Species-Specific Responses of Insectivorous Bats to Weather Conditions in Central Chile
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Bat Sampling
2.3. Weather Conditions
2.4. Statistical Analyses
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ramírez-Fráncel, L.A.; García-Herrera, L.V.; Losada-Prado, S.; Reinoso-Flórez, G.; Sánchez-Hernández, A.; Estrada-Villegas, S.; Lim, B.K.; Guevara, G. Bats and Their Vital Ecosystem Services: A Global Review. Integr. Zool. 2022, 17, 2–23. [Google Scholar] [CrossRef]
- Cleveland, C.; Betke, M.; Federico, P.; Frank, J.; Hallam, T.; Horn, J.; López, J.; McCracken, G.; Medellín, R.A.; Moreno-Valdez, A.; et al. Economic Value of the Pest Control Service Provided by Brazilian Free-Tailed Bats in South-Central Texas. Front. Ecol. Environ. 2006, 4, 238–243. [Google Scholar] [CrossRef]
- Rodríguez-San Pedro, A.; Allendes, J.L.; Beltrán, C.A.; Chaperon, P.N.; Saldarriaga-Córdoba, M.M.; Silva, A.X.; Grez, A.A. Quantifying Ecological and Economic Value of Pest Control Services Provided by Bats in a Vineyard Landscape of Central Chile. Agric. Ecosyst. Environ. 2020, 302, 107063. [Google Scholar]
- Maine, J.J.; Boyles, J.G. Bats Initiate Vital Agroecological Interactions in Corn. Proc. Natl. Acad. Sci. USA 2015, 112, 12438–12443. [Google Scholar] [CrossRef] [PubMed]
- Tuneu-Corral, C.; Puig-Montserrat, X.; Riba-Bertolín, D.; Russo, D.; Rebelo, H.; Cabeza, M.; López-Baucells, A. Pest Suppression by Bats and Management Strategies to Favour It a Global Review. Biol. Rev. 2023, 98, 1564–1582. [Google Scholar] [CrossRef]
- Frick, W.F.; Kingston, T.; Flanders, J. A Review of the Major Threats and Challenges to Global Bat Conservation. Ann. N. Y. Acad. Sci. 2020, 1469, 5–25. [Google Scholar] [CrossRef] [PubMed]
- Frick, W.F.; Stepanian, P.M.; Kelly, J.F.; Howard, K.W.; Kuster, C.M.; Kunz, T.H.; Chilson, P.B. Climate and Weather Impact Timing of Emergence of Bats. PLoS ONE 2012, 7, e42737. [Google Scholar] [CrossRef] [PubMed]
- Jones, G.; Jacobs, D.S.; Kunz, T.H.; Willig, M.R.; Racey, P.A. Carpe Noctem: The Importance of Bats as Bioindicators. Endanger. Species Res. 2009, 8, 93–115. [Google Scholar] [CrossRef]
- O’Shea, T.J.; Cryan, P.M.; Hayman, D.T.S.; Plowright, R.K.; Streicker, D.G. Multiple Mortality Events in Bats: A Global Review. Mamm. Rev. 2016, 46, 175–190. [Google Scholar] [CrossRef]
