Factor in Fear: Interference Competition in Polymorphic Spadefoot Toad Tadpoles and Its Potential Role in Disruptive Selection
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study System
2.2. Behavioral Trials
2.3. Competition Experiment
2.4. Data Analysis
3. Results
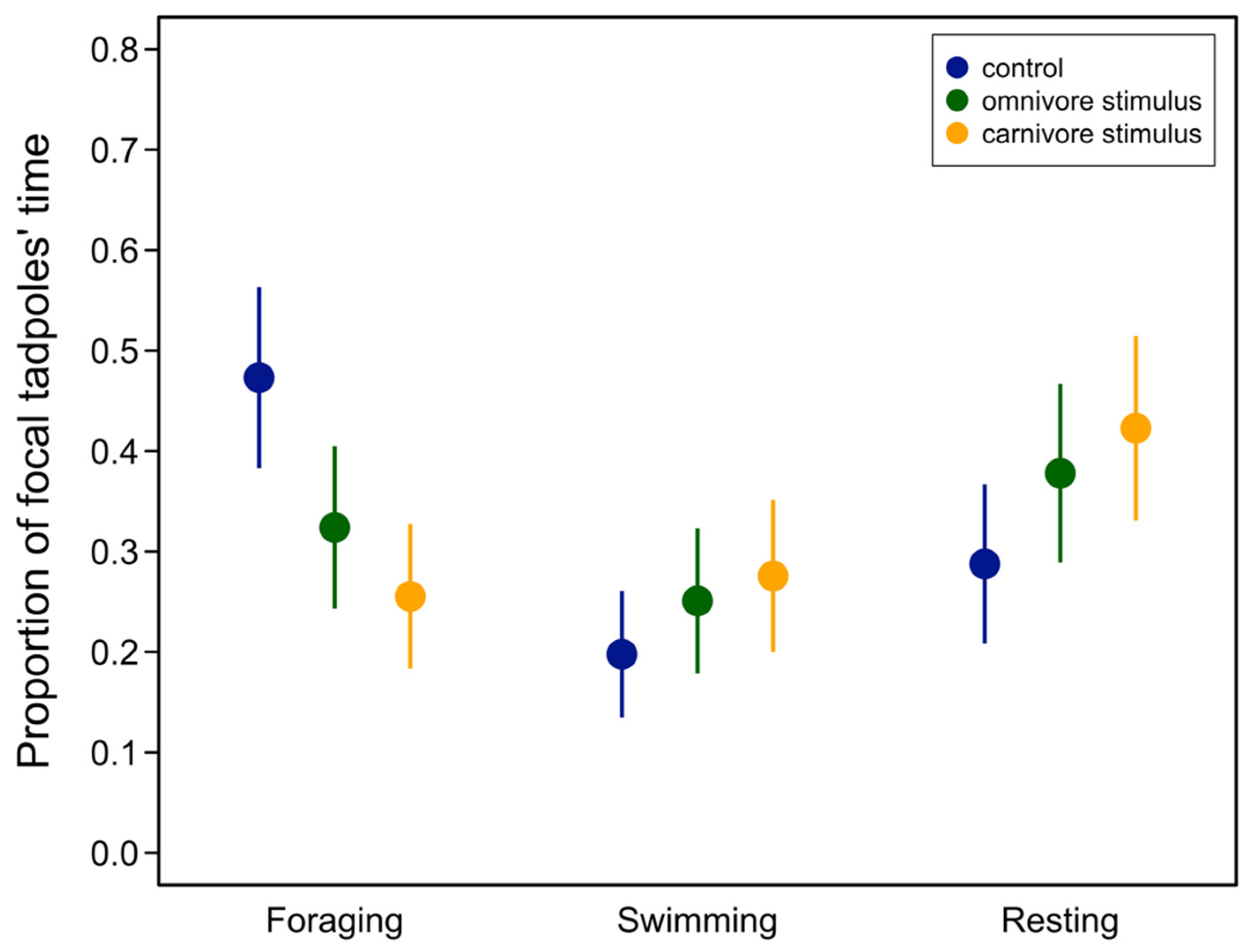
3.1. Behavioral Trials
3.2. Competition Experiment
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Darwin, C. On the Origin of Species by Means of Natural Selection, or the Preservation of Favoured Races in the Struggle for Life, 1st ed.; John Murray: London, UK, 1859. [Google Scholar]
- Pfennig, K.S.; Pfennig, D.W. Character Displacement: Ecological and Reproductive Responses to a Common Evolutionary Problem. Q. Rev. Biol. 2009, 84, 253–276. [Google Scholar] [CrossRef] [PubMed]
- Abrams, P.A.; Matsuda, H.; Harada, Y. Evolutionarily Unstable Fitness Maxima and Stable Fitness Minima of Continuous Traits. Evol. Ecol. 1993, 7, 465–487. [Google Scholar] [CrossRef]
- Bolnick, D.I. Can Intraspecfic Competition Drive Disruptive Selection? An Experimental Test in Natural Populations of Sticklebacks. Evolution 2004, 58, 608–618. [Google Scholar]
- Martin, R.A.; Pfennig, D.W. Disruptive Selection in Natural Populations: The Roles of Ecological Specialization and Resource Competition. Am. Nat. 2009, 174, 268–281. [Google Scholar] [CrossRef] [PubMed]
- Rueffler, C.; Van Dooren, T.J.M.; Leimar, O.; Abrams, P.A. Disruptive Selection and Then What? Trends Ecol. Evol. 2006, 21, 238–245. [Google Scholar] [CrossRef]
- Smith, T.B.; Skúlason, S. Evolutionary Significance of Resource Polymorphisms in Fishes, Amphibians, and Birds. Annu. Rev. Ecol. Syst. 1996, 27, 111–133. [Google Scholar] [CrossRef]
- Rosenzweig, M.L. Competitive Speciation. Biol. J. Linn. Soc. 1978, 10, 275–289. [Google Scholar] [CrossRef]
- Maynard Smith, J. Disruptive Selection, Polymorphism and Sympatric Speciation. Nature 1962, 195, 60–62. [Google Scholar] [CrossRef]
- Schluter, D. Adaptive Radiation in Sticklebacks: Trade-Offs in Feeding Performance and Growth. Ecology 1995, 76, 82–90. [Google Scholar] [CrossRef]
- Robinson, B.W.; Wilson, D.S.; Shea, G.O. Trade-Offs of Ecological Specialization: An Intraspecific Comparison of Pumpkinseed Sunfish Phenotypes. Ecology 1996, 77, 170–178. [Google Scholar] [CrossRef]
