Defining Short-Term Accommodation for Animals
Abstract
Simple Summary
Abstract
1. Introduction
Terminology
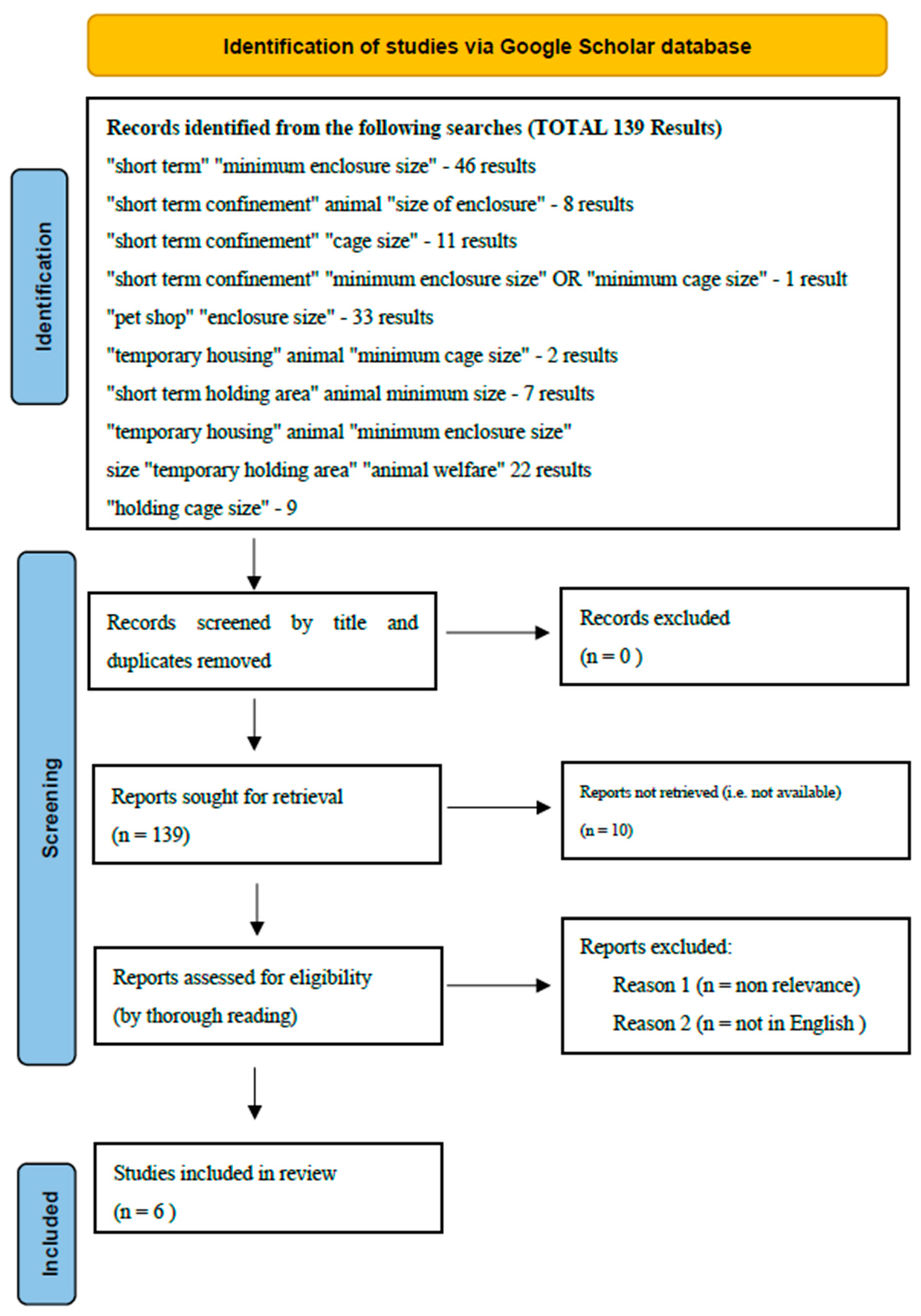
2. Methods
3. Results
4. Discussion
4.1. The Acute Stress-to-Chronic Stress Transition Paradigm and its Relevance to Short-Term Housing
4.2. Managing Stressors and Stress
5. Conclusions
6. Recommendations
- The stipulations for short-term, temporary, transitional, or other similarly intended conditions should infer periods of less than a single circadian cycle (typically <24 h).
- All animals at all facilities should be subject to the single circadian cycle as a principle for determining maximum short-term, temporary, or other transitional conditions.
- All animals at all facilities must be accommodated in higher or other similarly recognised conditions consistent with long-term husbandry and best practices wherever confinement persists beyond the single circadian principle.
- Best practice examples of short-term, temporary, or other transitional conditions should include higher standards of husbandry.
- Keeping animals in short-term, lower standard conditions should be minimised and only for recorded and essential reasons.
