Cotinine as a Sentinel of Canine Exposure to Tobacco Smoke
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Blood and Hair Sample Collection
2.3. Laboratory Procedure
2.4. Statistical Analysis
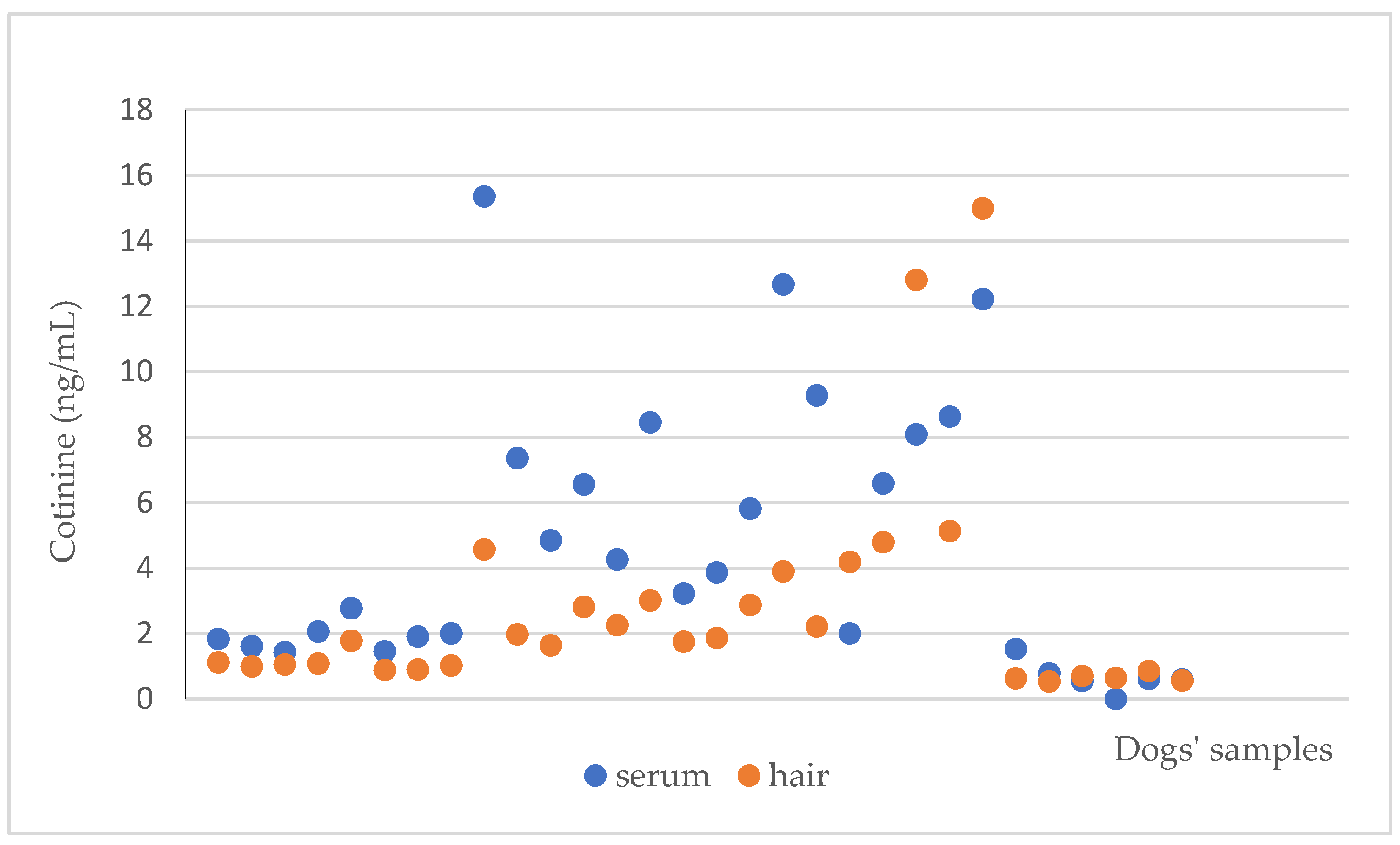
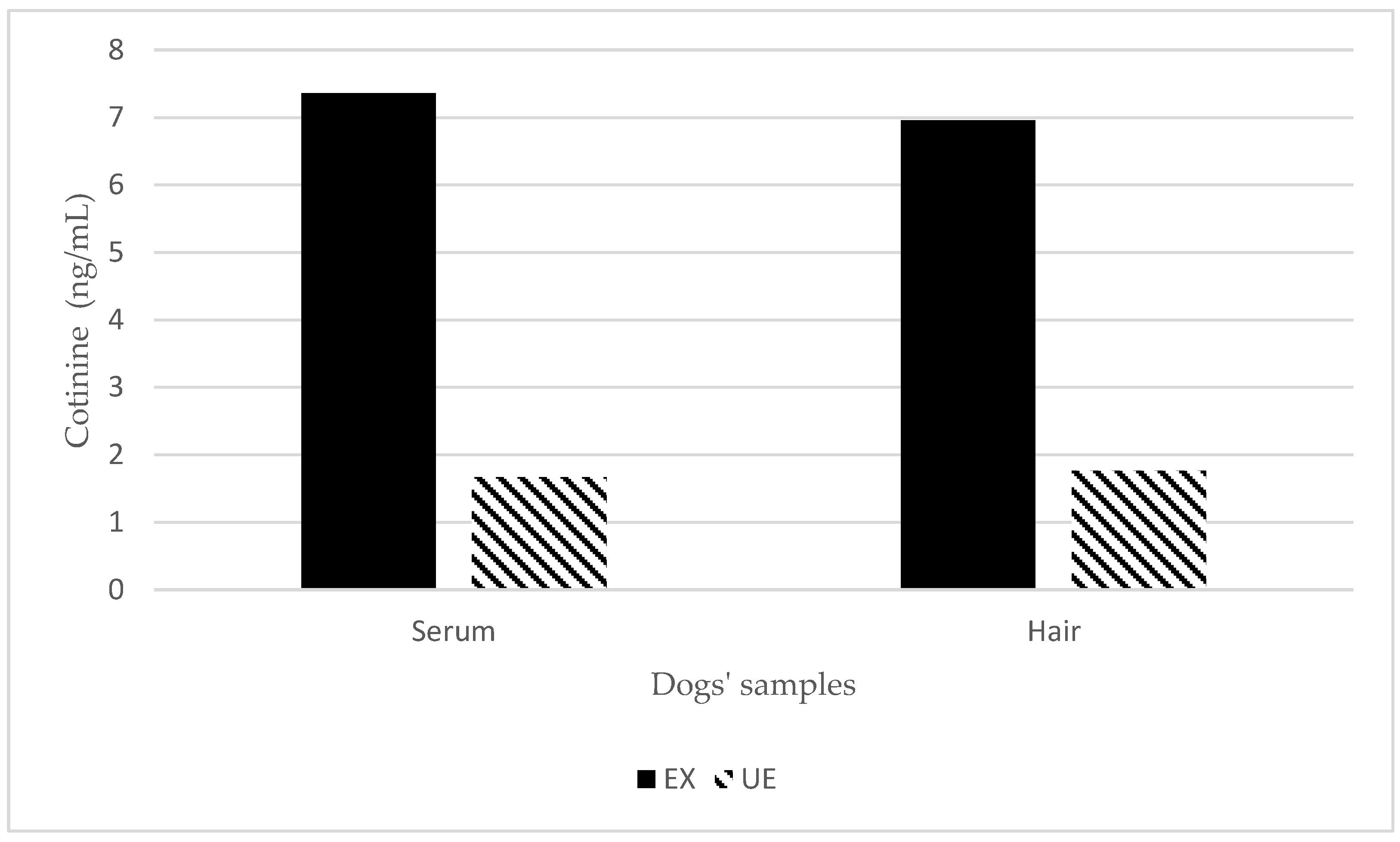
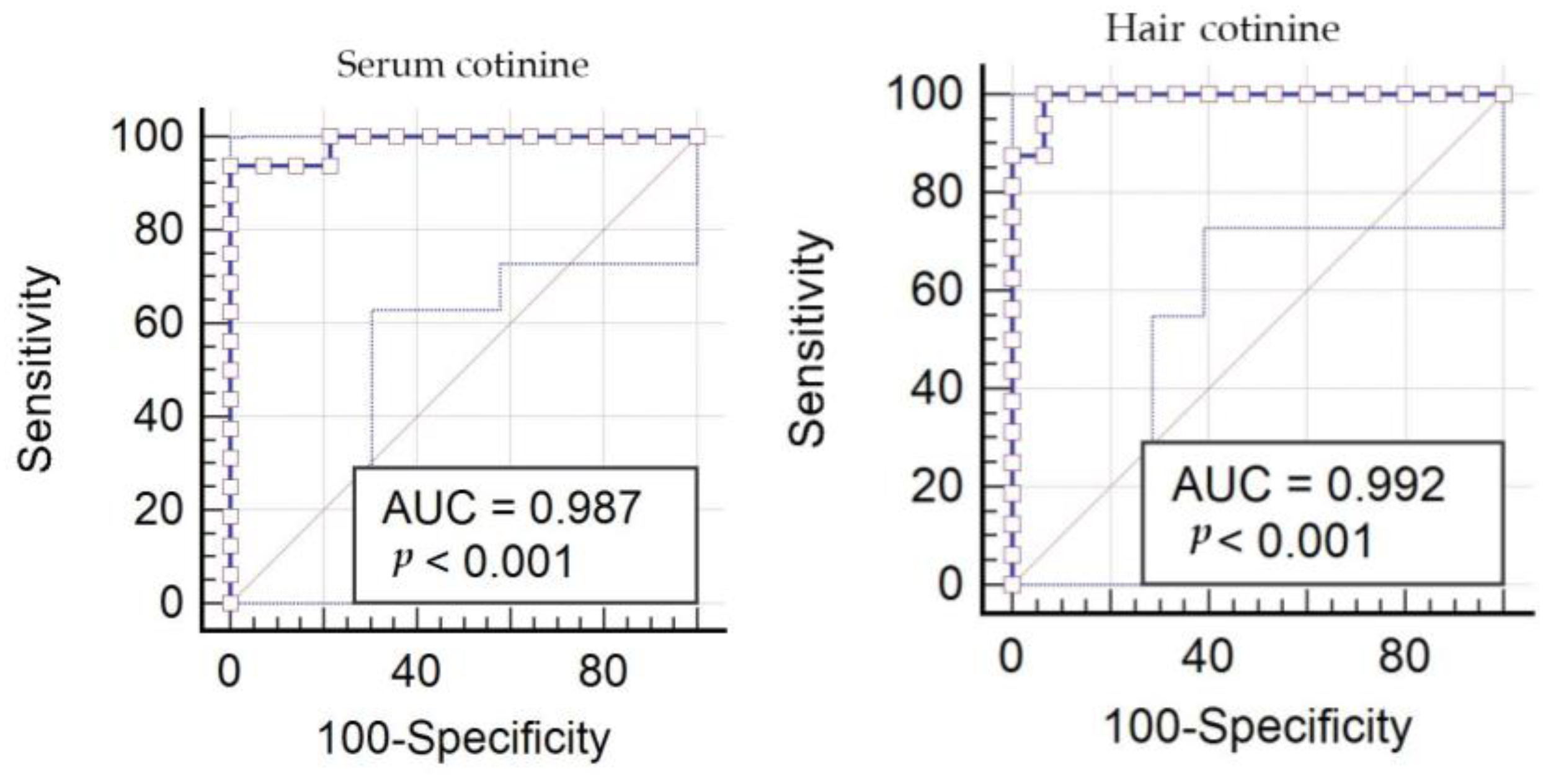
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lindsay, R.P.; Tsoh, J.Y.; Sung, H.-Y.; Max, W. Secondhand Smoke Exposure and Serum Cotinine Levels among Current Smokers in the USA. Tob. Control 2016, 25, 224–231. [Google Scholar] [CrossRef]
- Max, W.; Sung, H.-Y.; Shi, Y. Deaths from Secondhand Smoke Exposure in the United States: Economic Implications. Am. J. Public Health 2012, 102, 2173–2180. [Google Scholar] [CrossRef] [PubMed]
- Onor, I.O.; Stirling, D.L.; Williams, S.R.; Bediako, D.; Borghol, A.; Harris, M.B.; Darensburg, T.B.; Clay, S.D.; Okpechi, S.C.; Sarpong, D.F. Clinical Effects of Cigarette Smoking: Epidemiologic Impact and Review of Pharmacotherapy Options. Int. J. Environ. Res. Public Health 2017, 14, 1147. [Google Scholar] [CrossRef] [PubMed]
- Tan, X.; Vrana, K.; Ding, Z.-M. Cotinine: Pharmacologically Active Metabolite of Nicotine and Neural Mechanisms for Its Actions. Front. Behav. Neurosci. 2021, 15, 758252. [Google Scholar] [CrossRef] [PubMed]
- Jacob, P.; Benowitz, N.L.; Destaillats, H.; Gundel, L.; Hang, B.; Martins-Green, M.; Matt, G.E.; Quintana, P.J.E.; Samet, J.M.; Schick, S.F.; et al. Thirdhand Smoke: New Evidence, Challenges, and Future Directions. Chem. Res. Toxicol 2017, 30, 270–294. [Google Scholar] [CrossRef] [PubMed]