- Russo, D.; Jones, G. Bats as Bioindicators: An Introduction. Mamm. Biol. 2015, 80, 157–158. [Google Scholar] [CrossRef]
- Anthony, E.L.; Stack, M.; Kunz, T. Night Roosting and the Nocturnal Time Budget of the Little Brown Bat, Myotis lucifugus: Effects of Reproductive Status, Prey Density, and Environmental Conditions. Oecologia 1981, 51, 151–156. [Google Scholar] [CrossRef]
- Appel, G.; López-Baucells, A.; Magnusson, W.E.; Bobrowiec, P.E.D. Temperature, Rainfall, and Moonlight Intensity Effects on Activity of Tropical Insectivorous Bats. J. Mammal. 2019, 100, 1889–1900. [Google Scholar] [CrossRef]
- Erickson, J.L.; West, S.D. The Influence of Regional Climate and Nightly Weather Conditions on Activity Patterns of Insectivorous Bats. Acta Chiropterologica 2002, 4, 17–24. [Google Scholar] [CrossRef]
- O’Donnell, C.F. Influence of Season, Habitat, Temperature, and Invertebrate Availability on Nocturnal Activity of the New Zealand Long-Tailed Bat (Chalinolobus tuberculatus). N. Z. J. Zool. 2000, 27, 207–221. [Google Scholar] [CrossRef]
- Reichard, J.D.; Gonzalez, L.E.; Casey, C.M.; Allen, L.C.; Hristov, N.I.; Kunz, T.H. Evening Emergence Behaviour and Seasonal Dynamics in Large Colonies of Brazilian Free-Tailed Bats. J. Mammal. 2009, 90, 1478–1486. [Google Scholar] [CrossRef]
- Jonason, D.; Franzén, M.; Ranius, T. Surveying Moths Using Light Traps: Effects of Weather and Time of Year. PLoS ONE 2014, 9, e92453. [Google Scholar] [CrossRef] [PubMed]
- Meyer, G.A.; Senulis, J.A.; Reinartz, J.A. Effects of Temperature and Availability of Insect Prey on Bat Emergence from Hibernation in Spring. J. Mammal. 2016, 97, 1623–1633. [Google Scholar] [CrossRef]
- Milne, D.J.; Fisher, A.; Rainey, I.; Pavey, C.R. Temporal Patterns of Bats in the Top End of the Northern Territory, Australia. J. Mammal. 2005, 86, 909–920. [Google Scholar] [CrossRef]
- Wolbert, S.J.; Zellner, A.S.; Whidden, H.P. Bat Activity, Insect Biomass, and Temperature along an Elevational Gradient. Northeast. Nat. 2014, 21, 72–85. [Google Scholar] [CrossRef]
- Arias-Aguilar, A.; Chacón-Madrigal, E.; Laval, R.; Rodríguez-Herrera, B. Diversity and Activity Patterns of Aerial Insectivorous Bats along an Altitudinal Gradient in a Tropical Forest in Costa Rica. Hystrix 2020, 31, 58–63. [Google Scholar] [CrossRef]
- Voigt, C.C.; Schneeberger, K.; Voigt-Heucke, S.L.; Lewanzik, D. Rain Increases the Energy Cost of Bat Flight. Biol. Lett. 2011, 7, 793–795. [Google Scholar] [CrossRef]