- Hendry, A.P.; Huber, S.K.; De León, L.F.; Herrel, A.; Podos, J. Disruptive Selection in a Bimodal Population of Darwin’s Finches. Proc. R. Soc. B Biol. Sci. 2008, 276, 753–759. [Google Scholar] [CrossRef] [PubMed]
- Dijkstra, P.D.; Border, S.E. How Does Male–Male Competition Generate Negative Frequency-Dependent Selection and Disruptive Selection during Speciation? Curr. Zool. 2018, 64, 89–99. [Google Scholar] [CrossRef] [PubMed]
- Martin, R.A.; Pfennig, D.W. Field and Experimental Evidence That Competition and Ecological Opportunity Promote Resource Polymorphism. Biol. J. Linn. Soc. 2010, 100, 73–88. [Google Scholar] [CrossRef]
- Dubois, F.; Giraldeau, L.A. Fighting for Resources: The Economics of Defense and Appropriation. Ecology 2005, 86, 3–11. [Google Scholar] [CrossRef]
- Bolnick, D.I.; Svanbäck, R.; Fordyce, J.A.; Yang, L.H.; Davis, J.M.; Hulsey, C.D.; Forister, M.L. The Ecology of Individuals: Incidence and Implications of Individual Specialization. Am. Nat. 2003, 161, 1–28. [Google Scholar] [CrossRef]
- Rudolf, V.H.W. The Interaction of Cannibalism and Omnivory: Consequences for Community Dynamics. Ecology 2007, 88, 2697–2705. [Google Scholar] [CrossRef]
- Andersson, J.; Byström, P.; Claessen, D.; Persson, L.; Roos, A.M.D. Stabilization of Population Fluctuations Due to Cannibalism Promotes Resource Polymorphism in Fish. Am. Nat. 2007, 169, 820–829. [Google Scholar] [CrossRef]
- Brown, J.S.; Laundré, J.W.; Gurung, M. The Ecology of Fear: Optimal Foraging, Game Theory, and Trophic Interactions. J. Mammal. 1999, 80, 385–399. [Google Scholar] [CrossRef]
- Preisser, E.L.; Bolnick, D.I.; Benard, M.E. Scared to Death? The Effects of Intimidation and Consumption in Predator-Prey Interactions. Ecology 2005, 86, 501–509. [Google Scholar]
- Werner, E.E.; Peacor, S.D. A Review of Trait-Mediated Indirect Interactions in Ecological Communities. Ecology 2003, 84, 1083–1100. [Google Scholar] [CrossRef]
- Brock, K.M.; Bednekoff, P.A.; Pafilis, P.; Foufopoulos, J. Evolution of Antipredator Behavior in an Island Lizard Species, Podarcis erhardii (Reptilia: Lacertidae): The Sum Of All Fears? Evolution 2015, 69, 216–231. [Google Scholar] [CrossRef] [PubMed]
- du Preez, B.; Hart, T.; Loveridge, A.J.; Macdonald, D.W. Impact of Risk on Animal Behaviour and Habitat Transition Probabilities. Anim. Behav. 2015, 100, 22–37. [Google Scholar] [CrossRef]
