Non-Invasive Methods for Assessing the Welfare of Farmed White-Leg Shrimp (Penaeus vannamei)
Abstract
Simple Summary
Abstract
1. Introduction
1.1. Animal Welfare and Progress in Shrimp Farming
1.2. Shrimp Sentience
1.3. Shrimp Welfare and Legislation
2. Materials and Methods
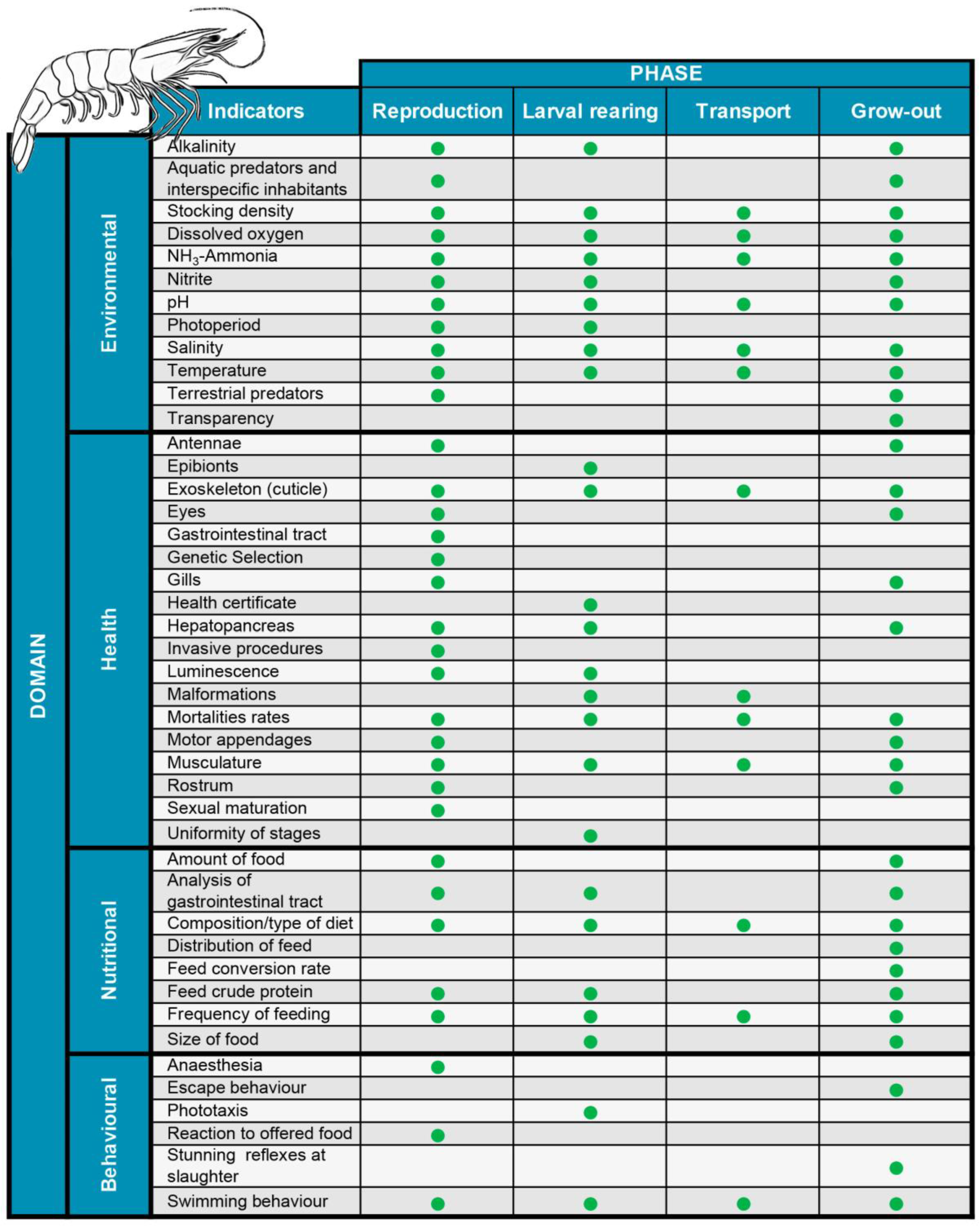
2.1. The Welfare Domains and Indicators
2.2. Identification and Selection of Documents for Bibliographic Review and Reference Levels
2.3. Field Evaluation of the Preselected Indicators
3. Results
3.1. Reproduction Stage
3.2. Larvae Rearing Phase
3.3. Postlarvae Transport
3.4. Grow-Out Stage
4. Discussion
4.1. Indicators for Shrimp Welfare
4.2. The Establishment of Farmed Shrimp Welfare Protocols
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Stonich, S.C. The Environmental Quality and Social Justice Implications of Shrimp Mariculture Development in Honduras. Hum. Ecol. 1995, 23, 143–168. [Google Scholar] [CrossRef]
- Swapan, M.S.H. Michael Gavin A Desert in the Delta: Participatory Assessment of Changing Livelihoods Induced by Commercial Shrimp Farming in Southwest Bangladesh. Ocean Coast. Manag. 2011, 54, 45–54. [Google Scholar] [CrossRef]
- Queiroz, L.; Rossi, S.; Meireles, J.; Coelho, C. Shrimp Aquaculture in the Federal State of Ceará, 1970–2012: Trends after Mangrove Forest Privatization in Brazil. Ocean Coast. Manag. 2013, 73, 54–62. [Google Scholar] [CrossRef]
- Jayanthi, M.; Thirumurthy, S.; Muralidhar, M.; Ravichandran, P. Impact of Shrimp Aquaculture Development on Important Ecosystems in India. Glob. Environ. Chang. 2018, 52, 10–21. [Google Scholar] [CrossRef]
- Treviño, M.; Murillo-Sandoval, P.J. Uneven Consequences: Gendered Impacts of Shrimp Aquaculture Development on Mangrove Dependent Communities. Ocean Coast. Manag. 2021, 210, 105688. [Google Scholar] [CrossRef]
- Nguyen, T.A.T.; Nguyen, K.A.T.; Jolly, C. Is Super-Intensification the Solution to Shrimp Production and Export Sustainability? Sustainability 2019, 11, 5277. [Google Scholar] [CrossRef]
- Joffre, O.M.; Klerkx, L.; Khoa, T.N.D. Aquaculture Innovation System Analysis of Transition to Sustainable Intensification in Shrimp Farming. Agron. Sustain. Dev. 2018, 38, 1–11. [Google Scholar] [CrossRef]
- Boyd, C.E.; Davis, R.P.; McNevin, A.A. Perspectives on the Mangrove Conundrum, Land Use, and Benefits of Yield Intensification in Farmed Shrimp Production: A Review. J. World Aquac. Soc. 2022, 53, 8–46. [Google Scholar] [CrossRef]
- Cozer, N.; Horodesky, A.; Rossi, V.G.; Pont, G.D.; Ostrensky, A. Challenges and Potentialities of the Integrated Production Regime Implementation in the Brazilian Marine Shrimp Farming: A Systematic Review. Aquac. Int. 2019, 27, 539–553. [Google Scholar] [CrossRef]
- Cozer, N.; Pont, G.D.; Horodesky, A.; Ostrensky, A. Infrastructure, Management and Energy Efficiency in a Hypothetical Semi-Intensive Shrimp Model Farm in Brazil: A Systematic Review and Meta-Analysis. Rev. Aquac. 2020, 12, 1072–1089. [Google Scholar] [CrossRef]
- Bush, S.R. Understanding the Potential of Eco-Certification in Salmon and Shrimp Aquaculture Value Chains. Aquaculture 2018, 493, 376–383. [Google Scholar] [CrossRef]
- Tlusty, M.F.; Thompson, M.; Tausig, H. Statistical Tools to Assess the Breadth and Depth of Shrimp Aquaculture Certification Schemes. Fish. Res. 2016, 182, 172–176. [Google Scholar] [CrossRef]
- Samerwong, P.; Bush, S.R.; Oosterveer, P. Implications of Multiple National Certification Standards for Thai Shrimp Aquaculture. Aquaculture 2018, 493, 319–327. [Google Scholar] [CrossRef]
- Vijayan, K.K. Domestication and Genetic Improvement of Indian White Shrimp, Penaeus Indicus: A Complimentary Native Option to Exotic Pacific White Shrimp, Penaeus Vannamei. J. Coast. Res. 2019, 86, 270–276. [Google Scholar] [CrossRef]
- Alday-Sanz, V.; Brock, J.; Flegel, T.W.; McIntosh, R.; Bondad-Reantaso, M.G.; Salazar, M.; Subasinghe, R. Facts, Truths and Myths about SPF Shrimp in Aquaculture. Rev. Aquac. 2020, 12, 76–84. [Google Scholar] [CrossRef]
- Sookying, D.; Davis, D.A.; Soller Dias da Silva, F. A Review of the Development and Application of Soybean-Based Diets for Pacific White Shrimp Litopenaeus Vannamei. Aquac. Nutr. 2013, 19, 441–448. [Google Scholar] [CrossRef]
- Butt, U.D.; Lin, N.; Akhter, N.; Siddiqui, T.; Li, S.; Wu, B. Overview of the Latest Developments in the Role of Probiotics, Prebiotics and Symbiotics in Shrimp Aquaculture. Fish Shellfish Immunol. 2021, 114, 263–281. [Google Scholar] [CrossRef] [PubMed]
- Hoseinifar, S.H.; Sun, Y.Z.; Wang, A.; Zhou, Z. Probiotics as Means of Diseases Control in Aquaculture, a Review of Current Knowledge and Future Perspectives. Front. Microbiol. 2018, 9, 2429. [Google Scholar] [CrossRef] [PubMed]
- Flegel, T.W. A Future Vision for Disease Control in Shrimp Aquaculture. J. World Aquac. Soc. 2019, 50, 249–266. [Google Scholar] [CrossRef]
- FAO. The State of the World Fisheries and Aquaculture 2020: Towards Blue Transformation; FAO (Food and Agriculture Organization of the United Nations): Rome, Italy, 2020. [Google Scholar] [CrossRef]
- Albalat, A.; Zacarias, S.; Coates, C.J.; Neil, D.M.; Planellas, S.R. Welfare in Farmed Decapod Crustaceans, With Particular Reference to Penaeus Vannamei. Front. Mar. Sci. 2022, 9, 677. [Google Scholar] [CrossRef]
- Waldhorn, D.R.; Autric, E. Shrimp Production: Understanding the Scope of the Problem; Center for Open Science: San Francisco, CA, USA, 2022; pp. 1–32. [Google Scholar] [CrossRef]
- Broom, D.M. A History of Animal Welfare Science. Acta Biotheor. 2011, 59, 121–137. [Google Scholar] [CrossRef] [PubMed]
- Ohl, F.; van der Staay, F.J. Animal Welfare: At the Interface between Science and Society. Vet. J. 2012, 192, 13–19. [Google Scholar] [CrossRef]
- Fernandes, J.N.; Hemsworth, P.H.; Coleman, G.J.; Tilbrook, A.J. Costs and Benefits of Improving Farm Animal Welfare. Agriculture 2021, 11, 104. [Google Scholar] [CrossRef]
- Kaygisiz, F.; Bolat, B.A.; Bulut, D. Determining Factors Affecting Consumer’s Decision to Purchase Organic Chicken Meat. Braz. J. Poult. Sci. 2019, 21. [Google Scholar] [CrossRef]
- Beaver, A.; Proudfoot, K.L.; von Keyserlingk, M.A.G. Symposium Review: Considerations for the Future of Dairy Cattle Housing: An Animal Welfare Perspective. J. Dairy Sci. 2020, 103, 5746–5758. [Google Scholar] [CrossRef] [PubMed]
- Vaärikkala, S.; Artukka, S.M.; Hänninen, L.; Nevas, M. Finnish Cattle and Pig Farmers’ Perceptions of Animal Welfare Inspections. Anim. Welf. 2018, 27, 369–377. [Google Scholar] [CrossRef]
- Yunes, M.C.; Osório-Santos, Z.; von Keyserlingk, M.A.G.; Hötzel, M.J. Gene Editing for Improved Animal Welfare and Production Traits in Cattle: Will This Technology Be Embraced or Rejected by the Public? Sustainability 2021, 13, 4966. [Google Scholar] [CrossRef]
- Ankamah-Yeboah, I.; Jacobsen, J.B.; Olsen, S.B.; Nielsen, M.; Nielsen, R. The Impact of Animal Welfare and Environmental Information on the Choice of Organic Fish: An Empirical Investigation of German Trout Consumers. Mar. Resour. Econ. 2019, 34, 248–266. [Google Scholar] [CrossRef]
- Maesano, G.; di Vita, G.; Chinnici, G.; Pappalardo, G.; D’amico, M. The Role of Credence Attributes in Consumer Choices of Sustainable Fish Products: A Review. Sustainability 2020, 12, 10008. [Google Scholar] [CrossRef]
- Pedrazzani, A.S.; Tavares, C.P.d.S.; Quintiliano, M.; Cozer, N.; Ostrensky, A. New Indices for the Diagnosis of Fish Welfare and Their Application to the Grass Carp (Ctenopharyngodon Idella) Reared in Earthen Ponds. Aquac. Res. 2022, 53, 5825–5845. [Google Scholar] [CrossRef]
- David, P.N.F.; Manipol, N.E.P.; Madamba, J.A.B.; Mariano, R.A. Good Aquaculture Practices Adoption and Certification of Shrimp Aquaculture Farms in Bulacan, Philippines: Status, Issues and Prospects. J Glob Bus Trade 2020, 15, 11–26. [Google Scholar] [CrossRef]
- Xuan, B.B.; Sandorf, E.D.; Ngoc, Q.T.K. Stakeholder Perceptions towards Sustainable Shrimp Aquaculture in Vietnam. J. Environ. Manag. 2021, 290, 112585. [Google Scholar] [CrossRef]
- Booncharoen, C.; Anal, A.K. Attitudes, Perceptions, and On-Farm Self-Reported Practices of Shrimp Farmers’ towards Adoption of Good Aquaculture Practices (GAP) in Thailand. Sustainability 2021, 13, 5194. [Google Scholar] [CrossRef]
- Cooper, J.J.; Tinarwo, A.; Ventura, B.A. Decapods as Food, Companions and Research Animals: Legal Impact of Ascribing Sentience. Anim. Sentience 2022, 7, 27. [Google Scholar] [CrossRef]
- Souza Valente, C.D. Decapod Sentience: Broadening the Framework. Anim. Sentience 2022, 7, 8. [Google Scholar] [CrossRef]
- Gorman, R. What Might Decapod Sentience Mean for Policy, Practice, and Public? Anim. Sentience 2022, 32, 1–4. [Google Scholar] [CrossRef]
- Passantino, A.; Elwood, R.W.; Coluccio, P. Why Protect Decapod Crustaceans Used as Models in Biomedical Research and in Ecotoxicology? Ethical and Legislative Considerations. Animals 2021, 11, 73. [Google Scholar] [CrossRef]
