A Gestational Pectin Diet Could Improve the Health of Multiparous Sows by Modulating the Gut Microbiota and Cytokine Level during Late Pregnancy
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals and Diets
2.2. Animal Sample Collection
2.2.1. Sow Blood Samples
2.2.2. Sow Fecal Samples
2.3. Performance Measurement
2.4. Feedstuff Samples
2.5. Chemical Analyses
2.5.1. Analysis of Plasma Inflammatory Cytokines and Intestinal Permeability Index
2.5.2. Microbiological Analysis of Sow Feces
2.5.3. Analysis of Plasma SCFA Levels in the Sows
2.6. Statistical Analyses
2.6.1. Reproductive Performance Data, Plasma SCFA and Cytokines Concentration Data Analysis
2.6.2. Microbiota Data Analysis
3. Results
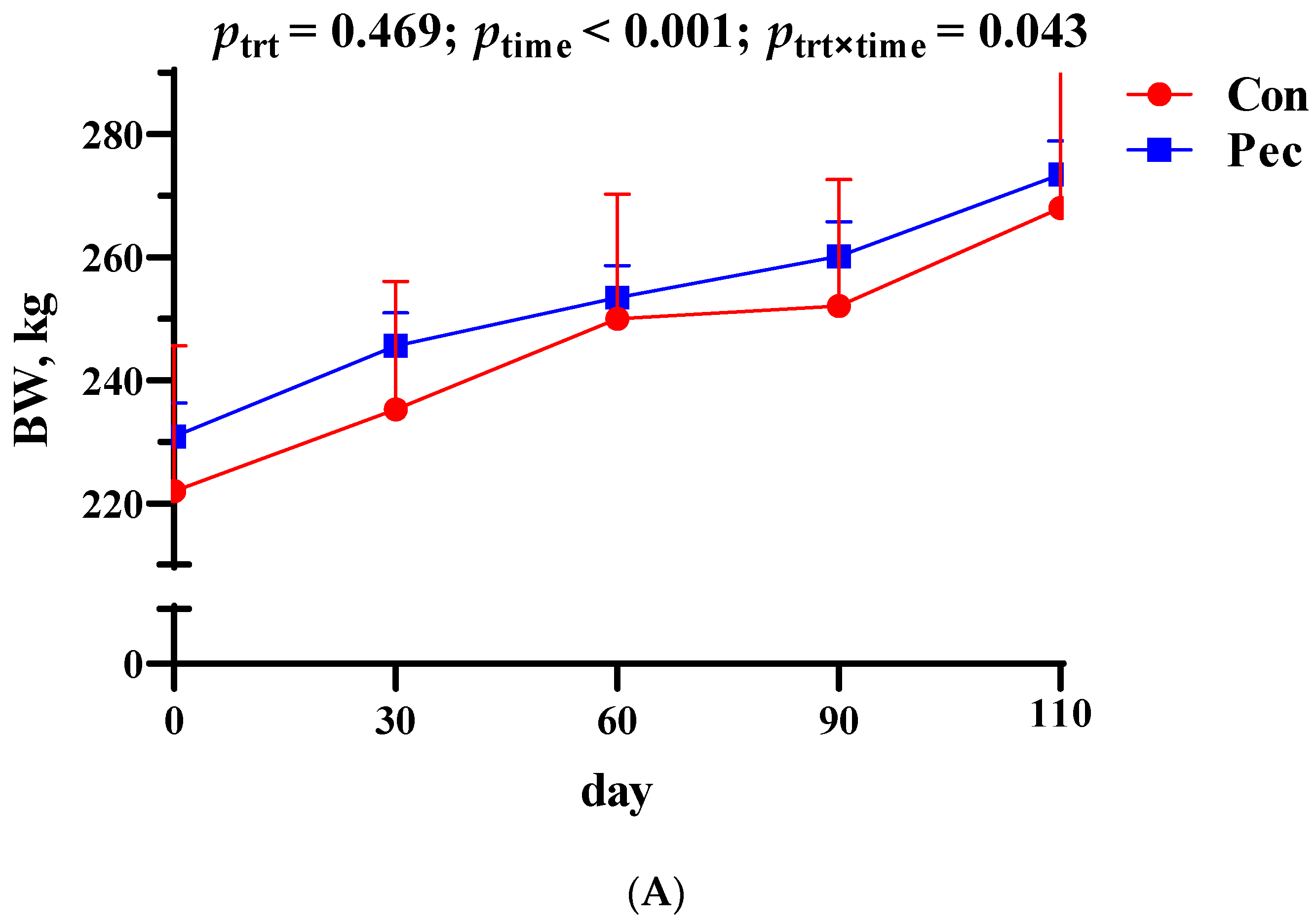
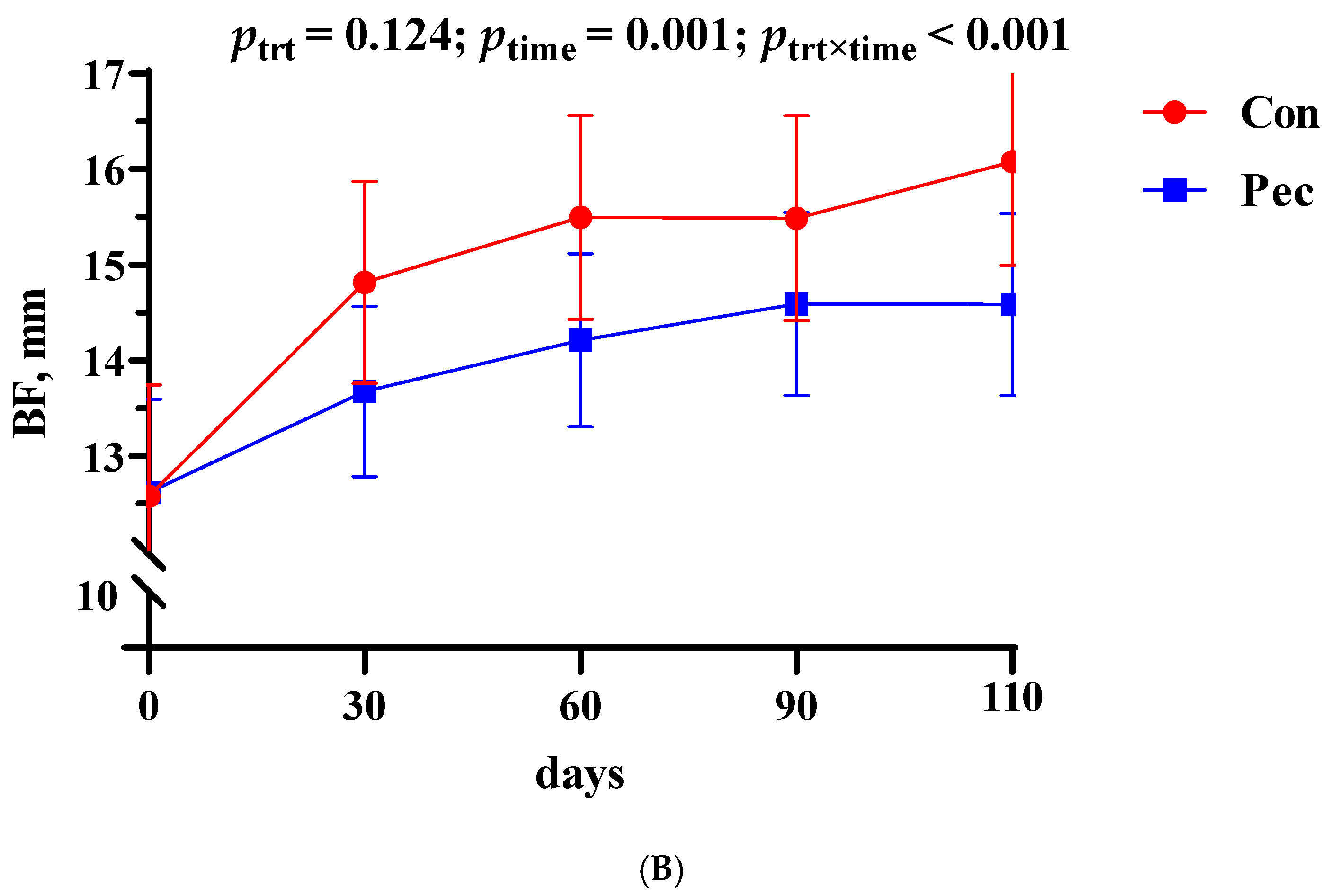
3.1. Body Weight and Backfat Thickness Changes in the Sows
3.2. Production Performance of the Sows
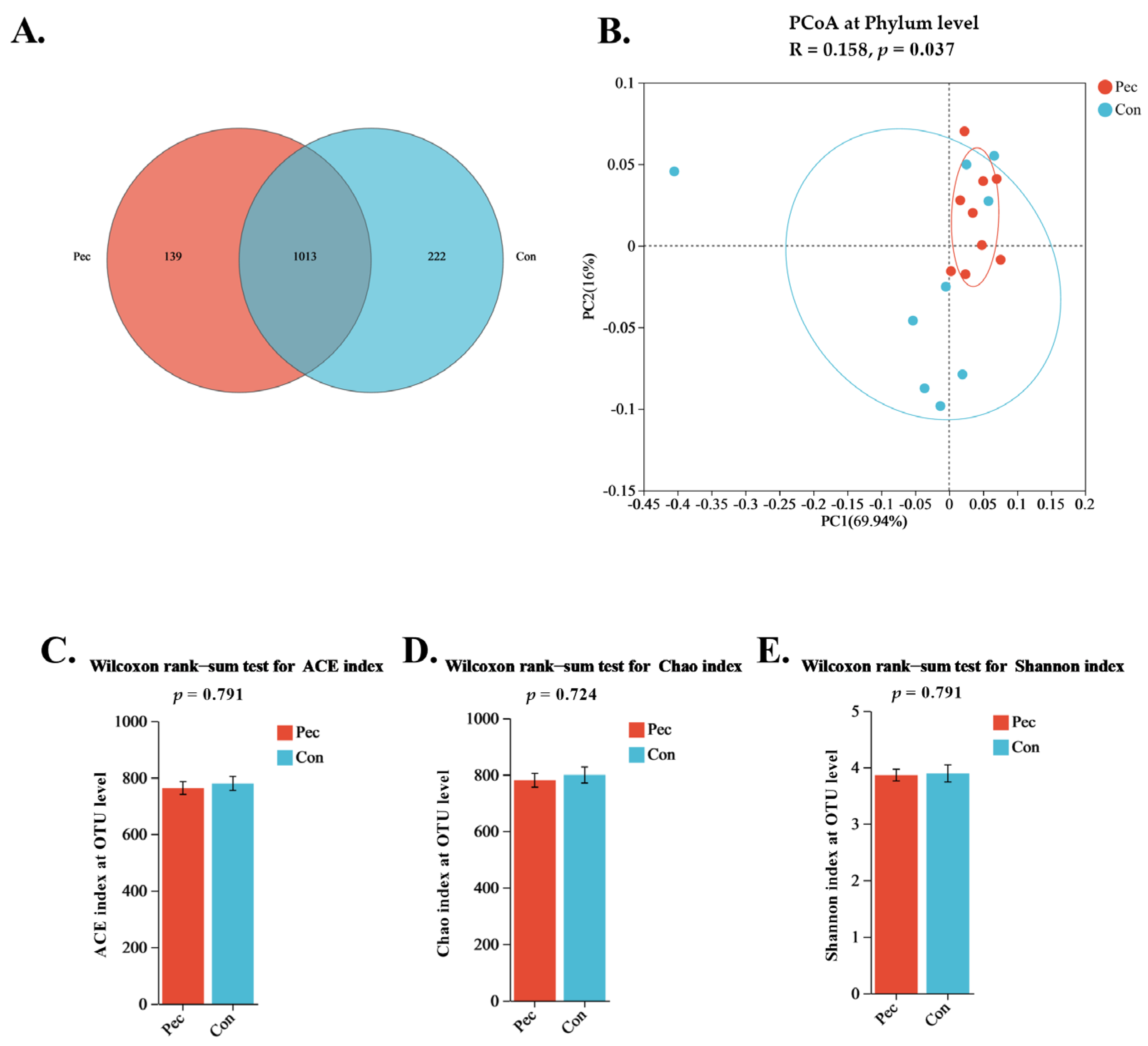
3.3. Microbiological Analysis of the Sow Fecal Samples
3.3.1. Differences in Sow Microbiota Depending on the Treatment
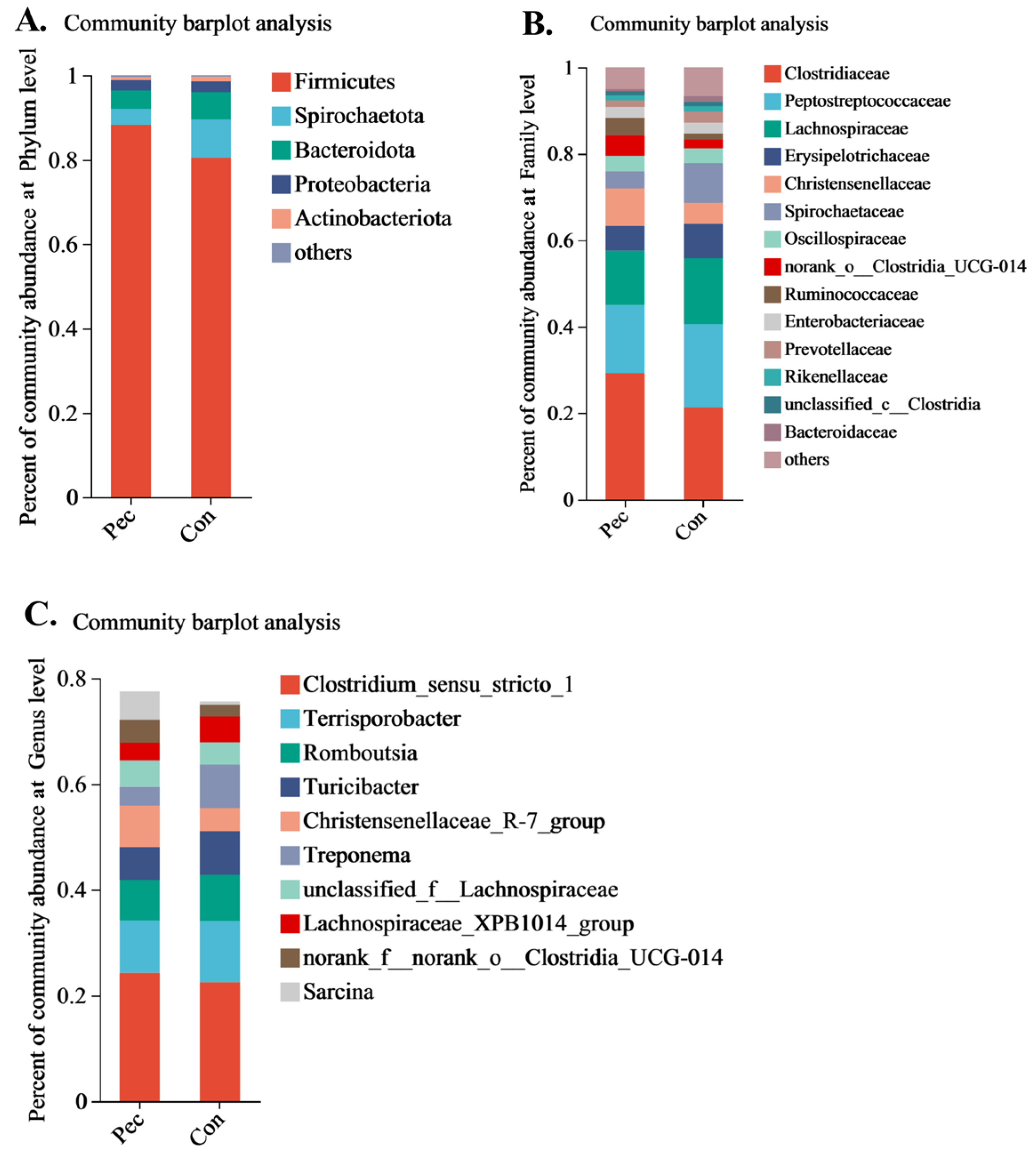
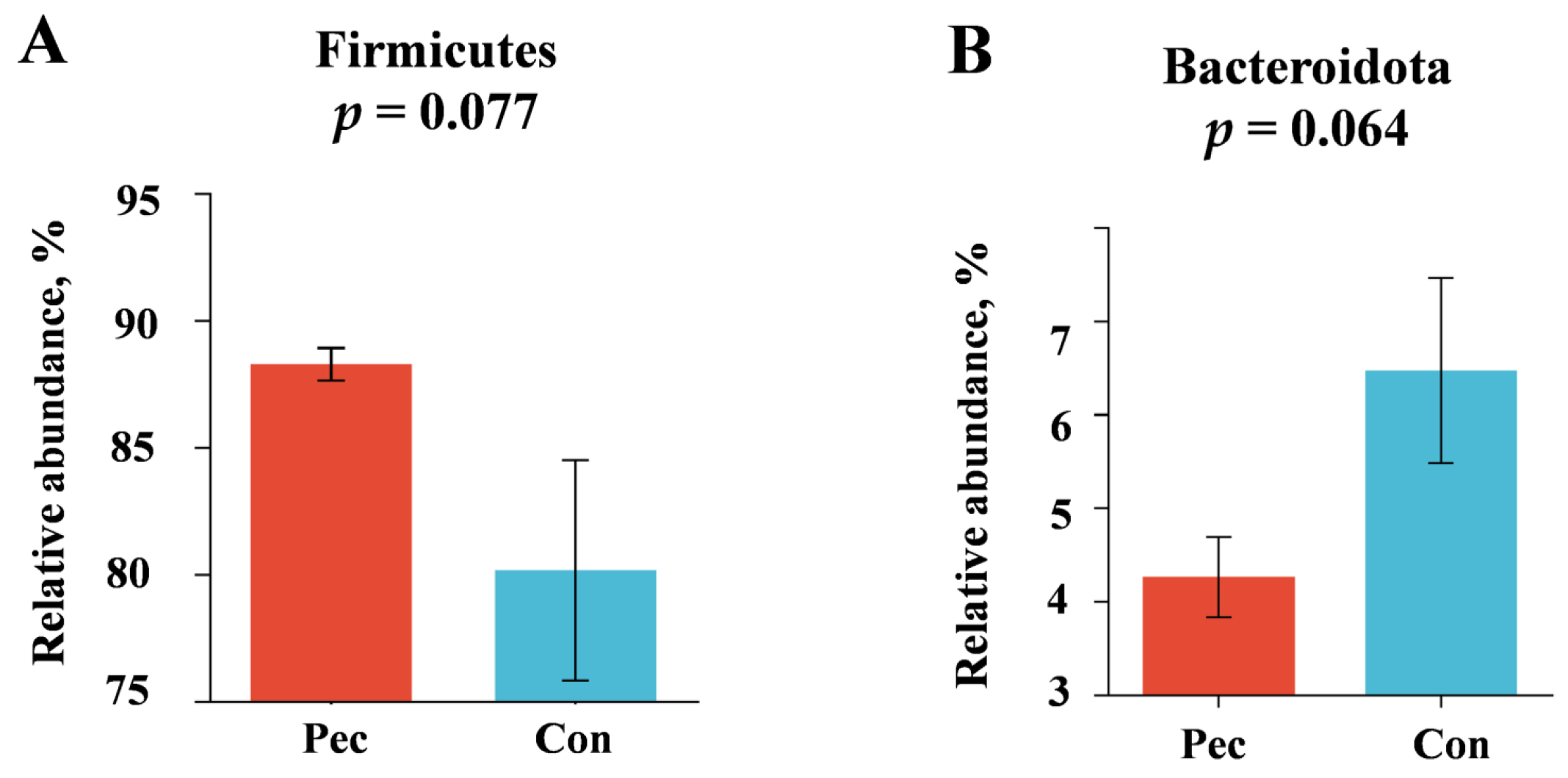
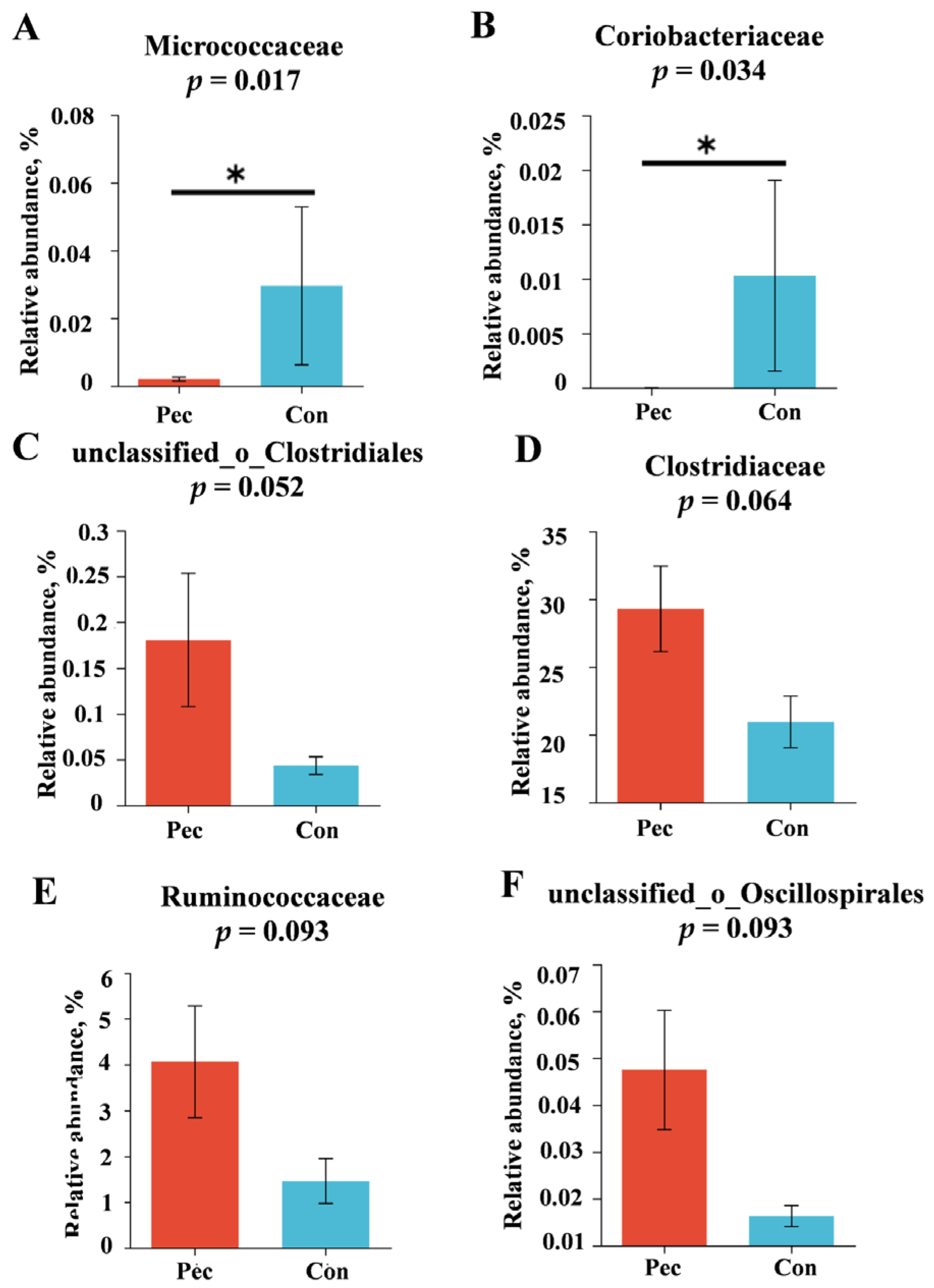
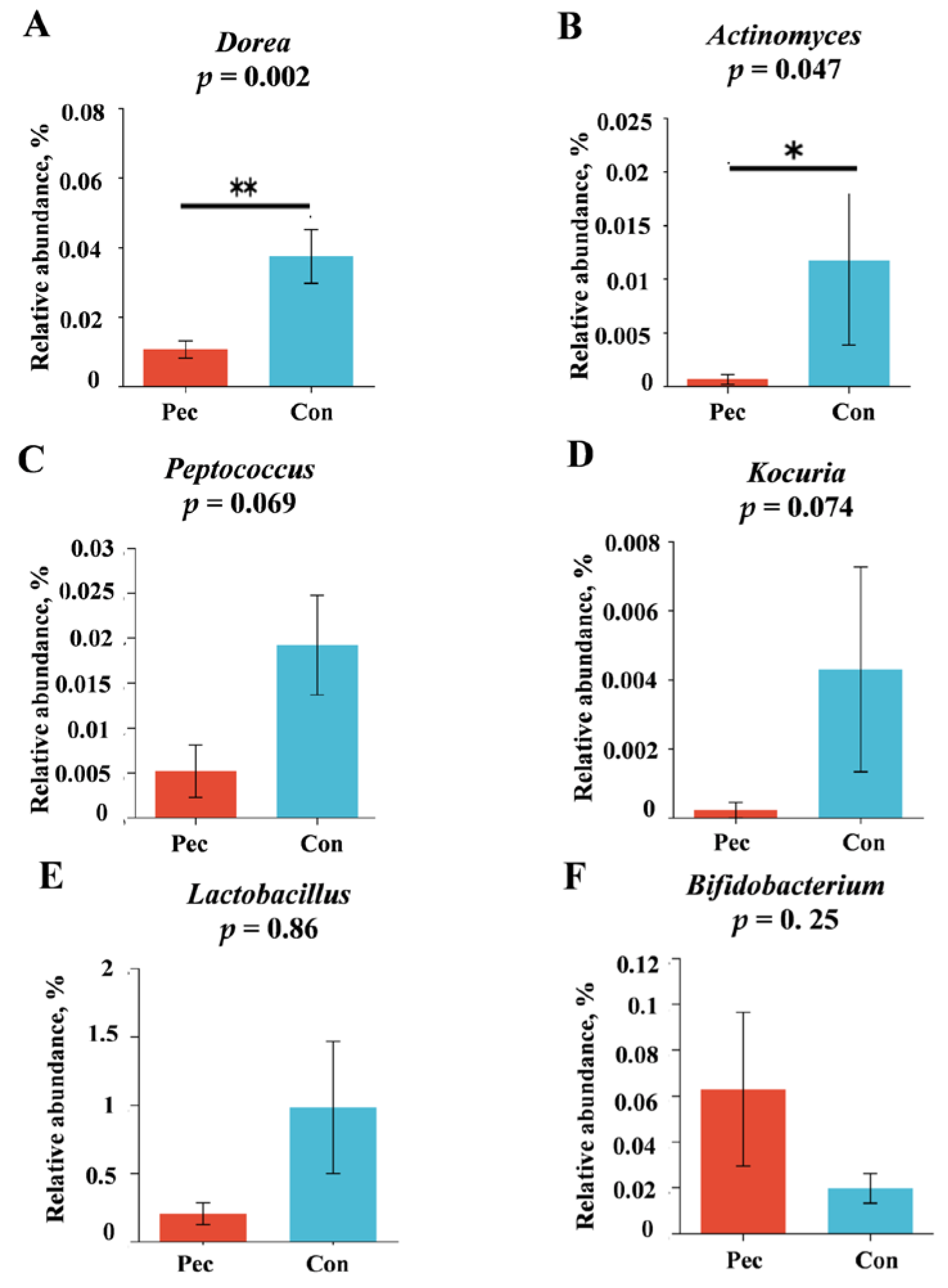
3.3.2. Changes in Relative Abundance at the Phylum, Family, and Genus Levels after the Two Treatments
3.4. Plasma Biochemical Indices
3.5. Plasma Inflammatory Cytokines
3.6. Changes in the Levels of Intestinal Cytokines and Zonulin in Sows during Late Gestation
3.7. Plasma SCFA Levels in Sows on Day 110 of Gestation
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cheng, C.; Wei, H.; Yu, H.; Xu, C.; Jiang, S.; Peng, J. Metabolic syndrome during perinatal period in sows and the link with gut microbiota and metabolites. Front. Microbiol. 2018, 9, 1989. [Google Scholar] [CrossRef] [PubMed]
- Newbern, D.; Freemark, M. Placental hormones and the control of maternal metabolism and fetal growth. Curr. Opin. Endocrinol. Diabetes Obes. 2011, 18, 409–416. [Google Scholar] [CrossRef] [PubMed]
- Sargent, I.L.; Borzychowski, A.M.; Redman, C.W. NK cells and human pregnancy–an inflammatory view. Trends Immunol. 2006, 27, 399–404. [Google Scholar] [CrossRef] [PubMed]
- Nelson, S.M.; Matthews, P.; Poston, L. Maternal metabolism and obesity: Modifiable determinants of pregnancy outcome. Hum. Reprod. Update 2010, 16, 255–275. [Google Scholar] [CrossRef] [PubMed]
- Redman, C.W.; Sacks, G.P.; Sargent, I.L. Preeclampsia: An excessive maternal inflammatory response to pregnancy. Am. J. Obstet. Gynecol. 1999, 180, 499–506. [Google Scholar] [CrossRef] [PubMed]
- Chiefari, E.; Arcidiacono, B.; Foti, D.; Brunetti, A. Gestational diabetes mellitus: An updated overview. J. Endocrinol. Investig. 2017, 40, 899–909. [Google Scholar] [CrossRef] [PubMed]