- Peckarsky, B.L.; Abrams, P.A.; Bolnick, D.I.; Dill, L.M.; Grabowski, J.H.; Luttbeg, B.; Orrock, J.L.; Peacor, S.D.; Preisser, E.L.; Schmitz, O.J.; et al. Revisiting the Classics: Considering Nonconsumptive Effects in Textbook Examples of Predator-Prey Interactions. Ecology 2008, 89, 2416–2425. [Google Scholar] [CrossRef] [PubMed]
- Sheriff, M.J.; Peacor, S.D.; Hawlena, D.; Thaker, M. Non-Consumptive Predator Effects on Prey Population Size: A Dearth of Evidence. J. Anim. Ecol. 2020, 89, 1302–1316. [Google Scholar] [CrossRef] [PubMed]
- Pfennig, D. The Adaptive Significance of an Environmentally-Cued Developmental Switch in an Anuran Tadpole. Oecologia 1990, 85, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Pfennig, D.W.; Rice, A.M.; Martin, R.A. Field and Experimental Evidence for Competition’s Role in Phenotypic Divergence. Evolution 2007, 61, 257–271. [Google Scholar] [CrossRef]
- Pomeroy, L.V. Developmental Polymorphism in the Tadpoles of the Spadefoot Toad Scaphiopus multiplicatus. Ph.D. Thesis, University of California, Riverside, CA, USA, 1981. [Google Scholar]
- Pfennig, D.W.; Frankino, W.A. Kin-Mediated Morphogenesis in Facultatively Cannibalistic Tadpoles. Evolution 1997, 51, 1993–1999. [Google Scholar]
- Pfennig, D.W. Proximate and Functional Causes of Polyphenism in an Anuran Tadpole. Funct. Ecol. 1992, 6, 167–174. [Google Scholar] [CrossRef]
- Pfennig, D.W. Polyphenism in Spadefoot Toad Tadpoles as a Logically Adjusted Evolutionarily Stable Strategy. Evolution 1992, 46, 1408–1420. [Google Scholar] [CrossRef]
- Martin, R.A.; Pfennig, D.W. Widespread Disruptive Selection in the Wild Is Associated with Intense Resource Competition. BMC Evol. Biol. 2012, 12, 136. [Google Scholar] [CrossRef]
- Morey, S.; Reznick, D. A Comparative Analysis of Plasticity in Larval Development in Three Species of Spadefoot Toads. Ecology 2000, 81, 1736–1749. [Google Scholar] [CrossRef]
- Bates, D.; Mäechler, M.; Bolker, B.; Walker, S. Mixed-effects models using lme4. J. Stat Softw. 2015, 67, 1–48. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Project for Statistical Computing: Vienna, Austria, 2009. [Google Scholar]
- Lenth, R.V.; Bolker, B.; Buerkner, P.; Giné-Vázquez, I.; Herve, M.; Jung, M.; Love, J.; Miguez, F.; Riebl, H.; Singmann, H. Emmeans: Estimated Marginal Means, Aka Least-Squares Means. 2018. Available online: https://github.com/rvlenth/emmeans (accessed on 10 March 2023).