- Mather, J.A. Philosophical Background of Attitudes toward and Treatment of Invertebrates. ILAR J. 2011, 52, 205–212. [Google Scholar] [CrossRef]
- Mikhalevich, I.; Powell, R. Minds without Spines: Evolutionarily Inclusive Animal Ethics. Anim. Sentience 2020, 29. [Google Scholar] [CrossRef]
- Beausoleil, N.J.; Mellor, D.J.; Beausoleil, N.J. Extending the “Five Domains” Model for Animal Welfare Assessment to Incorporate Positive Welfare States The Veterinarian’s Role in End-of-Life Management of Older and Chronically Ill Cats in New Zealand View Project Breathless Birds: Does Air Hunger Impact the Welfare of Poultry at Slaughter? View Project Extending the “Five Domains” Model for Animal Welfare Assessment to Incorporate Positive Welfare States. Anim. Welf. 2015, 24, 241–253. [Google Scholar] [CrossRef]
- Mellor, D.J. Welfare-Aligned Sentience: Enhanced Capacities to Experience, Interact, Anticipate, Choose and Survive. Animals 2019, 9, 440. [Google Scholar] [CrossRef]
- Crump, A.; Browning, H.; Schnell, A.; Burn, C.; Birch, J. Sentience in Decapod Crustaceans: A General Framework and Review of the Evidence. Anim. Sentience 2022, 7, 1. [Google Scholar] [CrossRef]
- Birch, J.; Burn, C.; Schnell, A.; Browning, H.; Crump, A. Review of the Evidence of Sentience in Cephalopod Molluscs and Decapod Crustaceans I; LSE Consulting. LSE Enterprise Ltd.; The London School of Economics and Political Science: London, UK, 2021. [Google Scholar]
- Feinberg, T.E.; Mallatt, J.M. The Ancient Origins of Consciousness: How the Brain Created Experience; MIT Press: Cambridge, MA, USA, 2016. [Google Scholar]
- Birch, J.; Broom, D.M.; Browning, H.; Crump, A.; Ginsburg, S.; Halina, M.; Harrison, D.; Jablonka, E.; Lee, A.Y.; Kammerer, F.; et al. How Should We Study Animal Consciousness Scientifically? J. Conscious Stud. 2022, 29, 8–28. [Google Scholar] [CrossRef]
- Conte, F.; Voslarova, E.; Vecerek, V.; Elwood, R.W.; Coluccio, P.; Pugliese, M.; Passantino, A. Humane Slaughter of Edible Decapod Crustaceans. Animals 2021, 11, 1089. [Google Scholar] [CrossRef]
- Langworthy, K.; Helluy, S.; Benton, J.; Beltz, B. Amines and Peptides in the Brain of the American Lobster: Immunocytochemical Localization Patterns and Implications for Brain Function. Cell Tissue Res. 1997, 288, 191–206. [Google Scholar] [CrossRef]
- Briffa, M. Sentience in Decapods: An Open Question. Anim. Sentience 2022, 7, 1–6. [Google Scholar] [CrossRef]
- European Union Council Regulation (EC) No 1099/2009 of 24 September 2009 on the Protection of Animals at the Time of Killing. Off. J. Eur. Union 2009, 7, 1–30. Available online: http://data.europa.eu/eli/reg/2009/1099/oj (accessed on 29 January 2023).
- EFSA Scientific Panel on Animal Health and Welfare. Aspects of the Biology and Welfare of Animals Used for Experimental and Other Scientific Purposes. EFSA J. 2005, 292, 1–46. [Google Scholar]
- Smith, J.A.; Andrews, P.L.R.; Hawkins, P.; Louhimies, S.; Ponte, G.; Dickel, L. Cephalopod Research and EU Directive 2010/63/EU: Requirements, Impacts and Ethical Review. J. Exp. Mar. Biol. Ecol. 2013, 447, 31–45. [Google Scholar] [CrossRef]
- French Ministry of Agriculture, Agrifood and Forestry. Service Note DGAL/SDSSA/N2012-8219, Dated 20 November 2012. In Authorisation and Health Inspection of Storage Tanks for Crustaceans and Seawater and Freshwater Fish; French Ministry of Agriculture, Agrifood and Forestry: Paris, France, 2012. [Google Scholar]
- Conte, F.; Voslarova, E.; Passantino, A. Destinozí Korýši: Otázka Welfare v Souvislosti s Jejich Využitím Jako Potraviny [Decapod Crustaceans: Some Issues Related to Welfare and Their Use as Food]. Maso 2018, 7, 51–54. [Google Scholar]
- Weineck, K.; Ray, A.J.; Fleckenstein, L.J.; Medley, M.; Dzubuk, N.; Piana, E.; Cooper, R.L. Physiological Changes as a Measure of Crustacean Welfare under Different Standardized Stunning Techniques: Cooling and Electroshock. Animals 2018, 8, 158. [Google Scholar] [CrossRef]
- Crump, A.; Birch, J. Animal Consciousness: The Interplay of Neural and Behavioural Evidence. J. Conscious Stud. 2022, 29, 104–128. [Google Scholar] [CrossRef]
- Johnston, C.; Jungalwalla, P. Aquatic Animal Welfare Guidelines: Guidelines on Welfare of Fish and Crustaceans in Aquaculture and/or in Live Holding Systems for Human Consumption; National Aquaculture Council of Australia: Deakin, ACT, Australia, 2005. [Google Scholar]
- PNS/BAFS 197:2017; BAFS Philippine National Standards Code of Good Aquaculture Practices for Shrimp and Crab. Bureau of Agriculture and Fisheries Standards: Quezon City, Philippines, 2017.
- Nguyen, T.B.T. Good Aquaculture Practices (VietGAP) and Sustainable Aquaculture Development in Viet Nam; Romana-Eguia, M.R.R., ParadoEstepa, F., Salayo, N., Lebata-Ramos, M.J.H., Eds.; Resource Enhancement and Sustainable Aquaculture Practices in Southeast Asia: Challenges in Responsible Production of Aquatic Species: Proceedings of the International Workshop on Resource Enhancement and Sustainable Aquaculture Practices in Southeast Asia 2014 (RESA); Aquaculture Dept., Southeast Asian Fisheries Development Center: Tigbauan, Philippines, 2015; pp. 85–92. Available online: http://hdl.handle.net/10862/2766 (accessed on 29 January 2023).
- Paschke, M. Applying U.S. Animal Law Extraterritoriality to Improve Animal Welfare Standards Abroad and Avoid a Race to the Bottom. Denver J. Int. Law Policy 2021, 49, 13. [Google Scholar]
- Vesilind, P.A. Continental Drift: Agricultural Trade and the Widening Gap Between European Union and United States Animal Welfare Laws. Int. Trade eJ 2010, 12, 223. [Google Scholar] [CrossRef]
- Alfnes, F.; Chen, X.; Rickertsen, K. Labeling Farmed Seafood: A Review. Aquac. Econ. Manag. 2018, 22, 1–26. [Google Scholar] [CrossRef]
- Alfnes, F. Selling Only Sustainable Seafood: Attitudes toward Public Regulation and Retailer Policies. Mar. Policy 2017, 78, 74–79. [Google Scholar] [CrossRef]
- Coates, C.J.; Söderhäll, K. The Stress–Immunity Axis in Shellfish. J. Invertebr. Pathol. 2021, 186, 107492. [Google Scholar] [CrossRef]
- Scherer, L.; Tomasik, B.; Rueda, O.; Pfister, S. Framework for Integrating Animal Welfare into Life Cycle Sustainability Assessment. Int. J. Life Cycle Assess. 2018, 23, 1476–1490. [Google Scholar] [CrossRef]
- Franks, B.; Ewell, C.; Jacquet, J. Animal Welfare Risks of Global Aquaculture. Sci. Adv. 2021, 7, eabg0677. [Google Scholar] [CrossRef]
- FAWC Prevention of Cruelty to Animals Act 1979. Available online: https://legislation.nsw.gov.au/view/html/inforce/current/act-1979-200 (accessed on 29 December 2022).
- Stien, L.H.; Bracke, M.B.M.; Folkedal, O.; Nilsson, J.; Oppedal, F.; Torgersen, T.; Kittilsen, S.; Midtlyng, P.J.; Vindas, M.A.; Øverli, Ø.; et al. Salmon Welfare Index Model (SWIM 1.0): A Semantic Model for Overall Welfare Assessment of Caged Atlantic Salmon: Review of the Selected Welfare Indicators and Model Presentation. Rev. Aquac. 2013, 5, 33–57. [Google Scholar] [CrossRef]
- Pedrazzani, A.S.; Quintiliano, M.H.; Bolfe, F.; Sans, E.C.d.O.; Molento, C.F.M. Tilapia On-Farm Welfare Assessment Protocol for Semi-Intensive Production Systems. Front. Vet. Sci. 2020, 7, 991. [Google Scholar] [CrossRef]
- Ceballos-Vázquez, B.P.; Palacios, E.; Aguilar-Villavicencio, J.; Racotta, I.S. Gonadal Development in Male and Female Domesticated Whiteleg Shrimp, Litopenaeus Vannamei, in Relation to Age and Weight. Aquaculture 2010, 308, 116–123. [Google Scholar] [CrossRef]
- Ceballos-Vázquez, B.P.; Rosas, C.; Racotta, I.S. Sperm Quality in Relation to Age and Weight of White Shrimp Litopenaeus Vannamei. Aquaculture 2003, 228, 141–151. [Google Scholar] [CrossRef]
- González, R.A.; Díaz, F.; Licea, A.; Denisse Re, A.; Noemí Sánchez, L.; García-Esquivel, Z. Thermal Preference, Tolerance and Oxygen Consumption of Adult White Shrimp Litopenaeus Vannamei (Boone) Exposed to Different Acclimation Temperatures. J. Therm. Biol. 2010, 35, 218–224. [Google Scholar] [CrossRef]
- Perez-Velazquez, M.; Bray, W.A.; Lawrence, A.L.; Gatlin, D.M.; Gonzalez-Felix, M.L. Effect of Temperature on Sperm Quality of Captive Litopenaeus Vannamei Broodstock. Aquaculture 2001, 198, 209–218. [Google Scholar] [CrossRef]
- Barbieri, C.; Ostrensky, A. Camarões Marinhos–Reprodução, Maturação e Larvicultura; Aprenda Fácil: Viçosa, Brazil, 2001; p. 370. [Google Scholar]
- Balasubramanian, C.P.; Anand, S.; Kannappan, S.; Biju, I.F. Training Manual on Recent Advances in Farming of Pacific White Shrimp, Penaeus Vannamei. Train. Man. Ser. 2018, 14, 126. [Google Scholar]
- Simpson, J.J.; Zirino, A. Biological Control of PH in the Peruvian Coastal Upwelling Area. Deep Sea Res. Part A Oceanogr. Res. Pap. 1980, 27, 733–743. [Google Scholar] [CrossRef]
- Zuta, S.; Guillén, O. Oceanografía de Las Aguas Costeras Del Perú. Bol. Inst. Mar. Perú. 1970, 2, 157–324. [Google Scholar]
- FAO Health Management and Biosecurity Maintenance in White Shrimp (Penaeus Vannamei) Hatcheries in Latin America. Fish TechPap 2003, 450, 62.