- Nadeau-Vallée, M.; Obari, D.; Palacios, J.; Brien, M.-È.; Duval, C.; Chemtob, S.; Girard, S. Sterile inflammation and pregnancy complications: A review. Reproduction 2016, 152, R277–R292. [Google Scholar] [CrossRef] [PubMed]
- Kabiru, W.; Raynor, B.D. Obstetric outcomes associated with increase in BMI category during pregnancy. Am. J. Obstet. Gynecol. 2004, 191, 928–932. [Google Scholar] [CrossRef] [PubMed]
- Madan, J.C.; Davis, J.M.; Craig, W.Y.; Collins, M.; Allan, W.; Quinn, R.; Dammann, O. Maternal obesity and markers of inflammation in pregnancy. Cytokine 2009, 47, 61–64. [Google Scholar] [CrossRef]
- Cheng, C.; Wu, X.; Zhang, X.; Zhang, X.; Peng, J. Obesity of sows at late pregnancy aggravates metabolic disorder of perinatal sows and affects performance and intestinal health of piglets. Animals 2019, 10, 49. [Google Scholar] [CrossRef]
- Li, S.; Zheng, J.; He, J.; Liu, H.; Huang, Y.; Huang, L.; Wang, K.; Zhao, X.; Feng, B.; Che, L.; et al. Dietary fiber during gestation improves lactational feed intake of sows by modulating gut microbiota. J. Anim. Sci. Biotechn. 2023, 14, 65. [Google Scholar] [CrossRef]
- Cani, P.D.; Bibiloni, R.; Knauf, C.; Waget, A.; Neyrinck, A.M.; Delzenne, N.M.; Burcelin, R.J.D. Changes in gut microbiota control metabolic endotoxemia-induced inflammation in high-fat diet–induced obesity and diabetes in mice. Diabetes 2008, 57, 1470–1481. [Google Scholar] [CrossRef] [PubMed]
- Koren, O.; Goodrich, J.K.; Cullender, T.C.; Spor, A.; Laitinen, K.; Bäckhed, H.K.; Gonzalez, A.; Werner, J.J.; Angenent, L.T.; Knight, R.J.C. Host remodeling of the gut microbiome and metabolic changes during pregnancy. Cell 2012, 150, 470–480. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Ji, Y.; Yin, C.; Deng, M.; Tang, T.; Deng, B.; Ren, W.; Deng, J.; Yin, Y.; Tan, C. Differential analysis of gut microbiota correlated with oxidative stress in sows with high or low litter performance during lactation. Front. Microbiol. 2018, 9, 1665. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Hou, C.; Li, N.; Zhang, X.; Zhang, G.; Yang, F.; Zeng, X.; Liu, Z.; Qiao, S. Microbial and metabolic alterations in gut microbiota of sows during pregnancy and lactation. FASEB J. 2019, 33, 4490–4501. [Google Scholar] [CrossRef]
- De Leeuw, J.; Bolhuis, J.; Bosch, G.; Gerrits, W. Effects of dietary fibre on behaviour and satiety in pigs: Symposium on ‘Behavioural nutrition and energy balance in the young’. Proc. Nutr. Soc. 2008, 67, 334–342. [Google Scholar] [CrossRef]
- Jarrett, S.; Ashworth, C.J. The role of dietary fibre in pig production, with a particular emphasis on reproduction. J. Anim. Sci. Biotechnol. 2018, 9, 59. [Google Scholar] [CrossRef]
- Nelssen, L.; Miller, E.R.; Pettigrew, J.E.; Ellersieck, M.R. The addition of ground wheat straw as a fiber source in the gestation diet of sows and the effect on sow and litter performance for three successive parities. J. Anim. Sci. 2009, 87, 1003–1012. [Google Scholar] [CrossRef] [PubMed]
