Exposure to Coxiella burnetii in Wild Lagomorphs in Spanish Mediterranean Ecosystems
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Sampling
2.2. Laboratory Analysis
2.3. Statistical Analysis
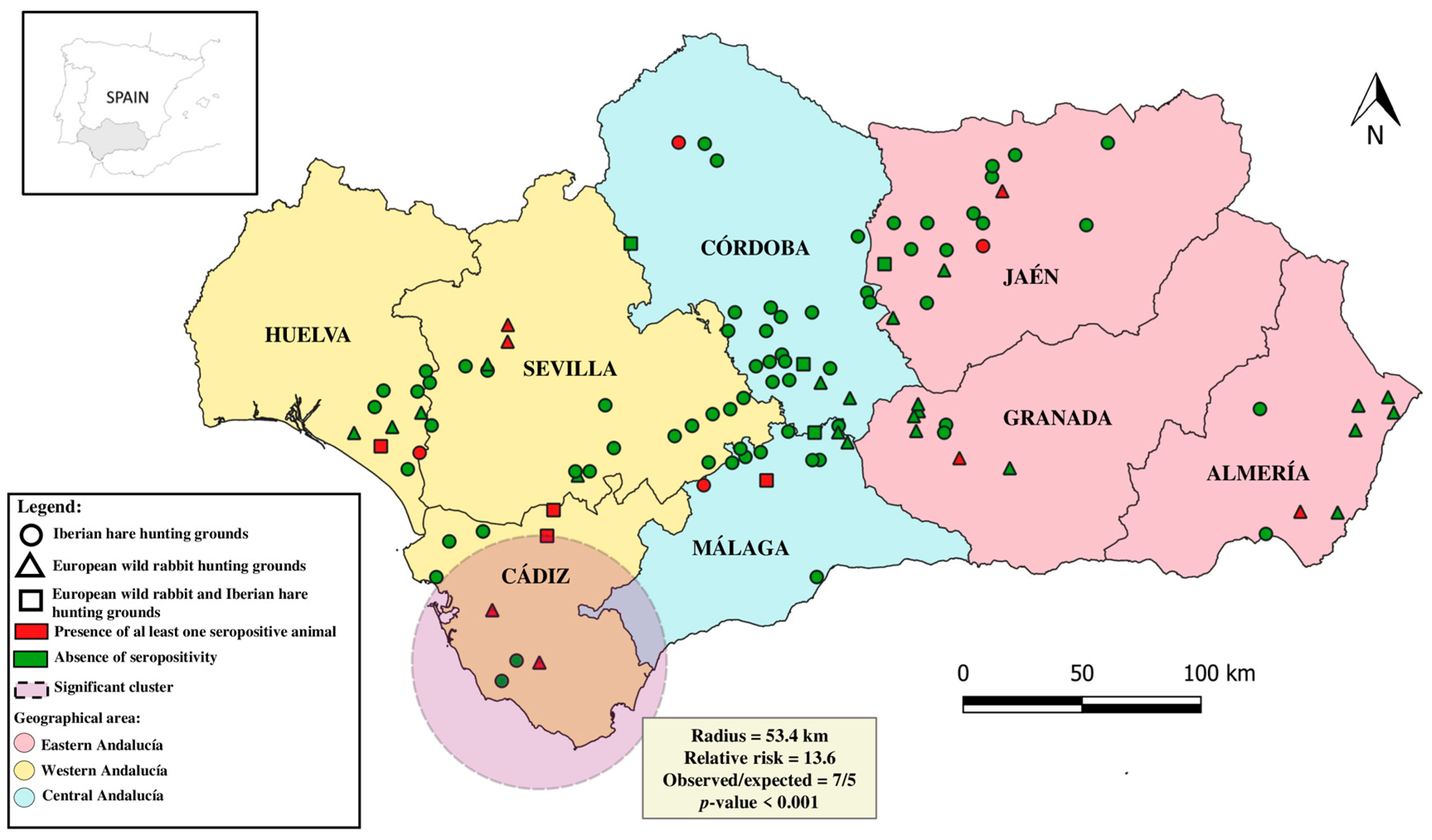
2.4. Spatial Cluster Analysis
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- EFSA (European Food Safety Authority); ECDC (European Centre for Disease Prevention and Control). The European Union One Health 2021 Zoonoses Report. EFSA J. 2022, 20, 7666. [Google Scholar]
- RENAVE (Red Nacional de Vigilancia Epidemiológica). Informe Epidemiológico sobre la situación de la Fiebre Q en España. Años 2019, 2020 y 2021; Centro Nacional de Epidemiología, Instituto de Salud Carlos III: Madrid, Spain, 2022. [Google Scholar]
- González, J.; González, M.G.; Valcárcel, F.; Sánchez, M.; Martín-Hernández, R.; Tercero, J.M.; Olmeda, A.S. Transstadial transmission from nymph to adult of Coxiella burnetii by naturally infected Hyalomma lusitanicum. Pathogens 2020, 9, 884. [Google Scholar] [CrossRef]
- Espejo, E.; Gil-Díaz, A.; Oteo, J.A.; Castillo-Rueda, R.; García-Alvarez, L.; Santana-Baez, S.; Bella, F. Clinical presentation of acute Q fever in Spain: Seasonal and geographical differences. Int. J. Infect. Dis. 2014, 26, 162–164. [Google Scholar] [CrossRef][Green Version]
- Van Asseldonk, M.A.P.M.; Prins, J.; Bergevoet, R.H.M. Economic assessment of Q fever in the Netherlands. Prev. Vet. Med. 2013, 112, 27–34. [Google Scholar] [CrossRef]
- González-Barrio, D.; Ruiz-Fons, F. Coxiella burnetii in wild mammals: A systematic review. Transbound Emerg. Dis. 2019, 66, 662–671. [Google Scholar] [CrossRef] [PubMed]
- Marrie, T.; Williams, J.; Schlech, W., III; Yates, L. Q fever pneumonia associated with exposure to wild rabbits. Lancet 1986, 327, 427–429. [Google Scholar] [CrossRef]
- Astobiza, I.; Barral, M.; Ruiz-Fons, F.; Barandika, J.F.; Gerrikagoitia, X.; Hurtado, A.; García-Pérez, A.L. Molecular investigation of the occurrence of Coxiella burnetii in wildlife and ticks in an endemic area. Vet. Microbiol. 2011, 147, 190–194. [Google Scholar] [CrossRef]
- Psaroulaki, A.; Chochlakis, D.; Angelakis, E.; Ioannou, I.; Tselentis, Y. Coxiella burnetii in wildlife and ticks in an endemic area. Trans. R. Soc. Trop. Med. Hyg. 2014, 108, 625–631. [Google Scholar] [CrossRef]
- Delibes-Mateos, M.; Redpath, S.M.; Angulo, E.; Ferreras, P.; Villafuerte, R. Rabbits as a keystone species in southern Europe. Biol. Conserv. 2007, 137, 149–156. [Google Scholar] [CrossRef]