- Treece, G.D. Shrimp Maturation and Spawning; UJNR Technical Report No. 28; Texas A&M University, Sea Grant College Program: Bryan, TX, USA, 2000; pp. 121–134. [Google Scholar]
- Ostrensky, A.; Stevanato, D.J.; Pont, G.D.; Castilho-Westphal, G.G.; Girotto, M.V.F.; Cozer, N.; García-Madrigal, R.F.d.A.; Silva, U. A Produção Integrada Na Carcinicultura Brasileira (Volume 1); Ostrensky, A., Cozer, N., Eds.; Instituto GIA: Curitiba, PR, Brasil, 2017. [Google Scholar]
- Peixoto, S.; Wasielesky, W.; Louzada, L. Comparative Analysis of Pink Shrimp, Farfantepenaeus Paulensis, and Pacific White Shrimp, Litopenaeus Vannamei, Culture in Extreme Southern Brazil. J. Appl. Aquac. 2003, 14, 101–111. [Google Scholar] [CrossRef]
- Venkateswarlu, V.; Seshaiah, P.; Arun, P.; Behra, P. A Study on Water Quality Parameters in Shrimp L. Vannamei Semi-Intensive Grow out Culture Farms in Coastal Districts of Andhra Pradesh, India. Int. J. Fish, Aquat. Stud. 2019, 7, 394–397. [Google Scholar]
- Fast, A.W.; Lester, L.J. Marine Shrimp Culture: Principles and Practices, 1st ed.; Elsevier: Amsterdam, The Netherlands, 1992; p. 862. ISBN 978-0-444-88606-4. [Google Scholar]
- Nunes, A.J.P. O cultivo de camarões marinhos no Nordeste do Brasil. Rev. Panor. Aqüic. 2001, 11, 65. [Google Scholar]
- Nogueira, D.B.; Souza, L.S.B.; Santos, W.R.; Júnior, G.d.N.A.; Souza, C.A.A.; Jardim, A.M.R.F.; Silva, T.G.F. Understanding the Influence of Photoperiod on the Cultivation of Litopenaeus Vannamei: Revisiting Studies Carried out for the 2005–2020 Cropping Period. Res. Soc. Dev. 2021, 10. [Google Scholar] [CrossRef]
- ABCC. MCR Apostila Técnica de Boas Práticas de Manejo Para a Capacitação de Pequenos Produtores, Natal-RN; Associação Brasileira de Criadores de Camarão: Candelária, Natal, RN, Brasil, 2010. [Google Scholar]
- Fonseca, C.S. Manual de Boas Práticas de Manejo e de Biossegurança Para a Carcinicultura Brasileira; Associação Brasileira de Criadores de Camarão: Candelária, RN, Brasil, 2021. [Google Scholar]
- Nunes AJ, P.; Gesteira TC, V.; Oliveira, G.G.; Lima, R.C.; Miranda PT, C.; Madrid, R.M. Princípios Para Boas Práticas de Manejo Na Engorda de Camarão Marinho No Estado Do Ceará; Universidade Federal do Ceará–LABOMAR: Fortaleza, CE, Brasil, 2005. [Google Scholar]
- Rojas, A.A.; Haws, M.C.; y Cabanillas, J.A. Buenas Prácticas de Manejo Para el Cultivo de Camarón, 1st ed.; The David and Lucile Packard Foundation; United States Agency for International Development: Washington, DC, USA, 2005; pp. 4–51. [Google Scholar]
- Zhang, P.; Zhang, X.; Li, J.; Huang, G. The Effects of Body Weight, Temperature, Salinity, PH, Light Intensity and Feeding Condition on Lethal DO Levels of Whiteleg Shrimp, Litopenaeus Vannamei (Boone, 1931). Aquaculture 2006, 256, 579–587. [Google Scholar] [CrossRef]
- Han, S.; Wang, B.; Wang, M.; Liu, Q.; Zhao, W.; Wang, L. Effects of Ammonia and Nitrite Accumulation on the Survival and Growth Performance of White Shrimp Litopenaeus Vannamei. Invert. Surv. J. 2017, 14, 221–232. [Google Scholar] [CrossRef]
- Chen, J. Acute Toxicity of Ammonia on Litopenaeus Vannamei Boone Juveniles at Different Salinity Levels. J. Exp. Mar. Biol. Ecol. 2001, 259, 109–119. [Google Scholar]
- Gross, A.; Abutbul, S.; Zilberg, D. Acute and Chronic Effects of Nitrite on White Shrimp, Litopenaeus Vannamei, Cultured in Low-Salinity Brackish Water. J. World Aquac. Soc. 2004, 35, 315–321. [Google Scholar] [CrossRef]
- Lin, Y.C.; Chen, J.C. Acute Toxicity of Nitrite on Litopenaeus Vannamei (Boone) Juveniles at Different Salinity Levels. Aquaculture 2003, 224, 193–201. [Google Scholar] [CrossRef]
- Tan, J.; Luan, S.; Cao, B.; Luo, K.; Meng, X.; Kong, J. Comparison of Growth and Reproduction Performance of Broodstock Pacific White Shrimp Litopenaeus Vannamei Reared in Oceanic and Brackish Water. Aquac. Res. 2019, 50, 1893–1902. [Google Scholar] [CrossRef]
- Flores, R.; Espino, M.; Luque, G.; Quispe, J. Patrones de Variabilidad Ambiental En El Mar Peruano. Rev. Peru Biol. 2013, 20, 021–028. [Google Scholar] [CrossRef]
- Aquacop. Penaeid Reared Brood Stock: Closing the Cycle. Meeting of the World Mariculture Society. J. World Maricult. Soc. 1979, 10, 445–452. [Google Scholar] [CrossRef]
- Alday-Sanz, V. The Shrimp Book, 1st ed.; Nottingham University Press: Nottingham, UK, 2012; ISBN 9781904761594. [Google Scholar]
- Chen, F.; Reid, B.; Arnold, C.R. Maturing, Spawning and Egg Collecting of the White Shrimp Penaeus Vannamei Boone in a Recirculating System. J. World Aquac. Soc. 1991, 22, 167–172. [Google Scholar] [CrossRef]
- Basso, E.; Drever, M.C.; Fonseca, J.; Navedo, J.G. Semi-intensive Shrimp Farms as Experimental Arenas for the Study of Predation Risk from Falcons to Shorebirds. Ecol. Evol. 2021, 11, 13379. [Google Scholar] [CrossRef]
- Kungvankij, P.; Chua, T.E.; Pudadera, B.J., Jr.; Corre, K.G. Shrimp Culture: Pond Design, Operation and Management. 1988. Available online: https://www.fao.org/3/ac210e/AC210E00.htm#TOC (accessed on 29 January 2023).
- Terazaki, M.; Tharnbuppa, P.; Nakayama, Y. Eradication of Predatory Fishes in Shrimp Farms by Utilization of Thai Tea Seed. Aquaculture 1980, 19, 235–242. [Google Scholar] [CrossRef]
- Palanikumar, P.; Velmurugan, S.; Citarasu, T. Factors Influencing in Success of Penaeus Vannamei Culture. Aquac. Asia Mag. 2011, 15, 10–15. [Google Scholar]
- Arulmoorthy, M.P.; Anandajothi, E.; Vasudevan, S.; Suresh, E. Major Viral Diseases in Culturable Penaeid Shrimps: A Review. Aquac. Int. 2020, 28, 1939–1967. [Google Scholar] [CrossRef]
- Furtado, P.S.; Campos, B.R.; Serra, F.P.; Klosterhoff, M.; Romano, L.A.; Wasielesky, W. Effects of Nitrate Toxicity in the Pacific White Shrimp, Litopenaeus Vannamei, Reared with Biofloc Technology (BFT). Aquac. Int. 2015, 23, 315–327. [Google Scholar] [CrossRef]
- Lee, D.; Yu, Y.B.; Choi, J.H.; Jo, A.H.; Hong, S.M.; Kang, J.C.; Kim, J.H. Viral Shrimp Diseases Listed by the OIE: A Review. Viruses 2022, 14, 585. [Google Scholar] [CrossRef]
- Johnson, S.K. Handbook of Shrimp Diseases; Texas A&M Univer sity Sea Grant College Program, the Texas A&M Department of Wildlife and Fisheries Sciences and the Texas Agricultural Extension Service: Bryan, TX, USA, 1995. [Google Scholar]
- Gunalan, B.; Soundarapandian, P.; Anand, T.; Kotiya, A.S.; Simon, N.T. Disease Occurrence in Litopenaeus Vannamei Shrimp Culture Systems in Different Geographical Regions of India. Int. J. Aquac. 2014. [Google Scholar] [CrossRef]
- Lightner, D.V. A Handbook of Shrimp Pathology and Diagnostic Procedures for Disease of Cultured Penaeid Shrimp; World Aqua.: Baton Rouge, LA, USA, 1996.111. Smith, P.T. Diseases of the Eye of Farmed Shrimp Penaeus Monodon. Dis. Aquat. Organ. 2000, 43, 159–173. [Google Scholar] [CrossRef]
- Bondad-Reantaso, M.G.; McGladdery, S.E.; East, I.; Subasinghe, R.P. Asia Diagnostic Guide to Aquatic Animal Diseases; FAO Fisheries Technical Paper 402, Supplement 2; FAO: Rome, Italy, 2001; 240p. [Google Scholar]
- Dewangan, N.K.; Gopalakrishnan, A.; Kannan, D.; Shettu, N.; Singh, R.R. Black Gill Disease of Pacific White Leg Shrimp (Litopenaeus Vannamei) by Aspergillus Flavus. J. Coast. Life Med. 2015, 3, 761–765. [Google Scholar] [CrossRef]
- Sivakamavalli, J.; Park, K.; Kwak, I.; Baskaralingam, V. Bacterial Disease Control Methods in Shrimp ( Penaeus, 1798) Farming Sector in Asian Countries. In Arthropods-Are They Beneficial for Mankind? IntechOpen: London, UK, 2021; ISBN 978-1-78984-166-4. [Google Scholar]
- Dey, B.K.; Dugassa, G.H.; Hinzano, S.M.; Bossier, P. Causative Agent, Diagnosis and Management of White Spot Disease in Shrimp: A Review. Rev. Aquac. 2020, 12, 822–865. [Google Scholar] [CrossRef]
- Zhang, Q.; Xu, T.; Wan, X.; Liu, S.; Wang, X.; Li, X.; Dong, X.; Yang, B.; Huang, J. Prevalence and Distribution of Covert Mortality Nodavirus (CMNV) in Cultured Crustacean. Virus Res. 2017, 233, 113–119. [Google Scholar] [CrossRef]
- Prasad, K.P.; Shyam, K.U.; Banu, H.; Jeena, K.; Krishnan, R. Infectious Myonecrosis Virus (IMNV)—An Alarming Viral Pathogen to Penaeid Shrimps. Aquaculture 2017, 477, 99–105. [Google Scholar] [CrossRef]
- Prachumwat, A.; Munkongwongsiri, N.; Eamsaard, W.; Lertsiri, K.; Flegel, T.W.; Stentiford, G.D.; Sritunyalucksana, K. A Potential Prokaryotic and Microsporidian Pathobiome That May Cause Shrimp White Feces Syndrome (WFS). bioRxiv 2021, 12120. [Google Scholar] [CrossRef]
- Kannan, D.; Thirunavukkarasu, P.; Jagadeesan, K.; Shettu, N.; Kumar, A. Procedure for Maturation and Spawning of Imported Shrimp Litopenaeus Vannamei in Commercial Hatchery, South East Coast of India. Fish. Aquac. J. 2015, 6. [Google Scholar] [CrossRef]
- Taylor, J.; Vinatea, L.; Ozorio, R.; Schuweitzer, R.; Andreatta, E.R. Minimizing the Effects of Stress during Eyestalk Ablation of Litopenaeus Vannamei Females with Topical Anesthetic and a Coagulating Agent. Aquaculture 2004, 233, 173–179. [Google Scholar] [CrossRef]
- Menezes, T.B.B. de Efeito Da Não Ablação No Processo de Reprodução de Fêmeas de Penaeus Vannamei. 54 f. Master’s Thesis, (Mestrado em Engenharia de Pesca)–Universidade Federal do Ceará, Fortaleza, Brazil, 2019. [Google Scholar]
- Ren, S.; Prentis, P.; Mather, P.B.; Li, Y.; Tang, B.; Hurwood, D.A. Genetic Parameters for Growth and Survival Traits in a Base Population of Pacific White Shrimp (Litopenaeus Vannamei) Developed from Domesticated Strains in China. Aquaculture 2020, 523, 735148. [Google Scholar] [CrossRef]
- Ogle, J.T.; Beaugez, K.; Lotz, J.M. Effects of Salinity on Survival and Growth of Postlarval Penaeus Vannamei. Gulf. Caribb Res. 1992, 8, 415–421. [Google Scholar] [CrossRef]
- Doyle, R.W. Inbreeding and Disease in Tropical Shrimp Aquaculture: A Reappraisal and Caution. Aquac. Res. 2016, 47, 21–35. [Google Scholar] [CrossRef]
- Pontes, C.S.; de F. Arruda, M. Acesso Ao Alimento Artificial e Enchimento Do Trato Digestivo de Juvenis Do Camarão Marinho Litopenaeus Vannamei (Boone) (Crustacea, Decapoda, Penaeidae) Durante as Fases Clara e Escura Do Período de 24 Horas. Rev. Bras. Zool. 2005, 22, 1039–1043. [Google Scholar] [CrossRef]
- Wyk, P. Van Nutrition and Feeding of Litopenaeus Vannamei in Intensive Culture System. Farming Mar. Shrimp Recirc. Freshw. Syst. 1999, 2, 125–139. [Google Scholar]
- Alencar, C.A. Relatório Sobre o Acompanhamento de Um Cultivo de Camarão Branco Do Pacífico, Litopenaeus Vannamei (Boone,1931), Em Viveiros Escavados, Realizado Na Fazenda Santa Isabel, No Município de Aracati–CE. Relatório de Estágio Supervisionado. Relatório supervisionado; Departamento de Engenharia de Pesca do Centro de Ciências Agrárias, Universidade Federal do Ceará: Fortaleza, CE, Brasil, 2003. [Google Scholar]
- de Sousa, R.R. Descrição Do Cultivo de Camarões Marinho Em Duas Fazendas Da CPH Aquacultura LTDA; Trabalho de Conclusão de Curso: Fortaleza, CE, Brasil, 2009. [Google Scholar]
- Felix, S. Advances in Shrimp Aquaculture Management, 1st ed.; Daya Publishing House: New Delhi, India, 2009; ISBN 9788170355472. [Google Scholar]
- Carvalho, E.A. Efeito Da Frequencia Alimentar Sovbre o Desempenho Zootécnico Do Camarão Branco Do Pacífico Litopenaeus Vannamei Em Condições de Cultivo. Diploma Thesis, Universidade de São Paulo–USP, São Paulo, SP, Brasil, 2005. [Google Scholar]
- Brito, M.D.L.S. Arraçoamento Do Litopenaeus Vannamei: Desempenho, Sobrevivência e Qualidade Do Efluente. Diploma Thesis, Universidade de São Paulo–USP, São Paulo, SP, Brasil, 2017. [Google Scholar]
- De Lima, P.P.; Pontes, C.S.; Arruda, M.D.F. Activity Pattern of the Marine Shrimp Litopenaeus Vannamei (Boone 1931) in Laboratory as a Function of Different Feeding Frequencies. Aquac. Res. 2009, 41, 53–60. [Google Scholar] [CrossRef]
- Wouters, R.; Lavens, P.; Nieto, J.; Sorgeloos, P. Penaeid Shrimp Broodstock Nutrition: An Updated Review on Research and Development. Aquaculture 2001, 202, 1–21. [Google Scholar] [CrossRef]
