying Hens: Selected Control Methods and Alternative Solutions. Med. Weter. 2021, 77, 6571. [Google Scholar] [CrossRef]
- Horvath-Papp, I. Zastosowanie Chemioprofilaktyki w Programach Kontroli Kokcydiozy Brojlerów Kurzych—Stan Obecny i Perspektywy. Magazyn Weterynaryjny 2010, 398–404. [Google Scholar]
- Bień, D.; Urban, J.; Żbikowska, K.; Michalczuk, M. Kokcydioza Drobiu—Problem Nadal Aktualny. Środowisko Przyrodnicze Jako Obszar Badań 2021, 3, 9–19. [Google Scholar]
- Clarke, L.; Fodey, T.L.; Crooks, S.R.H.; Moloney, M.; O’Mahony, J.; Delahaut, P.; O’Kennedy, R.; Danaher, M. A Review of Coccidiostats and the Analysis of Their Residues in Meat and Other Food. Meat Sci. 2014, 97, 358–374. [Google Scholar] [CrossRef] [PubMed]
- Bergen, W.G.; Bates, D.B. Ionophores: Their Effect on Production Efficiency and Mode of Action. J. Anim. Sci. 1984, 58, 1465–1483. [Google Scholar] [CrossRef] [PubMed]
- Rybicki, M. Coccidiostats in Treating Coccidiosis. Żywność Nauka—Technologia—Jakość 2020, 4, 127–137. [Google Scholar] [CrossRef]
- Kart, A.; Bilgili, A. Ionophore Antibiotics: Toxicity, Mode of Action and Neurotoxic Aspect of Carboxylic Ionophores. J. Anim. Vet. Adv. 2008, 7, 748–751. [Google Scholar]
- Chapman, H.D. Biochemical, Genetic and Applied Aspects of Drug Resistance in Eimeria Parasites of the Fowl. Avian Pathol. 1997, 26, 221–244. [Google Scholar] [CrossRef]
- Olejnik, M.; Szprengier-Juszkiewicz, T. Pozostałości Kokcydiostatyków w Tkankach Drobiu i Jajach. Med. Weter. 2007, 63, 1539–1545. [Google Scholar]
- Szprengier-Juszkiewicz, T. Pozostałości Kokcydiostatyków w Żywności Zwierzęcego Pochodzenia. Med. Weter. 1999, 55, 356–361. [Google Scholar]
- Martins, R.R.; Pereira, A.M.P.T.; Silva, L.J.G.; Esteves, A.; Duarte, S.C.; Freitas, A.; Pena, A. Risk Assessment of Nine Coccidiostats in Commercial and Home-Raised Eggs. Foods 2023, 12, 1225. [Google Scholar] [CrossRef]
- VKM. The Risk of Development of Antimicrobial Resistance with the Use of Coccidiostats in Poultry Diets. Opinion of the the Panel on Animal Feed of the Norwegian Scientific Committee for Food Safety; VKM: Oslo, Norway, 2015; Volume 5, p. 24. [Google Scholar]
- Joyner, L.P.; Davies, S.F.M.; Kendall, S.B. Chemotherapy of Coccidiosis. In Experimental Chemotherapy; Schnitzer, R., Hawking, F., Eds.; Academic Press: New York, NY, USA, 1963; pp. 445–486. [Google Scholar]
- Abbas, R.Z.; Iqbal, Z.; Blake, D.; Khan, M.N.; Saleemi, M.K. Anticoccidial Drug Resistance in Fowl Coccidia: The State of Play Revisited. Worlds Poult. Sci. J. 2011, 67, 337–350. [Google Scholar] [CrossRef]
- Waletzky, E.; Neal, R.; Hable, I. A Field Strain of Eimeria Tenella Resistant to Sulfonamides. J. Parasitol. 1954, 40, 24. [Google Scholar]
- Peek, H.W. Resistance to Anticoccidial Drugs. Alternative Strategies to Control Coccidiosis in Broilers. Ph.D. Thesis, University of Utrecht, Utrecht, The Netherlands, 2010. [Google Scholar]
- Kohls, R.E. Comparison of Shuttle and Single Drug Coccidiostat Programs. In Proceedings of the 15th World Poultry Congress, New Orleans, LA, USA, 11–16 August 1974; pp. 95–97. [Google Scholar]