- All animals at all facilities should be subject to government mandatory identification and registration on arrival and departure in order to accurately record their period of stay.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- French, F.; Williams, L.; Coe, J.C.; Single, L.; Fernandez, E.; Martin, C.F. ReptileJam 2021: Designing Enrichment for Reptiles in Captivity: This is a proposal for a ZooJam workshop around the theme of designing tech-enabled enrichment opportunities for zoo-housed and domestic reptiles. In Proceedings of the Eight International Conference on Animal-Computer Interaction (ACI’21), New York, NY, USA, 8–11 November 2021. [Google Scholar]
- Gygax, L.; Hillmann, E. “Naturalness” and its relation to animal welfare from an ethological perspective. Agriculture 2018, 8, 136. [Google Scholar] [CrossRef]
- Marliani, G.; Sprocatti, I.; Schiavoni, G.; Bellodi, A.; Accorsi, P.A. Evaluation of Horses’ Daytime Activity Budget in a Model of Ethological Stable: A Case Study in Italy. J. Appl. Anim. Welf. Sci. 2021, 24, 200–213. [Google Scholar] [CrossRef]
- Ryan, M.; Waters, R.; Wolfensohn, S. Assessment of the Welfare of Experimental Cattle and Pigs Using the Animal Welfare Assessment Grid. Animals 2021, 11, 999. [Google Scholar] [CrossRef]
- Sánchez-Casanova, R.; Sarmiento-Franco, L.; Phillips, C.; Zulkifli, I. Do free-range systems have potential to improve broiler welfare in the tropics? World’s Poult. Sci. J. 2020, 76, 34–48. [Google Scholar] [CrossRef]
- Zhuchaev, K.; Orlov, D.; Borisenko, E.; Kochneva, M.; Hart, V. Ethological evaluation of pig welfare under different keeping technologies in Siberian conditions. BIO Web Conf. 2021, 36, 06038. [Google Scholar] [CrossRef]
- Burghardt, G.M. Environmental enrichment and cognitive complexity in reptiles and amphibians: Concepts, review, and implications for captive populations. Appl. Anim. Behav. Sci. 2013, 147, 286–298. [Google Scholar] [CrossRef]
- King, L.A. Behavioral evaluation of the psychological welfare and environmental requirements of agricultural research animals: Theory, measurement, ethics, and practical implications. ILAR J. 2003, 44, 211–221. [Google Scholar] [CrossRef]
- Baumans, V. Science-based assessment of animal welfare: Laboratory animals. Rev. Sci. Tech. 2005, 24, 503. [Google Scholar] [CrossRef]
- Duncan, I.J. Science-based assessment of animal welfare: Farm animals. Rev. Sci. Tech. 2005, 24, 483–492. [Google Scholar] [CrossRef]
- Whitham, J.C.; Wielebnowski, N. Animal-based welfare monitoring: Using keeper ratings as an assessment tool. Zoo Biol. Publ. Affil. Am. Zoo Aquar. Assoc. 2009, 28, 545–560. [Google Scholar] [CrossRef]
- Alligood, C.A.; Dorey, N.R.; Mehrkam, L.R.; Leighty, K.A. Applying behavior-analytic methodology to the science and practice of environmental enrichment in zoos and aquariums. Zoo Biol. 2017, 36, 175–185. [Google Scholar] [CrossRef]
- Alligood, C.; Leighty, K. Putting the “E” in SPIDER: Evolving trends in the evaluation of environmental enrichment efficacy in zoological settings. Anim. Behav. Cogn. 2015, 2, 200–217. [Google Scholar] [CrossRef]
- Mendl, M.; Mason, G.; Paul, E.S. Animal welfare science. In APA Handbook of Comparative Psychology; Call, J., Burghardt, G.M., Pepperberg, I.M., Snowdon, C.T., Zentall, T., Eds.; American Psychological Association: Washington, DC, USA, 2017; Volume 2, pp. 793–811. [Google Scholar]
- Mendyk, R.W. Challenging folklore reptile husbandry in zoological parks. In Zoo Animals: Husbandry, Welfare and Public Interactions; Berger, M., Corbett, S., Eds.; CABI: Wallingford Oxon, UK, 2018; pp. 265–292. [Google Scholar]
- Mellor, D.J.; Beausoleil, N. Extending the ‘Five Domains’ model for animal welfare assessment to incorporate positive welfare states. Anim. Welf. 2015, 24, 241. [Google Scholar] [CrossRef]
- Mellor, D.J. Updating animal welfare thinking: Moving beyond the “Five Freedoms” towards “a Life Worth Living”. Animals 2016, 6, 21. [Google Scholar] [CrossRef]
- DEFRA. Selling Animals as Pets Licensing: Statutory Guidance for Local Authorities (Updated 2022). 2018. Available online: https://www.gov.uk/government/publications/animal-activities-licensing-guidance-for-local-authorities/selling-animals-as-pets-licensing-statutory-guidance-for-local-authorities--2 (accessed on 15 October 2022).
