Phytochemical-Mediated Modulation of Doramectin Pharmacology in Sheep: Exploring the Cinnamaldehyde–Pink Grapefruit Combination
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Evaluation of the Efficacy of the Coadministration of DRM and CNM–PGF
2.2. Ex Vivo Assessment of Intestinal Transport-Mediated Interactions
2.3. In Vivo Pharmacokinetic/Pharmacodynamic Assessment of DRM Modulation
2.4. Analytical Procedures
2.4.1. Chromatographic Analysis
2.4.2. Intestinal Efflux Analysis
2.5. Data Analysis
3. Results
3.1. Evaluation of the Efficacy of the Coadministration of DRM and CNM–PGF
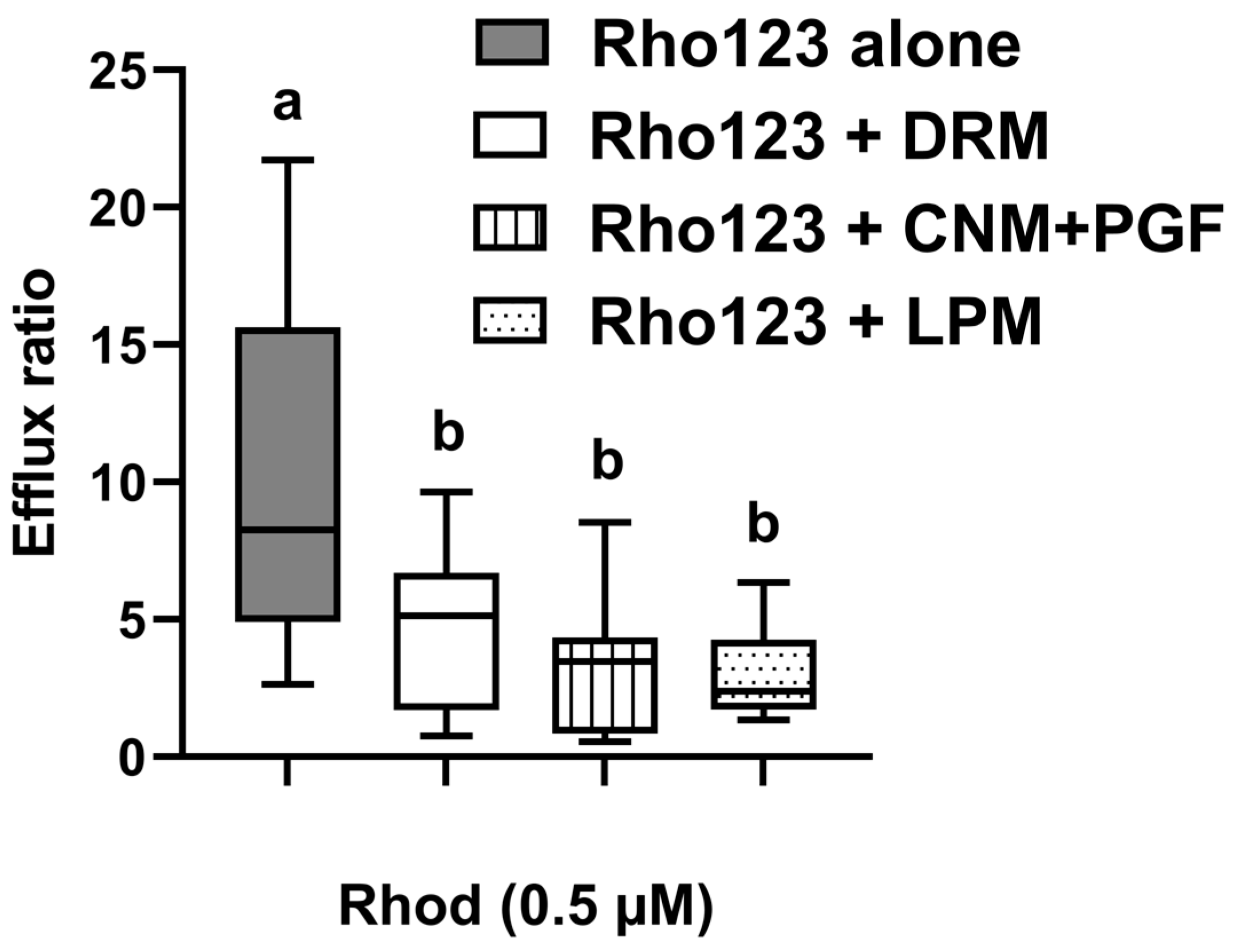
3.2. Ex Vivo Assessment of Intestinal Transport-Mediated Interactions
3.3. In Vivo Pharmacokinetic/Pharmacodynamic Assessment of DRM Modulation
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kaplan, R.M.; Vidyashankar, A.N. An inconvenient truth: Global worming and anthelmintic resistance. Vet. Parasitol. 2012, 186, 70–78. [Google Scholar] [CrossRef]
- Sutherland, I.A.; Leathwick, D.M. Anthelmintic resistance in nematode parasites of cattle: A global issue? Trends Parasitol. 2011, 27, 176–181. [Google Scholar] [CrossRef]
- Sauermann, C.; Waghorn, T.; Miller, C.; Leathwick, D. Simultaneous resistance to multiple anthelmintic classes in nematode parasites of cattle in New Zealand. Vet. Parasitol. 2024, 325, 110079. [Google Scholar] [CrossRef]
- Ghosh, R.; Andersen, E.C.; Shapiro, J.A.; Gerke, J.P.; Kruglyak, L. Natural variation in a chloride channel subunit confers avermectin resistance in C. elegans. Science 2012, 335, 574–578. [Google Scholar] [CrossRef]
- Kotze, A.C.; Prichard, R.K. Anthelmintic Resistance in Haemonchus contortus: History, Mechanisms and Diagnosis. Adv. Parasitol. 2016, 93, 397–428. [Google Scholar] [CrossRef]
- David, M.; Lebrun, C.; Duguet, T.; Talmont, F.; Beech, R.; Orlowski, S.; André, F.; Prichard, R.K.; Lespine, A. Structural model, functional modulation by ivermectin and tissue localization of Haemonchus contortus P-glycoprotein-13. Int. J. Parasitol. Drugs Drug Resist. 2018, 8, 145–157. [Google Scholar] [CrossRef] [PubMed]
- Lespine, A.; Blancfuney, C.; Prichard, R.; Alberich, M. P-glycoproteins in anthelmintic safety, efficacy, and resistance. Trends Parasitol. 2024, 40, 896–913. [Google Scholar] [CrossRef]
