Overexpression of DGAT2 Regulates the Differentiation of Bovine Preadipocytes
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Bovine Preadipocytes Isolation, Culture, and Differentiation
2.2. Vector Construction and Adenovirus Packaging
2.3. Optimisation of Conditions for DGAT2 Overexpression/Knockdown
2.4. RNA Extraction and Quantitative Real-Time PCR (qRT-PCR) Detection
2.5. Western Blotting
2.6. Oil Red O Staining and Triglyceride Determination
2.7. Determination of Adiponectin (ADP) Concentration
2.8. RNA Sequencing (RNA-seq)
2.9. Gene Ontology (GO) and Kyoto Encyclopaedia of Genes and Genomes (KEGG) Enrichment Analysis
2.10. Statistical Analyses
3. Results
3.1. Optimal MOI Value and Infection Efficiency of the Shuttle Viruses
3.2. DGAT2 Affects TAG Accumulation, ADP Content, and LD Formation in Bovine Preadipocyte
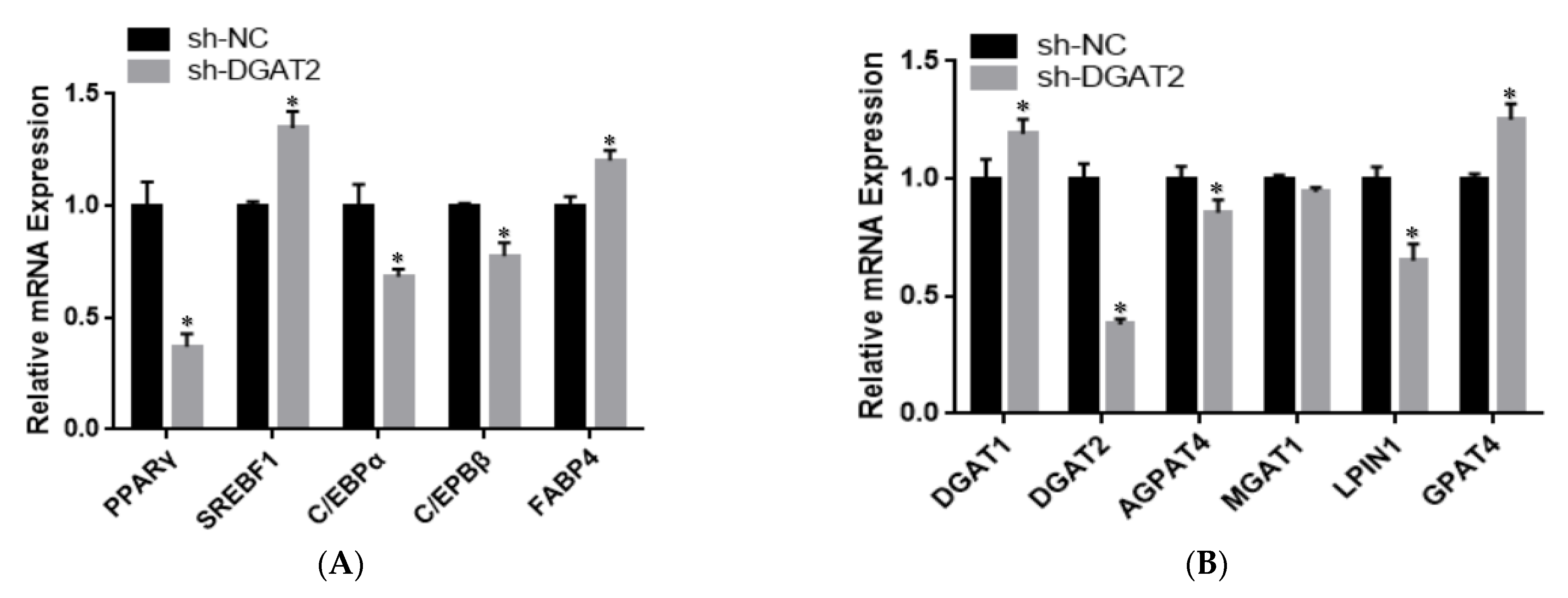
3.3. DGAT2 Affects the Differentiation of Bovine Preadipocytes
3.4. Differential Analysis of Ad–DGAT2/sh–DGAT2 Infected Bovine Preadipocytes
3.5. GO Function Analysis
3.6. KEGG Pathway Enrichment Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wang, X.; Li, G.P. Beef quality traits and their influencing factors. Chin. J. Anim. Nutr. 2019, 31, 4949–4958. [Google Scholar]
- Martins, T.S.; Sanglard, L.M.P.; Silva, W.; Chizzotti, M.L.; Rennó, L.N.; Serão, N.V.L.; Silva, F.F.; Guimarães, S.E.F.; Ladeira, M.M.; Dodson, M.V.; et al. Molecular factors underlying the deposition of intramuscular fat and collagen in skeletal muscle of Nellore and Angus Cattle. PLoS ONE 2015, 10, e0139943. [Google Scholar] [CrossRef] [PubMed]
- Billecke, N.; Bosma, M.; Rock, W.; Fleissner, F.; Best, G.; Schrauwen, P.; Kersten, S.; Bonn, M.; Hesselink, M.K.C.; Parekh, S.H. Perilipin 5 mediated lipid droplet remodelling revealed by coherent Raman imaging. Integr. Biol. 2015, 7, 467–476. [Google Scholar] [CrossRef] [PubMed]
- Coleman, R.A.; Lee, D.P. Enzymes of triacylglycerol synthesis and their regulation. Prog. Lipid Res. 2004, 43, 134–176. [Google Scholar] [CrossRef] [PubMed]
- Turchetto-Zolet, A.C.; Maraschin, F.S.; de Morais, G.L.; Cagliari, A.; Andrade, C.M.; Margis-Pinheiro, M.; Margis, R. Evolutionary view of acyl-CoA diacylglycerol acyltransferase (DGAT), a key enzyme in neutral lipid biosynthesis. BMC Evol. Biol. 2011, 11, 263. [Google Scholar] [CrossRef]