- Avila-Tang, E.; Al-Delaimy, W.K.; Ashley, D.L.; Benowitz, N.; Bernert, J.T.; Kim, S.; Samet, J.M.; Hecht, S.S. Assessing Secondhand Smoke Using Biological Markers. Tob. Control 2013, 22, 164–171. [Google Scholar] [CrossRef]
- Benowitz, N.L. Cotinine as a Biomarker of Environmental Tobacco Smoke Exposure. Epidemiol. Rev. 1996, 18, 188–204. [Google Scholar] [CrossRef] [PubMed]
- Al-Delaimy, W.K. Hair as a Biomarker for Exposure to Tobacco Smoke. Tob. Control 2002, 11, 176–182. [Google Scholar] [CrossRef]
- Puzycki, K.; Ekin, U.; Bidaisee, S.; Keku, E. Tobacco Smoke Exposure and Household Pets: A Systematic Literature Review Examining the Health Risk to Household Pets and New Indications of Exposed Pets Affecting Human Health. Int. Public Health J. 2018, 10, 11–24. [Google Scholar]
- Bukowski, J.A.; Wartenberg, D. An Alternative Approach for Investigating the Carcinogenicity of Indoor Air Pollution: Pets as Sentinels of Environmental Cancer Risk. Environ. Health Perspect. 1997, 105, 1312–1319. [Google Scholar] [CrossRef]
- Gollenberg, A.L.; Bertone-Johnson, E.R. Passive Smoking and Health Research; Passive Smoking and Cancer Risk in Companion Animals: Sentinels for Health Effects in Humans; Jeorgensen, N.A., Ed.; Nova Science Publishers: Hauppauge, NY, USA, 2006. [Google Scholar]
- Gunay, F.; Ilarslan, N.E.C.; Bakar-Ates, F.; Deniz, K.; Kadioglu, Y.K.; Kiran, S.; Bakirarar, B.; Cobanoglu, N. Evaluation of Hair Cotinine and Toxic Metal Levels in Children Who Were Exposed to Tobacco Smoke. Pediatr. Pulmonol. 2020, 55, 1012–1019. [Google Scholar] [CrossRef] [PubMed]