- Schluter, D. The Ecology of Adaptive Radiation; Oxford University Press: Oxford, UK, 2000. [Google Scholar]
- Pfennig, D.; Pfennig, K. Evolution’s Wedge: Competition and the Origins of Diversity; University of California Press: Berkeley, CA, USA, 2012. [Google Scholar]
- Pfennig, D.W.; Reeve, H.K.; Sherman, P.W. Kin Recognition and Cannibalism in Spadefoot Toad Tadpoles. Anim. Behav. 1993, 46, 87–94. [Google Scholar] [CrossRef]
- Benard, M.F. Predator-Induced Phenotypic Plasticity in Organisms with Complex Life Histories. Annu. Rev. Ecol. Evol. Syst. 2004, 35, 651–673. [Google Scholar] [CrossRef]
- Relyea, R.A. Competitor-Induced Plasticity in Tadpoles: Consequences, Cues, and Connections to Predator-Induced Plasticity. Ecol. Monogr. 2002, 72, 523–540. [Google Scholar] [CrossRef]
- Yu, T.L.; Lambert, M.R. Conspecific Visual Cues: The Relative Importance of Interference and Exploitation Competition among Tadpoles of Rana kukunoris. Ethol. Ecol. Evol. 2017, 29, 193–199. [Google Scholar] [CrossRef]
- Rot-Nikcevic, I.; Taylor, C.N.; Wassersug, R.J. The Role of Images of Conspecifics as Visual Cues in the Development and Behavior of Larval Anurans. Behav. Ecol. Sociobiol. 2006, 60, 19–25. [Google Scholar] [CrossRef]
- Sutherland, M.; Gouchie, G.; Wassersug, R. Can Visual Stimulation Alone Induce Phenotypically Plastic Responses in Rana Sylvatica Tadpole Oral Structures. J. Herpetol. 2009, 43, 165–1681. [Google Scholar] [CrossRef]
- Pfennig, D.W. Kin recognition among spadefoot toad tadpoles: A side-effect of habitat selection? Evolution 1990, 44, 785–798. [Google Scholar] [PubMed]
- Pfennig, K.S.; Pfennig, D.W. Dead Spadefoot Tadpoles Adaptively Modify Development in Future Generations: A Novel Form of Nongenetic Inheritance? Copeia 2020, 108, 116–121. [Google Scholar] [CrossRef]
- Pfennig, D.W. Cannibalistic Tadpoles That Pose the Greatest Threat to Kin Are Most Likely to Discriminate Kin. Proc. R. Soc. B Biol. Sci. 1999, 266, 57. [Google Scholar] [CrossRef]
- Dugas, M.B.; McCormack, L.; Gadau, A.; Martin, R.A. Choosy Cannibals Preferentially Consume Siblings with Relatively Low Fitness Prospects. Am. Nat. 2016, 188, 124–131. [Google Scholar] [CrossRef]
- Martin, R.A.; Garnett, S.C. Relatedness and Resource Diversity Interact to Influence the Intensity of Competition. Biol. J. Linn. Soc. 2013, 110, 689–695. [Google Scholar] [CrossRef]
- Frankino, W.A.; Pfennig, D.W. Condition-Dependent Expression of Trophic Polyphenism: Effects of Individual Size and Competitive Ability. Evol. Ecol. Res. 2001, 3, 939–951. [Google Scholar]
- Polis, G.A. The Evolution and Dynamics of Intraspecific Predation. Annu. Rev. Ecol. Syst. 1981, 12, 225–251. [Google Scholar] [CrossRef]
- Pfennig, D.W.; Sherman, P.W.; Collins, J.P. Kin Recognition and Cannibalism in Polyphenic Salamanders. Behav. Ecol. 1994, 5, 225–232. [Google Scholar] [CrossRef]
- Svanbäck, R.; Persson, L. Individual Diet Specialization, Niche Width and Population Dynamics: Implications for Trophic Polymorphisms. J. Anim. Ecol. 2004, 73, 973–982. [Google Scholar] [CrossRef]
- Persson, L.; Bystrom, P.; Wahlstrom, E. Cannibalism and Competition in Eurasian Perch: Population Dynamics of an Ontogenetic Omnivore. Ecology 2000, 81, 1058–1071. [Google Scholar] [CrossRef]
- Pfennig, D.W. Evolution, Development, and Behavior of Alternative Amphibian Morphologies. Ph.D. Thesis, University of Texas, Austin, TX, USA, 1989. [Google Scholar]
- Sutherland, W.J. Why Do Animals Specialize? Nature 1987, 325, 483–484. [Google Scholar] [CrossRef]
- Goss-Custard, J.D.; Durell, S.L.V.D. Individual and Age Differences in the Feeding Ecology of Oystercatchers Haematopus ostralegus Wintering on the Exe Estuary, Devon. Ibis 1983, 125, 155–171. [Google Scholar] [CrossRef]