- Gortazar, C.; Millán, J.; Acevedo, P.; Escudero, M.A.; Marco, J.; De Luco, D.F. A large-scale survey of brown hare Lepus europaeus and Iberian hare L. granatensis populations at the limit of their ranges. Wildl. Biol. 2007, 13, 244–250. [Google Scholar] [CrossRef]
- Ferrer, M.; Negro, J.J. The near extinction of two large European predators: Super specialists pay a price. Conserv. Biol. 2004, 18, 344–349. [Google Scholar] [CrossRef]
- Gratz, N. Vector-and Rodent-Borne Diseases in Europe and North America: Distribution, Public Health Burden, and Control; Cambridge University Press: Cambridge, UK, 2006. [Google Scholar]
- Fernández-Aguilar, X.; Alzaga, V.; Villanúa, D.; Cabezón, O.; García-Bocanegra, I.; Dubey, J.P.; Almería, S. Epidemiology and prevalence of Toxoplasma gondii infection in the Iberian hare (Lepus granatensis). Vet. Parasitol. 2013, 196, 194–198. [Google Scholar] [CrossRef]
- Jiménez, M.; González, E.; Martín-Martín, I.; Hernández, S.; Molina, R. Could wild rabbits (Oryctolagus cuniculus) be reservoirs for Leishmania infantum in the focus of Madrid, Spain? Vet. Parasitol. 2014, 202, 296–300. [Google Scholar] [CrossRef]
- Martín-Sánchez, J.; Torres-Medina, N.; Morillas-Márquez, F.; Corpas-López, V.; Díaz-Sáez, V. Role of wild rabbits as reservoirs of leishmaniasis in a non-epidemic Mediterranean hot spot in Spain. Acta Trop. 2021, 222, 106036. [Google Scholar] [CrossRef]
- ENETWILD-consortium; Gavier-Widen, D.; Ferroglio, E.; Smith, G.; Gonçalves, C.; Vada, R.; Zanet, S.; Gethöffer, F.; Keuling, O.; Staubach, C.; et al. Recommendations and technical specifications for sustainable surveillance of zoonotic pathogens where wildlife is implicated. EFSA Support. Publ. 2023, 20, 7812E. [Google Scholar] [CrossRef]
- European Food Safety Authority (EFSA); Berezowski, J.; de Balogh, K.; Dórea, F.C.; Rüegg, S.; Broglia, A.; Gervelmeyer, A.; Kohnle, L. Prioritisation of zoonotic diseases for coordinated surveillance systems under the One Health approach for cross-border pathogens that threaten the Union. EFSA J. 2023, 21, e07853. [Google Scholar] [PubMed]
- Agencia Estatal de Meteorología (AEMET). Visor del Atlas Climático de la Península y Baleares. 2023. Available online: https://www.aemet.es/es/serviciosclimaticos/datosclimatologicos/atlas_climatico/visor_atlas_climatico (accessed on 8 January 2023).