- Bertone-Johnson, E.R.; Procter-Gray, E.; Gollenberg, A.L.; Ryan, M.B.; Barber, L.G. Environmental Tobacco Smoke and Canine Urinary Cotinine Level. Environ. Res. 2008, 106, 361–364. [Google Scholar] [CrossRef] [PubMed]
- Almeida, M.I.G.S.; Barreiros, L.; Kolev, S.D.; Segundo, M.A. Chapter 50—Salivary Cotinine Assays. In Neuroscience of Nicotine; Preedy, V.R., Ed.; Academic Press: Cambridge, MA, USA, 2019; pp. 411–418. ISBN 978-0-12-813035-3. [Google Scholar]
- Bowman, E.R.; McKennis, H. Studies on the Metabolism of (-)-Cotinine in the Human. J. Pharmacol. Exp. Ther. 1962, 135, 306–311. [Google Scholar] [PubMed]
- Brazell, R.S.; Stiff, A.C.; Henderson, G.M.; Jenkins, R.A.; Romig, P.L.; Auerbach, O. Plasma Nicotine and Cotinine in Tobacco Smoke Exposed Beagle Dogs. Toxicol. Appl. Pharmacol. 1984, 73, 152–158. [Google Scholar] [CrossRef]
- Bawazeer, S.; Watson, D.G.; Knottenbelt, C. Determination of Nicotine Exposure in Dogs Subjected to Passive Smoking Using Methanol Extraction of Hair Followed by Hydrophilic Interaction Chromatography in Combination with Fourier Transform Mass Spectrometry. Talanta 2012, 88, 408–411. [Google Scholar] [CrossRef]
- Knottenbelt, C.M.; Bawazeer, S.; Hammond, J.; Mellor, D.; Watson, D.G. Nicotine Hair Concentrations in Dogs Exposed to Environmental Tobacco Smoke: A Pilot Study. J. Small Anim. Pract. 2012, 53, 623–626. [Google Scholar] [CrossRef] [PubMed]
- Al-Delaimy, W.K.; Crane, J.; Woodward, A. Is the Hair Nicotine Level a More Accurate Biomarker of Environmental Tobacco Smoke Exposure than Urine Cotinine? J. Epidemiol. Community Health 2002, 56, 66–71. [Google Scholar] [CrossRef]