- Welsh Government. The Animal Welfare (Licensing of Activities Involving Animals) (Wales) Regulation 2021. Guidance Notes for Conditions for Selling Animals as Pets. 2021. Available online: https://gov.wales/sites/default/files/publications/2021-10/the-animal-welfare-licensing-of-activities-involving-animals-wales-regulation-2021-guidance.pdf (accessed on 15 October 2022).
- UK Government. Wildlife and Countryside Act. 1981. Available online: https://www.legislation.gov.uk/ukpga/1981/69 (accessed on 11 December 2020).
- DEFRA. Keeping or Training Animals for Exhibition Licensing: Statutory Guidance for Local Authorities. 2022. Available online: https://www.gov.uk/government/publications/animal-activities-licensing-guidance-for-local-authorities/keeping-or-training-animals-for-exhibition-licensing-statutory-guidance-for-local-authorities (accessed on 15 October 2022).
- UK Government. Guidance on the Operation of the Animals (Scientific Procedures) Act 1986. 2014. Available online: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/662364/Guidance_on_the_Operation_of_ASPA.pdf (accessed on 15 October 2022).
- DEFRA. Cat Boarding Licensing: Statutory Guidance for Local Authorities. 2022. Available online: https://www.gov.uk/government/publications/animal-activities-licensing-guidance-for-local-authorities/cat-boarding-licensing-statutory-guidance-for-local-authorities (accessed on 15 October 2022).
- Broom, D.M. Indicators of poor welfare. Br. Vet. J. 1986, 142, 524–526. [Google Scholar] [CrossRef]
- Crane, M. Without the wisdom of Solomon or his ring: Setting standards for exhibited animals in New South Wales. J. Vet. Behav. 2007, 2, 223–229. [Google Scholar] [CrossRef]
- Hashizume, C.; Buote, P. Emergent Planning for the Veterinary Care and Short-Term Housing of Companion Animals Evacuated due to a Wildfire in Alberta, Canada. Prehospital Disaster Med. 2017, 32, S242. [Google Scholar] [CrossRef]
- Hobby, A.; Pankhurst, N.; Haddy, J. The effect of short term confinement stress on binding characteristics of sex steroid binding protein (SBP) in female black bream (Acanthopagrus butcheri) and rainbow trout (Oncorhynchus mykiss). Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2000, 125, 85–94. [Google Scholar] [CrossRef]
- McGarigal, C.R.; Galima, M.M.; Topping, D.; Goldman, K.; Cartamil, D.; Kelley, K.M.; Lowe, C.G. Physiological and behavioral effects of angling on California sheephead (semicossychus pulcher): Response, recovery, and captivity stress of an important gamefish in Southern California. J. Fish Res. 2020, 4, 1–11. [Google Scholar] [CrossRef]
- Narayan, E.J.; Molinia, F.C.; Cockrem, J.F.; Hero, J.-M. Changes in urinary testosterone and corticosterone metabolites during short-term confinement with repeated handling in wild male cane toads (Rhinella marina). Aust. J. Zool. 2012, 59, 264–269. [Google Scholar] [CrossRef]
- Arena, P.C.; Warwick, C. Miscellaneous factors affecting health and welfare. In Health and Welfare of Captive Reptiles; Warwick, C., Frye, F.L., Murphy, J.B., Eds.; Chapman & Hall/Kluwer: London, UK; New York, NY, USA, 1995; pp. 263–283. [Google Scholar]
- Arena, P.C.; Bashaw, M.J.; Grant, R.; Howell, T.; Martínez-Silvestre, A.; Warwick, C. Miscellaneous factors for health and welfare. In Health and Welfare of Captive Reptiles, 2nd ed.; Warwick, C., Arena, P.C., Burghardt, G.M., Eds.; Springer: Cham, Switzerland, 2023; pp. 583–617. [Google Scholar] [CrossRef]
- Tyrrell, C.; Cree, A. Relationships between corticosterone concentration and season, time of day and confinement in a wild reptile (tuatara, Sphenodon punctatus). Gen. Comp. Endocrinol. 1998, 110, 97–108. [Google Scholar] [CrossRef]