- Leclere, L.; Van Cutsem, P.; Michiels, C. Anti-cancer activities of pH-or heat-modified pectin. Front. Pharmacol. 2013, 4, 66284. [Google Scholar] [CrossRef]
- Ridley, B.L.; O’Neill, M.A.; Mohnen, D. Pectins: Structure, biosynthesis, and oligogalacturonide-related signaling. Phytochemistry 2001, 57, 929–967. [Google Scholar] [CrossRef]
- Salman, H.; Bergman, M.; Djaldetti, M.; Orlin, J.; Bessler, H. Citrus pectin affects cytokine production by human peripheral blood mononuclear cells. Biomed. Pharmacother. 2008, 62, 579–582. [Google Scholar] [CrossRef] [PubMed]
- Sanders, L.M.; Henderson, C.E.; Hong, M.Y.; Barhoumi, R.; Burghardt, R.C.; Wang, N.; Spinka, C.M.; Carroll, R.J.; Turner, N.D.; Chapkin, R.S. An increase in reactive oxygen species by dietary fish oil coupled with the attenuation of antioxidant defenses by dietary pectin enhances rat colonocyte apoptosis. J. Nutr. 2004, 134, 3233–3238. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Dong, M.; Yang, Z.; Pan, S. Anti-diabetic effect of citrus pectin in diabetic rats and potential mechanism via PI3K/Akt signaling pathway. Int. J. Biol. Macromol. 2016, 89, 484–488. [Google Scholar] [CrossRef] [PubMed]
- Sahasrabudhe, N.M.; Beukema, M.; Tian, L.; Troost, B.; Bruininx, E.; van den Berg, M.; Schols, H.A.; Faas, M.M.; de Vos, P. Dietary fiber pectin directly blocks toll-like receptor 2–1 and prevents doxorubicin-induced ileitis. Front. Immunol. 2018, 9, 383. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.-H.; Sheu, M.-T.; Chen, T.-F.; Wang, Y.-C.; Hou, W.-C.; Liu, D.-Z.; Chung, T.-C.; Liang, Y.-C. Suppression of endotoxin-induced proinflammatory responses by citrus pectin through blocking LPS signaling pathways. Biochem. Pharmacol. 2006, 72, 1001–1009. [Google Scholar] [CrossRef] [PubMed]
- Ishisono, K.; Yabe, T.; Kitaguchi, K. Citrus pectin attenuates endotoxin shock via suppression of Toll-like receptor signaling in Peyer’s patch myeloid cells. J. Nutr. Biochem. 2017, 50, 38–45. [Google Scholar] [CrossRef] [PubMed]
- Wiese, M. The potential of pectin to impact pig nutrition and health: Feeding the animal and its microbiome. FEMS Microbiol. Lett. 2019, 366, fnz029. [Google Scholar] [CrossRef]
- Knudsen, K.E.B.; Lærke, H.N.; Hedemann, M.S. The role of fibre in piglet gut health. In Gut Efficiency; the Key Ingredient in Pig and Poultry Production; Wageningen Academic: Wageningen, The Netherlands, 2008; pp. 65–95. [Google Scholar]
- Larsen, N.; Cahú, T.B.; Saad, S.M.I.; Blennow, A.; Jespersen, L. The effect of pectins on survival of probiotic Lactobacillus spp. in gastrointestinal juices is related to their structure and physical properties. Food Microbiol. 2018, 74, 11–20. [Google Scholar] [CrossRef]