- Thrusfield, M.V.; Christley, R. Veterinary Epidemiology, 4th ed.; Wiley-Blackwell Ltd.: Hoboken, NJ, USA, 2018. [Google Scholar]
- Morris, P. A review of mammalian age determination methods. Mammal Rev. 1972, 2, 69–104. [Google Scholar] [CrossRef]
- Caraguel, C.; Bassett, S.; González-Barrio, D.; Elsworth, P.; Chaber, A.L. Comparison of three serological tests for the detection of Coxiella burnetii specific antibodies in European wild rabbits. BMC Vet Res. 2020, 16, 315. [Google Scholar] [CrossRef] [PubMed]
- Dohoo, I.R.; Martin, W.; Stryhn, H.E. Veterinary Epidemiologic Research; Charlottetown, P.E.I., Ed.; University of Prince Edward Island: Charlottetown, PE, Canada, 2003. [Google Scholar]
- Kulldorff, M.; Huang, L.; Pickle, L.; Duczmal, L. An elliptic spatial scan statistic. Stat. Med. 2006, 25, 3929–3943. [Google Scholar] [CrossRef] [PubMed]
- Alves, P.C.; Acevedo, P.; Melo-Ferreira, J. Iberian Hare Lepus granatensis Rosenhauer, 1856. In Handbook of the Mammals of Europe; Springer International Publishing: Cham, Switzerland, 2023; pp. 1–23. [Google Scholar]
- Mykytowycz, R. Social behaviour of an experimental colony of wild rabbits, Oryctolagus cuniculus (L.) I. Establishment of the colony. CSIRO Wildl. Res. 1958, 3, 7–25. [Google Scholar] [CrossRef]
- González-Barrio, D.; Maio, E.; Vieira-Pinto, M.; Ruiz-Fons, F. European rabbits as reservoir for Coxiella burnetii. Emerg. Infect. Dis. 2015, 21, 1055–1058. [Google Scholar] [CrossRef]
- Sánchez, M.; Valcárcel, F.; González, J.; González, M.G.; Martín-Hernández, R.; Tercero, J.M.; González-Jara, P.; Olmeda, A.S. Seasonality of Coxiella burnetii among Wild Rabbits (Oryctolagus cuniculus) and the Hyalomma lusitanicum (Acari: Ixodidae) in a Meso-Mediterranean Ecosystem. Pathogens 2022, 11, 36. [Google Scholar] [CrossRef] [PubMed]
- González-Barrio, D.; Velasco Ávila, A.L.; Boadella, M.; Beltrán-Beck, B.; Barasona, J.Á.; Santos, J.P.V.; Queirós, J.; García-Pérez, A.L.; Barral, M.; Ruiz-Fons, F. Host and environmental factors modulate the exposure of free-ranging and farmed red deer (Cervus elaphus) to Coxiella burnetii. Appl. Environ. Microbiol. 2015, 81, 6223–6231. [Google Scholar] [CrossRef] [PubMed]
- Dedek, J.; Nattermann, H.; Loepelmann, H.; Rinka, E.; Knoepke, C. Results obtained from serological investigations of hare (Lepus europaeus Pallan, 1778) for selected infections. Monatshefte Fuer Veterinaermedizin 1990, 45, 833–836. [Google Scholar]