- Ebrahimi, M.; Bull, C.M. Determining the success of varying short-term confinement time during simulated translocations of the endangered pygmy bluetongue lizard (Tiliqua adelaidensis). Amphibia-Reptilia 2013, 34, 31–39. [Google Scholar] [CrossRef]
- Mathies, T.; Felix, T.A.; Lance, V.A. Effects of trapping and subsequent short-term confinement stress on plasma corticosterone in the brown treesnake (Boiga irregularis) on Guam. Gen. Comp. Endocrinol. 2001, 124, 106–114. [Google Scholar] [CrossRef]
- Adams, N.J.; Farnworth, M.J.; Rickett, J.; Parker, K.A.; Cockrem, J.F. Behavioural and corticosterone responses to capture and confinement of wild blackbirds (Turdus merula). Appl. Anim. Behav. Sci. 2011, 134, 246–255. [Google Scholar] [CrossRef]
- Wichman, A.; Norring, M.; Pastell, M.; Algers, B.; Pösö, R.; Valros, A.; Saloniemi, H.; Hänninen, L. Effect of crate height during short-term confinement on the welfare and behaviour of turkeys. Appl. Anim. Behav. Sci. 2010, 126, 134–139. [Google Scholar] [CrossRef]
- Lombardo, M.P.; Thorpe, P.A. Captivity affects sperm production, testes size and beak color in house sparrows (Passer domesticus). Intern. Stud. Sparrows 2009, 33, 5–16. [Google Scholar]
- Mal, M.; Friend, T.; Lay, D.; Vogelsang, S.; Jenkins, O. Behavioral responses of mares to short-term confinement and social isolation. Appl. Anim. Behav. Sci. 1991, 31, 13–24. [Google Scholar] [CrossRef]
- Weiler, U.; Salloum, B.A.; Claus, R. Influence of short-term confinement and exercise on tibia development in growing pigs. J. Vet. Med. Ser. A 2006, 53, 450–455. [Google Scholar] [CrossRef]
- Zhang, M.-Y.; Li, X.; Zhang, X.-H.; Liu, H.-G.; Li, J.-H.; Bao, J. Effects of confinement duration and parity on stereotypic behavioral and physiological responses of pregnant sows. Physiol. Behav. 2017, 179, 369–376. [Google Scholar] [CrossRef]
- Zeiler, G.E.; Fosgate, G.T.; Van Vollenhoven, E.; Rioja, E. Assessment of behavioural changes in domestic cats during short-term hospitalisation. J. Feline Med. Surg. 2014, 16, 499–503. [Google Scholar] [CrossRef]
- Griffin, B. Population wellness: Keeping cats physically and behaviorally healthy. In The Cat; Little, S.E., Ed.; W.B. Sauders: Philadelphia, PA, USA, 2012; pp. 1312–1356. [Google Scholar] [CrossRef]
- Uetake, K.; Goto, A.; Koyama, R.; Kikuchi, R.; Tanaka, T. Effects of single caging and cage size on behavior and stress level of domestic neutered cats housed in an animal shelter. Anim. Sci. J. 2013, 84, 272–274. [Google Scholar] [CrossRef]
- Judge, P.G.; de Waal, F.B. Conflict avoidance among rhesus monkeys: Coping with short-term crowding. Anim. Behav. 1993, 46, 221–232. [Google Scholar] [CrossRef]
- AWC. Establishing Minimum Length Size for Snake Vivariums in Pet Selling Establishments (Working Title); Animal Welfare Commission: London, UK, (in press).
- Warwick, C.; Grant, R.; Steedman, C.; Howell, T.J.; Arena, P.C.; Lambiris, A.J.; Nash, A.-E.; Jessop, M.; Pilny, A.; Amarello, M. Getting It Straight: Accommodating rectilinear behavior in captive snakes—A review of recommendations and their evidence base. Animals 2021, 11, 1459. [Google Scholar] [CrossRef]
- Cargill, B.; Benato, L.; Rooney, N.J. A survey exploring the impact of housing and husbandry on pet snake welfare. Anim. Welf. 2022, 31, 193–208. [Google Scholar] [CrossRef]
- McCullock, S.P. A Wolf in Sheep’s Clothing. 2020. Available online: https://theecologist.org/2020/feb/11/wolf-sheeps-clothing (accessed on 20 October 2022).
- Warwick, C.; Steedman, C. Exotic pet trading and keeping: Proposing a model government consultation and advisory protocol. J. Vet. Behav. 2021, 43, 66–76. [Google Scholar] [CrossRef]
- DEFRA (UK Department for Environment, Food & Rural Affairs) (London, UK); Amess, D. (London, UK). Personal communication, 2019.