- Shinohara, K.; Ohashi, Y.; Kawasumi, K.; Terada, A.; Fujisawa, T. Effect of apple intake on fecal microbiota and metabolites in humans. Anaerobe 2010, 16, 510–515. [Google Scholar] [CrossRef]
- Onumpai, C.; Kolida, S.; Bonnin, E.; Rastall, R.A. Microbial utilization and selectivity of pectin fractions with various structures. Appl. Environ. Microbiol. 2011, 77, 5747–5754. [Google Scholar] [CrossRef]
- Martens, E.C.; Lowe, E.C.; Chiang, H.; Pudlo, N.A.; Wu, M.; McNulty, N.P.; Abbott, D.W.; Henrissat, B.; Gilbert, H.J.; Bolam, D.N. Recognition and degradation of plant cell wall polysaccharides by two human gut symbionts. PLoS Biol. 2011, 9, e1001221. [Google Scholar] [CrossRef] [PubMed]
- Grondin, J.M.; Tamura, K.; Déjean, G.; Abbott, D.W.; Brumer, H. Polysaccharide utilization loci: Fueling microbial communities. J. Bacteriol. 2017, 199, 15. [Google Scholar] [CrossRef]
- Kimura, I.; Inoue, D.; Hirano, K.; Tsujimoto, G. The SCFA receptor GPR43 and energy metabolism. Front. Endocrinol. 2014, 5, 93736. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; van Esch, B.C.; Wagenaar, G.T.; Garssen, J.; Folkerts, G.; Henricks, P.A. Pro-and anti-inflammatory effects of short chain fatty acids on immune and endothelial cells. Eur. J. Pharmacol. 2018, 831, 52–59. [Google Scholar] [CrossRef]
- Ramachandran, C.; Wilk, B.; Melnick, S.J.; Eliaz, I. Synergistic antioxidant and anti-inflammatory effects between modified citrus pectin and honokiol. Evid.-Based Complement. Altern. Med. 2017, 2017, 8379843. [Google Scholar] [CrossRef]
- Li, S.; Zheng, J.; Deng, K.; Chen, L.; Zhao, X.L.; Jiang, X.; Fang, Z.; Che, L.; Xu, S.; Feng, B.J. Supplementation with organic acids showing different effects on growth performance, gut morphology, and microbiota of weaned pigs fed with highly or less digestible diets. J. Anim. Sci. 2018, 96, 3302–3318. [Google Scholar] [CrossRef]
- Fardet, A. New hypotheses for the health-protective mechanisms of whole-grain cereals: What is beyond fibre? Nutr. Res. Rev. 2010, 23, 65–134. [Google Scholar] [CrossRef] [PubMed]
- Aune, D.; Chan, D.S.; Lau, R.; Vieira, R.; Greenwood, D.C.; Kampman, E.; Norat, T. Dietary fibre, whole grains, and risk of colorectal cancer: Systematic review and dose-response meta-analysis of prospective studies. Bmj 2011, 343, d6617. [Google Scholar] [CrossRef]
- Reynolds, A.; Mann, J.; Cummings, J.; Winter, N.; Mete, E.; Te Morenga, L. Carbohydrate quality and human health: A series of systematic reviews and meta-analyses. Lancet 2019, 393, 434–445. [Google Scholar] [CrossRef]
- Wang, Y.; Zhou, P.; Liu, H.; Li, S.; Zhao, Y.; Deng, K.; Cao, D.D.; Che, L.Q.; Fang, Z.F.; Wu, D.; et al. Effects of inulin supplementation in low-or high-fat diets on reproductive performance of sows and antioxidant defence capacity in sows and offspring. Reprod. Domest. Anim. 2016, 51, 492–500. [Google Scholar] [CrossRef]