- Hubalek, Z.; Juticová, Z.; Svobodová, Š.; Halouzka, J. A serologic survey for some bacterial and viral zoonoses in game animals in the Czech Republic. J. Wildl. Dis. 1993, 29, 604–607. [Google Scholar] [CrossRef] [PubMed]
- Tsokana, C.N.; Sokos, C.; Giannakopoulos, A.; Birtsas, P.; Athanasiou, L.V.; Valiakos, G.; Sofia, M.; Chatzopoulos, D.C.; Athanasakopoulou, Z.; Kantere, M.; et al. Investigation of the Role of the European Brown Hare in the Epidemiology of Bacterial Zoonotic Pathogens: A Serological and Molecular Survey in Greece. Vector Borne Zoonotic Dis. 2020, 20, 252–257. [Google Scholar] [CrossRef] [PubMed]
- Enright, J.B.; Franti, C.E.; Behymer, D.E.; Longhurst, W.M.; Dutson, V.J.; Wright, M.E. Coxiella burneti in a wildlife-livestock environment: Distribution of Q fever in wild mammals. Am. J. Epidemiol. 1971, 94, 79–90. [Google Scholar] [CrossRef] [PubMed]
- Clark, N.J.; Soares-Magalhães, R.J. Airborne geographical dispersal of Q fever from livestock holdings to human communities: A systematic review and critical appraisal of evidence. BMC Infect. Dis. 2018, 18, 218. [Google Scholar] [CrossRef]
- Grøtan, V.; SÆther, B.-E.; Engen, S.; Solberg, E.J.; Linnell, J.D.C.; Andersen, R.; Brøseth, H.; Lund, E. Climate causes large-scale spatial synchrony in population fluctuations of a temperate herbivore. Ecology 2005, 86, 1472–1482. [Google Scholar] [CrossRef]
- CAPMA. Consejería de Agricultura Pesca y Medio Ambiente. Programa de Vigilancia Epidemiológica de la Fauna Silvestre. 2013. Available online: https://www.juntadeandalucia.es/medioambiente/portal/landing-page-%C3%ADndice/-/asset_publisher/zX2ouZa4r1Rf/content/programa-de-vigilancia-epidemiol-c3-b3gica-de-la-fauna-silvestre-en-andaluc-c3-ada-pve-1/20151?categoryVal= (accessed on 5 January 2024).
- Dabaja, M.F.; Greco, G.; Villari, S.; Vesco, G.; Bayan, A.; El Bazzal, B.; Ibrahim, E.; Gargano, V.; Sciacca, C.; Lelli, R.; et al. Occurrence and risk factors of Coxiella burnetii in domestic ruminants in Lebanon. Comp. Immunol. Microb. 2019, 64, 109–116. [Google Scholar] [CrossRef]
- CAPADR. Consejería de Agricultura, Pesca, Agua y Desarrollo Rural. Distribución Geográfica de las Explotaciones Ganaderas Extensivas e Intensivas de Andalucía. 2020. Available online: https://www.juntadeandalucia.es/organismos/agriculturapescaaguaydesarrollorural/servicios/estadistica-cartografia/estadisticas-ganaderas/paginas/explotaciones-ganaderas-extensivas.html (accessed on 26 January 2024).