- Lloberas, M.; Alvarez, L.; Entrocasso, C.; Virkel, G.; Ballent, M.; Mate, L.; Lanusse, C.; Lifschitz, A. Comparative tissue pharmacokinetics and efficacy of moxidectin, abamectin and ivermectin in lambs infected with resistant nematodes: Impact of drug treatments on parasite P-glycoprotein expression. Int. J. Parasitol. Drugs Drug Resist. 2013, 3, 20–27. [Google Scholar] [CrossRef] [PubMed]
- Godoy, P.; Lian, J.; Beech, R.N.; Prichard, R.K. Haemonchus contortus P-glycoprotein-2: In situ localisation and characterisation of macrocyclic lactone transport. Int. J. Parasitol. 2015, 45, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Bartley, D.J.; McAllister, H.; Bartley, Y.; Dupuy, J.; Ménez, C.; Alvinerie, M.; Jackson, F.; Lespine, A. P-glycoprotein interfering agents potentiate ivermectin susceptibility in ivermectin sensitive and resistant isolates of Teladorsagia circumcincta and Haemonchus contortus. Parasitology 2009, 136, 1081–1088. [Google Scholar] [CrossRef]
- Lifschitz, A.L.; Entrocasso, C.; Alvarez, L.I.; Lloberas, M.; Ballent, M.; Manazza, G.; Virkel, G.; Borda, B.; Lanusse, C.E. Interference with P-glycoprotein improves ivermectin activity against adult resistant nematodes in sheep. Vet. Parasitol. 2010, 172, 291–298. [Google Scholar] [CrossRef]
- Raza, A.; Kopp, S.R.; Kotze, A.C. Synergism between ivermectin and the tyrosine kinase/P-glycoprotein inhibitor crizotinib against Haemonchus contortus larvae in vitro. Vet. Parasitol. 2016, 227, 64–68. [Google Scholar] [CrossRef]
- Nixon, S.A.; Welz, C.; Woods, D.J.; Costa-Junior, L.; Zamanian, M.; Martin, R.J. Where are all the anthelmintics? Challenges and opportunities on the path to new anthelmintics. Int. J. Parasitol. Drugs Drug Resist. 2020, 14, 8–16. [Google Scholar] [CrossRef]
- Ferreira, L.E.; Benincasa, B.I.; Fachin, A.L.; França, S.C.; Contini, S.S.H.T.; Chagas, A.C.S.; Beleboni, R.O. Thymus vulgaris L. essential oil and its main component thymol: Anthelmintic effects against Haemonchus contortus from sheep. Vet. Parasitol. 2016, 228, 70–76. [Google Scholar] [CrossRef]
- Katiki, L.M.; Araujo, R.C.; Ziegelmeyer, L.; Gomes, A.C.P.; Gutmanis, G.; Rodrigues, L.; Bueno, M.S.; Veríssimo, C.J.; Louvandini, H.; Ferreira, J.F.S.; et al. Evaluation of encapsulated anethole and carvone in lambs artificially-and naturally-infected with Haemonchus contortus. Exp. Parasitol. 2019, 197, 36–42. [Google Scholar] [CrossRef]