- Gard, D.; Schlegel, B.; Garcia, E.; Murphy, C.; Tonkinsin, L. Continuous Monensin Programs vs Shuttle Programs for Broilers. In Proceedings of the World Poultry Congress, Rio de Janeiro, Brazil, 17–21 September 1978; pp. 896–902. [Google Scholar]
- Watkins, K.L.; Bafundo, K.W. Effect of Anticoccidial Programs on Broiler Performance. J. Appl. Poult. Res. 1993, 2, 55–60. [Google Scholar] [CrossRef]
- Chapman, H. Rotation Programmes for Coccidiosis Control. Chapman H. Rotation programmes for coccidiosis control. Int. Poult. Product 2007, 15, 7–9. [Google Scholar]
- Chapman, H.D. Evaluation of the Efficacy of Anticoccidial Drugs against Eimeria Species in the Fowl. Int. J. Parasitol. 1998, 28, 1141–1144. [Google Scholar] [CrossRef]
- Chapman, H.D.; Roberts, B.; Shirley, M.W.; Williams, R.B. Guidelines for Evaluating the Efficacy and Safety of Live Anticoccidial Vaccines, and Obtaining Approval for Their Use in Chickens and Turkeys. Avian Pathol. 2005, 34, 279–290. [Google Scholar] [CrossRef]
- Chapman, H.D.; Cherry, T.E.; Danforth, H.D.; Richards, G.; Shirley, M.W.; Williams, R.B. Sustainable Coccidiosis Control in Poultry Production: The Role of Live Vaccines. Int. J. Parasitol. 2002, 32, 617–629. [Google Scholar] [CrossRef]
- Joyner, L.P.; Norton, C.C. The Immunity Arising from Continuous Low-Level Infection with Eimeria tenella. Parasitology 1973, 67, 333–340. [Google Scholar] [CrossRef]
- Bobusia, K.; Bobrek, K.; Gaweł, A. Wybrane Zagadnienia Immunoprofilaktyki Kokcydiozy Kur i Kurcząt Brojlerów. Życie Weterynaryjne 2014, 89, 759–762. [Google Scholar]
- Chapman, H.D.; Barta, J.R.; Blake, D.; Gruber, A.; Jenkins, M.; Smith, N.C.; Suo, X.; Tomley, F.M. A Selective Review of Advances in Coccidiosis Research. Adv. Parasitol. 2013, 83, 93–171. [Google Scholar]
- Williams, R.B. Anticoccidial Vaccines for Broiler Chickens: Pathways to Success. Avian Pathol. 2002, 31, 317–353. [Google Scholar] [CrossRef]
- Vermeulen, A.N.; Schaap, D.C.; Schetters, T.P.M. Control of Coccidiosis in Chickens by Vaccination. Vet. Parasitol. 2001, 100, 13–20. [Google Scholar] [CrossRef]
- Chapman, H.D. Practical Use of Vaccines for the Control of Coccidiosis in the Chicken. Worlds Poult. Sci. J. 2000, 56, 7–20. [Google Scholar] [CrossRef]
- Trout, J.M.; Lillehoi, H.S. Evidence of a Role for Intestinal CD8+ Lymphocytes and Macrophages in Transport of Eimeria acervulina Sporozoites. J. Parasitol. 1993, 79, 790–792. [Google Scholar] [CrossRef]
- Vervelde, L.; Vermeulen, A.N.; Jeurissen, S.H. Common Epitopes on Eimeria tenella Sporozoites and Cecal Epithelium of Chickens. Infect. Immun. 1993, 61, 4504–4506. [Google Scholar] [CrossRef]
- Hong, Y.H.; Lillehoj, H.S.; Lee, S.H.; Dalloul, R.A.; Lillehoj, E.P. Analysis of Chicken Cytokine and Chemokine Gene Expression Following Eimeria acervulina and Eimeria tenella Infections. Vet. Immunol. Immunopathol. 2006, 114, 209–223. [Google Scholar] [CrossRef] [PubMed]
- Williams, R.B. Epidemiological Studies of Coccidiosis in the Domesticated Fowl (Gallus gallus): IV. Reciprocity between the Immune Status of Floor-Reared Chickens and Their Excretion of Oocysts. Appl. Parasitol. 1995, 36, 290–298. [Google Scholar] [PubMed]