- Hawkins, B.A.; Holyoak, M. Transcontinental Crashes of Insect Populations? Am. Nat. 1998, 152, 480–484. [Google Scholar] [CrossRef]
- Snell-Rood, E. The Effect of Climate on Acoustic Signals: Does Atmospheric Sound Absorption Matter for Bird Song and Bat Echolocation? J. Acoust. Soc. Am. 2012, 131, 1650–1658. [Google Scholar] [CrossRef]
- Chaverri, G.; Quirós, O.E. Variation in Echolocation Call Frequencies in Two Species of Free-Tailed Bats According to Temperature and Humidity. J. Acoust. Soc. Am. 2017, 142, 146–150. [Google Scholar] [CrossRef]
- Wu, H.; Gong, L.; Jiang, T.; Feng, J.; Lin, A. Echolocation Call Frequencies of Bats Vary with Body Temperature and Weather Conditions. Anim. Behav. 2021, 180, 51–61. [Google Scholar] [CrossRef]
- Lacki, M.J. Temperature and Humidity-Induced Shifts in the Flight Activity of Little Brown Bats. J. Sci. 1984, 84, 264–266. [Google Scholar]
- Amorim, F.; Rebelo, H.; Rodrigues, L. Factors Influencing Bat Activity and Mortality at a Wind Farm in the Mediterranean Region. Acta Chiropterologica 2012, 14, 439–457. [Google Scholar] [CrossRef]
- Rocha, A.D.; Bichuette, M.E. Influence of Abiotic Variables on the Bat Fauna of a Granitic Cave and Its Surroundings in the State of São Paulo, Brazil. Biota Neotrop. 2016, 16, e20150032. [Google Scholar] [CrossRef]
- Thomas, A.J.; Jacobs, D.S. Factors Influencing the Emergence Times of Sympatric Insectivorous Bat Species. Acta Chiropterologica 2013, 15, 121–132. [Google Scholar] [CrossRef]
- Rodríguez-San Pedro, A.; Allendes, J.L.; Ossa, G. Lista Actualizada de Los Murciélagos de Chile Con Comentarios Sobre Taxonomía, Ecología, y Distribución. Biodivers. Nat. Hist. 2016, 2, 18–41. [Google Scholar]
- Rodríguez-San Pedro, A.; Rodríguez-Herbach, C.; Allendes, J.L.; Chaperon, P.N.; Beltrán, C.A.; Grez, A.A. Responses of Aerial Insectivorous Bats to Landscape Composition and Heterogeneity in Organic Vineyards. Agric. Ecosyst. Environ. 2019, 277, 74–82. [Google Scholar] [CrossRef]
- Muñoz, A.E.; Ossa, G.; Zaviezo, T.; Bonacic, C. Diversity and Foraging Activity of Bats in Cultivated and Uncultivated Areas in Agroecosystems of a Mediterranean-Climate Hotspot. Neotrop. Biodivers. 2019, 5, 36–40. [Google Scholar] [CrossRef]
- Vilches-Piñones, K.I.; Matus-Olivares, C.; Catalán, G.; Lisón, F. Effect of Forest Areas Configuration on the Community of Insectivorous Bats in Agroforestry Landscapes. Austral Ecol. 2024, 49, e13268. [Google Scholar]