- Bayazit, V. Evaluation of cortisol and stress in captive animals. Aust. J. Basic Appl. Sci. 2009, 3, 1022–1031. [Google Scholar]
- Morgan, K.N.; Tromborg, C.T. Sources of stress in captivity. Appl. Anim. Behav. Sci. 2007, 102, 262–302. [Google Scholar] [CrossRef]
- Gelling, M.; Montes, I.; Moorhouse, T.P.; Macdonald, D.W. Captive housing during water vole (Arvicola terrestris) reintroduction: Does short-term social stress impact on animal welfare? PLoS ONE 2010, 5, e9791. [Google Scholar] [CrossRef]
- Zhang, K. Environmental stressor, stress response, and disease. Environ. Dis. 2018, 3, 1. [Google Scholar] [CrossRef]
- Garner, J.P. Stereotypies and other abnormal repetitive behaviors: Potential impact on validity, reliability, and replicability of scientific outcomes. ILAR J. 2005, 46, 106–117. [Google Scholar] [CrossRef]
- Moberg, G.P. Biological response to stress: Implications for animal welfare. In The Biology of Animal Stress: Basic Principles and Implications for Animal Welfare; Moberg, G.P., Mench, J.A., Eds.; CABI: Oxon, UK, 2000; pp. 1–21. [Google Scholar]
- Willner, P. The chronic mild stress (CMS) model of depression: History, evaluation and usage. Neurobiol. Stress 2017, 6, 78–93. [Google Scholar] [CrossRef]
- Chmitorz, A.; Kurth, K.; Mey, L.K.; Wenzel, M.; Lieb, K.; Tüscher, O.; Kubiak, T.; Kalisch, R. Assessment of microstressors in adults: Questionnaire development and ecological validation of the Mainz inventory of microstressors. JMIR Ment. Health 2020, 7, e14566. [Google Scholar] [CrossRef]
- Greenberg, N. The saurian psyche revisited: Lizards in research. In The Care and Use of Amphibians, Reptiles, and Fish in Research; Schaeffer, D.O., Kleinow, K.M., Krulisch, L., Eds.; Scientists Center Animal Welfare: Greenbelt, MD, USA, 1992; pp. 75–91. [Google Scholar]
- Zanette, L.Y.; Clinchy, M. Predator–prey interactions: Integrating fear effects. In APA Handbook of Comparative Psychology: Basic Concepts, Methods, Neural Substrate, and Behavior, Vol. 1; American Psychological Association: Washington, DC, USA, 2017; pp. 815–831. [Google Scholar]
- Korzan, W.J.; Summers, T.R.; Ronan, P.J.; Summers, C.H. Visible sympathetic activity as a social signal in Anolis carolinensis: Changes in aggression and plasma catecholamines. Horm. Behav. 2000, 38, 193–199. [Google Scholar] [CrossRef]
- Martínez-Silvestre, A. How to assess stress in reptiles. J. Exot. Pet Med. 2014, 23, 240–243. [Google Scholar] [CrossRef]
- Gangloff, E.; Greenberg, N.B. Biology of stress. In Health and Welfare of Captive Reptiles, 2nd ed.; Warwick, C., Arena, P.C., Burghardt, G.M., Eds.; Springer: Cham, Switzerland, 2023; pp. 93–142. [Google Scholar] [CrossRef]
- Mancera, K.F.; Phillips, C.J.C. Effects of noise and light. In Health and Welfare of Captive Reptiles, 2nd ed.; Warwick, C., Arena, P.C., Burghardt, G.M., Eds.; Springer: Cham, Switzerland, 2023; pp. 357–378. [Google Scholar] [CrossRef]
- Breuning, L.G. Stimulating dopamine, serotonin, oxytocin and endorphin by learning how they’re stimulated in animals. J. Med. -Clin. Res. Rev. 2018, 2, 1–3. [Google Scholar] [CrossRef]
- Cowan, D.R. Adaptation, maladaptation and disease of captive reptiles. In Contributions to Herpetology; Murphy, J.B., Collins, J.T., Eds.; Society for the Study of Amphibians and Reptiles: Lawrence, KS, USA, 1980; pp. 191–196. [Google Scholar]
- Nelson, R.J.; Demas, G.E.; Klein, S.L.; Kriegsfeld, L.J. Seasonal Patterns of Stress, Immune Function, and Disease; Cambridge University Press: Cambridge, UK, 2002. [Google Scholar]
- Schneiderman, N.; Ironson, G.; Siegel, S.D. Stress and health: Psychological, behavioral, and biological determinants. Annu. Rev. Clin. Psychol. 2005, 1, 607–628. [Google Scholar] [CrossRef]
- Hoshaw, B.A.; Evans, J.C.; Mueller, B.; Valentino, R.J.; Lucki, I. Social competition in rats: Cell proliferation and behavior. Behav. Brain Res. 2006, 175, 343–351. [Google Scholar] [CrossRef]