- Lillehoj, H.S. Effects of Immunosuppression on Avian Coccidiosis: Cyclosporin A but Not Hormonal Bursectomy Abrogates Host Protective Immunity. Infect. Immun. 1987, 55, 1616–1621. [Google Scholar] [CrossRef]
- Shirley, M.W.; Bedrník, P. Live Attenuated Vaccines against Avian Coccidiosis: Success with Precocious and Egg-Adapted Lines of Eimeria. Parasitol. Today 1997, 13, 481–484. [Google Scholar] [CrossRef]
- McDonald, V.; Shirley, M.W.; Millard, B.J. A Comparative Study of Two Lines of Eimeria tenella Attenuated Either by Selection for Precocious Development in the Chicken or by Growth in Chicken Embryos. Avian Pathol. 1986, 15, 323–335. [Google Scholar] [CrossRef] [PubMed]
- Shirley, M.W.; Millard, B.J. Studies on the Immunogenicity of Seven Attenuated Lines of Eimeria given as a Mixture to Chickens. Avian Pathol. 1986, 15, 629–638. [Google Scholar] [CrossRef] [PubMed]
- Williams, R.B.; Carlyle, W.W.H.; Bond, D.R.; Brown, I.A.G. The Efficacy and Economic Benefits of Paracox®, Alive Attenuated Anticoccidial Vaccine, in Commercial Trials with Standard Broiler Chickens in the United Kingdom. Int. J. Parasitol. 1999, 29, 341–355. [Google Scholar] [CrossRef]
- Hashemipour, H.; Kermanshahi, H.; Golian, A.; Veldkamp, T. Effect of Thymol and Carvacrol Feed Supplementation on Performance, Antioxidant Enzyme Activities, Fatty Acid Composition, Digestive Enzyme Activities, and Immune Response in Broiler Chickens. Poult. Sci. 2013, 92, 2059–2069. [Google Scholar] [CrossRef]
- Urban, J.; Bień, D.; Matuszewski, A.; Ciborowska, P.; Zalewska, A.; Lutostański, K.; Batorska, M.; Michalczuk, M. Fitobiotyki—Naturalne Dodatki Poprawiające Zdrowotność. Hodowca Drobiu 2023, 4, 20–23. [Google Scholar]
- Cowan, M.M. Plant Products as Antimicrobial Agents. Clin. Microbiol. Rev. 1999, 12, 564–582. [Google Scholar] [CrossRef]
- Makała, H. Herbs and Phytogenic Feed Additives in Poultry Nutrition. Med. Weter. 2022, 77, 6589. [Google Scholar] [CrossRef]
- Windisch, W.; Schedle, K.; Plitzner, C.; Kroismayr, A. Use of Phytogenic Products as Feed Additives for Swine and Poultry. J. Anim. Sci. 2008, 86, E140–E148. [Google Scholar] [CrossRef]
- Stajnder, E. Wykorzystanie roślin wysokobiałkowych w żywieniu drobiu. In Dział Technologii Produkcji Rolniczej i Doświadczalnictwa. MODR z s. w Karniowicach; Małopolski Ośrodek Doradztwa Rolniczego: Karnowice, Poland, 2019; pp. 1–14. [Google Scholar]
- Grela, E.R. Alternatywne Dla Soi Pasze Białkowe w Żywieniu Świń i Drobiu. Życie Weterynaryjne 2020, 95, 480–486. [Google Scholar]
- Frankić, T.; Volić, M.; Salobir, J.; Rezar, V. Use of Herbs and Spices and Their Extracts in Animal Nutrition. Acta Agric. Slov. 2009, 94, 95–102. [Google Scholar] [CrossRef]
- Fawaz, M.A.; Ismail, Z.S.H.; Hassan, H.A.; Abdel-Wareth, A.A.A. Effect of Thyme Essential Oil on Productive Performance of Broiler Chickens A-Review. SVU-Int. J. Environ. Res. 2021, 3, 8–18. [Google Scholar] [CrossRef]