- Quesnel, H.; Meunier-Salaun, M.-C.; Hamard, A.; Guillemet, R.; Etienne, M.; Farmer, C.; Dourmad, J.-Y.; Père, M.-C. Dietary fiber for pregnant sows: Influence on sow physiology and performance during lactation. J. Anim. Sci. 2009, 87, 532–543. [Google Scholar] [CrossRef] [PubMed]
- Che, L.; Feng, D.; Wu, D.; Fang, Z.; Lin, Y.; Yan, T. Effect of dietary fibre on reproductive performance of sows during the first two parities. Reprod. Domest. Anim. 2011, 46, 1061–1066. [Google Scholar] [CrossRef] [PubMed]
- Holscher, H.D. Dietary fiber and prebiotics and the gastrointestinal microbiota. Gut Microbes 2017, 8, 172–184. [Google Scholar] [CrossRef]
- Chung, W.S.F.; Meijerink, M.; Zeuner, B.; Holck, J.; Louis, P.; Meyer, A.S.; Wells, J.M.; Flint, H.J.; Duncan, S.H. Prebiotic potential of pectin and pectic oligosaccharides to promote anti-inflammatory commensal bacteria in the human colon. FEMS Microbiol. Ecol. 2017, 93, fix127. [Google Scholar] [CrossRef]
- Luo, Y.; Liu, Y.; Li, H.; Zhao, Y.; Wright, A.-D.G.; Cai, J.; Tian, G.; Mao, X. Differential effect of dietary fibers in intestinal health of growing pigs: Outcomes in the gut microbiota and immune-related indexes. Front. Microbiol. 2022, 13, 843045. [Google Scholar] [CrossRef] [PubMed]
- Tian, L.; Bruggeman, G.; van den Berg, M.; Borewicz, K.; Scheurink, A.J.; Bruininx, E.; de Vos, P.; Smidt, H.; Schols, H.A.; Gruppen, H. Effects of pectin on fermentation characteristics, carbohydrate utilization, and microbial community composition in the gastrointestinal tract of weaning pigs. Mol. Nutr. Food Res. 2017, 61, 1600186. [Google Scholar] [CrossRef] [PubMed]
- Tian, L.; Scholte, J.; Borewicz, K.; van den Bogert, B.; Smidt, H.; Scheurink, A.J.; Gruppen, H.; Schols, H.A. Effects of pectin supplementation on the fermentation patterns of different structural carbohydrates in rats. Mol. Nutr. Food Res. 2016, 60, 2256–2266. [Google Scholar] [CrossRef] [PubMed]
- Bianchi, F.; Larsen, N.; de Mello Tieghi, T.; Adorno, M.A.T.; Kot, W.; Saad, S.M.I.; Jespersen, L.; Sivieri, K. Modulation of gut microbiota from obese individuals by in vitro fermentation of citrus pectin in combination with Bifidobacterium longum BB-46. Appl. Microbiol. Biotechnol. 2018, 102, 8827–8840. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Fang, Q.; Fan, L.; Hong, T.; Tan, H.; Nie, S. Pectin with various degrees of esterification differentially alters gut microbiota and metabolome of healthy adults. Efood 2022, 3, e5. [Google Scholar] [CrossRef]
- Wu, Y.; Zhang, X.; Han, D.; Pi, Y.; Tao, S.; Zhang, S.; Wang, S.; Zhao, J.; Chen, L.; Wang, J. Early life administration of milk fat globule membrane promoted SCFA-producing bacteria colonization, intestinal barriers and growth performance of neonatal piglets. Anim. Nutr. 2021, 7, 346–355. [Google Scholar] [CrossRef]