- Kataoka, H.; Kaji, S.; Moai, M. Risk Assessment of Passive Smoking Based on Analysis of Hair Nicotine and Cotinine as Exposure Biomarkers by In-Tube Solid-Phase Microextraction Coupled On-Line to LC-MS/MS. Molecules 2021, 26, 7356. [Google Scholar] [CrossRef] [PubMed]
- Tzatzarakis, M.N.; Vardavas, C.I.; Terzi, I.; Kavalakis, M.; Kokkinakis, M.; Liesivuori, J.; Tsatsakis, A.M. Hair Nicotine/Cotinine Concentrations as a Method of Monitoring Exposure to Tobacco Smoke among Infants and Adults. Hum. Exp. Toxicol. 2012, 31, 258–265. [Google Scholar] [CrossRef]
- Dehn, D.L.; Claffey, D.J.; Duncan, M.W.; Ruth, J.A. Nicotine and Cotinine Adducts of a Melanin Intermediate Demonstrated by Matrix-Assisted Laser Desorption/Ionization Time-of-Flight Mass Spectrometry. Chem. Res. Toxicol. 2001, 14, 275–279. [Google Scholar] [CrossRef]
- Pirkle, J.L.; Flegal, K.M.; Bernert, J.T.; Brody, D.J.; Etzel, R.A.; Maurer, K.R. Exposure of the US Population to Environmental Tobacco Smoke: The Third National Health and Nutrition Examination Survey, 1988 to 1991. JAMA 1996, 275, 1233–1240. [Google Scholar] [CrossRef]
- Hukkanen, J.; Jacob, P.; Benowitz, N.L. Metabolism and Disposition Kinetics of Nicotine. Pharmacol. Rev. 2005, 57, 79–115. [Google Scholar] [CrossRef]
- Kalkbrenner, A.E.; Hornung, R.W.; Bernert, J.T.; Hammond, S.K.; Braun, J.M.; Lanphear, B.P. Determinants of Serum Cotinine and Hair Cotinine as Biomarkers of Childhood Secondhand Smoke Exposure. J. Expo Sci. Environ. Epidemiol. 2010, 20, 615–624. [Google Scholar] [CrossRef]