- Giorgi, F.; Lionello, P. Climate Change Projections for the Mediterranean Region. Glob. Planet. Change 2008, 63, 90–104. [Google Scholar] [CrossRef]
- Reidsma, P.; Ewert, F.; Boogaard, H.; van Diepen, K. Regional Crop Modelling in Europe: The Impact of Climatic Conditions and Farm Characteristics on Maize Yields. Agric. Syst. 2009, 100, 51–60. [Google Scholar] [CrossRef]
- Pörtner, H.-O.; Roberts, D.C.; Tignor, M.; Poloczanska, E.S.; Mintenbeck, K.; Alegría, A.; Craig, M.; Langsdorf, S.; Löschke, S.; Möller, V.; et al. (Eds.) IPCC Climate Change 2022: Impacts, Adaptation, and Vulnerability. Contribution of Working Group II to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2022. [Google Scholar]
- Marquet, P.A.; Altamirano, A.; Arroyo, M.T.K.; Fernández, M.; Gelcich, S.; Górski, K.; Habit, E.; Lara, A.; Maass, A.; Pauchard, A.; et al. (Eds.) Biodiversidad y Cambio Climático en Chile: Evidencia Científica Para la Toma de Decisiones; Comité Científico COP25; Ministerio de Ciencia, Tecnología, Conocimiento e Innovación: Santiago, Chile, 2019.
- Marquet, P.A.; Vianna, J.A.; Pliscoff, P. Impactos y Adaptación En Ecosistemas y Biodiversidad. In Cambio Climático en Chile: Ciencia, Mitigación y Adaptación; Castilla, J.C., Meza, F.J., Vicuña, S., Marquet, P.A., Montero, J.P., Eds.; Ediciones Universidad Católica de Chile: Santiago, Chile, 2019; pp. 335–359. [Google Scholar]
- Luebert, F.; Pliscoff, P. Sinopsis Bioclimática y Vegetacional de Chile; Editorial Universitaria: Santiago, Chile, 2017. [Google Scholar]
- Mann, G. Los Pequeños Mamíferos de Chile. Gayana 1978, 40, 1–356. [Google Scholar]
- Rodríguez-San Pedro, A.; Simonetti, J.A. Acoustic Identification of Four Species of Bats (Order Chiroptera) in Central Chile. Bioacoustics 2013, 22, 165–172. [Google Scholar] [CrossRef]
- Miller, B. A Method for Determining Relative Activity of Free Flying Bats Using a New Activity for Acoustic Monitoring A Method for Determining Relative Activity of Free Flying Bats Using a New Activity Index for Acoustic Monitoring. Acta Chiropterologica 2001, 3, 93–105. [Google Scholar]
- Wood, S.N. Low-Rank Scale-Invariant Tensor Product Smooths for Generalized Additive Mixed Models. Biometrics 2006, 62, 1025–1036. [Google Scholar] [CrossRef]
- Quinn, G.; Keough, M. Experimental Design and Data Analysis for Biologists; Cambridge University Press: Cambridge, UK, 2002. [Google Scholar]