- Bellahcene, M.; O’Dowd, J.F.; Wargent, E.T.; Zaibi, M.S.; Hislop, D.C.; Ngala, R.A.; Smith, D.M.; Cawthorne, M.A.; Stocker, C.J.; Arch, J.R. Male mice that lack the G-protein-coupled receptor GPR41 have low energy expenditure and increased body fat content. Br. J. Nutr. 2013, 109, 1755–1764. [Google Scholar] [CrossRef] [PubMed]
- Hara, T.; Kashihara, D.; Ichimura, A.; Kimura, I.; Tsujimoto, G.; Hirasawa, A. Role of free fatty acid receptors in the regulation of energy metabolism. Biochim. Et Biophys. Acta Mol. Cell Biol. Lipids 2014, 1841, 1292–1300. [Google Scholar] [CrossRef] [PubMed]
- Manderson, K.; Pinart, M.; Tuohy, K.; Grace, W.; Hotchkiss, A.; Widmer, W.; Yadhav, M.; Gibson, G.; Rastall, R. In vitro determination of prebiotic properties of oligosaccharides derived from an orange juice manufacturing by-product stream. Appl. Environ. Microbiol. 2005, 71, 8383–8389. [Google Scholar] [CrossRef] [PubMed]
- Bang, S.-J.; Kim, G.; Lim, M.Y.; Song, E.-J.; Jung, D.-H.; Kum, J.-S.; Nam, Y.-D.; Park, C.-S.; Seo, D.-H. The influence of in vitro pectin fermentation on the human fecal microbiome. AMB Express 2018, 8, 98. [Google Scholar] [CrossRef]
- Li, W.; Zhang, K.; Yang, H. Pectin alleviates high fat (lard) diet-induced nonalcoholic fatty liver disease in mice: Possible role of short-chain fatty acids and gut microbiota regulated by pectin. J. Agric. Food Chem. 2018, 66, 8015–8025. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Arango, L.F.; Barrett, H.L.; McIntyre, H.D.; Callaway, L.K.; Morrison, M.; Dekker Nitert, M. Connections between the gut microbiome and metabolic hormones in early pregnancy in overweight and obese women. Diabetes 2016, 65, 2214–2223. [Google Scholar] [CrossRef] [PubMed]
- Könönen, E.; Wade, W.G. Actinomyces and related organisms in human infections. Clin. Microbiol. Rev. 2015, 28, 419–442. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.; Song, M.; Gu, M.; Ren, D.; Zhu, X.; Cao, X.; Li, F.; Wang, W.; Cai, X.; Yuan, B. Dietary intake of whole strawberry inhibited colonic inflammation in dextran-sulfate-sodium-treated mice via restoring immune homeostasis and alleviating gut microbiota dysbiosis. J. Agric. Food Chem. 2019, 67, 9168–9177. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Zhu, X.; Cui, Y.; Wang, W.; Liu, H.; Li, Z.; Guo, Z.; Ma, S.; Li, D.; Wang, C. Consumption of dietary fiber from different sources during pregnancy alters sow gut microbiota and improves performance and reduces inflammation in sows and piglets. Msystems 2021, 6, 10-1128. [Google Scholar] [CrossRef]
- Huang, C.; Song, P.; Fan, P.; Hou, C.; Thacker, P.; Ma, X. Dietary sodium butyrate decreases postweaning diarrhea by modulating intestinal permeability and changing the bacterial communities in weaned piglets. J. Nutr. 2015, 145, 2774–2780. [Google Scholar] [CrossRef]