- Przybyszewski, J.; Urban, J.; Michalczuk, M. Rośliny Lecznicze Jako Alternatywa Dla Antybiotykowych Stymulatorów Wzrostu w Odchowie Brojlerów Kurzych. Natl. Res. Inst. Anim. Prod. 2025, 52, 49–60. [Google Scholar]
- Hassan, A.H.A.; Youssef, I.M.I.; Abdel-Atty, N.S.; Abdel-Daim, A.S.A. Effect of Thyme, Ginger, and Their Nano-Particles on Growth Performance, Carcass Characteristics, Meat Quality and Intestinal Bacteriology of Broiler Chickens. BMC Vet. Res. 2024, 20, 269. [Google Scholar] [CrossRef] [PubMed]
- Alfaro, D.M.; Silva, A.V.F.; Borges, S.A.; Maiorka, F.A.; Vargas, S.; Santin, E. Use of Yucca Schidigera Extract in Broiler Diets and Its Effects on Performance Results Obtained with Different Coccidiosis Control Methods. J. Appl. Poult. Res. 2007, 16, 248–254. [Google Scholar] [CrossRef]
- Yan, Y.; Li, X.; Zhang, C.; Lv, L.; Gao, B.; Li, M. Research Progress on Antibacterial Activities and Mechanisms of Natural Alkaloids: A Review. Antibiotics 2021, 10, 318. [Google Scholar] [CrossRef]
- Hascoët, A.-S.; Torres-Celpa, P.; Riquelme-Neira, R.; Hidalgo-Olate, H. Evaluation of the Effectiveness of a Phytogenic Supplement (Alkaloids and Flavonoids) in the Control of Eimeria Spp. in Experimentally Challenged Broiler Chickens. Animals 2025, 15, 847. [Google Scholar] [CrossRef]
- Li, J.-Y.; Huang, H.-B.; Pan, T.-X.; Wang, N.; Shi, C.-W.; Zhang, B.; Wang, C.-F.; Yang, G.-L. Sanguinarine Induces Apoptosis in Eimeria tenella Sporozoites via the Generation of Reactive Oxygen Species. Poult. Sci. 2022, 101, 101771. [Google Scholar] [CrossRef]
- Nguyen, B.T.; Flores, R.A.; Cammayo, P.L.T.; Kim, S.; Kim, W.H.; Min, W. Anticoccidial Activity of Berberine against Eimeria-Infected Chickens. Korean J. Parasitol. 2021, 59, 403–408. [Google Scholar] [CrossRef]
- Wang, M.L.; Suo, X.; Gu, J.H.; Zhang, W.W.; Fang, Q.; Wang, X. Influence of Grape Seed Proanthocyanidin Extract in Broiler Chickens: Effect on Chicken Coccidiosis and Antioxidant Status. Poult. Sci. 2008, 87, 2273–2280. [Google Scholar] [CrossRef]
- Jang, S.I.; Jun, M.-H.; Lillehoj, H.S.; Dalloul, R.A.; Kong, I.-K.; Kim, S.; Min, W. Anticoccidial Effect of Green Tea-Based Diets against Eimeria maxima. Vet. Parasitol. 2007, 144, 172–175. [Google Scholar] [CrossRef]
- Abd El-Hack, M.E.; Mahgoub, S.A.; Alagawany, M.; Ashour, E.A. Improving Productive Performance and Mitigating Harmful Emissions from Laying Hen Excreta via Feeding on Graded Levels of Corn DDGS with or without Bacillus subtilis Probiotic. J. Anim. Physiol. Anim. Nutr. 2017, 101, 904–913. [Google Scholar] [CrossRef] [PubMed]
- Alagawany, M.; Abd El-Hack, M.E.; Farag, M.R.; Sachan, S.; Karthik, K.; Dhama, K. The Use of Probiotics as Eco-Friendly Alternatives for Antibiotics in Poultry Nutrition. Environ. Sci. Pollut. Res. 2018, 25, 10611–10618. [Google Scholar] [CrossRef] [PubMed]
- Świątkiewicz, S.; Świątkiewicz, M. Zastosowanie Fruktanów o Właściwościach Prebiotycznych w Żywieniu Zwierząt Gospodarskich. Med. Weter. 2008, 8, 987–990. [Google Scholar]
- Gibson, G.R.; Roberfroid, M.B. Dietary Modulation of the Human Colonic Microbiota: Introducing the Concept of Prebiotics. J. Nutr. 1995, 125, 1401–1412. [Google Scholar] [CrossRef]