- Yang, L.; Liu, Z.; Ou, K.; Wang, T.; Li, Z.; Tian, Y.; Wang, Y.; Kang, X.; Li, H.; Liu, X. Evolution, dynamic expression changes and regulatory characteristics of gene families involved in the glycerophosphate pathway of triglyceride synthesis in chicken (Gallus gallus). Sci. Rep. 2019, 9, 12735. [Google Scholar] [CrossRef]
- Bhatt-Wessel, B.; Jordan, T.W.; Miller, J.H.; Peng, L. Role of DGAT enzymes in triacylglycerol metabolism. Arch. Biochem. Biophys. 2018, 655, 1–11. [Google Scholar] [CrossRef]
- Smith, S.J.; Cases, S.; Jensen, D.R.; Chen, H.C.; Sande, E.; Tow, B.; Sanan, D.A.; Raber, J.; Eckel, R.H., Jr.; Farese, R.V. Obesity resistance and multiple mechanisms of triglyceride synthesis in mice lacking DGAT. Nat. Genet. 2000, 25, 87–90. [Google Scholar] [CrossRef]
- Stone, S.J.; Myers, H.M.; Watkins, S.M.; Brown, B.E.; Feingold, K.R.; Elias, P.M.; Farese, R.V. Lipopenia and skin barrier abnormalities in DGAT2-deficient mice. J. Biol. Chem. 2004, 279, 11767–11776. [Google Scholar] [CrossRef]
- Ramsay, T.G. Porcine preadipocyte proliferation and differentiation: A role for leptin? J. Anim. Sci. 2005, 83, 2066–2074. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and 2-ΔΔCT method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- McFie, P.J.; Banman, S.L.; Kary, S.; Stone, S.J. Murine diacylglycerol acyltransferase-2 (DGAT2) can catalyze triacylglycerol synthesis and promote lipid droplet formation independent of its localization to the endoplasmic reticulum. J. Biol. Chem. 2011, 286, 28235–28246. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.H.; Ginsberg, H.N. The role of acyl-CoA: Diacylglycerol acyltransferase (DGAT) in energy metabolism. Ann. Med. 2004, 36, 252–261. [Google Scholar] [CrossRef] [PubMed]
- van Rijn, J.M.; van Hoesel, M.; de Heus, C.; van Vugt, A.H.M.; Klumperman, J.; Nieuwenhuis, E.E.S.; Houwen, R.H.J.; Middendorp, S. DGAT2 partially compensates for lipid-induced ER stress in human DGAT1-deficient intestinal stem cells. J. Lipid Res. 2019, 60, 1787–1800. [Google Scholar] [CrossRef]
- Ibrahimi, A.; Sfeir, Z.; Magharaie, H.; Amri, E.Z.; Grimaldi, P.; Abumrad, N.A. Expression of the CD36 homolog (FAT) in fibroblast cells: Effects on fatty acid transport. Proc. Natl Acad. Sci. USA 1996, 93, 2646–2651. [Google Scholar] [CrossRef]