- Feldman, L.Y.; Thacher, J.D.; van Hage, M.; Kull, I.; Melén, E.; Pershagen, G.; Wickman, M.; To, T.; Protudjer, J.L.; Bergström, A. Early-Life Secondhand Smoke Exposure and Food Hypersensitivity through Adolescence. Allergy 2018, 73, 1558–1561. [Google Scholar] [CrossRef] [PubMed]
- Kim, A.-S.; Ko, H.-J.; Kwon, J.-H.; Lee, J.-M. Exposure to Secondhand Smoke and Risk of Cancer in Never Smokers: A Meta-Analysis of Epidemiologic Studies. Int. J. Environ. Res. Public Health 2018, 15, 1981. [Google Scholar] [CrossRef] [PubMed]
- Saulyte, J.; Regueira, C.; Montes-Martínez, A.; Khudyakov, P.; Takkouche, B. Active or Passive Exposure to Tobacco Smoking and Allergic Rhinitis, Allergic Dermatitis, and Food Allergy in Adults and Children: A Systematic Review and Meta-Analysis. PLoS Med. 2014, 11, e1001611. [Google Scholar] [CrossRef] [PubMed]
- Cao, S.; Yang, C.; Gan, Y.; Lu, Z. The Health Effects of Passive Smoking: An Overview of Systematic Reviews Based on Observational Epidemiological Evidence. PLoS ONE 2015, 10, e0139907. [Google Scholar] [CrossRef] [PubMed]
- Baltar, V.T.; Xun, W.W.; Chuang, S.-C.; Relton, C.; Ueland, P.M.; Vollset, S.E.; Midttun, Ø.; Johansson, M.; Slimani, N.; Jenab, M.; et al. Smoking, Secondhand Smoke, and Cotinine Levels in a Subset of EPIC Cohort. Cancer Epidemiol. Biomark. Prev. 2011, 20, 869–875. [Google Scholar] [CrossRef]
- Connor Gorber, S.; Schofield-Hurwitz, S.; Hardt, J.; Levasseur, G.; Tremblay, M. The Accuracy of Self-Reported Smoking: A Systematic Review of the Relationship between Self-Reported and Cotinine-Assessed Smoking Status. Nicotine Tob. Res. 2009, 11, 12–24. [Google Scholar] [CrossRef]