- Hoa, N.D.; Wouters, R.; Wille, M.; Thanh, V.; Dong, T.K.; Van Hao, N.; Sorgeloos, P. A Fresh-Food Maturation Diet with an Adequate HUFA Composition for Broodstock Nutrition Studies in Black Tiger Shrimp Penaeus Monodon (Fabricius, 1798). Aquaculture 2009, 297, 116–121. [Google Scholar] [CrossRef]
- Racotta, I.S.; Palacios, E.; Ibarra, A.M. Shrimp Larval Quality in Relation to Broodstock Condition. Aquaculture 2003, 227, 107–130. [Google Scholar] [CrossRef]
- Harrison, K.E. The Role of Nutrition in Maturation Reproduction and Embryonic Development of Decapod Crustaceans a Review. J. Shellfish. Res. 1990, 9, 1–28. [Google Scholar] [CrossRef]
- Vaghei, G.R.; Abolhasani, M.H.; Matinfar, A.; Dadgar, S.; Ghorbani, R. Production of Artificial Diets for Female Broodstock of Western White Shrimp (Litopenaeus Vannamei) and Study on Their Singular Effect. Iran J. Fish. Sci. 2017, 16, 1204–1213. [Google Scholar]
- Rocha, N.S. Maturação Do Camarão Marinho Litopenaeus Vannamel: O Modelo de Produção Da Aquacrusta Ltda (Acarau-Ce). Diploma Thesis, Universidade Federal do Ceará, Aracau, CE, Brasil, 2007. [Google Scholar]
- de Oliveira, L.L. Larvicultura Do Camarão Marinho Litopenaeus Vannamei; Graduation thesis in Fishing Engineering; Universidade Federal do Ceará: Aracaú, CE, Brasil, 2006. [Google Scholar]
- Cuzon, G.; Lawrence, A.; Gaxiola, G.; Rosas, C.; Guillaume, J. Nutrition of Litopenaeus Vannamei Reared in Tanks or in Ponds. Aquaculture 2004, 235, 513–551. [Google Scholar] [CrossRef]
- Bardera, G.; Owen, M.A.G.; Façanha, F.N.; Sloman, K.A.; Alexander, M.E. The Influence of Sex on Feeding Behaviour in Pacific White Shrimp (Litopenaeus Vannamei). Appl. Anim. Behav. Sci. 2020, 224, 104946. [Google Scholar] [CrossRef]
- Venero, J.A.; Davis, D.A.; Rouse, D.B. Variable Feed Allowance with Constant Protein Input for the Pacific White Shrimp Litopenaeus Vannamei Reared under Semi-Intensive Conditions in Tanks and Ponds. Aquaculture 2007, 269, 490–503. [Google Scholar] [CrossRef]
- Severino, D.V. Formação de Reprodutores de Camarões Marinhos (Litopenaeus Vannamei) Em Sistema Biosseguro de Bioflocos. Diploma Thesis, Universidade Federal de Santa Catarina, Florianópolis, SC, Brasil, 2014. [Google Scholar]
- Zheng, C.; Zhao, Q.; Li, E.; Zhao, D.; Sun, S. Role of Hypoxia in the Behaviour, Physiology, Immunity and Response Mechanisms of Crustaceans: A Review. Rev. Aquac. 2022, 14, 676–687. [Google Scholar] [CrossRef]
- Darodes de Tailly, J.B.; Keitel, J.; Owen, M.A.G.; Alcaraz-Calero, J.M.; Alexander, M.E.; Sloman, K.A. Monitoring Methods of Feeding Behaviour to Answer Key Questions in Penaeid Shrimp Feeding. Rev. Aquac. 2021, 13, 1828–1843. [Google Scholar] [CrossRef]
- Parodi, T.V.; Cunha, M.A.; Heldwein, C.G.; De Souza, D.M.; Martins, Á.C.; Garcia, L.D.O.; Junior, W.W.; Monserrat, J.M.; Schmidt, D.; Caron, B.O.; et al. The Anesthetic Efficacy of Eugenol and the Essential Oils of Lippia Alba and Aloysia Triphylla in Post-Larvae and Sub-Adults of Litopenaeus Vannamei (Crustacea, Penaeidae). Comp. Biochem. Physiol. C Toxicol. Pharmac. 2012, 155, 462–468. [Google Scholar] [CrossRef] [PubMed]
- Becker, A.J.; Vaz, L.J.; De Garcia, L.O.; Wasielesky, W.; Heinzmann, B.M.; Baldisserotto, B. Anesthetic Potential of Different Essential Oils for Two Shrimp Species, Farfantepenaeus Paulensis and Litopenaeus Vannamei (Decapoda, Crustacea). Cienc. Rural. 2021, 51. [Google Scholar] [CrossRef]
- de Souza Valente, C. Anaesthesia of Decapod Crustaceans. Vet. Anim. Sci. 2022, 16, 100252. [Google Scholar] [CrossRef]
- Serviço Nacional de Aprendizagem Rural. Larvicultura de Camarão Marinho: Do Náuplio a Pós-Larva, 1st ed.; Serviço Nacional de Aprendizagem Rural: Brasília, DF, Brasil, 2016; Volume 1. [Google Scholar]
- Bermudes-Lizárraga, J.; Nieves-Soto, M.; Medina-Jasso, A.; Piña-Valdez, P.; Bermudes-Lizárraga, J.; Nieves-Soto, M.; Medina-Jasso, A.; Piña-Valdez, P. Effect of Temperature and Salinity on Larval Survival and Development of Litopenaeus Vannamei. Rev. MVZ Cordoba 2017, 22, 5844–5853. [Google Scholar] [CrossRef]
- Ravichandran, P.; Devarajan, K.; Pandian, S.K. Unit-7 Seed Production and Larval Rearing of Shrimps. In Block-2 Hatchery Technology for Prawns and Fishes; Indira Gandhi National Open University New Delhi: New Delhi, India, 2004; p. 11. [Google Scholar]
- Nunes, H.R.; Andreatta, E.R. Efeito Da Salinidade e Temperatura Sobre a Taxa de Metamorfose de Náuplios Para Protozoea e Sobre a Qualidade Das Larvas de Litopenaeus Vannamei. Atlântica 2011, 33, 87–96. [Google Scholar] [CrossRef]
- Ponce-Palafox, J.; Martinez-Palacios, C.A.; Ross, L.G. The Effects of Salinity and Temperature on the Growth and Survival Rates of Juvenile White Shrimp, Penaeus Vannamei, Boone, 1931. Aquaculture 1997, 157, 107–115. [Google Scholar] [CrossRef]
- Viet, L.Q.; Hai, T.N.; Phu, T.M.; Ngan, T.V. Effects of Photoperiods on Growth and Quality of White Leg Shrimp (Litopenaeus van-Namei) in Biofloc System. Can Tho Univ. J. Sci. 2017, 6, 83. [Google Scholar] [CrossRef]
- Sanudin, N.; Daning Tuzan, A.; Seok, A.; Yong, K. Feeding Activity and Growth Performance of Shrimp Post Larvae Litopenaeus Vannamei Under Light and Dark Condition. J. Agric. Sci. 2014, 6, p103. [Google Scholar] [CrossRef]
- Cobo, M.D.L.; Sonnenholzner, S.; Wille, M.; Sorgeloos, P. Ammonia Tolerance of Litopenaeus Vannamei (Boone) Larvae. Aquac. Res. 2014, 45, 470–475. [Google Scholar] [CrossRef]
- Valencia-Castañeda, G.; Frías-Espericueta, M.G.; Vanegas-Pérez, R.C.; Pérez-Ramírez, J.A.; Chávez-Sánchez, M.C.; Páez-Osuna, F. Acute Toxicity of Ammonia, Nitrite and Nitrate to Shrimp Litopenaeus Vannamei Postlarvae in Low-Salinity Water. Bull. Environ. Contam. Toxicol. 2018, 101, 229–234. [Google Scholar] [CrossRef]
- Frías-Espericueta, M.G.; Harfush-Melendez, M.; Páez-Osuna, F. Effects of Ammonia on Mortality and Feeding of Postlarvae Shrimp Litopenaeus Vannamei. Bull. Environ. Contam. Toxicol. 2000, 65, 98–103. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, B.L.F. de Adição Do Nitrito de Sódio Em Sistema de Biofloco e Toxicidade Do Nitrito Para o Camarão Litopenaeus Vannamei. Diploma Thesis, Universidade Federal Rural de Pernambuco, Recife, PE, Brasil, 2013. [Google Scholar]
- Branco, L.F.C. Avaliação Da Taxa de Sobrevivência Final Na Larvicultura Do Camarão Marinho Litopenaeus Vannamei Através de Diferentes Densidades de Estocagem. Diploma Thesis, Universidade Federal Rural do Semi-Árido, Mossoró, RN, Brasil, 2021. [Google Scholar]
- López Peraza, D.J.; Bermudes Lizárraga, J.F.; Flores Alarcón, J.M.; Nieves Soto, M. Effect of the Larval Density and Food Ratio on Litopenaeus Vannamei (Boone, 1931) Zoea. Agro Prod. 2022. [Google Scholar] [CrossRef]
- Knoll, R.C. Sistema de Avaliação Da Qualidade de Pós-Larvas Do Camarão Marinho Litopenaeus Vannamei (Boone, 1931). Diploma Thesis, Universidade Federal de Santa Catarina, Florianópolis, SC, Brasil, 2012. [Google Scholar]
- Magalhães, M.E. Cultivo do Camarão Marinho Litopenaeus Vannamei (BOONE,1931) em Sistema Multifásico–Pesquisa Google. Diploma Thesis, Universidade Federal Rural de Pernambuco, Recife, Brazil, 2004. [Google Scholar]
- Peregrino, L.H.S.; Correia, E.S.; Oliveira, A. Revista da ABCC; Associação Brasileira de Criadores de Camarão: Recife, PE, Brazil, 2002; pp. 1–70. [Google Scholar]
- Alencar, C.A. Acompanhamento de um Cultivo Intensivo de Camarão Branco do Pacífico, Litopenaeus Vannamei (BOONE,1931), em Viveiros Escavados, Realizado na Fazenda Santa Isabel, No Municipio de Aracati-CE. Diploma Thesis, Universidade Federal do Ceará, Fortaleza, CE, Brazil, 2003. [Google Scholar]
- Gelabert, R.; Pacheco, A. Selectivity of Particle Size by the Shrimp Litopenaeus Vannamei (Boone, 1931) Larvae. Aquac. Nutr. 2011, 17, 244–247. [Google Scholar] [CrossRef]
- Barbieri, R.C.; Ostrensky, A. Camarões Marinhos–Engorda Ostrensky; Viçosa: Aprenda Fácil, Brazil, 2002; 326p. [Google Scholar]
- Van Wyk, P.; Davis-Hodgkins, M.; Laramore, R.; Main, K.L.; Mountain, J.; Scarpa, J. Farming Marine Shrimp in Recirculating Freshwater Systems Harbor Branch Oceanographic Institution. In Farming Marine Shrimp in Recirculating Freshwater Systems; van Wyk, P., Davis-Hodgkins, M., Laramore, R., Main, K.L., Mountain, J., Scarpa, J., Eds.; Harbor Branch Oceanographic Institution: Fort Pierce, FL, USA, 1999; Volume 220, pp. 125–140. [Google Scholar]
- Jiménez-Yan, L.; Brito, A.; Cuzon, G.; Gaxiola, G.; García, T.; Taboada, G.; Soto, L.A.; Brito, R. Energy Balance of Litopenaeus Vannamei Postlarvae Fed on Animal or Vegetable Protein Based Compounded Feeds. Aquaculture 2006, 260, 337–345. [Google Scholar] [CrossRef]
- Colvin, L.B.; Brand, C.W. The protein requirement of penaeid shrimp at various life-cycle stages in controlled environment systems. Proc. Annu. Meet. World Maric. Soc. 1977, 8, 821–840. [Google Scholar] [CrossRef]
- Robertson, L.; Wrence, A.L.L.; Castille, F.L. Effect of Feeding Frequency and Feeding Time on Growth of Penaeus Vannamei (Boone). Aquac. Res. 1993, 24, 1–6. [Google Scholar] [CrossRef]
- Aalimahmoudi, M.; Reyshahri, A.; Salehipour Bavarsad, S.; Maniat, M.; Mazdak Aalimahmoudi, C. Effects of Feeding Frequency on Growth, Feed Conversion Ratio, Survival Rate and Water Quality of White Leg Shrimp (Litopenaeus Vannamei, Boone, 1931). Int. J. Fish. Aquat. Stud. 2016, 4, 293–297. [Google Scholar]
- Kuban, F.D.; Lawrence, A.L.; Wilkenfeld, J.S. Survival, Metamorphosis and Growth of Larvae from Four Penaeid Species Fed Six Food Combinations. Aquaculture 1985, 47, 151–162. [Google Scholar] [CrossRef]
- Chu, K.H.; Shing, C.K. Feeding Behavior of the Shrimp, Metapenaeus Ensis, on Artemia Nauplii. Aquaculture 1986, 58, 175–184. [Google Scholar] [CrossRef]
- Piña, P.; Nieves, M.; Ramos-Brito, L.; Chavira-Ortega, C.O.; Voltolina, D. Survival, Growth and Feeding Efficiency of Litopenaeus Vannamei Protozoea Larvae Fed Different Rations of the Diatom Chaetoceros Muelleri. Aquaculture 2005, 249, 431–437. [Google Scholar] [CrossRef]
- Martín, L.; Arenal, A.; Fajardo, J.; Pimentel, E.; Hidalgo, L.; Pacheco, M.; García, C.; Santiesteban, D. Complete and Partial Replacement of Artemia Nauplii by Moina Micrura during Early Postlarval Culture of White Shrimp (Litopenaeus Schmitti). Aquac. Nutr. 2006, 12, 89–96. [Google Scholar] [CrossRef]
- Kolkovski, S. Digestive Enzymes in Fish Larvae and Juveniles—Implications and Applications to Formulated Diets. Aquaculture 2001, 200, 181–201. [Google Scholar] [CrossRef]
- Barajas, F.J.M.; Villegas, R.S.; Clark, G.P.; Moreno, B.L. Litopenaeus Vannamei (Boone) Post-Larval Survival Related to Age, Temperature, PH and Ammonium Concentration. Aquac. Res. 2006, 37, 492–499. [Google Scholar] [CrossRef]
- Soares, M.; Evangelista, D.K.R.; Pereira, A.M.L. Boas Práticas de Manejo e de Biossegurança Na Carcinicultura Para Convivência Com Enfermidades; Embrapa Pesca e Aquicultura: Brasília, DF, Brasil, 2021; ISSN 2318-1400. [Google Scholar]
- Moraes, R.d.C. Influência Da Densidade e Idade No Transporte de Pós-Larvas Do Camarão Marinho Litopenaeus Vannamei. Diploma Thesis, Universidade Federal de Santa Catarina, Florianópolis, SC, Brasil, 2004. [Google Scholar]
- Saputra, H.K.; Hamka, M.S.; Kurniaji, A.; Susanti, L.; Firman, S.W.; Dwiarto, A.; Alam, H.S. The Technology of Shrimp Larvae Transportation: Ecophysiology and Bioeconomy Effects on High Stocking Density Shrimp Litopenaeus Vannamei. E3S Web Conf. 2022, 348, 1–38. [Google Scholar] [CrossRef]
- ABCC. Projeto de Desenvolvimento Tecnológico Com Boas Práticas de Manejo e Biossegurança Para a Carcinicultura No Nordeste; Associação Brasileira de Criadores de Camarão: Candelária, RN, Brazil, 2017; p. 75. [Google Scholar]
- Vinza, A.; Emma, G.; Loaiza, L.; Nicole, P. Elaboración de Una Guía de Transporte Para Postlarvas de Penaeus Vannamei. 2020. Graduation Thesis in Aquaculture Engineering. Escuela Superior Politécnica del Litoral, Guayaquil, Ecuador. Available online: http://www.dspace.espol.edu.ec/xmlui/handle/123456789/50598 (accessed on 29 January 2023).