- Miró, M.V.; E Silva, C.R.; Viviani, P.; Luque, S.; Lloberas, M.; Costa-Júnior, L.M.; Lanusse, C.; Virkel, G.; Lifschitz, A. Combination of bioactive phytochemicals and synthetic anthelmintics: In vivo and in vitro assessment of the albendazole-thymol association. Vet. Parasitol. 2020, 281, 109121. [Google Scholar] [CrossRef]
- Miró, M.V.; Costa-Júnior, L.M.; Lloberas, M.; Cardozo, P.; Lanusse, C.; Virkel, G.; Lifschitz, A. Phytochemicals in Gastrointestinal Nematode Control: Pharmacokinetic-Pharmacodynamic Evaluation of the Ivermectin plus Carvone Combination. Animals 2023, 13, 1287. [Google Scholar] [CrossRef]
- Aguiar, A.A.R.M.; Filho, J.V.A.; Pinheiro, H.N.; Campelo, M.D.S.; Ribeiro, W.L.C.; Melo, A.C.F.L.; Rocha, L.O.D.; Ribeiro, M.E.N.P.; Ricardo, N.M.P.S.; Abreu, F.O.M.D.S.; et al. In vitro anthelmintic activity of an R-carvone nanoemulsions towards multiresistant Haemonchus contortus. Parasitology 2022, 149, 1631–1641. [Google Scholar] [CrossRef]
- Wang, S.; Liu, Y.; Cui, M.; Zou, X.; Pan, M.; Jiang, D.; Chang, T.; Xu, H.; Wu, D.; Chu, X. Cinnamaldehyde enhances the intervention effect of puerarin on stroke from the perspectives of pharmacokinetics and pharmacodynamics. Eur. J. Pharmacol. 2025, 1000, 177732. [Google Scholar] [CrossRef]
- Wang, E.J.; Casciano, C.N.; Clement, R.P.; Johnson, W.W. Inhibition of P-glycoprotein transport function by grapefruit juice psoralen. Pharm. Res. 2001, 18, 432–438. [Google Scholar] [CrossRef]
- Pacheco, P.A.; Louvandini, H.; Giglioti, R.; Wedy, B.C.R.; Ribeiro, J.C.; Verissimo, C.J.; Ferreira, J.F.D.S.; Amarante, A.F.T.D.; Katiki, L.M. Phytochemical modulation of P-Glycoprotein and its gene expression in an ivermectin-resistant Haemonchus contortus isolate in vitro. Vet. Parasitol. 2022, 305, 109713. [Google Scholar] [CrossRef]
- Katiki, L.M.; Giglioti, R.; Ferreira, J.F.S.; Pacheco, P.A.; Barbosa, H.Z.; Rodrigues, L.; Veríssimo, C.J.; Braga, P.A.C.; Amarante, A.F.T.; Louvandini, H. Combined effects of Limonene and Ivermectin on P-glycoprotein-9 gene expression of lambs Infected with Haemonchus contortus. Vet. Parasitol. 2023, 324, 110069. [Google Scholar] [CrossRef]
- Lespine, A.; Martin, S.; Dupuy, J.; Roulet, A.; Pineau, T.; Orlowski, S.; Alvinerie, M. Interaction of macrocyclic lactones with P-glycoprotein: Structure-affinity relationship. Eur. J. Pharm. Sci. 2007, 30, 84–94. [Google Scholar] [CrossRef]
- MAFF (Ministry of Agriculture, Fisheries and Food). Manual of Veterinary Parasitological Laboratory Techniques, 3rd ed.; Her Majesty’s Stationery Office: London, UK, 1986.