- Zhang, J.F.; Choi, S.H.; Li, Q.; Wang, Y.; Sun, B.; Tang, L.; Wang, E.Z.; Hua, H.; Li, X.Z. Overexpression of DGAT2 stimulates lipid droplet formation and triacylglycerol accumulation in bovine satellite cells. Animals 2022, 12, 1847. [Google Scholar] [CrossRef] [PubMed]
- Zhou, F.; Fan, X.; Miao, Y. LPIN1 promotes triglycerides synthesis and is transcriptionally regulated by PPARG in buffalo mammary epithelial cells. Sci. Rep. 2022, 12, 2390. [Google Scholar] [CrossRef]
- Singh, A.B.; Liu, J. Identification of hepatic lysophosphatidylcholine acyltransferase 3 as a novel target gene regulated by peroxisome proliferator-activated receptor δ. J. Biol. Chem. 2017, 292, 884–897. [Google Scholar] [CrossRef] [PubMed]
- Man, W.C.; Miyazaki, M.; Chu, K.; Ntambi, J. Colocalization of SCD1 and DGAT2: I mplying preference for endogenous monounsaturated fatty acids in triglyceride synthesis. J. Lipid Res. 2006, 47, 1928–1939. [Google Scholar] [CrossRef] [PubMed]
- Hotamisligil, G.S.; Bernlohr, D.A. Metabolic functions of FABPs -mechanisms and therapeutic implications. Nat. Rev. Endocrinol. 2015, 11, 592–605. [Google Scholar] [CrossRef]
- Hu, S.; Gao, S.; Zhu, J.; Gan, X.; Chen, X.; He, H.; Liang, L.; Hu, B.; Hu, J.; Liu, H.; et al. Differential actions of diacylglycerol acyltransferase (DGAT) 1 and 2 in regulating lipid metabolism and progesterone secretion of goose granulosa cells. J. Steroid Biochem. Mol. Biol. 2020, 202, 105721. [Google Scholar] [CrossRef]
- McLaren, D.G.; Han, S.; Murphy, B.A.; Wilsie, L.; Stout, S.J.; Zhou, H.; Roddy, T.P.; Gorski, J.N.; Metzger, D.E.; Shin, M.K.; et al. DGAT2 inhibition alters aspects of triglyceride metabolism in rodents but not in non-human primates. Cell Metab. 2018, 27, 1236–1248.e6. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Shu, G.; Zhu, X.; Guo, J.; Cai, H.; Wang, S.; Wang, L.; Gao, P.; Xi, Q.; Zhang, Y.; et al. Effect of diacylglycerol acyltransferase 2 overexpression in 3T3-L1 is associated to an increase in mono-unsaturated fatty acid accumulation. J. Anim. Sci. Biotechnol. 2014, 5, 29. [Google Scholar] [CrossRef] [PubMed]