- Akaike, H. On the Likelihood of a Time Series Model. J. R. Stat. Soc. Ser. D (Stat.) 1978, 27, 217–235. [Google Scholar] [CrossRef]
- Burnham, K.P.; Anderson, D.R. Multimodel Inference: Understanding AIC and BIC in Model Selection. Sociol. Methods Res. 2004, 33, 261–304. [Google Scholar] [CrossRef]
- Elith, J.; Leathwick, J.R.; Hastie, T. A Working Guide to Boosted Regression Trees. J. Anim. Ecol. 2008, 77, 802–813. [Google Scholar] [CrossRef]
- Lee, Y.-F.; McCracken, G.F. Timing and Variation in the Emergence and Return of Mexican Free-Tailed Bats, Tadarida brasiliensis mexicana. Zool. Stud. 2001, 40, 309–316. [Google Scholar]
- Arndt, R.J.; O’Keefe, J.M.; Mitchell, W.A.; Holmes, J.B.; Lima, S.L. Do Predators Influence the Behaviour of Temperate-Zone Bats? An Analysis of Competing Models of Roost Emergence Times. Anim. Behav. 2018, 145, 161–170. [Google Scholar] [CrossRef]
- Welbergen, J.A. Timing of the Evening Emergence from Day Roosts of the Grey-Headed Flying Fox, Pteropus poliocephalus: The Effects of Predation Risk, Foraging Needs, and Social Context. Behav. Ecol. Sociobiol. 2006, 60, 311–322. [Google Scholar] [CrossRef]
- Jones, G.; Rydell, J. Foraging Strategy and Predation Risk as Factors Influencing Emergence Time in Echolocating Bats. Philos. Trans. R. Soc. B Biol. Sci. 1994, 346, 445–455. [Google Scholar] [CrossRef]
- Denzinger, A.; Schnitzler, H.-U. Bat Guilds, a Concept to Classify the Highly Diverse Foraging and Echolocation Behaviors of Microchiropteran Bats. Front. Physiol. 2013, 4, 164. [Google Scholar] [CrossRef]
- Canals, M.; Grossi, B.; Iriarte-Diaz, J.; Veloso, C. Biomechanical and Ecological Relationships of Wing Morphology of Eight Chilean Bats. Rev. Chil. Hist. Nat. 2005, 78, 207–2014. [Google Scholar] [CrossRef]
- Beilke, E.A.; Blakey, R.V.; O’Keefe, J.M. Bats Partition Activity in Space and Time in a Large, Heterogeneous Landscape. Ecol. Evol. 2021, 11, 6513–6526. [Google Scholar] [CrossRef]
- Salinas-Ramos, V.B. Interspecific Competition in Bats: State of Knowledge and Research Challenges. Mammal Rev. 2020, 50, 68–81. [Google Scholar] [CrossRef]
- Rodríguez-San Pedro, A.; Allendes, J.L.; Carrasco-Lagos, P.; Moreno, R.A. Murciélagos de La Región Metropolitana de Santiago, Chile, 1st ed.; SEREMI Metropolitana del Medio Ambiente; Universidad Santo Tomás: Santiago, Chile, 2014; ISBN 9789567204496. [Google Scholar]