- Eneroth, A.; Astrom, E.; Hoogstraate, J.; Schrenk, D.; Conrad, S.; Kauffmann, H.M.; Gjellan, K. Evaluation of a vincristine resistant Caco-2 cell line for use in a calcein AM extrusion screening assay for P-glycoprotein interaction. Eur. J. Pharm. Sci. 2001, 12, 205–214. [Google Scholar] [CrossRef]
- Wandel, C.; Kim, R.; Wood, M.; Wood, A. Interaction of morphine, fentanyl, sufentanil, alfentanil, and loperamide with the efflux drug transporter P-glycoprotein. Anesthesiology 2002, 96, 913–920. [Google Scholar] [CrossRef]
- Alvinerie, M.; Sutra, J.F.; Galtier, P. Ivermectin in goat plasma and milk after subcutaneous injection. Vet. Res. 1993, 24, 417–421. [Google Scholar]
- Lifschitz, A.; Virkel, G.; Pis, A.; Imperiale, F.; Sanchez, S.; Alvarez, L.; Kujanek, R.; Lanusse, C. Ivermectin disposition kinetics after subcutaneous and intramuscular administration of an oil-based formulation to cattle. Vet. Parasitol. 1999, 86, 203–215. [Google Scholar] [CrossRef]
- de Montigny, P.; Shim, J.S.K.; Pivnichny, J.V. Liquid chromatographic determination of ivermectin in animal plasma with trifluoroacetic anhydride and N-methylimidazole as the derivatization reagent. J. Pharm. Biomed. Anal. 1990, 8, 507–511. [Google Scholar] [CrossRef]
- Ballent, M.; Maté, L.; Virkel, G.; Sallovitz, J.; Viviani, P.; Lanusse, C.; Lifschitz, A. Intestinal drug transport: Ex vivo evaluation of the interactions between ABC transporters and anthelmintic molecules. J. Vet. Pharmacol. Ther. 2014, 37, 332–337. [Google Scholar] [CrossRef]
- Lennernäs, H. Human jejunal effective permeability and its correlation with preclinical drug absorption models. J. Pharm. Pharmacol. 1997, 49, 627–638. [Google Scholar] [CrossRef]
- Gibaldi, M.; Perrier, D. Pharmacokinetics, 2nd ed.; Marcel Dekker: New York, NY, USA, 1982; pp. 45–109. [Google Scholar]
- McKenna, P. The detection of anthelmintic resistance by the faecal egg count reduction test: An examination of some of the factors affecting performance and interpretation. N. Z. Vet. J. 1990, 38, 142–147. [Google Scholar] [CrossRef]
- Kaplan, R.M.; Denwood, M.J.; Nielsen, M.K.; Thamsborg, S.M.; Torgerson, P.R.; Gilleard, J.S.; Dobson, R.J.; Vercruysse, J.; Levecke, B. World Association for the Advancement of Veterinary Parasitology (WAAVP) guideline for diagnosing anthelmintic resistance using the faecal egg count reduction test in ruminants, horses and swine. Vet. Parasitol. 2023, 318, 109936. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.W.; Kim, M.J.; Chung, B.Y.; Bang, D.Y.; Lim, S.K.; Choi, S.M.; Lim, D.S.; Cho, M.C.; Yoon, K.; Kim, H.S.; et al. Safety evaluation and risk assessment of d-Limonene. J. Toxicol. Environ. Health Part B Crit. Rev. 2013, 16, 17–38. [Google Scholar] [CrossRef] [PubMed]
- Han, R.; Li, X.; Gao, X.; Lv, G. Cinnamaldehyde: Pharmacokinetics, anticancer properties and therapeutic potential (Review). Mol. Med. Rep. 2024, 30, 163. [Google Scholar] [CrossRef]
- Štrbac, F.; Bosco, A.; Maurelli, M.P.; Ratajac, R.; Stojanović, D.; Simin, N.; Orčić, D.; Pušić, I.; Krnjajić, S.; Sotiraki, S.; et al. Anthelmintic Properties of Essential Oils to Control Gastrointestinal Nematodes in Sheep-In Vitro and In Vivo Studies. Vet. Sci. 2022, 9, 93. [Google Scholar] [CrossRef]