- Blander, J.M.; Longman, R.S.; Iliev, I.D.; Sonnenberg, G.F.; Artis, D. Regulation of inflammation by microbiota interactions with the host. Nat. Immunol. 2017, 18, 851–860. [Google Scholar] [CrossRef] [PubMed]
| Item, % | Gestation | Lactation | |
|---|---|---|---|
| Con | Pec | ||
| Corn | 79.59 | 76.72 | 62.89 |
| Dehulled soybean meal | 14.0 | 14.0 | 22.13 |
| Wheat bran | - | - | 6.0 |
| Fish meal | 1.5 | 1.5 | 2.6 |
| Soybean oil | 1.5 | 1.5 | 2.0 |
| Pectin | - | 3.0 | - |
| L-Lysine HCl (98%) | 0.08 | 0.06 | 0.27 |
| D,L-Methionine (99%) | - | - | 0.13 |
| L-Threonine (98.5%) | 0.08 | 0.07 | - |
| Limestone | 1.1 | 1.06 | 0.98 |
| Dicalcium phosphate | 1.2 | 1.16 | 1.5 |
| Sodium chloride | 0.4 | 0.39 | 0.4 |
| Choline chloride (50%) | 0.15 | 0.14 | 0.15 |
| Vitamin and mineral premix | 0.4 1 | 0.4 1 | 0.5 2 |
| Total | 100.00 | 100.00 | 100.00 |
| Nutrient levels 3 | |||
| DE, Mcal/kg | 3.38 | 3.28 | 3.27 |
| NE, Mcal/kg | 2.52 | 2.45 | - |
| CP, % | 14.05 | 13.81 | 17.50 |
| EE, % | 4.59 | 4.49 | |
| SDF, % | 1.72 | 4.65 | |
| IDF, % | 10.36 | 10.13 | |
| IDF/SDF | 6.02 | 2.18 | |
| DF, % | 12.08 | 14.78 | |
| Ca, % | 0.88 | 0.86 | 0.90 |
| AP, % | 0.38 | 0.37 | 0.90 |
| SID-Lys, % | 0.65 | 0.63 | 0.98 |
| SID-Met, % | 0.20 | 0.20 | |
| SID-Thr, % | 0.51 | 0.49 | |
| SID-Trp, % | 0.13 | 0.13 | |
| Item | Day of Gestation (d) | ||
|---|---|---|---|
| 0–30 | 30–90 | 90 Farrowing | |
| Daily feed allowances | |||
| Con, kg/d | 2.40 | 2.30 | 2.70 |
| Pec, kg/d | 2.49 | 2.37 | 2.78 |
| Item | Con | Pec |
|---|---|---|
| ADFI, kg/d | 2.41 | 2.49 |
| NE, Mcal/d | 6.08 | 6.09 |
| CP, g/d | 338.77 | 343.50 |
| SDF, g/d | 41.37 | 115.74 |
| IDF, g/d | 249.71 | 252.08 |
| IDF/SDF | 6.03 | 2.18 |
| DF, g/d | 291.08 | 367.82 |
| Ca, g/d | 21.27 | 21.29 |
| AP, g/d | 9.07 | 9.11 |
| SID-Lys, g/d | 15.68 | 15.73 |
| SID-Met, g/d | 4.92 | 4.99 |
| SID-Thr, g/d | 12.29 | 12.28 |
| SID-Trp, g/d | 3.08 | 3.15 |
| Items | Treatment | p-Value | |
|---|---|---|---|
| Con | Pec | ||
| Litter size at birth, No/litter | 15 | 15 | |
| Total born, n | 15.09 ± 0.04 | 15.46 ± 0.03 | 0.836 |
| Born alive, n | 13.36 ± 0.06 | 13.85 ± 0.05 | 0.812 |
| Stillborn, n | 0.33 ± 0.14 | 0.13 ± 0.09 | 0.250 |
| Mummified fetuses, n | 2.67 ± 0.17 | 1.00 ± 0.29 | 0.099 |
| Low-birth-weight piglet 1, n | 0.74 ± 0.15 | 0.41 ± 0.17 | 0.155 |
| Piglet weight at birth, kg | 1.41 ± 0.19 | 1.43 ± 0.16 | 0.653 |
| Litter weight at birth, kg | 18.77 ± 1.21 | 21.85 ± 0.95 | 0.058 |
| CVbw 2, % | 21.73 ± 1.72 | 19.97 ± 1.45 | 0.442 |
| ADFI during lactation, kg/d | |||
| d 1–7 | 3.19 ± 0.32 | 3.50 ± 0.26 | 0.448 |
| d 8–14 | 6.21 ± 0.32 | 6.02 ± 0.20 | 0.599 |
| d 15–21 | 6.26 ± 0.37 | 6.25 ± 0.25 | 0.981 |
| Total | 5.18 ± 0.22 | 5.09 ± 0.13 | 0.711 |
| Items | Treatment | p-Value | |
|---|---|---|---|
| Con | Pectin | ||
| TP, g/L | 78.04 ± 1.57 | 85.33 ± 2.05 | 0.009 |
| GLU, mmol/L | 3.96 ± 0.11 | 3.89 ± 0.14 | 0.675 |