- Kim, S.; Apelberg, B.J.; Avila-Tang, E.; Hepp, L.; Yun, D.; Samet, J.M.; Breysse, P.N. Utility and Cutoff Value of Hair Nicotine as a Biomarker of Long-Term Tobacco Smoke Exposure, Compared to Salivary Cotinine. Int. J. Environ. Res. Public Health 2014, 11, 8368–8382. [Google Scholar] [CrossRef] [PubMed]
- Audrain-McGovern, J.; Benowitz, N.L. Cigarette Smoking, Nicotine, and Body Weight. Clin. Pharmacol. Ther. 2011, 90, 164–168. [Google Scholar] [CrossRef] [PubMed]
- Chiolero, A.; Faeh, D.; Paccaud, F.; Cornuz, J. Consequences of Smoking for Body Weight, Body Fat Distribution, and Insulin Resistance. Am. J. Clin. Nutr. 2008, 87, 801–809. [Google Scholar] [CrossRef] [PubMed]
- Robinson, N.E. Airway Physiology. Vet. Clin. N. Am. Small Anim. Pract. 1992, 22, 1043–1064. [Google Scholar] [CrossRef] [PubMed]
- Lippi, D.; Bianucci, R.; Donell, S. Gender Medicine: Its Historical Roots. Postgrad. Med. J. 2020, 96, 480–486. [Google Scholar] [CrossRef] [PubMed]
- Baggio, G.; Corsini, A.; Floreani, A.; Giannini, S.; Zagonel, V. Gender Medicine: A Task for the Third Millennium. Clin. Chem. Lab. Med. 2013, 51, 713–727. [Google Scholar] [CrossRef] [PubMed]
- Chen, A.; Krebs, N.M.; Zhu, J.; Sun, D.; Stennett, A.; Muscat, J.E. Sex/Gender Differences in Cotinine Levels Among Daily Smokers in the Pennsylvania Adult Smoking Study. J. Womens Health 2017, 26, 1222–1230. [Google Scholar] [CrossRef] [PubMed]
- Flores, R.J.; Uribe, K.P.; Swalve, N.; O’Dell, L.E. Sex Differences in Nicotine Intravenous Self-Administration: A Meta-Analytic Review. Physiol. Behav. 2019, 203, 42–50. [Google Scholar] [CrossRef]