- Goss-Custard, J.D.; Clarke, R.T.; Dit Durell, S.L.V. Rates of Food Intake and Aggression of Oystercatchers Haematopus ostralegus on the Most and Least Preferred Mussel Mytilus Edulis Beds of the Exe Estuary. J. Anim. Ecol. 1984, 53, 233–245. [Google Scholar] [CrossRef]
- Sinervo, B.; Lively, C.M. The Rock-Paper-Scissors Game and the Evolution of Alternative Male Strategies. Nature 1996, 380, 240–243. [Google Scholar] [CrossRef]
- Grether, G.F.; Losin, N.; Anderson, C.N.; Okamoto, K. The Role of Interspecific Interference Competition in Character Displacement and the Evolution of Competitor Recognition. Biol. Rev. 2009, 84, 617–635. [Google Scholar] [CrossRef] [PubMed]
- Ledón-Rettig, C.C.; Pfennig, D.W. Antipredator Behavior Promotes Diversification of Feeding Strategies. Integr. Comp. Biol. 2012, 52, 53–63. [Google Scholar] [CrossRef]
- Amarasekare, P. Interference Competition and Species Coexistence. Proc. R. Soc. Lond. B Biol. Sci. 2002, 269, 2541–2550. [Google Scholar] [CrossRef]
- Greig, D.; Travisano, M. Density-Dependent Effects on Allelopathic Interactions in Yeast. Evolution 2008, 62, 521–527. [Google Scholar] [CrossRef] [PubMed]
- Rendueles, O.; Amherd, M.; Velicer, G.J. Positively Frequency-Dependent Interference Competition Maintains Diversity and Pervades a Natural Population of Cooperative Microbes. Curr. Biol. 2015, 25, 1673–1681. [Google Scholar] [CrossRef]
- Czárán, T.L.; Hoekstra, R.F.; Pagie, L. Chemical Warfare between Microbes Promotes Biodiversity. Proc. Natl. Acad. Sci. USA 2002, 99, 786–790. [Google Scholar] [CrossRef]

| Behavior | Contrast | Estimate | SE | Z | p |
|---|---|---|---|---|---|
| Foraging | carnivore–control | −0.217 | 0.0349 | −6.24 | <0.0001 |
| carnivore–omnivore | −0.0685 | 0.0324 | −2.11 | 0.0347 | |
| control–omnivore | −0.149 | 0.0352 | 4.23 | <0.0001 | |
| Swimming | carnivore–control | 0.0778 | 0.300 | 2.58 | 0.0289 |
| carnivore–omnivore | 0.0247 | 0.309 | 0.801 | 0.423 | |
| control–omnivore | −0.0531 | 0.029 | −1.82 | 0.102 | |
| Resting | carnivore–control | 0.135 | 0.0346 | 3.89 | 0.003 |
| carnivore–omnivore | 0.0448 | 0.0355 | 1.26 | 0.206 | |
| control–omnivore | −0.0902 | 0.0339 | −2.65 | 0.0118 |
| Contrast | Estimate | SE | T Ratio | p |
|---|---|---|---|---|
| e,c—i,c | 0.575 | 0.0906 | 6.35 | <0.0001 |
| e,c—e,o | −0.0000625 | 0.0906 | −0.001 | 0.999 |
| e,c—i,o | −0.104 | 0.0906 | −1.15 | 0.305 |
| i,c—e,o | −0.575 | 0.0906 | −6.35 | <0.0001 |
| i,c—i,o | −0.679 | 0.0906 | −7.50 | <0.0001 |
| e,o—i,o | −0.104 | 0.0906 | −1.15 | 0.304 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Strugariu, A.; Martin, R.A. Factor in Fear: Interference Competition in Polymorphic Spadefoot Toad Tadpoles and Its Potential Role in Disruptive Selection. Animals 2023, 13, 1264. https://doi.org/10.3390/ani13071264
Strugariu A, Martin RA. Factor in Fear: Interference Competition in Polymorphic Spadefoot Toad Tadpoles and Its Potential Role in Disruptive Selection. Animals. 2023; 13(7):1264. https://doi.org/10.3390/ani13071264
Chicago/Turabian StyleStrugariu, Alexandru, and Ryan Andrew Martin. 2023. "Factor in Fear: Interference Competition in Polymorphic Spadefoot Toad Tadpoles and Its Potential Role in Disruptive Selection" Animals 13, no. 7: 1264. https://doi.org/10.3390/ani13071264
APA StyleStrugariu, A., & Martin, R. A. (2023). Factor in Fear: Interference Competition in Polymorphic Spadefoot Toad Tadpoles and Its Potential Role in Disruptive Selection. Animals, 13(7), 1264. https://doi.org/10.3390/ani13071264