- Varga-Visi, É.; Nagy, G.; Csivincsik, Á.; Tóth, T. Evaluation of a Phytogenic Feed Supplement Containing Carvacrol and Limonene on Sheep Performance and Parasitological Status on a Hungarian Milking Sheep Farm. Vet. Sci. 2023, 10, 369. [Google Scholar] [CrossRef] [PubMed]
- Štrbac, F.; Krnjajić, S.; Ratajac, R.; Rinaldi, L.; Musella, V.; Castagna, F.; Stojanović, D.; Simin, N.; Orčić, D.; Bosco, A. Anthelmintic activity of winter savory (Satureja montana L.) essential oil against gastrointestinal nematodes of sheep. BMC Vet. Res. 2025, 21, 405. [Google Scholar] [CrossRef]
- Katiki, L.M.; Barbieri, A.M.E.; Araujo, R.C.; Veríssimo, C.J.; Louvandini, H.; Ferreira, J.F.S. Synergistic interaction of ten essential oils against Haemonchus contortus in vitro. Vet. Parasitol. 2017, 243, 47–51. [Google Scholar] [CrossRef]
- Lu, L.; Shu, C.; Chen, L.; Yang, Y.; Ma, S.; Zhu, K.; Shi, B. Insecticidal activity and mechanism of cinnamaldehyde in C. elegans. Fitoterapia 2020, 146, 104687. [Google Scholar] [CrossRef]
- Hernando, G.; Turani, O.; Rodriguez Araujo, N.; Pulido Carrasquero, A.; Bouzat, C. Unraveling anthelmintic targets and mechanisms of action of trans-cinnamaldehyde from cinnamon essential oil. Sci. Rep. 2025, 15, 5422. [Google Scholar] [CrossRef]
- Marjanović, D.; Bogunović, D.; Milovanović, M.; Marinković, D.; Zdravković, N.; Magaš, V.; Trailović, S. Antihelminic Activity of Carvacrol, Thymol, Cinnamaldehyde and P-Cymen Against the Free-Living Nematode Caenorhabditis elegans and Rat Pinworm Syphacia muris. Acta Vet.-Beogr. 2018, 68, 445–456. [Google Scholar] [CrossRef]
- Williams, A.R.; Ramsay, A.; Hansen, T.V.; Ropiak, H.M.; Mejer, H.; Nejsum, P.; Mueller-Harvey, I.; Thamsborg, S.M. Anthelmintic activity of trans-cinnamaldehyde and A- and B-type proanthocyanidins derived from cinnamon (Cinnamomum verum). Sci. Rep. 2015, 5, 14791. [Google Scholar] [CrossRef]
- Piao, X.; Sun, M.; Yi, F. Evaluation of Nematocidal Action against Caenorhabditis elegans of Essential Oil of Flesh Fingered Citron and Its Mechanism. J. Chem. 2020, 2020, 1740938. [Google Scholar] [CrossRef]
- Nordi, E.C.; Costa, R.L.; David, C.M.; Parren, G.A.; Freitas, A.C.; Lameirinha, L.P.; Katiki, L.M.; Bueno, M.S.; Quirino, C.R.; Gama, P.E.; et al. Supplementation of moist and dehydrated citrus pulp in the diets of sheep artificially and naturally infected with gastrointestinal nematodes on the parasitological parameters and performance. Vet. Parasitol. 2014, 205, 532–539. [Google Scholar] [CrossRef] [PubMed]
- Ballent, M.; Lifschitz, A.; Virkel, G.; Sallovitz, J.; Maté, L.; Lanusse, C. In vivo and ex vivo assessment of the interaction between ivermectin and danofloxacin in sheep. Vet. J. 2012, 192, 422–427. [Google Scholar] [CrossRef]
- Zhang, H.; Yao, M.; Morrison, R.A.; Chong, S. Commonly used surfactant, Tween 80, improves absorption of P-glycoprotein substrate, digoxin, in rats. Arch. Pharm. Res. 2003, 26, 768–772. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.T.; Duong, V.A.; Maeng, H.J. Pharmaceutical Formulations with P-Glycoprotein Inhibitory Effect as Promising Approaches for Enhancing Oral Drug Absorption and Bioavailability. Pharmaceutics 2021, 13, 1103. [Google Scholar] [CrossRef] [PubMed]