- Del Giudice, M.; Ellis, B.J.; Shirtcliff, E.A. Making sense of stress: An evolutionary—Developmental framework. In Adaptive and Maladaptive Aspects of Developmental Stress; Springer: Berlin/Heidelberg, Germany, 2013; pp. 23–43. [Google Scholar]
- Koch, C.; Leinweber, B.; Drengberg, B.; Blaum, C.; Oster, H. Interaction between circadian rhythms and stress. Neurobiol. Stress 2017, 6, 57–67. [Google Scholar] [CrossRef]
- Oster, H. The interplay between stress, circadian clocks, and energy metabolism. J. Endocrinol. 2020, 247, R13–R25. [Google Scholar] [CrossRef]
- Douma, E.H.; de Kloet, E.R. Stress-induced plasticity and functioning of ventral tegmental dopamine neurons. Neurosci. Biobehav. Rev. 2020, 108, 48–77. [Google Scholar] [CrossRef]
- Russell, G.; Lightman, S. The human stress response. Nat. Rev. Endocrinol. 2019, 15, 525–534. [Google Scholar] [CrossRef]
- Martino, T.A.; Oudit, G.Y.; Herzenberg, A.M.; Tata, N.; Koletar, M.M.; Kabir, G.M.; Belsham, D.D.; Backx, P.H.; Ralph, M.R.; Sole, M.J. Circadian rhythm disorganization produces profound cardiovascular and renal disease in hamsters. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2008, 294, R1675–R1683. [Google Scholar] [CrossRef] [PubMed]
- Evans, J.A.; Davidson, A.J. Health consequences of circadian disruption in humans and animal models. Prog. Mol. Biol. Transl. Sci. 2013, 119, 283–323. [Google Scholar] [CrossRef] [PubMed]
- Healy, K.; McNally, L.; Ruxton, G.D.; Cooper, N.; Jackson, A.L. Metabolic rate and body size are linked with perception of temporal information. Anim. Behav. 2013, 86, 685–696. [Google Scholar] [CrossRef]
- Coria-Avila, G.A.; Pfaus, J.G.; Orihuela, A.; Domínguez-Oliva, A.; José-Pérez, N.; Hernández, L.A.; Mota-Rojas, D. The Neurobiology of Behavior and Its Applicability for Animal Welfare: A Review. Animals 2022, 12, 928. [Google Scholar] [CrossRef] [PubMed]
- Tort, L.; Cockrem, J.F.; Narayan, E.J. Editorial: Comparative endocrine stress responses in vertebrates. Comp. Endocr. Stress Responses Vertebr. 2020, 10, 652. [Google Scholar] [CrossRef] [PubMed]
- Ralph, C.; Tilbrook, A. Invited review: The usefulness of measuring glucocorticoids for assessing animal welfare. J. Anim. Sci. 2016, 94, 457–470. [Google Scholar] [CrossRef] [PubMed]
- Romero, M.L.; Butler, L.K. Endocrinology of stress. Int. J. Comp. Psychol. 2007, 20, 89–95. [Google Scholar] [CrossRef]
- van der Laan, J.E.; Vinke, C.M.; Arndt, S.S. Evaluation of hair cortisol as an indicator of long-term stress responses in dogs in an animal shelter and after subsequent adoption. Sci. Rep. 2022, 12, 1–10. [Google Scholar] [CrossRef]
- Wunderink, Y.S.; Engels, S.; Halm, S.; Yúfera, M.; Martínez-Rodríguez, G.; Flik, G.; Klaren, P.H.; Mancera, J.M. Chronic and acute stress responses in Senegalese sole (Solea senegalensis): The involvement of cortisol, CRH and CRH-BP. Gen. Comp. Endocrinol. 2011, 171, 203–210. [Google Scholar] [CrossRef]
- Ashley, S.; Brown, S.; Ledford, J.; Martin, J.; Nash, A.E.; Terry, A.; Tristan, T.; Warwick, C. Morbidity and mortality of invertebrates, amphibians, reptiles, and mammals at a major exotic companion animal wholesaler. J. Appl. Anim. Welf. Sci. 2014, 17, 308–321. [Google Scholar] [CrossRef]
- Fischer, C.P.; Romero, L.M. Chronic captivity stress in wild animals is highly species-specific. Conserv. Physiol. 2019, 7, coz093. [Google Scholar] [CrossRef]
- Peng, S.; Broom, D.M. The Sustainability of Keeping Birds as Pets: Should Any Be Kept? Animals 2021, 11, 582. [Google Scholar] [CrossRef]
- Sinclair, J.S.; Stringham, O.C.; Udell, B.; Mandrak, N.E.; Leung, B.; Romagosa, C.M.; Lockwood, J.L. The International Vertebrate Pet Trade Network and insights from US imports of exotic pets. BioScience 2021, 71, 977–990. [Google Scholar] [CrossRef]
- Grant, R.A.; Montrose, V.T.; Wills, A.P. ExNOTic: Should We Be Keeping Exotic Pets? Animals 2017, 7, 47. [Google Scholar] [CrossRef]
- Farm Animal Welfare Council. Farm Animal Welfare Council Press Statement. 1979. Available online: https://webarchive.nationalarchives.gov.uk/20121010012428/http://www.fawc.org.uk/pdf/fivefreedoms1979.pdf (accessed on 23 February 2020).