- Ejercito, C.L.A.; Cai, L.; Htwe, K.K.; Taki, M.; Inoshima, Y.; Kondo, T.; Hirai, K. Serological evidence of Coxiella burnetii infection in wild animals in Japan. J. Wildl. Dis. 1993, 29, 481–484. [Google Scholar] [CrossRef] [PubMed]
| Variable | Categories | No. Positives/ Overall 1 | Seroprevalence (%) | p |
|---|---|---|---|---|
| Data recorded from the sampled animals | ||||
| Species | Wild rabbit | 53/471 | 11.3 | <0.001 |
| Iberian hare | 4/167 | 2.4 | ||
| Age | Adult | 53/460 | 11.5 | 0.001 |
| Subadult | 3/135 | 2.2 | ||
| Young | 0/36 | 0.0 | ||
| Sex | Male | 28/307 | 9.1 | 0.516 |
| Female | 29/326 | 8.9 | ||
| Kidney fat index | 0 | 19/154 | 12.3 | 0.541 |
| 1 | 16/148 | 10.8 | ||
| 2 | 10/114 | 8.8 | ||
| 3 | 7/99 | 7.1 | ||
| Bodyweight (kg) | 0.4–0.9 | 13/166 | 7.8 | 0.420 |
| 1.0–1.2 | 7/144 | 4.9 | ||
| 1.3–3.1 | 7/147 | 4.8 | ||
| Body length (cm) | 19–37 | 6/161 | 3.7 | 0.075 |
| 38–40 | 14/143 | 9.8 | ||
| 41–59 | 7/136 | 5.1 | ||
| Hunting season | 2018/2019 | 2/60 | 3.3 | <0.001 |
| 2019/2020 | 2/48 | 4.2 | ||
| 2020/2021 | 24/391 | 6.1 | ||
| 2021/2022 | 29/139 | 20.9 | ||
| Hunting ground’s characteristics | ||||
| Geographical area | Western | 45/217 | 20.7 | <0.001 |
| Central | 3/196 | 1.5 | ||
| Eastern | 9/225 | 4.0 | ||
| Burrow density | High | 43/418 | 10.3 | <0.001 |
| Medium | 10/37 | 27.0 | ||
| Low | 0/76 | 0.0 | ||
| High abundance of ticks in the hunting ground | Yes | 24/297 | 8.1 | 0.001 |
| No | 1/142 | 0.7 | ||
| High abundance of fleas in the hunting ground | Yes | 16/273 | 5.9 | 0.514 |
| No | 9/166 | 5.4 | ||
| Fenced hunting ground | Yes | 5/37 | 13.5 | 0.305 |
| No | 48/494 | 9.7 | ||
| Presence of rabbit feeders | Yes | 26/310 | 8.4 | 0.097 |
| No | 27/221 | 12.2 | ||
| Feed supplementation in rabbits | Yes | 32/217 | 14.7 | 0.002 |
| No | 21/314 | 6.7 | ||
| Presence of swamps | Yes | 18/85 | 21.2 | 0.001 |
| No | 35/446 | 7.8 | ||
| Presence of troughs | Yes | 50/442 | 11.3 | 0.012 |
| No | 3/89 | 3.4 | ||
| Presence of streams | Yes | 23/277 | 8.3 | 0.115 |
| No | 30/254 | 11.8 | ||
| The hunting ground is weeded | Yes | 17/118 | 14.4 | 0.054 |
| No | 36/413 | 8.7 | ||
| Presence of artificial burrows | Yes | 4/37 | 10.8 | 0.517 |
| No | 49/494 | 9.9 | ||
| Detection of clinical cases of other infectious diseases | ||||
| Outbreaks of myxomatosis in the last year | Yes | 53/508 | 10.4 | 0.084 |
| No | 0/23 | 0.0 | ||
| Outbreaks of RHD 2 in the last year | Yes | 47/435 | 10.8 | 0.120 |
| No | 6/96 | 6.3 | ||
| Outbreaks of myxomatosis in the last month | Yes | 51/434 | 11.8 | 0.001 |
| No | 2/97 | 2.1 | ||
| Outbreaks of RHD 2 in the last month | Yes | 18/121 | 14.9 | 0.034 |
| No | 35/410 | 8.5 | ||
| Presence of other sympatric species in the hunting ground | ||||
| Presence of wild boar (Sus scrofa) | Yes | 18/333 | 5.4 | 0.003 |
| No | 34/286 | 11.9 | ||
| Presence of red deer (Cervus elaphus) | Yes | 3/63 | 4.8 | 0.199 |
| No | 49/556 | 8.8 | ||