| UREA, mmol/L | 3.24 ± 0.20 | 3.16 ± 0.15 | 0.763 |
| TC, mmol/L | 131.47 ± 3.06 | 142.46 ± 3.39 | 0.070 |
| HDL-C, mmol/L | 0.45 ± 0.04 | 0.59 ± 0.02 | 0.001 |
| LDL-C, mmol/L | 0.65 ± 0.02 | 0.65 ± 0.03 | 0.939 |
| TBA, μmol/L | 15.10 ± 1.72 | 21.39 ± 2.85 | 0.082 |
| NEFA, mmol/L | 0.26 ± 0.03 | 0.30 ± 0.03 | 0.375 |
| TG, mmol/L | 0.36 ± 0.03 | 0.31 ± 0.02 | 0.228 |
| Estradiol, pg/mL | 78.04 ± 1.57 | 85.33 ± 2.08 | 0.027 |
| Items | Treatment | p Value | |
|---|---|---|---|
| Con | Pec | ||
| IL-6, pg/mL | 756.212 ± 24.76 | 647.781 ± 19.69 | 0.002 |
| IL-10, pg/mL | 142.400 ± 5.047 | 135.371 ± 1.82 | 0.155 |
| IL-1β, pg/mL | 660.178 ± 16.70 | 562.002 ± 12.35 | <0.01 |
| TNF-α, pg/mL | 200.516 ± 6.67 | 179.009 ± 1.80 | <0.01 |
| IFN-γ, pg/mL | 35.861 ± 1.33 | 33.402 ± 0.74 | 0.097 |
| Items | Treatment | p Value | |
|---|---|---|---|
| Con | Pec | ||
| LCN-2, pg/mL | 10.458 ± 0.38 | 9.341 ± 0.43 | 0.079 |
| Zonulin, ng/L | 610.452 ± 34.15 | 522.237 ± 21.29 | 0.046 |
| ZO-1, pg/mL | 148.759 ± 8.08 | 143.044 ± 8.52 | 0.651 |
| Occludin, pg/mL | 257.742 ± 7.74 | 249.183 ± 10.55 | 0.533 |
| Items | Treatment | p Value | |
|---|---|---|---|
| Con | Pec | ||
| Acetate, μmol/L | 424.69 ± 12.20 | 486.54 ± 14.59 | 0.004 |
| Propionate, μmol/L | 85.14 ± 16.10 | 166.08 ± 12.06 | 0.001 |
| Butyrate, μmol/L | 22.91 ± 5.97 | 44.24 ± 8.09 | 0.085 |
| Total SCFAs, μmol/L | 441.97 ± 57.09 | 673.87 ± 26.46 | 0.001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zheng, J.; Li, S.; He, J.; Liu, H.; Huang, Y.; Jiang, X.; Zhao, X.; Li, J.; Feng, B.; Che, L.; et al. A Gestational Pectin Diet Could Improve the Health of Multiparous Sows by Modulating the Gut Microbiota and Cytokine Level during Late Pregnancy. Animals 2024, 14, 1559. https://doi.org/10.3390/ani14111559
Zheng J, Li S, He J, Liu H, Huang Y, Jiang X, Zhao X, Li J, Feng B, Che L, et al. A Gestational Pectin Diet Could Improve the Health of Multiparous Sows by Modulating the Gut Microbiota and Cytokine Level during Late Pregnancy. Animals. 2024; 14(11):1559. https://doi.org/10.3390/ani14111559
Chicago/Turabian StyleZheng, Jie, Shuang Li, Jiaqi He, Hao Liu, Yingyan Huang, Xuemei Jiang, Xilun Zhao, Jian Li, Bin Feng, Lianqiang Che, and et al. 2024. "A Gestational Pectin Diet Could Improve the Health of Multiparous Sows by Modulating the Gut Microbiota and Cytokine Level during Late Pregnancy" Animals 14, no. 11: 1559. https://doi.org/10.3390/ani14111559
APA StyleZheng, J., Li, S., He, J., Liu, H., Huang, Y., Jiang, X., Zhao, X., Li, J., Feng, B., Che, L., Fang, Z., Xu, S., Lin, Y., Hua, L., Zhuo, Y., & Wu, D. (2024). A Gestational Pectin Diet Could Improve the Health of Multiparous Sows by Modulating the Gut Microbiota and Cytokine Level during Late Pregnancy. Animals, 14(11), 1559. https://doi.org/10.3390/ani14111559