| Animal(s) | Context | Short-Term Stipulation | Source |
|---|---|---|---|
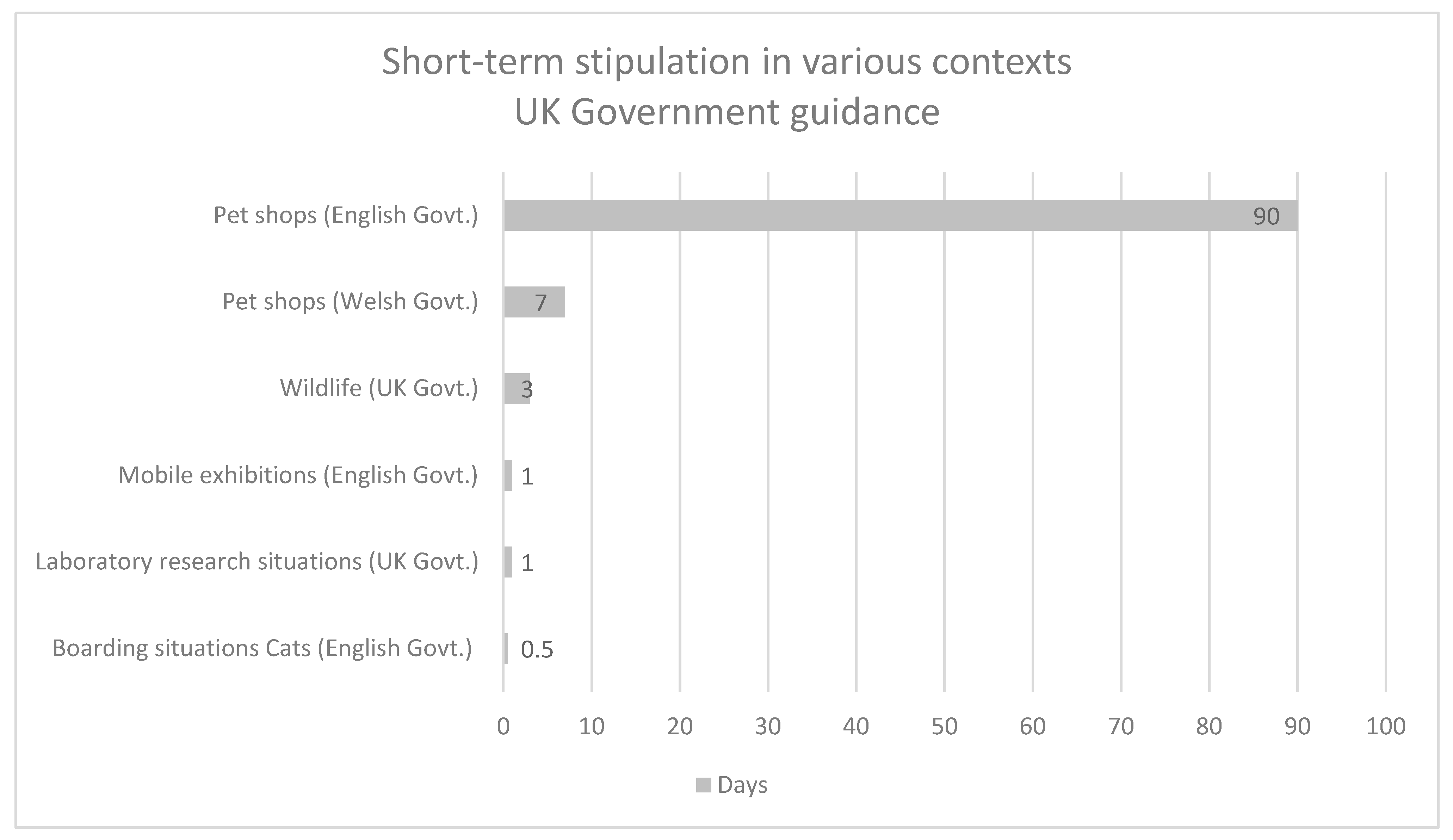
| All animals | Pet shops | 3 months | English Government [18] |
| All animals | Pet shops | 7 days | Welsh Government [19] |
| All animals | Wildlife | 72 h | UK Government [20] |
| All animals | Mobile exhibitions | 24 h | English Government [21] |
| All animals | Laboratory research situations | <24 h | UK Government [22] |
| Cats | Boarding situations | 12 h | English Government [23] |
| Animal(s) | Context | Short-Term Stipulation | Source |
|---|---|---|---|
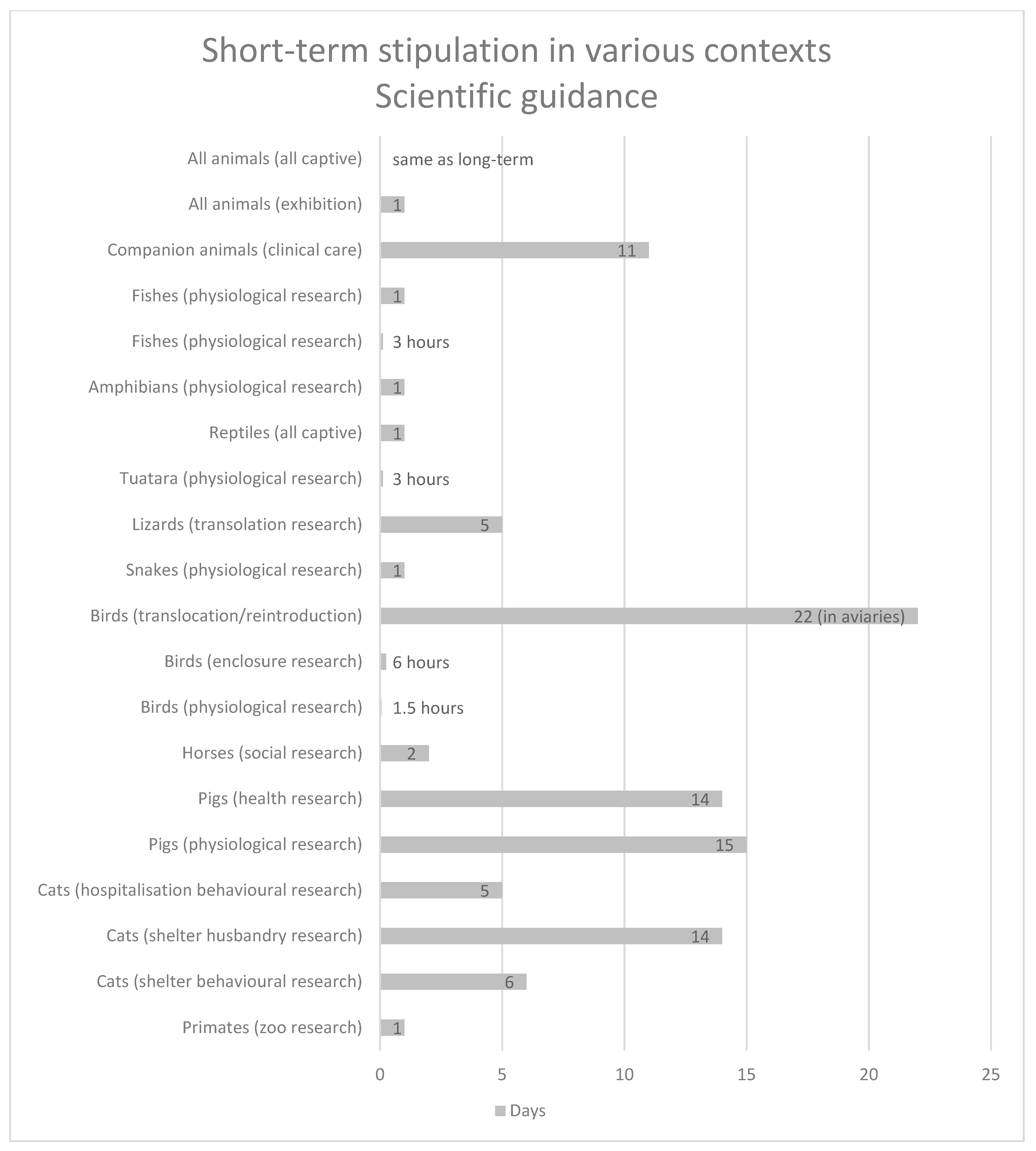
| All animals | All captive situations | Short-term captives are to have the same benefits as long-term captives | [24] |
| All animals | Exhibition situations | <24 h | [25] |
| Companion animals | Clinical care situation | 11 h | [26] |
| Fishes | Physiological research situation | <24 h | [27] |
| Physiological research situation | ≤3 h | [28] | |
| Amphibians | Physiological research situation | 24 h | [29] |
| Reptiles general | All captive situations | 24 h | [30,31] |
| Tuatara | Physiological research situation | 3 h | [32] |
| Lizards | Translocation research situation | 1–5 days | [33] |
| Snakes | Physiological research situation | 1 night | [34] |