- Ostrensky, A.; Stevanato, D.J.; Pont, G.D.; Castilho-Westphal, G.G.; Girotto, M.V.F.; Cozer, N.; García-Madrigal, R.F.d.A.; Silva, U. A Produção Integrada Na Carcinicultura Brasileira (Volume 2), 1st ed.; Ostrensky, A., Cozer, N., Eds.; Instituto GIA: Curitiba, PR, Brazil, 2017. [Google Scholar]
- Yonghui, Y.; Guangbin, H.; Zhong, P.; Zulei, J. Analysis of Mortality Rate of Penaeus Vannamei Larvae Transferred from Large Culture Ponds to Small Culture Tanks. J. Fish. Livest Prod. 2021, 9, 1–4. [Google Scholar]
- Liu, H.L.; Yang, S.P.; Wang, C.G.; Chan, S.M.; Wang, W.X.; Feng, Z.H.; Sun, C.B. Effect of Air Exposure and Resubmersion on the Behavior and Oxidative Stress of Pacific White Shrimp Litopenaeus Vannamei. N. Am. J. Aquac. 2015, 77, 43–49. [Google Scholar] [CrossRef]
- Akbari, S.; Khoshnod, M.J.; Rajaian, H.; Afsharnasab, M. The Use of Eugenol as an Anesthetic in Transportation of With Indian Shrimp (Fenneropenaeus Indicus) Post Larvae. Turk. J. Fish. Aquat. Sci. 2010, 10, 423–429. [Google Scholar] [CrossRef]
- Kumlu, M.; Kumlu, M.; Turkmen, S. Combined Effects of Temperature and Salinity on Critical Thermal Minima of Pacific White Shrimp Litopenaeus Vannamei (Crustacea: Penaeidae). J. Therm. Biol. 2010, 35, 302–304. [Google Scholar] [CrossRef]
- Chamberlain, G.W.; Hutchins, D.L.; Lawrence, A.L.; Parker, J.C. Winter Culture of Penaeus Vannamei in Ponds Receiving Thermal Effluent at Different Rates. Proceed World Maricul. Soc. 2009, 11, 30–43. [Google Scholar] [CrossRef]
- Wyban, J.; Walsh, W.A.; Godin, D.M. Temperature Effects on Growth, Feeding Rate and Feed Conversion of the Pacific White Shrimp (Penaeus Vannamei). Aquaculture 1995, 138, 267–279. [Google Scholar] [CrossRef]
- Abdelrahman, H.A.; Abebe, A.; Boyd, C.E. Influence of Variation in Water Temperature on Survival, Growth and Yield of Pacific White Shrimp Litopenaeus Vannamei in Inland Ponds for Low-Salinity Culture. Aquac. Res. 2019, 50, 658–672. [Google Scholar] [CrossRef]
- Bückle, F.L.; Barón, B.; Hernández, M. Osmoregulatory Capacity of the Shrimp Litopenaeus Vannamei at Different Temperatures and Salinities, and Optimal Culture Environment. Available online: https://www.scielo.sa.cr/scielo.php?script=sci_arttext&pid=S0034-77442006000300005 (accessed on 26 June 2022).
- Hernández, R.M.; Bückle, R.L.F.; Palacios, E.; Barón, S.B. Preferential Behavior of White Shrimp Litopenaeus Vannamei (Boone 1931) by Progressive Temperature–Salinity Simultaneous Interaction. J. Therm. Biol. 2006, 31, 565–572. [Google Scholar] [CrossRef]
- Lima, P.P. de Influência Da Salinidade e Temperatura Da Água Nas Respostas Comportamental e Fisiológica de Camarões Marinhos Litopenaeus Vannamei (BOONE 1931); Universidade Federal do Rio Grande do Norte: Lagoa Nova, RN, Brazil, 2011. [Google Scholar]
- Boyd, C.E. Management Practices for Reducing the Environmental Impacts of Shrimp Farming in Methods for Improving Shrimp Farming in Central Americ UCA University Press Managua, Nicaragua. In Bottom Soils, Sediment, and Pond Aquaculture; Chapman and Hall: New York, NY, USA, 2001; p. 348. [Google Scholar]
- Furtado, P.S.; Fugimura, M.M.S.; Monserrat, J.M.; Souza, D.M.; Garcia, L.d.O.; Wasielesky, W. Acute Effects of Extreme PH and Its Influences on the Survival and Biochemical Biomarkers of Juvenile White Shrimp, Litopenaeus Vannamei. Mar. Fresh Behav. Physiol. 2015, 48, 417–429. [Google Scholar] [CrossRef]
- Girotto, M.V.F. Efeitos Da Amônia Sobre Juvenis de Litopenaeus Vannamei (BOONE, 1931) e Litopenaeus Schmitti ( BURKENROAD, 1936): Excreção e Toxicidade. Diploma Thesis, Universidade Federal do Paraná, Curitiba, PR, Brazil, 2010. [Google Scholar]
- Re, A.D.; Díaz, F.; Ponce-Rivas, E.; Giffard, I.; Muñoz-Marquez, M.E.; Sigala-Andrade, H.M. Combined effect of temperature and salinity on the thermotolerance and osmotic pressure of juvenile white shrimp Litopenaeus vannamei (Boone). J. Therm. Biol. 2012, 37, 413–418. [Google Scholar] [CrossRef]
- da Fonseca, S.B.; de Paula Mendes, P.; de Lyra Albertim, C.J.; Bittencourt, C.F.; da Silva, J.H.V. Cultivo Do Camarão Marinho Em Água Doce Em Diferentes Densidades de Estocagem. Pesq. Agropec. Bras. 2009, 44, 1352–1358. [Google Scholar] [CrossRef]
- Kotiya, A.S.; Vadher, K.H. Effect of Different Stocking Density on Growth, Survival on Litopenaeus Vannamei (Boone, 1931) in Summer and Monsoon Crop in Province of Gujarat States in India. J. Surv. Fish. Sci. 2021, 7, 71–99. [Google Scholar] [CrossRef]
- Mena-Herrera, A.; Gutierrez-Corona, C.; Linan-Cabello, M.; Sumano-Lopez, H. Effects of Stocking Densities on Growth of the Pacific White Shrimp (Litopenaeus Vannamei) in Earthen Ponds. Isr. J. Aquac. Bamidgeh 2006, 58, 205–213. [Google Scholar] [CrossRef]
- Sookying, D.; Silva, F.S.D.; Davis, D.A.; Hanson, T.R. Effects of Stocking Density on the Performance of Pacific White Shrimp Litopenaeus Vannamei Cultured under Pond and Outdoor Tank Conditions Using a High Soybean Meal Diet. Aquaculture 2011, 319, 232–239. [Google Scholar] [CrossRef]
- Wyban, J.A.; Lee, C.S.; Sato, V.T.; Sweeney, J.N.; Richards, W.K. Effect of Stocking Density on Shrimp Growth Rates in Manure-Fertilized Ponds. Aquaculture 1987, 61, 23–32. [Google Scholar] [CrossRef]
- Bardera, G.; Owen, M.A.G.; Façanha, F.N.; Alcaraz-Calero, J.M.; Alexander, M.E.; Sloman, K.A. The Influence of Density and Dominance on Pacific White Shrimp (Litopenaeus Vannamei) Feeding Behaviour. Aquaculture 2021, 531, 735949. [Google Scholar] [CrossRef]
- da Costa, F.P.; Gomes, B.S.F.; Pereira, S.D.d.N.A.; de Fátima Arruda, M. Influence of Stocking Density on the Behaviour of Juvenile Litopenaeus Vannamei (Boone, 1931). Aquac. Res. 2016, 47, 912–924. [Google Scholar] [CrossRef]
- Barbieri, R.C.; Ostrensky, A. Camarões Marinhos: Engorda II; Aprenda Fácil Editora: Viçosa, Brasil, 2020. [Google Scholar]
- Martinez-Cordova, L.R.; Porchas-Cornejo, M.A.; Villarreal-Colemnares, H.; Calderon-Perez, J.A.; Naranjo-Paramo, J. Evaluation of Three Feeding Strategies on the Culture of White Shrimp Penaeus Vannamei Boone 1931 in Low Water Exchange Ponds. Aquac. Eng. 1998, 17, 21–28. [Google Scholar] [CrossRef]
- Carvalho, E.A.; Nunes, A.J.P. Effects of Feeding Frequency on Feed Leaching Loss and Grow-out Patterns of the White Shrimp Litopenaeus Vannamei Fed under a Diurnal Feeding Regime in Pond Enclosures. Aquaculture 2006, 252, 494–502. [Google Scholar] [CrossRef]
- Sousa, P. Análise Da Eficiência Da Produção. 2003. [Google Scholar]
- Amaya, E.; Davis, D.A.; Rouse, D.B. Alternative Diets for the Pacific White Shrimp Litopenaeus Vannamei. Aquaculture 2007, 262, 419–425. [Google Scholar] [CrossRef]
- Albertim-Santos, C.J.; Santos, D.L.; Mendes, P. pAULA Uso de Modelos Matemáticos Para Avaliação Das Variáveis de Manejo Do Litopenaeus Vannamei (Boone, 1931)/Use of Mathematical Models for Evolution of the Management Variables Litopenaeus Vannamei (Boone, 1931). Acta Fish. Aquatic. Resour. 2014, 2, 28–39. [Google Scholar] [CrossRef]
- Maia, E.P. Produzindo Camarão Em Sistema Trifásico. Rev. Panor. Aquic. 2017, 1, 5–32. [Google Scholar]
- Boyd, C.E.; Davis, R.P.; Wilson, A.G.; Marcillo, F.; Brian, S.; McNevin, A.A. Resource Use in Whiteleg Shrimp Litopenaeus Vannamei Farming in Ecuador. J. World Aquac. Soc. 2021, 52, 772–788. [Google Scholar] [CrossRef]
- Nunes, A.J.P.; Sandoval, P.F.C. Dados de Produção e Qualidade de Água de Um Cultivo Comercial Semi-Intensivo Dos Camarões Penaeus Subtilis e P. Vannamei Com a Utilização de Bandejas de Alimentação. Rev. Panor. Aquic. 1997, 1, 12–15. [Google Scholar]
- Farias, G.R. Eficiência de Bandejas Na Alimentação de Juvenis Do Camarão Branco, Litopenaeus Vannamei (BOONE, 1931) Avaliado Por Meio de Imagens Subaquáticas. Diploma Thesis, Universidade Federal do Ceará, Fortaleza, CE, Brasil, 2013. [Google Scholar]
- Nunes, A.J.P. Alimentação Para Camarões Marinhos–Parte II. Panor. Aquic. 2001, 63, 17–21. [Google Scholar]
- Pontes, C.S.; Arruda, M.F. Comportamento de Litopenaeus vannamei (Boone)(Crustacea, Decapoda, Penaeidae) em função da oferta do alimento artificial nas fases clara e escura do período de 24 horas. Rev. Bras. Zool. 2005, 22, 648–652. [Google Scholar] [CrossRef]
- Robles-Romo, A.; Zenteno-Savín, T.; Racotta, I.S. Bioenergetic Status and Oxidative Stress during Escape Response until Exhaustion in Whiteleg Shrimp Litopenaeus Vannamei. J. Exp. Mar. Biol. Ecol. 2016, 478, 16–23. [Google Scholar] [CrossRef]
- Becker, A.J.; Ramos, P.B.; Monserrat, J.M.; Wasielesky, W.; Baldisserotto, B. Behavioural and Biochemical Responses in Adult Pacific White Shrimp, Litopenaeus Vannamei, Exposed to the Essential Oil of Cymbopogon Citratus. Aquac. Res. 2021, 52, 6205–6217. [Google Scholar] [CrossRef]
- Stoner, A.W. Evaluating Vitality and Predicting Mortality in Spot Prawn, Pandalus Platyceros, Using Reflex Behaviors. Fish. Res. 2012, 119–120, 108–114. [Google Scholar] [CrossRef]
- Tomlinson, M. Crustacean Compassion: Should decapod crustaceans be legally defined as animals? Fish. Vet. J. 2018, 16, 30–35. [Google Scholar]
- Keeling, L.; Tunón, H.; Olmos Antillón, G.; Berg, C.; Jones, M.; Stuardo, L.; Swanson, J.; Wallenbeck, A.; Winckler, C.; Blokhuis, H. Animal Welfare and the United Nations Sustainable Development Goals. Front. Vet. Sci. 2019, 6, 336. [Google Scholar] [CrossRef] [PubMed]
- Saraiva, J.; Volstorf, J. Whiteleg Shrimp Litopenaeus Vannamei. Available online: https://fishethobase.net/db/21/farm/shortprofile/ (accessed on 6 January 2023).