- Shendure, J.; Ji, H. Next-generation DNA sequencing. Nat. Biotechnol. 2008, 26, 1135–1145. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Gerstein, M.; Snyder, M. RNA-Seq: A revolutionary tool for transcriptomics. Nat. Rev. Genet. 2009, 10, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Lv, Y. Effects of ACSL3 Gene on Preadipocyte Differentiation and Its Genetic Polymorphism in Chinese Steppe Red Cattle. Ph.D. Thesis, Jilin Agricultural University, Jilin, China, 2019. [Google Scholar]
- Guo, L.; Kang, J.S.; Park, Y.H.; Je, B.I.; Lee, Y.J.; Kang, N.J.; Park, S.Y.; Hwang, D.Y.; Choi, Y.W. Spetasin inhibits lipid accumulation in oleic acid-induced Hep G2 cells through activation of the AMPK signaling pathway. Food Funct. 2020, 11, 5664–5673. [Google Scholar] [CrossRef]
- Suburu, J.; Shi, L.; Wu, J.; Wang, S.; Chen, Y.Q. Fatty acid synthase is required for mammary gland development and milk production during lactation. Am. J. Physiol.-Endoc. Metab. 2014, 306, 1132–1143. [Google Scholar] [CrossRef]
- Li, N.; Zhao, F.; Wei, C.; Liang, M.; Zhang, N.; Wang, C.; Li, Q.Z.; Gao, X.J. Function of SREBP1 in the milk fat synthesis of dairy cow mammary epithelial cells. Int. J. Mol. Sci. 2014, 15, 16998–17013. [Google Scholar] [CrossRef]
- Li, J.; Luo, J.; Xu, H.; Wang, M.; Zhu, J.; Shi, H.; Haile, A.B.; Wang, H.; Sun, Y. Fatty acid synthase promoter: Characterization, and transcriptional regulation by sterol regulatory element binding protein-1 in goat mammary epithelial cells. Gene 2015, 561, 157–164. [Google Scholar] [CrossRef]
- Ji, F.; Fu, S.J.; Shen, S.L.; Zhang, L.J.; Cao, Q.H.; Li, S.Q.; Peng, B.G.; Liang, L.J.; Hua, Y.P. The prognostic value of combined TGF-β1 and ELF in hepatocellular carcinoma. BMC Cancer 2015, 15, 116. [Google Scholar] [CrossRef]














| Scheme 5 | Sense Strand (5′-3′) | Anti-Sense Strand (3′-5′) | Target Sequence |
|---|---|---|---|
| DGAT2-sh-108 | AATTCGGTAGAGAAGCAGCTCCAAGTTTCAAGAGAACTTGGAGCTGCTTCTCTACCTTTTTTG | GATCCAAAAAAGGTAGAGAAGCAGCTCCAAGTTCTCTTGAAACTTGGAGCTGCTTCTCTACCG | GGTAGAGAAGCAGCTCCAAGT |
| DGAT2-sh-320 | AATTCGCTACTTTCGAGACTACTTTCTTCAAGAGAGAAAGTAGTCTCGAAAGTAGCTTTTTTG | GATCCAAAAAAGCTACTTTCGAGACTACTTTCTCTCTTGAAGAAAGTAGTCTCGAAAGTAGCG | GCTACTTTCGAGACTACTTTC |
| DGAT2-sh-687 | AATTCGCGCAATCGCAAGGGCTTTGTTTCAAGAGAACAAAGCCCTTGCGATTGCGCTTTTTTG | GATCCAAAAAAGCGCAATCGCAAGGGCTTTGTTCTCTTGAAACAAAGCCCTTGCGATTGCGCG | GCGCAATCGCAAGGGCTTTGT |