- Meynard, C.N.; Soto-Gamboa, M.; Heady, P.a.; Frick, W.F. Bats of the Chilean Temperate Rainforest: Patterns of Landscape Use in a Mosaic of Native Forests, Eucalyptus Plantations and Grasslands within a South American Biodiversity Hotspot. Biodivers. Conserv. 2014, 23, 1949–1963. [Google Scholar] [CrossRef]
- Neuweiler, G. Foraging Ecology and Audition in Echolocating Bats. Trends Ecol. Evol. 1989, 4, 160–166. [Google Scholar] [CrossRef]
- Murray, K.L.; Fraser, E.; Davy, C.; Fleming, T.H.; Fenton, M.B. Characterization of the Echolocation Calls of Bats from Exuma, Bahamas. Acta Chiropterologica 2009, 11, 415–424. [Google Scholar] [CrossRef]
- Duchamp, J.E.; Yates, M.; Muzika, R.-M.; Swihart, R.K. Estimating Probabilities of Detection for Bat Echolocation Calls: An Application of the Double-Observer Method. Wildl. Soc. Bull. 2006, 34, 408–412. [Google Scholar] [CrossRef]
- Dekker, J.; Steen, W.; Bouman, H.B.; van der Vliet, R.E. Differences in Acoustic Detectibility of Bat Species Hamper Environmental Impact Assessment Studies. Eur. J. Wildl. Res. 2022, 68, 14. [Google Scholar] [CrossRef]
- Speakman, J.R.; Thomas, D.W.; Kunz, T.H.; Fenton, M.B. Physiological Ecology and Energetics of Bats. In Bat Ecology; Kunz, T.H., Fenton, M.B., Eds.; The University of Chicago Press: Chicago, IL, USA, 2003; pp. 430–490. [Google Scholar]
- Ormsbee, P.C.; Kiser, J.D.; Perimeter, S.I. Importance of Night Roosts to the Ecology of Bats. In Bats in Forest: Conservation and Management; Johns Hopkins University Press: Baltimore, MD, USA, 2007; pp. 129–165. [Google Scholar]
- Holland, R.A.; Meyer, C.F.J.; Kays, R. Emergence Time and Foraging Activity in Pallas’ Mastiff Bat, Molossus molossus (Chiroptera: Molossidae) in Relation to Sunset/Sunrise and Phase of the Moon. Acta Chiropterologica 2011, 13, 399–404. [Google Scholar] [CrossRef]
- Adam, M.D.; Hayes, J.P. Use of Bridges as Night Roosts by Bats in the Oregon Coast Range. J. Mammal. 2000, 81, 402–407. [Google Scholar] [CrossRef]
- Korine, C.; Adams, R.; Russo, D.; Fisher-Phelps, M.; Jacobs, D. Bats and Water: Anthropogenic Alterations Threaten Global Bat Populations. In Bats in the Anthropocene: Conservation of Bats in a Changing World; Springer: Cham, Switzerland, 2016; pp. 215–241. [Google Scholar]
- Froidevaux, J.S.P.; Jones, G.; Kerbiriou, C.; Park, K.J. Acoustic Activity of Bats at Power Lines Correlates with Relative Humidity: A Potential Role for Corona Discharges. Proc. R. Soc. B Biol. Sci. 2023, 290, 20222510. [Google Scholar] [CrossRef] [PubMed]