| Presence of wildcat (Felis silvestris) | Yes | 14/170 | 8.2 | 0.224 |
| No | 39/361 | 10.8 | ||
| Presence of Iberian lynx (Lynx pardinus) | Yes | 14/86 | 16.3 | 0.032 |
| No | 39/445 | 8.8 | ||
| Presence of domestic cat (Felis silvestris catus) | Yes | 45/476 | 9.5 | 0.168 |
| No | 8/55 | 14.5 | ||
| Presence of dog (Canis familiaris) | Yes | 34/396 | 8.6 | 0.051 |
| No | 19/135 | 14.1 | ||
| Presence of cattle (Bos taurus) | Yes | 5/25 | 20.0 | 0.012 |
| No | 19/372 | 5.1 | ||
| Presence of goat (Capra aegagrus hircus) | Yes | 5/149 | 3.4 | 0.060 |
| No | 19/248 | 7.7 | ||
| Presence of sheep (Ovis aries) | Yes | 21/192 | 10.9 | <0.001 |
| No | 3/205 | 1.5 | ||
| Presence of farmed rabbit (Oryctolagus cuniculus) | Yes | 7/60 | 11.7 | 0.390 |
| No | 46/471 | 9.8 | ||
| Presence of domestic pig (Sus scrofa domesticus) | Yes | 5/40 | 12.5 | 0.081 |
| No | 19/357 | 5.3 | ||
| Climate characteristics of the hunting ground | ||||
| Mean temperature (°C) | 12.3–16.8 | 2/230 | 0.9 | <0.001 |
| 16.9–17.4 | 7/106 | 6.6 | ||
| 17.5–18.5 | 17/163 | 10.4 | ||
| Max temperature (°C) | 18.9–23.0 | 10/184 | 5.4 | 0.060 |
| 23.1–24.2 | 8/218 | 3.7 | ||
| 24.3–27.4 | 8/73 | 11.0 | ||
| Mean annual rainfall (mm) | 273.3–563.8 | 3/169 | 1.8 | 0.002 |
| 563.9–597.9 | 9/199 | 4.5 | ||
| 598.0–1134.6 | 14/131 | 10.7 | ||
| Humidity (g/m3) | 33–56 | 0/26 | 0.0 | 0.346 |
| 57–65 | 9/132 | 6.8 | ||
| 66–100 | 6/75 | 8.0 | ||
| Variable | Categories | p-Value | OR 95% CI |
|---|---|---|---|
| Presence of sheep | Yes | 0.023 | 4.6 (1.2–17.0) |
| No | a | a | |
| Geographical area | Western | 0.006 | 19.9 (2.3–170.4) |
| Eastern | 0.133 | 5.2 (0.6–43.8) | |
| Central | a | a |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Castro-Scholten, S.; Caballero-Gómez, J.; Cano-Terriza, D.; Jiménez-Martín, D.; Rouco, C.; Beato-Benítez, A.; Camacho-Sillero, L.; García-Bocanegra, I. Exposure to Coxiella burnetii in Wild Lagomorphs in Spanish Mediterranean Ecosystems. Animals 2024, 14, 749. https://doi.org/10.3390/ani14050749
Castro-Scholten S, Caballero-Gómez J, Cano-Terriza D, Jiménez-Martín D, Rouco C, Beato-Benítez A, Camacho-Sillero L, García-Bocanegra I. Exposure to Coxiella burnetii in Wild Lagomorphs in Spanish Mediterranean Ecosystems. Animals. 2024; 14(5):749. https://doi.org/10.3390/ani14050749
Chicago/Turabian StyleCastro-Scholten, Sabrina, Javier Caballero-Gómez, David Cano-Terriza, Débora Jiménez-Martín, Carlos Rouco, Adrián Beato-Benítez, Leonor Camacho-Sillero, and Ignacio García-Bocanegra. 2024. "Exposure to Coxiella burnetii in Wild Lagomorphs in Spanish Mediterranean Ecosystems" Animals 14, no. 5: 749. https://doi.org/10.3390/ani14050749
APA StyleCastro-Scholten, S., Caballero-Gómez, J., Cano-Terriza, D., Jiménez-Martín, D., Rouco, C., Beato-Benítez, A., Camacho-Sillero, L., & García-Bocanegra, I. (2024). Exposure to Coxiella burnetii in Wild Lagomorphs in Spanish Mediterranean Ecosystems. Animals, 14(5), 749. https://doi.org/10.3390/ani14050749