| sh-NC | AATTCGTTCTCCGAACGTGT CACGTTTCAAGAGAACGTGAC ACGTTCGGAGAACTTTTTTG | GATCCAAAAAAGTTCTCCGA ACGTGTCACGTTCTCTTGAA ACGTGACACGTTCGGAGAACG | TTCTCCGAACGTGTCACGT |
| Gene | Sense Strand (5′-3′) | Length (bp) | Gene ID |
|---|---|---|---|
| GAPDH | F:ACTCTGGCAAAGTGGATGTTGTC R:GCATCACCCCACTTGATGTTG | 143 | NM_001034034 |
| DGAT1 | F:CTACACCATCCTCTTCCTCAAG R:AGTAGTAGAGATCGCGGTAGGTC | 176 | NM_174693.2 |
| DGAT2 | F:GACCCTCATAGCCTCCTACTCC R:GACCCATTGTAGCACCGAGATGAC | 145 | NM_205793.2 |
| AGPAT4 | F:TGTTCTCGTCTTCTTTGTGGCTTCC R:TCGCTATGTTTCTGCTTGCTGTCC | 111 | NM_001015537.1 |
| MGAT1 | F:AGCCGTGGTGGTAGAGGATGATC R:TGCTCCTTGCCATTGTCGTTCC | 132 | NM_001015653 |
| LPIN1 | F:AGTCCTCGCCACACAAGATG R:AGATGCCCTGACCAGTGTTG | 137 | NM_001206156 |
| GPAT4 | F:ATGCGGTCCAGTTTGCCAATAGG R:GCTTCTGCTGCTCCTCCTTGAAC | 129 | NM_001083669.1 |
| PPARγ | F-ATCTGCTGCAAGCCTTGGA R-TGGAGCAGCTTGGCAAAGA | 138 | NM_181024 |
| C/EBPα | F-CCAGAAGAAGGTGGAGCAACTG R-TCGGGCAGCGTCTTGAAC | 69 | NM_176788 |
| C/EBPβ | F-CAACCTGGAGACGCAGCACAAG R-CGGAGGAGGCGAGCAGAGG | 143 | NM_176788 |
| SREBF1 | F-CTGCTGACCGACATAGAAGACAT R-GTAGGGCGGGTCAAACAGG | 81 | NM_001113302 |
| FABP4 | F-AAACTTAGATGAAGGTGCTCTGG R-CATAAACTCTGGTGGCAGTGA | 134 | NM_174314.2 |
| FASN | F-CGCTTGCTGCTGGAGGTCAC R-GGTCTCAGGGTCTCGGCTCAG | 141 | NM_001012669 |
| SCD | F-TGCCCACCACAAGTTTTCAG R-GCCAACCCACGTGAGAGAAG | 80 | NM_173959 |
| ACACA | F-GCCAAACCTCTGGAGCTGAA R-CGAGCTTCACCAGGTTGCTA | 97 | NM_174224 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guo, P.-P.; Jin, X.; Zhang, J.-F.; Li, Q.; Yan, C.-G.; Li, X.-Z. Overexpression of DGAT2 Regulates the Differentiation of Bovine Preadipocytes. Animals 2023, 13, 1195. https://doi.org/10.3390/ani13071195
Guo P-P, Jin X, Zhang J-F, Li Q, Yan C-G, Li X-Z. Overexpression of DGAT2 Regulates the Differentiation of Bovine Preadipocytes. Animals. 2023; 13(7):1195. https://doi.org/10.3390/ani13071195
Chicago/Turabian StyleGuo, Pan-Pan, Xin Jin, Jun-Fang Zhang, Qiang Li, Chang-Guo Yan, and Xiang-Zi Li. 2023. "Overexpression of DGAT2 Regulates the Differentiation of Bovine Preadipocytes" Animals 13, no. 7: 1195. https://doi.org/10.3390/ani13071195
APA StyleGuo, P.-P., Jin, X., Zhang, J.-F., Li, Q., Yan, C.-G., & Li, X.-Z. (2023). Overexpression of DGAT2 Regulates the Differentiation of Bovine Preadipocytes. Animals, 13(7), 1195. https://doi.org/10.3390/ani13071195