- Wright, J. Seajoy’s Ablation-Free Shrimp Answers Emerging Welfare Concern-Responsible Seafood Advocate. Available online: https://www.globalseafood.org/advocate/seajoys-ablation-free-shrimp-answers-emerging-welfare-concern/ (accessed on 6 January 2023).
- Uawisetwathana, U.; Leelatanawit, R.; Klanchui, A.; Prommoon, J.; Klinbunga, S.; Karoonuthaisiri, N. Insights into Eyestalk Ablation Mechanism to Induce Ovarian Maturation in the Black Tiger Shrimp. PLoS ONE 2011, 6, e24427. [Google Scholar] [CrossRef]
- Okumura, T. Perspectives on Hormonal Manipulation of Shrimp Reproduction. Jpn. Agric. Res. Q. JARQ 2004, 38, 49–54. [Google Scholar] [CrossRef]
- Segner, H.; Sundh, H.; Buchmann, K.; Douxfils, J.; Sundell, K.S.; Mathieu, C.; Ruane, N.; Jutfelt, F.; Toften, H.; Vaughan, L. Health of Farmed Fish: Its Relation to Fish Welfare and Its Utility as Welfare Indicator. Fish. Physiol. Biochem. 2012, 38, 85–105. [Google Scholar] [CrossRef]
- Millard, R.S.; Ellis, R.P.; Bateman, K.S.; Bickley, L.K.; Tyler, C.R.; van Aerle, R.; Santos, E.M. How Do Abiotic Environmental Conditions Influence Shrimp Susceptibility to Disease? A Critical Analysis Focussed on White Spot Disease. J. Invertebr. Pathol. 2021, 186, 1–13. [Google Scholar] [CrossRef]
- ASC. ASC Shrimp Standard Version 1.1; Aquaculture Stewardship Council: Utrecht, The Netherlands, 2019. [Google Scholar]
- Stien, L.H.; Bracke, M.; Noble, C.; Kristiansen, T.S. Assessing Fish Welfare in Aquaculture. In The Welfare of Fish; Springer: Cham, Switzerland, 2020; pp. 303–321. [Google Scholar]
- Noble, C.; Gismervik, K.; Iversen, M.H.; Kolarevic, J.; Nilsson, J.; Stien, L.H.; Turnbull, J.F. Welfare Indicators for Farmed Atlantic Salmon–Part A. Knowledge and Theoretical Background. In Welfare Indicators for Farmed Atlantic Salmon: Tools for Assessing Fish Welfare; NOFIMA: Tromsø, Norway, 2018; pp. 10–145. ISBN 9788282965569. [Google Scholar]
- Sivaraman, I.; Krishnan, M.; Radhakrishnan, K. Better Management Practices for Sustainable Small-Scale Shrimp Farming. J. Clean. Prod. 2019, 214, 559–572. [Google Scholar] [CrossRef]
- Kim, N.T.Q.; van Hien, H.; Doan Khoi, L.N.; Yagi, N.; Lerøy Riple, A.K. Quality Management Practices of Intensive Whiteleg Shrimp (Litopenaeus Vannamei) Farming: A Study of the Mekong Delta, Vietnam. Sustainability 2020, 12, 4520. [Google Scholar] [CrossRef]
- Florina, P.; Hartoyo, S.; Kunci, K.; Vannamei, L.; Udang, T.; Management Practice, G.; Nilai, R.; Kasus Berganda, S. Adoption of Good Management Practice (GMP) in Small and Medium Scale Vannamei Shrimp Farms on the Northern Shore of East Java. J. Manaj. Agribis 2012, 9, 79–88. [Google Scholar]
- Madhu, K.; Madhu, R. School Course Manual on “Recent Advances in Breeding and Larviculture of Marine Finfish and Shellfish”; Central Marine Fisheries Research Institute (Indian Council of Agricultural Research): Cochin, Kerala, India, 2008; Volume 12, pp. 1–12. [Google Scholar]
- Otta, S.K.; Patil, P.K. Training Programme on Management of Emerging Diseases of Shrimp with Special Reference to Pacific White Shrimp, Litopenaeus Vannamei; Indian Council of Agricultural Research: Chennai, Tamil Nadu, India, 2012; Volume 111, pp. 42–88. [Google Scholar]
- Barreto, M.O.; Rey Planellas, S.; Yang, Y.; Phillips, C.; Descovich, K. Emerging Indicators of Fish Welfare in Aquaculture. Rev. Aquac. 2022, 14, 343–361. [Google Scholar] [CrossRef]
- Lefort, M.-C.; Cruickshank, R.H.; Descovich, K.; Adams, N.J.; Barun, A.; Emami-Khoyi, A.; Ridden, J.; Smith, V.R.; Sprague, R.; Waterhouse, B.; et al. Blood, Sweat and Tears: A Review of Non-Invasive DNA Sampling. Peer Community J. 2022, 2. [Google Scholar] [CrossRef]
- Garshelis, D.L. On the Allure of Noninvasive Genetic Sampling–Putting a Face to the Name-Web of Science Core Collection. URSUS 2006, 17, 109–123. [Google Scholar] [CrossRef]
- Tu, H.T.; Silvestre, F.; Wang, N.; Thome, J.P.; Phuong, N.T.; Kestemont, P. A Multi-Biomarker Approach to Assess the Impact of Farming Systems on Black Tiger Shrimp (Penaeus Monodon). Chemosphere 2010, 81, 1204–1211. [Google Scholar] [CrossRef]
- Battison, A.L.; Horney, B.; Ciaramella, M.A. Measurement of Tissue Lipid Reserves in the American Lobster (Homarus Americanus): Hemolymph Metabolites as Potential Biomarkers of Nutritional Status. J. Crustac. Biol. 2014, 34, 629–638. [Google Scholar] [CrossRef]
- Albalat, A.; Johnson, L.; Coates, C.J.; Dykes, G.C.; Hitte, F.; Morro, B.; Dick, J.; Todd, K.; Neil, D.M. The Effect of Temperature on the Physiological Condition and Immune-Capacity of European Lobsters (Homarus Gammarus) During Long-Term Starvation. Front. Mar. Sci. 2019, 6, 281. [Google Scholar] [CrossRef]
- Nguyen, T.; Alfaro, A.C.; Rodríguez, J.; Bayot, B.; Sonnenholzner, S. Changes in Metabolic Profiling of Whiteleg Shrimp (Penaeus Vannamei) under Hypoxic Stress. J. Invertebr. Pathol. 2022, 193, 107798. [Google Scholar] [CrossRef] [PubMed]
- Moberg, G.P. Biological Response to Stress: Key to Assessment of Animal Well-Being? In Animal Stress; Moberg, G.P., Ed.; Springer: New York, NY, USA, 1985; pp. 27–49. [Google Scholar]
- Muhammad, F.; Zhang, Z.F.; Shao, M.Y.; Dong, Y.P.; Muhammad, S.; Balouch, W.A. Early Development of Nervous System in Litopenaeus Vannamei (Boone, 1931) (Crustacea: Decapoda) Larval Nervous System, Genesis, Differentiation. Sindh. Univ. Res. J. (Sci. Ser.) 2012, 44, 29–34. [Google Scholar]
- Birch, J. Animal Sentience and the Precautionary Principle. Anim. Sentience 2017, 16. [Google Scholar] [CrossRef]
- Chong-Robles, J.; Charmantier, G.; Boulo, V.; Lizárraga-Valdéz, J.; Enríquez-Paredes, L.M.; Giffard-Mena, I. Osmoregulation Pattern and Salinity Tolerance of the White Shrimp Litopenaeus Vannamei (Boone, 1931) during Post-Embryonic Development. Aquaculture 2014, 422–423, 261–267. [Google Scholar] [CrossRef]
- Medina-Reyna, C.E. Growth and Emigration of White Shrimp, Litopenaeus Vannamei, in the Mar Muerto Lagoon, Southern Mexico. Naga Iclarm Q. 2001, 24, 30–34. [Google Scholar]
- McKenney, C.L.; Celestial, D.M. Interactions among Salinity, Temperature, and Age on Growth of the Estuarine Mysid Mysidopsis Bahia Reared in the Laboratory through a Complete Life Cycle. I.Body Mass and Age-Specific Growth Rate. J. Crustac. Biol. 1995, 15, 169–178. [Google Scholar] [CrossRef]
- O’brien, C.J. The effects of temperature and salinity on growth and survival of juvenile tiger prawns Penaeus esculentus (Haswell). J. Exp. Mar. Biol. Ecol. 1994, 183, 133–145. [Google Scholar] [CrossRef]
- Boyd, C.E.; Pillai, V.K. Water Quality Management in Aquaculture; CMFRI Special Publication, 22; Central Marine Fisheries Research Institute: Cochin, India, 1985; p. 71. [Google Scholar]
- Rosas, C.; López, N.; Mercado, P.; Martínez, E. Effect of Salinity Acclimation on Oxygen Consumption of Juveniles of the White Shrimp Litopenaeus Vannamei. J. Crustac. Biol. 2001, 21, 912–922. [Google Scholar] [CrossRef]
- Zhang, P.; Zhang, X.; Li, J.; Gao, T. Effect of salinity on survival, growth, oxygen consumption and ammonia-N excretion of juvenile whiteleg shrimp, Litopenaeus vannamei. Aquac. Res. 2009, 40, 1419–1427. [Google Scholar] [CrossRef]
- Kautsky, N.; Rönnbäck, P.; Tedengren, M.; Troell, M. Ecosystem Perspectives on Management of Disease in Shrimp Pond Farming. Aquaculture 2000, 191, 145–161. [Google Scholar] [CrossRef]
- Moss, K.R.K.; Moss, S.M. Effects of Artificial Substrate and Stocking Density on the Nursery Production of Pacific White Shrimp Litopenaeus Vannamei. J. World Aquac. Soc. 2004, 35, 536–542. [Google Scholar] [CrossRef]
- Krummenauer, D.; Peixoto, S.; Cavalli, R.O.; Poersch, L.H.; Wasielesky, W. Superintensive Culture of White Shrimp, Litopenaeus Vannamei, in a Biofloc Technology System in Southern Brazil at Different Stocking Densities. J. World Aquac. Soc. 2011, 42. [Google Scholar] [CrossRef]
- Samocha, T.M.; Hamper, L.; Emberson, C.R.; Davis, A.D.; McIntosh, D.; Lawrence, A.L.; van Wyk, P.M. Review of Some Recent Developments in Sustainable Shrimp Farming Practices in Texas, Arizona, and Florida. J. Appl. Aquac. 2002, 12, 1–42. [Google Scholar] [CrossRef]
- Liu, G.; Zhu, S.; Liu, D.; Guo, X.; Ye, Z. Effects of Stocking Density of the White Shrimp Litopenaeus Vannamei (Boone) on Immunities, Antioxidant Status, and Resistance against Vibrio Harveyi in a Biofloc System. Fish Shellfish. Immunol. 2017, 67, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Lightner, D.; Redman, R.M. Shrimp Diseases and Current Diagnostic Methods. Aquaculture 1998, 164, 201–220. [Google Scholar] [CrossRef]
- Corsin, F.; Mohan, C.V. Towards Responsible Shrimp Farming. In The Shrimp Book; Aldey-Sanz, V., Ed.; Nottingham University Press: Nottingham, UK, 2010; pp. 353–375. [Google Scholar]
- O’Donncha, F.; Stockwell, C.L.; Planellas, S.R.; Micallef, G.; Palmes, P.; Webb, C.; Filgueira, R.; Grant, J. Data Driven Insight Into Fish Behaviour and Their Use for Precision Aquaculture. Front. Anim. Sci. 2021, 2, 30. [Google Scholar] [CrossRef]
- Antonucci, F. Corrado Costa Precision Aquaculture: A Short Review on Engineering Innovations. Aquac. Int. 2020, 28, 41–57. [Google Scholar] [CrossRef]
- de Avila, L.R.; da Costa Botelho, S.S.; Pias, M.R.; de Vargas Guterres, B.; Junior, J.N.J. On Aquaculture Enhancements Through Robotic Behaviour Actuation. In Proceedings of the 2021 Latin American Robotics Symposium (LARS), 2021 Brazilian Symposium on Robotics (SBR), and 2021 Workshop on Robotics in Education (WRE), Natal, Brazil, 11–15 October 2021; pp. 294–299. [Google Scholar] [CrossRef]
- Hung, C.C.; Tsao, S.C.; Huang, K.H.; Jang, J.P.; Chang, H.K.; Dobbs, F.C. A Highly Sensitive Underwater Video System for Use in Turbid Aquaculture Ponds. Sci. Rep. 2016, 6, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Sreepada, R.A.; Kuzkamil, S.; Suryavanshi2, U.; Ingole, B.S.; Drensgstigl, A.; Braa, B. Water Quality Management in Shrimp Aquaculture Ponds Using Remote Water Quality Logging System; School of Industrial Fisheries, CUSAT: Kerala, India, 2006. [Google Scholar]
- Simbeye, D.S.; Zhao, J.; Yang, S. Design and Deployment of Wireless Sensor Networks for Aquaculture Monitoring and Control Based on Virtual Instruments. Comput. Electron. Agric. 2014, 102, 31–42. [Google Scholar] [CrossRef]
- Gusmawati, N.; Soulard, B.; Selmaoui-Folcher, N.; Proisy, C.; Mustafa, A.; le Gendre, R.; Laugier, T.; Lemonnier, H. Surveying Shrimp Aquaculture Pond Activity Using Multitemporal VHSR Satellite Images–Case Study from the Perancak Estuary, Bali, Indonesia. Mar. Pollut. Bull. 2018, 131, 49–60. [Google Scholar] [CrossRef] [PubMed]
- Irawan, Y.; Fonda, H.; Sabna, E.; Febriani, A. Intelligent Quality Control of Shrimp Aquaculture Based On Real-Time System and IoT Using Mobile Device. Int. J. Eng. Trends Tech. 2021, 69, 49–56. [Google Scholar] [CrossRef]