- Tamošiunaite, M.; Tamošiunas, S.; Žilinskas, M.; Tamošiuniene, M.; Tamošiunaite, M.; Tamošiunas, S.; Žilinskas, M.; Tamošiuniene, M. Atmospheric Attenuation Due to Humidity. In Electromagnetic Waves; IntechOpen: London, UK, 2011. [Google Scholar] [CrossRef]
- Lawrence, B.D.; Simmons, J.A. Measurements of Atmospheric Attenuation at Ultra- Sonic Frequencies and the Significance for Echolocation by Bats. J. Acoust. Soc. Am. 1982, 71, 585–590. [Google Scholar] [CrossRef] [PubMed]
- Stilz, W.P.; Schnitzler, H.U. Estimation of the Acoustic Range of Bat Echolocation for Extended Targets. J. Acoust. Soc. Am. 2012, 132, 1765–1775. [Google Scholar] [CrossRef] [PubMed]
- Luo, J.; Koselj, K.; Zsebok, S.; Siemers, B.M.; Goerlitz, H.R. Global Warming Alters Sound Transmission: Differential Impact on the Prey Detection Ability of Echolocating Bats. J. R. Soc. Interface 2014, 11, 20130961. [Google Scholar] [CrossRef] [PubMed]
- Griffin, D.R. The Importance of Atmospheric Attenuation for the Echolocation of Bats (Chiroptera). Anim. Behav. 1971, 19, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Chaperon, P.; Rodríguez-San Pedro, A.; Beltrán, C.; Allendes, J.L.; Barahona-Segovia, R.M.; Urra, F.; Grez, A.A. Effects of Adjacent Habitat on Nocturnal Flying Insects in Vineyards and Implications for Bat Foraging. Agric. Ecosyst. Environ. 2022, 326, 107780. [Google Scholar] [CrossRef]
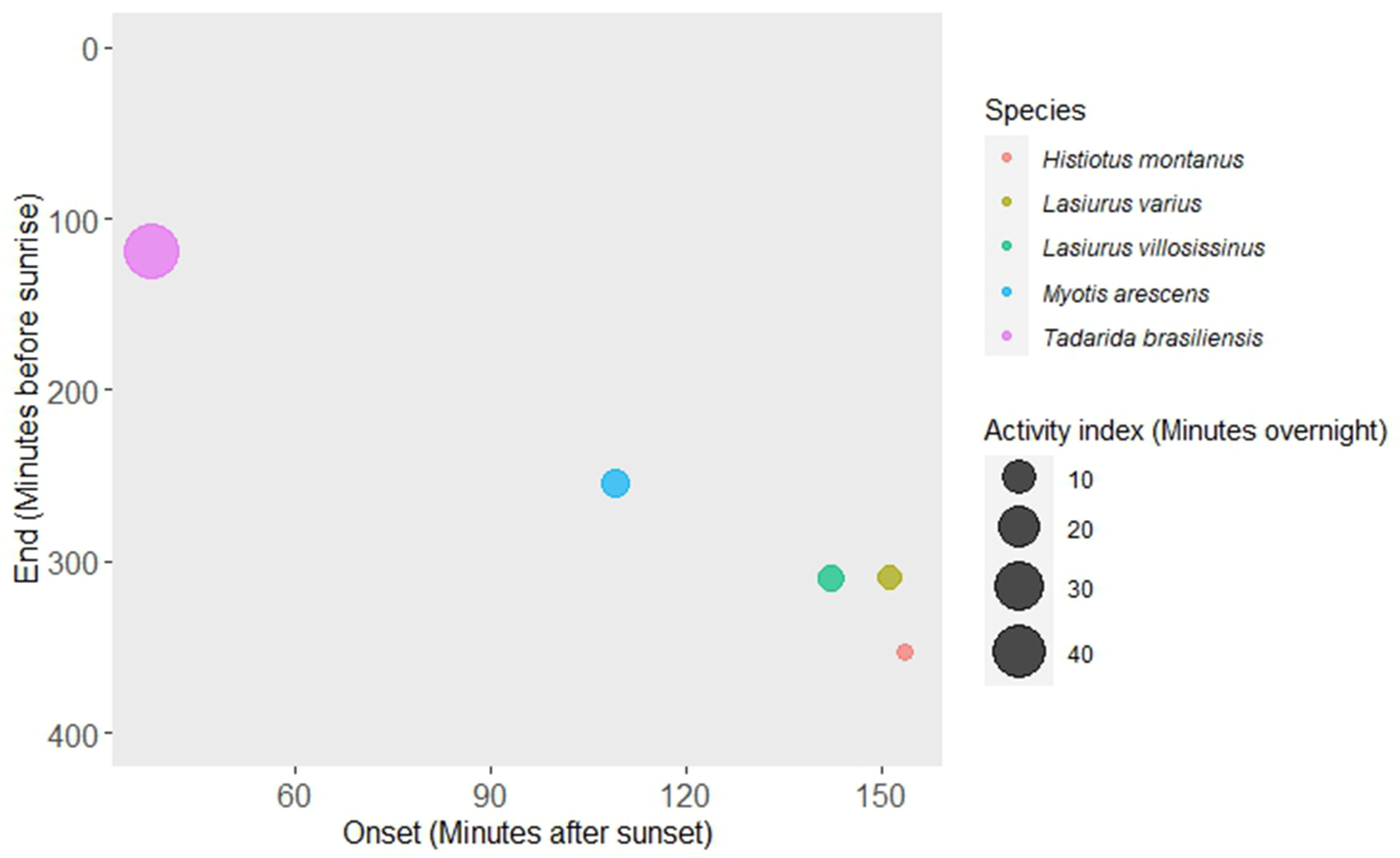
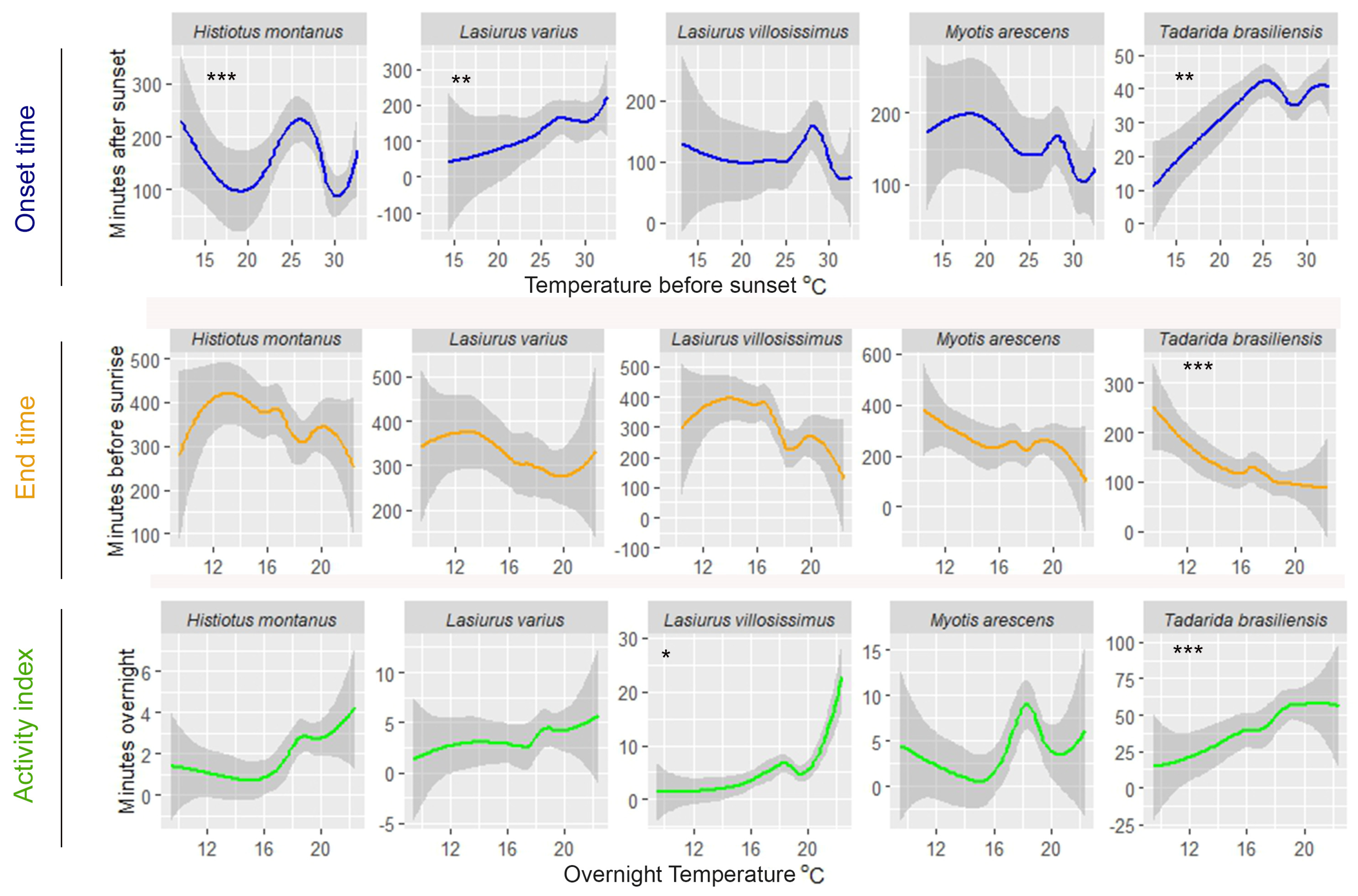
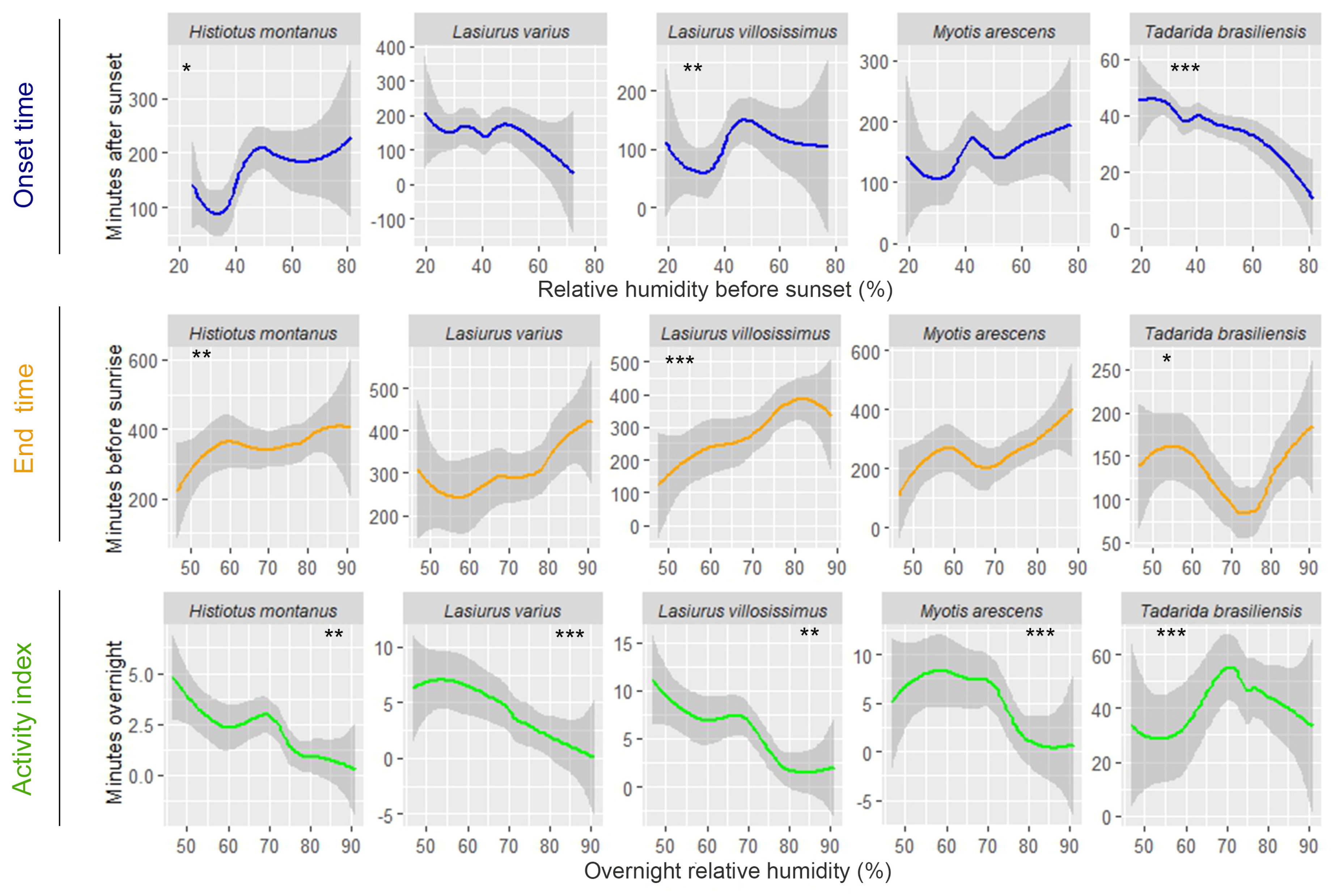
| Onset Time (Minutes after Sunset) | End Time (Minutes before Sunrise) | Activity Index (Minutes Overnight) | |
|---|---|---|---|
| Tadarida brasiliensis | 37.0 ± 1.31 (113–0) | 118.44 ± 8.21 (541–22) | 44.77 ± 3.38 (271–1) |
| Myotis arescens | 142.29 ± 9.56 (529–17) | 254.43 ± 13.45 (618–50) | 4.41 ± 0.76 (54–0) |
| Lasiurus varius | 151.07 ± 11.84 (514–24) | 309.84 ± 15.77 (600–55) | 3.50 ± 0.54 (41–0) |
| Lasiurus villosissimus | 109.00 ± 9.63 (592–27) | 309.06 ± 15.00 (620–23) | 4.62 ± 0.55 (32–0) |
| Histiotus montanus | 153.49 ± 9.50 (528–41) | 353.21 ± 12.14 (595–91) | 1.89 ± 0.24 (19–0) |
| Deviance Explained (%) | Predictor Variable | edf | χ2 | p | |
|---|---|---|---|---|---|
| Tadarida brasiliensis | 26.9 | Onset | 2.36 | 6.91 | 0.100 |
| HuNo | 1.92 | 16.75 | ≤0.05 | ||