- El-Sawah, A.A.; Aboelhadid, S.M.; El-Nahass, E.N.; Helal, H.E.; Korany, A.M.; El-Ashram, S. Efficacy of Probiotic Enterococcus faecium in Combination with Diclazuril against Coccidiosis in Experimentally Infected Broilers. J. Appl. Microbiol. 2020, 129, 1020–1028. [Google Scholar] [CrossRef]
- Lee, S.H.; Lillehoj, H.S.; Dalloul, R.A.; Park, D.W.; Hong, Y.H.; Lin, J.J. Influence of Pediococcus-Based Probiotic on Coccidiosis in Broiler Chickens. Poult. Sci. 2007, 86, 63–66. [Google Scholar] [CrossRef]
- Dalloul, R.; Lillehoj, H.; Shellem, T.; Doerr, J. Enhanced Mucosal Immunity against Eimeria Acervulina in Broilers Fed a Lactobacillus-Based Probiotic. Poult. Sci. 2003, 82, 62–66. [Google Scholar] [CrossRef]
- Ritzi, M.M.; Abdelrahman, W.; van-Heerden, K.; Mohnl, M.; Barrett, N.W.; Dalloul, R.A. Combination of Probiotics and Coccidiosis Vaccine Enhances Protection against an Eimeria Challenge. Vet. Res. 2016, 47, 111. [Google Scholar] [CrossRef]
- Elmusharaf, M.A.; Bautista, V.; Nollet, L.; Beynen, A.C. Effect of a Mannanoligosaccharide Preparation on Eimeria tenella Infection in Broiler Chickens. Int. J. Poult. Sci. 2006, 5, 583–588. [Google Scholar] [CrossRef]
- Fernandez, F.; Hinton, M.; Gils, B. Van Dietary Mannan-Oligosaccharides and Their Effect on Chicken Caecal Microflora in Relation to Salmonella Enteritidis Colonization. Avian Pathol. 2002, 31, 49–58. [Google Scholar] [CrossRef]
- Ahmed, S.T.; Islam, M.M.; Mun, H.-S.; Sim, H.-J.; Kim, Y.-J.; Yang, C.-J. Effects ofBacillus amyloliquefaciens as a Probiotic Strain on Growth Performance, Cecal Microflora, and Fecal Noxious Gas Emissions of Broiler Chickens. Poult. Sci. 2014, 93, 1963–1971. [Google Scholar] [CrossRef]
- Giannenas, I.; Papadopoulos, E.; Tsalie, E.; Triantafillou, E.; Henikl, S.; Teichmann, K.; Tontis, D. Assessment of Dietary Supplementation with Probiotics on Performance, Intestinal Morphology and Microflora of Chickens Infected with Eimeria tenella. Vet. Parasitol. 2012, 188, 31–40. [Google Scholar] [CrossRef] [PubMed]
- Zaghari, M.; Zahroojian, N.; Riahi, M.; Parhizkar, S. Effect of Bacillus subtilis Spore (GalliPro®) Nutrients Equivalency Value on Broiler Chicken Performance. Ital. J. Anim. Sci. 2015, 14, 3555. [Google Scholar] [CrossRef]
- Lutful Kabir, S.M. The Role of Probiotics in the Poultry Industry. Int. J. Mol. Sci. 2009, 10, 3531–3546. [Google Scholar] [CrossRef] [PubMed]
- Urban, J.; Roguski, M.; Rygało-Galewska, A. The use of pro- and prebiotics in poultry nutrition. In Problems of Sustainable Development in Agriculture Including Renewable Energy Production and Environment Protection; Romaniuk, W., Mazur, K., Eds.; Institute of Technology and Life Sciences National Research Institute Branch Warsaw: Warsaw, Poland, 2023; Volume 29, pp. 90–98. [Google Scholar]
- Ricke, S.C. Prebiotics and Alternative Poultry Production. Poult. Sci. 2021, 100, 101174. [Google Scholar] [CrossRef]
- Mizak, L.; Gryko, R.; Kwiatek, M.; Parasion, S. Probiotyki w Żywieniu Zwierząt. Życie Weterynaryjne 2012, 87, 736–742. [Google Scholar]