- Capelo, J.; Ruiz, E.; Asanza, V.; Toscano-Quiroga, T.; Sánchez-Pozo, N.N.; Lorente-Leyva, L.L.; Peluffo-Ordóñez, D.H. Raspberry Pi-Based IoT for Shrimp Farms Real-Time Remote Monitoring with Automated System. In Proceedings of the International Conference on Applied Electronics, University of West Bohemia, Pilsen, Czech Republic, 7 September 2021; IEEE Computer Society: Pilsen, Czech Republic, 2021; pp. 1–4. ISBN 978-80-261-0973-0. [Google Scholar]
| Domain | Specific Indicators |
|---|---|
| Environmental | water quality; temperature; pH; transparency; dissolved oxygen; ammonia; nitrite; salinity; alkalinity; predators; competitors; photoperiod; stocking density |
| Health | health; diseases; eyes; exoskeleton; gills; hepatopancreas; antennae; rostrum; mortality |
| Nutritional | protein; feeding frequency; feed amount; food distribution |
| Behavioural | behaviour; respiratory frequency; swimming; feed intake; anaesthesia; handling; slaughter |
| Indicators | Score | Reference Values | References |
|---|---|---|---|
| Temperature (°C) | 1 | 24.5–32.5 | [73,74,75,76,77,78,79,80] |
| 2 | 15.6–24.4 or 32.6–35.4 | ||
| 3 | ≤15.5 or ≥35.5 | ||
| pH | 1 | 7.5–8.5 | [75,76,77,79,81,82,83,84] |
| 2 | 5.0–7.4 or 8.6–9.0 | ||
| 3 | ≤4.9 or ≥9.1 | ||
| Photoperiod (Light: Dark) | 1 | Natural or 12L:12D–14L:10D | [75,76,79,80,85,86] |
| 2 | 15L:9D–16L:8D | ||
| 3 | 17L:7D or clearer; 11L:13D or darker | ||
| Alkalinity (mg/L CaCO3) | 1 | 100–140 | [81,83,87,88,89] |
| 2 | 51–99 or 141–199 | ||
| 3 | ≤50 or ≥200 | ||
| Dissolved oxygen (% saturation) | 1 | ≥62 | [80,81,83,87,88,90,91] |
| 2 | 46–61 | ||
| 3 | ≤45 | ||
| Non-ionised ammonia (mg/L NH3) | 1 | 0.00–0.10 | [81,87,88,89,92,93] |
| 2 | 0.11–0.30 | ||
| 3 | ≥0.31 | ||
| Nitrite (mg/L NO2−) | 1 | 0–0.6 | [81,83,87,88,89,92,94,95] |
| 2 | 0.7–1.6 | ||
| 3 | ≥1.7 | ||
| Salinity (psu) | 1 | 28–35 | [75,76,78,79,84,96,97] |
| 2 | 10–27 or 36–40 | ||
| 3 | ≤9 or ≥41 | ||
| Stocking density (g/m2) | 1 | ≤150 | [76,79,98,99,100] |
| 2 | 151–299 | ||
| 3 | ≥300 | ||
| Terrestrial predators * | 1 | Absence | [101,102] |
| 2 | Controlled presence | ||
| 3 | Uncontrolled presence | ||
| Aquatic predators and interspecific inhabitants ** | 1 | Absence | [102,103,104] |
| 2 | Controlled presence | ||
| 3 | Uncontrolled presence |
| Indicators | Score | Description or Reference Values | References |
|---|---|---|---|
| Antennae | 1 | Healthy appearance, no changes | [105,106,107] |
| 2 | Focal lesion, shortening or darkening | ||
| 3 | Absence, blueness, wrinkling, multifocal dark spots | ||
| Rostrum | 1 | Healthy appearance, no changes | [107,108,109] |
| 2 | Mild injury, erosion or necrosis | ||
| 3 | Severe injury, erosion or necrosis, deformity, bending to one side, upwards or downwards | ||
| Eyes | 1 | Healthy appearance, no changes | [108,110,111] |
| 2 | Unilateral lightening, injury, softening or swelling | ||
| 3 | Bilateral lightening, injury, softening or swelling, absence of one or both organs | ||
| Gills | 1 | Healthy appearance, no changes | [112,113,114] |
| 2 | Focal lesion or darkening | ||
| 3 | Pale, yellowish, general redness, darkening, whitish spots, or erosion | ||
| Hepatopancreas | 1 | Healthy appearance, no changes | [107,112,115,116] |
| 2 | Discrete volume reduction | ||
| 3 | Atrophy, stiffness, flaccidity, colour change (abnormal pallor or darkening, appearance of streaks of different shades), volume change, tearing, presence of worm-like structures, presence of fluid (oedema) | ||
| Motor appendages (pereiopods/uropods/pleopods) | 1 | Healthy appearance, no changes | [112,115,117] |
| 2 | Focal absence or erosion | ||
| 3 | Severe or complete absence, colour change, necrosis (lightening), dark spots, rough and darkened edges | ||
| Exoskeleton (cuticle) | 1 | Healthy appearance, no changes | [88,112,115] |
| 2 | Slight lesion or focal darkening, presence of debris | ||
| 3 | Tissue loss, necrosis, focal or generalised colour change, deformity, calcified round white patches, crusting | ||
| Musculature | 1 | Healthy appearance, no changes | [105,108,117] |
| 2 | Focal necrosis (lightening) | ||
| 3 | Generalised necrosis, colour change (yellowing, redness, opacity, milky appearance), atrophy, swelling, stiffness (constant bending), zigzag shape | ||
| Gastrointestinal tract | 1 | Healthy appearance, no changes | [88,108,118] |
| 2 | Presence of sand particles or debris | ||
| 3 | Atrophy, empty, pale, whitish bowel, whitish stools, faecal streaks | ||
| Luminescence | 1 | No luminescence is observed in breeders in complete darkness | [79] |
| 2 | Not applicable | ||
| 3 | Luminescence is observed in breeders in absolute darkness | ||
| Sexual maturation | 1 | Females: Enlarged ovaries, distinct olive-green colour (stage IV). Males: Shiny sperm ampullae | [75,81] |
| 2 | Not applicable | ||
| 3 | Females: Small, translucent ovaries (stage I); opaque and yellowish (stage II); bright yellow (stage III); spawned (stage V). Males: opaque or brownish (necrotic or melanised) sperm ampullae | ||
| Invasive procedures (ablation of the eyestalk, extrusion of the spermatophore) * | 1 | No invasive intervention | [75,119,120,121] |
| 2 | Invasive procedure with effective anaesthesia and postoperative analgesia | ||
| 3 | Invasive procedure without effective anaesthesia and postoperative analgesia | ||
| Mortality during breeding and spawning (%) ** | 1 | ≤10 | [70,122,123] |
| 2 | 11–25 | ||
| 3 | ≥26 | ||
| Genetic selection | 1 | Genetic selection and or improvement programme with inbreeding control | [124] |
| 2 | Genetic selection and or improvement programme without inbreeding control | ||
| 3 | No genetic improvement programme |
| Indicators | Score | Reference Values | References |
|---|---|---|---|
| Filling of the digestive tract * | 1 | Full | [125] |
| 2 | Medium | ||
| 3 | Empty | ||
| Frequency of feeding (times/day) | 1 | ≥3 | [87,126,127,128,129,130,131,132] |
| 2 | 2 | ||
| 3 | ≤1 | ||
| Composition/type of diet | 1 | Special artificial feed for breeders + fresh feed | [133,134,135] |
| 2 | Special artificial feed for breeders only | ||
| 3 | Fresh feed only | ||
| Amount of food (% of biomass **) | 1 | ≥4.0 | [75,136,137,138,139,140] |
| 2 | 2.4–3.9 | ||
| 3 | ≤2.3 | ||
| Crude protein in the breeders’ artificial diet (%) | 1 | ≥35 | [128,137,140,141,142,143] |
| 2 | 29–34 | ||
| 3 | ≤28 |
| Management | Indicators | Score | Description | References |
|---|---|---|---|---|
| Routine management | Swimming behaviour | 1 | Animals at the bottom that stop, walk or feed, or normally swim in the water column | [91,144,145] |
| 2 | Few animals with irregular swimming or with partial loss of balance at the tank bottom | |||
| 3 | Many animals with irregular swimming (off balance or in a spiral), crowding of shrimp in certain parts of the tank | |||
| Feeding behaviour | Reaction to offered food | 1 | Most animals react quickly by moving to the newly offered food | Authors’ suggestion |
| 2 | Only some of the animals react to the newly offered food | |||
| 3 | Most animals do not react to the newly offered food | |||
| Invasive methods | Anaesthesia—surgical phase * (loss of balance and response to external stimuli) | 1 | Induction and recovery in 3–5 min | [146,147,148] |
| 2 | Induction and or recovery in 6–9 min | |||
| 3 | Absence of anaesthetic procedures Induction and or recovery ≤ 2 min or ≥10 min; death |
| Indicators | Score | Reference Values | References |
|---|---|---|---|
| Temperature (°C) | 1 | 26.5–32.4 | [79,99,149,150,151,152,153] |
| 2 | 19.5–26.4 or 32.5–35.4 | ||
| 3 | ≤19.4 or ≥35.5 | ||
| pH | 1 | 7.5–8.5 | [79,99,149,151] |
| 2 | 5.0–7.4 or 8.6–9.0 | ||
| 3 | ≤4.9 or ≥9.1 | ||
| Photoperiod (Light:Dark) | 1 | 18L:6D–24L:0D | [86,154,155] |
| 2 | 17L:7D–12L:12D | ||
| 3 | 11L:13D or darker | ||
| Alkalinity (mg/L as CaC O3) | 1 | 100–140 | [81,83,87,88,89] |
| 2 | 51–99 or 141–199 | ||
| 3 | ≤50 or ≥200 | ||
| Dissolved oxygen (% saturation) | 1 | ≥64 | [79,99] |
| 2 | 49–63 | ||
| 3 | ≤48 | ||
| Non-ionised ammonia (mg/L NH3) | 1 | 0.00–0.01 | [99,156,157,158] |
| 2 | 0.02–0.06 | ||
| 3 | ≥0.07 | ||
| Nitrite (mg/L NO2−) | 1 | 0.0–0.1 | [99,157,159] |
| 2 | 0.2–0.7 | ||
| 3 | ≥0.8 | ||
| Salinity (psu) | 1 | 30–36 | [79,99,149,150,151,152] |
| 2 | 20–29 or 37–44 | ||
| 3 | ≤19 or ≥45 | ||
| Stocking density (Larvae or postlarvae/L) | 1 | ≤250 (larvae) ≤100 (postlarvae) | [79,160,161] |
| 2 | 251–300 (larvae) 101–210 (postlarvae) | ||
| 3 | ≥301 (larvae) ≥211 (postlarvae) |
| Indicators | Scores | Reference Values or Description | References |
|---|---|---|---|
| Health certificate | 1 | SPF 1 + SPT 2 and or SPR 3 | [79,88] |
| 2 | SPF 1 | ||
| 3 | Does not have a health certificate | ||
| Luminescence | 1 | No luminescence is observed in the larvae tank under absolute darkness | [79] |
| 2 | Not applicable | ||
| 3 | luminescence is observed in the larvae tank under absolute darkness | ||
| Uniformity of larval stages in a tank * | 1 | ≥75% of the sampled larvae are in the same larval stage | [79,88] |
| 2 | 50–74% of the sampled larvae are in the same larval stage | ||
| 3 | ≤49% of the sampled larvae are in the same larval stage | ||
| Malformations (%) | 1 | Low (≤5% of the sampled larvae have deformities) | [79,88,152] |
| 2 | Moderate (6–10% of the sampled larvae have deformities) | ||
| 3 | Severe (≥11% of the sampled larvae have deformities) | ||
| Staining of the hepatopancreas * | 1 | ≥90% of larvae have darkened hepatopancreas | [79,152,162] |
| 2 | 70–89% of larvae have darkened hepatopancreas | ||
| 3 | ≤69% of larvae have darkened hepatopancreas | ||
| Condition of the hepatopancreas * | 1 | ≥90% of sampled larvae have abundant lipid vacuoles | [79,152,162] |
| 2 | 70–89% of sampled larvae have abundant lipid vacuoles | ||
| 3 | ≤69% of the sampled larvae have abundant lipid vacuoles | ||
| Epibiont encrustation on the exoskeleton, appendages and gills * | 1 | ≤5% of sampled larvae have fouling | [79,152] |
| 2 | 6–10% of sampled larvae have encrustations | ||
| 3 | ≥11% of sampled larvae suffer from fouling | ||
| Necrosis * (%) | 1 | Absence of necrosis in larvae | [79,88] |
| 2 | ≤15% of larvae show opacities of the muscles or limbs | ||
| 3 | ≥16% of larvae show necrosis in muscles or limbs | ||
| Melanisation * (%) | 1 | ≥90% of larvae show up to 5% melanisation | [79] |
| 2 | 70–89% of larvae show up to 5% melanisation | ||
| 3 | ≤69% of larvae show up to 5% melanisation | ||
| Mortality at the end of larval rearing ** | 1 | ≤30 | [79,163,164,165] |
| 2 | 31–49 | ||
| 3 | ≥50 |
| Indicators | Score | Reference Values | References | ||
|---|---|---|---|---|---|
| 0.002–0.08 g | 0.081–1.0 g | 1.1–2.5 g | |||
| Size of the food (µm) | 1 | Fed mash-700 | 701–1800 | 1801 µm –3.2 mm | [125,148,166] |