- Hennessy, D.R.; Page, S.W.; Gottschall, D. The behaviour of doramectin in the gastrointestinal tract, its secretion in bile and pharmacokinetic disposition in the peripheral circulation after oral and intravenous administration to sheep. J. Vet. Pharmacol. Ther. 2000, 23, 203–213. [Google Scholar] [CrossRef]

| Treatment | Day 0 | Day 14 | FECR (%) (LCL-UCL) |
|---|---|---|---|
| DRM | 2716 ± 2641 aA | 910 ± 2548 abB | 66 |
| (0–98) | |||
| CNM–PGF | 1949 ± 1093 aA | 1615 ± 1129 aA | 17 |
| (0–53) | |||
| DRM + CNM–PGF | 2422 ± 1288 aA | 529 ± 1587 bB | 78 |
| (34–99) |
| CNM–PGF | DRM | DRM + CNM–PGF | |
|---|---|---|---|
| Haemonchus spp. | 37 | 0 | 79 |
| Teladorsagia spp. | 5.7 | 100 | 82 |
| Trichostrongylus spp. | 0 | 100 | 76 |
| Cooperia spp. | 0 | 100 | 100 |
| Kinetic Parameters | DRM | DRM + CNM–PGF | DRM + LPM |
|---|---|---|---|
| Cmax (ng/mL) | 25.76 ± 10.01 a | 23.73 ± 6.55 a | 26.95 ± 13.32 a |
| T max (days) | 0.93 ± 0.47 a | 0.80 ± 0.34 a | 0.86 ± 0.43 a |
| AUC0–t (ng d/mL) | 90.84 ± 33.61 a | 72.03 ± 18.99 a | 80.72 ± 31.29 a |
| T ½ ab (days) | 0.26 ± 0.13 a | 0.42 ± 0.15 a | 0.43 ± 0.33 a |
| T ½ el (days) | 2.19 ± 0.41 a | 2.52 ± 0.43 a | 2.66 ± 0.43 a |
| Treatment | Day 0 | Day 14 | FECR (%) (LCL-UCL) |
|---|---|---|---|
| DRM | 1586 ± 984 aA | 694 ± 944 aB | 56 |
| (9–88) | |||
| DRM + CNM–PGF | 1263 ± 736 aA | 762 ± 1565 aA | 40 |
| (0–95) | |||
| DRM + LPM | 1584 ± 782 aA | 397 ± 520 aB | 75 |
| (50–92) |
| DRM | DRM + CNM-GF | DRM + LPM | |
|---|---|---|---|
| Haemonchus spp. | 0 | 29 | 79 |
| Teladorsagia spp. | 82 | 49 | 86 |
| Trichostrongylus spp. | 100 | 100 | 95 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Miró, M.V.; Ichinose, P.; Lloberas, M.; Moriones, L.; Lanusse, C.; Virkel, G.; Lifschitz, A. Phytochemical-Mediated Modulation of Doramectin Pharmacology in Sheep: Exploring the Cinnamaldehyde–Pink Grapefruit Combination. Animals 2025, 15, 2539. https://doi.org/10.3390/ani15172539
Miró MV, Ichinose P, Lloberas M, Moriones L, Lanusse C, Virkel G, Lifschitz A. Phytochemical-Mediated Modulation of Doramectin Pharmacology in Sheep: Exploring the Cinnamaldehyde–Pink Grapefruit Combination. Animals. 2025; 15(17):2539. https://doi.org/10.3390/ani15172539
Chicago/Turabian StyleMiró, María Victoria, Paula Ichinose, Mercedes Lloberas, Lucila Moriones, Carlos Lanusse, Guillermo Virkel, and Adrián Lifschitz. 2025. "Phytochemical-Mediated Modulation of Doramectin Pharmacology in Sheep: Exploring the Cinnamaldehyde–Pink Grapefruit Combination" Animals 15, no. 17: 2539. https://doi.org/10.3390/ani15172539
APA StyleMiró, M. V., Ichinose, P., Lloberas, M., Moriones, L., Lanusse, C., Virkel, G., & Lifschitz, A. (2025). Phytochemical-Mediated Modulation of Doramectin Pharmacology in Sheep: Exploring the Cinnamaldehyde–Pink Grapefruit Combination. Animals, 15(17), 2539. https://doi.org/10.3390/ani15172539