| Birds | Capture and confinement translocation or reintroduction research situation | 22 days (in aviaries) = long-term conditions | [35] |
| Enclosure research situation | 6 h | [36] | |
| Physiological research situation | <90 min | [37] | |
| Horses | Social research situation | 48 h | [38] |
| Pigs | Health research situation | 2 weeks | [39] |
| Physiological research situation | 1–15 days | [40] | |
| Cats | Hospitalisation behavioural research situation | 3–5 days | [41] |
| Shelter husbandry research situation | <2 weeks | [42] | |
| Shelter behavioural research situation | 6 days | [43] | |
| Primates | Zoo research situation | 1 day | [44] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Warwick, C.; Steedman, C.; Jessop, M.; Grant, R. Defining Short-Term Accommodation for Animals. Animals 2023, 13, 732. https://doi.org/10.3390/ani13040732
Warwick C, Steedman C, Jessop M, Grant R. Defining Short-Term Accommodation for Animals. Animals. 2023; 13(4):732. https://doi.org/10.3390/ani13040732
Chicago/Turabian StyleWarwick, Clifford, Catrina Steedman, Mike Jessop, and Rachel Grant. 2023. "Defining Short-Term Accommodation for Animals" Animals 13, no. 4: 732. https://doi.org/10.3390/ani13040732
APA StyleWarwick, C., Steedman, C., Jessop, M., & Grant, R. (2023). Defining Short-Term Accommodation for Animals. Animals, 13(4), 732. https://doi.org/10.3390/ani13040732