| TeNo | 1.00 | 25.94 | ≤0.001 | ||
| Myotis arescens | 16.3 | Onset | 3.01 | 9.52 | ≤0.05 |
| HuNo | 1.00 | 3.80 | 0.051 | ||
| Lasiurus varius | 18.3 | Onset | 1.00 | 14.21 | ≤0.001 |
| HuNo | 1.40 | 1.92 | 0.223 | ||
| Lasiurus villosissimus | 21.9 | Onset | 2.73 | 21.13 | ≤0.001 |
| HuNo | 1.00 | 18.18 | ≤0.001 | ||
| Histiotus montanus | 31.0 | Onset | 1.57 | 41.49 | ≤0.001 |
| HuNo | 1.00 | 10.63 | ≤0.01 |
| Deviance Explained (%) | Predictor Variable | edf | χ2 | p | |
|---|---|---|---|---|---|
| Tadarida brasiliensis | 31.9 | HuSu | 1.00 | 52.55 | ≤0.001 |
| TeSu | 1.86 | 39.91 | ≤0.001 | ||
| Myotis arescens | 3.35 | TeSu | 1.00 | 3.20 | 0.074 |
| Lasiurus varius | 8.89 | HuSu | 1.00 | 2.55 | 0.110 |
| TeSu | 1.00 | 10.28 | ≤0.01 | ||
| Lasiurus villosissimus | 11.1 | HuSu | 2.99 | 10.35 | ≤0.01 |
| Histiotus montanus | 35.2 | HuSu | 1.82 | 7.52 | ≤0.05 |
| TeSu | 5.46 | 23.87 | ≤0.001 |
| Deviance Explained (%) | Predictor Variable | edf | χ2 | p | |
|---|---|---|---|---|---|
| Tadarida brasiliensis | 28.7 | HuNo | 1.00 | 40.86 | ≤0.001 |
| TeNo | 1.00 | 67.88 | ≤0.001 | ||
| Myotis arescens | 29.3 | HuNo | 2.98 | 22.01 | ≤0.001 |
| TeNo | 5.56 | 5.57 | 0.081 | ||
| Lasiurus varius | 16.4 | HuNo | 2.46 | 23.17 | ≤0.001 |
| TeNo | 1.03 | 8.11 | 0.078 | ||
| Lasiurus villosissimus | 24.8 | HuNo | 3.49 | 19.66 | ≤0.001 |
| TeNo | 4.94 | 12.44 | ≤0.05 | ||
| Histiotus montanus | 13.2 | HuNo | 2.01 | 20.29 | ≤0.001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rodríguez-San Pedro, A.; Allendes, J.L.; Bruna, T.; Grez, A.A. Species-Specific Responses of Insectivorous Bats to Weather Conditions in Central Chile. Animals 2024, 14, 860. https://doi.org/10.3390/ani14060860
Rodríguez-San Pedro A, Allendes JL, Bruna T, Grez AA. Species-Specific Responses of Insectivorous Bats to Weather Conditions in Central Chile. Animals. 2024; 14(6):860. https://doi.org/10.3390/ani14060860
Chicago/Turabian StyleRodríguez-San Pedro, Annia, Juan Luis Allendes, Tamara Bruna, and Audrey A. Grez. 2024. "Species-Specific Responses of Insectivorous Bats to Weather Conditions in Central Chile" Animals 14, no. 6: 860. https://doi.org/10.3390/ani14060860
APA StyleRodríguez-San Pedro, A., Allendes, J. L., Bruna, T., & Grez, A. A. (2024). Species-Specific Responses of Insectivorous Bats to Weather Conditions in Central Chile. Animals, 14(6), 860. https://doi.org/10.3390/ani14060860