- Strous, G.J.; Dekker, J. Mucin-Type Glycoproteins. Crit. Rev. Biochem. Mol. Biol. 1992, 27, 57–92. [Google Scholar] [CrossRef]
- Mustafa, A.; Bai, S.; Zeng, Q.; Ding, X.; Wang, J.; Xuan, Y.; Su, Z.; Zhang, K. Effect of Organic Acids on Growth Performance, Intestinal Morphology, and Immunity of Broiler Chickens with and without Coccidial Challenge. AMB Express 2021, 11, 140. [Google Scholar] [CrossRef]
- Ali, A.M.; Seddiek, S.A.; Khater, H.F. Effect of Butyrate, Clopidol and Their Combination on the Performance of Broilers Infected with Eimeria Maxima. Br. Poult. Sci. 2014, 55, 474–482. [Google Scholar] [CrossRef]
- Abdullahi, A.Y.; Yu, X.G.; Fu, Y.Q.; Wang, M.W.; Qi, N.S.; Xia, M.H.; Kallon, S.; Pan, W.D.; Shi, X.L.; Fang, Y.; et al. Effects of Dietary Supplement of Organic Acids Induced Protective Immunity against Coccidiosis. Iran. J. Appl. Anim. Sci. 2020, 10, 119–129. [Google Scholar]
- Ziobro, D.; Wengerska, K.; Rokicka, K.; Vasiukov, K.; Bagrowski, R.; Batkowska, J. Alternatywy dla antybiotyków jonoforowych w zwalczaniu pasożytów Eimeria spp. Available online: https://www.researchgate.net/publication/355210926_Alternatywy_dla_antybiotykow_jonoforowych_w_zwalczaniu_pasozytow_Eimeria_spp (accessed on 28 July 2025).
- Galli, G.M.; Aniecevski, E.; Petrolli, T.G.; Rosa, G.d.; Boiago, M.M.; Simões, C.A.D.P.; Wagner, R.; Copetti, P.M.; Morsch, V.M.; Araujo, D.N.; et al. Growth Performance and Meat Quality of Broilers Fed with Microencapsulated Organic Acids. Anim. Feed. Sci. Technol. 2021, 271, 114706. [Google Scholar] [CrossRef]
- Taherpour, K.; Moravej, H.; Reza Taheri, H.; Shivazad, M. Effect of Dietary Inclusion of Probiotic, Prebiotic and Butyric Acid Glycerides on Resistance against Coccidiosis in Broiler Chickens. J. Poult. Sci. 2012, 49, 57–61. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rosłoń, M.; Majewski, E.; Gębska, M.; Grontkowska, A.; Motrenko, M.; Żbikowski, A.; Michalczuk, M.; Urban, J. Vaccinations and Functional Feed Supplements as Alternatives to Coccidiostats for the Control of Coccidiosis in Raising Broiler Chickens. Animals 2025, 15, 2548. https://doi.org/10.3390/ani15172548
Rosłoń M, Majewski E, Gębska M, Grontkowska A, Motrenko M, Żbikowski A, Michalczuk M, Urban J. Vaccinations and Functional Feed Supplements as Alternatives to Coccidiostats for the Control of Coccidiosis in Raising Broiler Chickens. Animals. 2025; 15(17):2548. https://doi.org/10.3390/ani15172548
Chicago/Turabian StyleRosłoń, Maciej, Edward Majewski, Monika Gębska, Anna Grontkowska, Michał Motrenko, Artur Żbikowski, Monika Michalczuk, and Jakub Urban. 2025. "Vaccinations and Functional Feed Supplements as Alternatives to Coccidiostats for the Control of Coccidiosis in Raising Broiler Chickens" Animals 15, no. 17: 2548. https://doi.org/10.3390/ani15172548
APA StyleRosłoń, M., Majewski, E., Gębska, M., Grontkowska, A., Motrenko, M., Żbikowski, A., Michalczuk, M., & Urban, J. (2025). Vaccinations and Functional Feed Supplements as Alternatives to Coccidiostats for the Control of Coccidiosis in Raising Broiler Chickens. Animals, 15(17), 2548. https://doi.org/10.3390/ani15172548