| 2 | ≥701 | ≤701 | ≤1801 | ||
| 3 | Not applicable | ≥1801 | ≥3.3 | ||
| Crude protein in the artificial feed (%) | 1 | ≥25 | ≥25 | ≥40 | [140,167,168,169,170] |
| 2 | 20–24 | 20–24 | 25–40 | ||
| 3 | ≤19 | ≤19 | ≤24 | ||
| Analysis of PL gastrointestinal tract (% of sampled animals with complete tract) | 1 | Does not apply | Does not apply | ≥90 | [79] |
| 2 | 70–89 | ||||
| 3 | ≤69 | ||||
| Frequency of feeding—artificial feed (times/day) | 1 | ≥6 | [87,171,172] | ||
| 2 | 3–5 | ||||
| 3 | ≤2 | ||||
| Composition of the diet (type of food) | 1 | Specific artificial food for each larval stage + live food | [173,174,175,176,177] | ||
| 2 | Only specific artificial food for each larval stage | ||||
| 3 | Only live food | ||||
| Indicators | Score | Description or Reference Values | References |
|---|---|---|---|
| Phototaxis of nauplii and zoea | 1 | ≥95% of sampled larvae are attracted to the light from the water surface | [79,152] |
| 2 | 70–94% of sampled larvae are attracted to the light from the water surface | ||
| 3 | ≤69% of sampled larvae are attracted to the light from the water surface | ||
| Swimming behaviour of larvae and postlarvae | 1 | ≥95% of the sampled larvae are active | [79,152,162] |
| 2 | 70–94% of the sampled larvae are active | ||
| 3 | ≤69% of the sampled larvae are active |
| Category | Indicators | Score | Description or Reference Values | References |
|---|---|---|---|---|
| Environmental | Temperature (°C) | 1 | 22.5–25.4 | [81,88,149,178,179] |
| 2 | 19.5–22.4 or 25.5–29.5 | |||
| 3 | ≤19.4 or ≥29.6 | |||
| pH | 1 | 6.5–8.0 | [178] | |
| 2 | 5.0–6.4 or 8.1–8.5 | |||
| 3 | ≤4.9 or ≥8.6 | |||
| Salinity (psu) | 1 | Acclimatised to the salinity of the farm | [79,99,149,150,151,152] | |
| 2 | Need to acclimatise for up to 5 ups | |||
| 3 | Need to acclimatise for more than 5 ups | |||
| Dissolved oxygen (% saturation) | 1 | ≥80 | [81] | |
| 2 | 60–79 | |||
| 3 | ≤59 | |||
| Transport density (postlarvae/L) * | 1 | ≤750 | [81,179,180,181] | |
| 2 | 751–1000 | |||
| 3 | ≥1001 | |||
| Non-ionised ammonia (mg/L NH3) | 1 | 0.00–0.01 | [99,156,157,158] | |
| 2 | 0.02–0.06 | |||
| 3 | ≥0.07 | |||
| Health | Lesions, necrosis and/or malformations (rostrum, appendages and or gills) (%) | 1 | ≤5 | [182] |
| 2 | 6–10 | |||
| 3 | ≥11 | |||
| Mortality (%) | 1 | ≤5 | [178,180] | |
| 2 | 6–10 | |||
| 3 | ≥11 | |||
| Nutritional | Feeding (time interval) ** | 1 | ≤3 h during transport | [88,149,179,183,184] |
| 2 | ≥4 h during transport | |||
| 3 | No feeding during transport | |||
| Type of food added | 1 | Artemia nauplii + industrial feed | [179,183] | |
| 2 | Only industrial feed or artemia nauplii | |||
| 3 | No additional feed during transport | |||
| Behavioural | Swimming behaviour after transport (no anaesthesia) *** | 1 | ≥95% of active postlarvae and normal swimming ability | [79,152,162,185,186] |
| 2 | 70–94% of active postlarvae and normal swimming ability | |||
| 3 | ≤69% of active postlarvae and normal swimming ability | |||
| Swimming behaviour after transport (with anaesthesia) | 1 | Sedation (loss of balance with reaction to external stimuli) | [146,187] | |
| 2 | Anaesthesia (complete loss of equilibrium with no response to external stimuli) | |||
| 3 | No induction or recovery, death |
| Indicators | Score | Reference Values | References |
|---|---|---|---|
| Temperature (°C) | 1 | 25.5–32.4 | [82,88,188,189,190,191,192,193,194] |
| 2 | 14.5–25.4 or 32.5–35.4 | ||
| 3 | ≤14.4 or ≥35.5 | ||
| pH | 1 | 6.5–8.5 | [81,82,83,88,195,196] |
| 2 | 5.0–6.4 or 8.6–9.0 | ||
| 3 | ≤4.9 or ≥9.1 | ||
| Transparency (cm) | 1 | 35–50 | [81,87,88,89,90] |
| 2 | 21–34 or 51–59 | ||
| 3 | ≤20 or ≥60 | ||
| Alkalinity (mg/L CaCO3) | 1 | 100–140 | [81,83,87,88,89] |
| 2 | 51–99 or 141–199 | ||
| 3 | ≤50 or ≥200 | ||
| Non-ionised ammonia (mg/L de NH3) | 1 | 0.00–0.10 | [81,87,88,89,92,93,197] |
| 2 | 0.11–0.30 | ||
| 3 | ≥0.31 | ||
| Dissolved oxygen (% saturation) | 1 | ≥65 | [81,83,87,88,90,91] |
| 2 | 49–64 | ||
| 3 | ≤48 | ||
| Nitrite (mg/L NO2) | 1 | 0.0–0.6 | [81,83,87,88,89,92,94,95] |
| 2 | 0.7–1.6 | ||
| 3 | ≥1.7 | ||
| Salinity (psu) | 1 | 10.0–40.9 | [81,87,88,91,192,194,198] |
| 2 | 0.6–9.9 or 41.0–59.0 | ||
| 3 | ≤0.5 or ≥60.0 | ||
| Stocking density (shrimp/m2) | 1 | ≤40 | [199,200,201,202,203,204,205] |
| 2 | 41–60 | ||
| 3 | ≥61 | ||
| Terrestrial predators * | 1 | Absence | [101,102] |
| 2 | Controlled presence | ||
| 3 | Uncontrolled presence | ||
| Aquatic predators and interspecific inhabitants ** | 1 | Absence | [102,103,104] |
| 2 | Controlled presence | ||
| 3 | Uncontrolled presence |
| Indicators | Score | Description or Reference Values | References |
|---|---|---|---|
| Antennae | 1 | Healthy appearance, no changes | [105,106,107] |
| 2 | A focal lesion, shortening, or darkening | ||
| 3 | Absence, blueness, wrinkling, multifocal dark spots | ||
| Rostrum | 1 | Healthy appearance, no changes | [107,108,109] |
| 2 | Mild injury, erosion, or necrosis | ||
| 3 | Severe injury, erosion or necrosis, deformity, bending to one side, upwards or downwards | ||
| Eyes | 1 | Healthy appearance, no changes | [108,110,111] |
| 2 | Unilateral lightening, injury, softening or swelling | ||
| 3 | Bilateral lightening, injury, softening or swelling, absence of one or both organs | ||
| Gills | 1 | Healthy appearance, no changes | [112,113,114] |
| 2 | Focal lesion or darkening | ||
| 3 | Pale, yellowish, general redness or darkening, whitish spots, erosion | ||
| Hepatopancreas | 1 | Healthy appearance, no changes | [107,112,115,116] |
| 2 | Discrete volume reduction | ||
| 3 | Atrophy, stiffness, flaccidity, colour change (abnormal pallor or darkening, appearance of streaks of different shades), volume change, tearing, presence of worm-like structures, presence of fluid (oedema) | ||
| Motor appendages (pereiopods/uropods /pleopods/telson) | 1 | Healthy appearance, no changes | [112,115,117] |
| 2 | Focal absence or erosions | ||
| 3 | Severe or complete absence, colour change, necrosis (lightening), dark spots, rough and darkened edges | ||
| Exoskeleton (cuticle) | 1 | Healthy appearance, no changes | [88,112,115] |
| 2 | Slight lesion or focal darkening, presence of debris | ||
| 3 | Tissue loss, necrosis, focal or generalised colour change, deformity, calcified round white spots, encrustation | ||
| Musculature | 1 | Healthy appearance, no changes | [105,108,117] |
| 2 | Focal necrosis (lightening) | ||
| 3 | Generalised necrosis, colour change (yellowing, redness, opacity, milky appearance), atrophy, swelling, stiffness (constant bending), zigzag shape | ||
| Mortality (%) * | 1 | ≤10 | [70,122,123] |
| 2 | 11–25 | ||
| 3 | ≥26 |
| Indicators | Score | Weight (g) | References | ||||
|---|---|---|---|---|---|---|---|
| ≤0.9 | 1.0–3.9 | 4.0–8.9 | 9.0–15.0 | ||||
| Size of food (mm) | 1 | 0.1–0.5 | 0.6–1.0 | 1.1–2.0 | 2.1–3.0 | [149,206] | |
| 2 | ≥0.6 | ≥1.1 | ≥2.1 | ≥3.1 | |||
| 3 | <0.1 | ≤0.5 | ≤1.0 | ≤2.0 | |||
| Amount of initial food (% of biomass) | 1 | 6.0–10.9 | 4.0–6.9 | 4.0–6.9 | 2.0–3.9 | [75,87,126,206,207] | |
| 2 | 4.1–5.9 | 2.1–3.9 | 2.1–3.9 | 1.1–1.9 | |||
| 3 | ≤4.0 or ≥11.0 | ≤2.0 or ≥7.0 | ≤2.0 or ≥ 7.0 | ≤1.0 or ≥4.0 | |||
| Frequency of feeding in the ponds (times/day) | 1 | ≥4 | ≥2 | ≥2 | ≥2 | [87,126,127,128,129,130,131,132,208] | |
| 2 | 2–3 | 1 | 1 | 1 | |||
| 3 | ≤1 | <1 | <1 | <1 | |||
| Feed crude protein (%) | 1 | ≥35 | ≥35 | ≥32 | ≥32 | [87,126,127,128,140,142,206] | |
| 2 | 32–34 | 32–34 | 25–31 | 25–31 | |||
| 3 | ≤31 | ≤31 | ≤24 | ≤24 | |||
| Apparent feed conversion rate (FCR) * | 1 | Does not apply | Does not apply | ≤1.5 | ≤1.7 | [89,126,209,210,211,212,213] | |
| 2 | 1.6–2.0 | 1.8–2.0 | |||||
| 3 | ≥2.1 | ≥2.1 | |||||
| Feed distribution | Distribution of feed over the surface (% of pond surface) OR | 1 | ≥75 | [87,127,207,209,214,215,216] | |||
| 2 | 50–74 | ||||||
| 3 | ≤49 or no control of coverage area | ||||||
| Use of trays (number of trays/ha) | 1 | ≥20 | |||||
| 2 | 15–19 | ||||||
| 3 | ≤14 | ||||||
| Digestive tract filling index ** | 1 | Full | [217] | ||||
| 2 | Medium | ||||||
| 3 | Empty | ||||||
| Management | Indicators | Score | Reference Values | References |
|---|---|---|---|---|
| Routine management | Swimming behaviour | 1 | No shrimp on the pond surface or irregular swimming | [91,144,145] |
| 2 | Few animals on the pond surface or irregular swimming | |||
| 3 | Reduced, irregular or “spiral” swimming, accumulation of shrimp at the edges of the pond or near the water inlet, many animals exposing their bodies at the water surface | |||
| Partial or complete harvesting | Escape behaviour (successive tail movements by flexion and extension of the abdomen) | 1 | Few jumping shrimps during harvest, with low frequency and intensity | [218] |
| 2 | Few jumping shrimps, but with high frequency and/or intensity during harvesting | |||
| 3 | Many jumping shrimps, high frequency and/or intensity during harvesting | |||
| Stunning at slaughter * | Clinical reflexes | 1 | Immediate loss of response to external stimuli; balance (with cephalothorax in horizontal and descending position); movement of pleopods and pereiopods; and movement of scaphognathites | [146,147,186,219,220,221] |
| 2 | Progressive loss of response to external stimuli; balance (with cephalothorax in horizontal and descending position); movement of pleopods and pereiopods; and movement of scaphognathites in ≤30 s | |||
| 3 | Progressive loss of: Response to external stimuli; balance (with cephalothorax in horizontal and descending position); movement of pleopods and pereiopods; and movement of scaphognathites in >30 s |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pedrazzani, A.S.; Cozer, N.; Quintiliano, M.H.; Tavares, C.P.d.S.; da Silva, U.d.A.T.; Ostrensky, A. Non-Invasive Methods for Assessing the Welfare of Farmed White-Leg Shrimp (Penaeus vannamei). Animals 2023, 13, 807. https://doi.org/10.3390/ani13050807
Pedrazzani AS, Cozer N, Quintiliano MH, Tavares CPdS, da Silva UdAT, Ostrensky A. Non-Invasive Methods for Assessing the Welfare of Farmed White-Leg Shrimp (Penaeus vannamei). Animals. 2023; 13(5):807. https://doi.org/10.3390/ani13050807
Chicago/Turabian StylePedrazzani, Ana Silvia, Nathieli Cozer, Murilo Henrique Quintiliano, Camila Prestes dos Santos Tavares, Ubiratã de Assis Teixeira da Silva, and Antonio Ostrensky. 2023. "Non-Invasive Methods for Assessing the Welfare of Farmed White-Leg Shrimp (Penaeus vannamei)" Animals 13, no. 5: 807. https://doi.org/10.3390/ani13050807
APA StylePedrazzani, A. S., Cozer, N., Quintiliano, M. H., Tavares, C. P. d. S., da Silva, U. d. A. T., & Ostrensky, A. (2023). Non-Invasive Methods for Assessing the Welfare of Farmed White-Leg Shrimp (Penaeus vannamei). Animals, 13(5), 807. https://doi.org/10.3390/ani13050807