- Florescu, A.; Ferrence, R.; Einarson, T.; Selby, P.; Soldin, O.; Koren, G. Methods for Quantification of Exposure to Cigarette Smoking and Environmental Tobacco Smoke: Focus on Developmental Toxicology. Ther. Drug Monit. 2009, 31, 14–30. [Google Scholar] [CrossRef]
- Orasan, M.S.; Roman, I.I.; Coneac, A.; Muresan, A.; Orasan, R.I. Hair Loss and Regeneration Performed on Animal Models. Clujul Med. 2016, 89, 327–334. [Google Scholar] [CrossRef]
- Welle, M.M.; Wiener, D.J. The Hair Follicle: A Comparative Review of Canine Hair Follicle Anatomy and Physiology. Toxicol. Pathol. 2016, 44, 564–574. [Google Scholar] [CrossRef]
| Breed | Age (Years) | BW (kg) | Sex | Smoke Exposure | Exposure Intensity | Group | |
|---|---|---|---|---|---|---|---|
| 1 | Entlebucher Mountain Dog | 2.5 | 27.4 | Male | Yes | +++ | EX |
| 2 | Kurzhaar | 5 | 39.7 | Male | Yes | ++ | EX |
| 3 | Labrador retriever | 2 | 42.5 | Male | Yes | +++ | EX |
| 4 | Labrador retriever | 2 | 26.5 | Male | Yes | +++ | EX |
| 5 | Labrador retriever | 4 | 37.1 | Male | Yes | +++ | EX |
| 6 | Bassethound | 2.5 | 21.8 | Male | Yes | +++ | EX |
| 7 | Great dane | 5.5 | 77.7 | Male | Yes | + | EX |
| 8 | Afgan hound | 4.5 | 30.9 | Male | Yes | ++ | EX |
| 9 | Pharaoh hound | 4 | 29.2 | Male | Yes | ++ | EX |
| 10 | Bassethound | 4 | 28 | Male | Yes | ++ | EX |
| 11 | Bassethound | 6 | 23 | Female | Yes | +++ | EX |
| 12 | Bassethound | 3.5 | 24 | Female | Yes | +++ | EX |
| 13 | Bassethound | 1.5 | 21 | Female | Yes | +++ | EX |
| 14 | Bassethound | 2 | 28 | Female | Yes | ++ | EX |
| 15 | Mongrel | 12 | 15 | Female | Yes | +++ | EX |
| 16 | Mongrel | 11 | 30 | Female | Yes | +++ | EX |
| 17 | Kurzhaar | 2.5 | 30 | Male | No | none | UE |
| 18 | Rhodesian ridgeback | 7 | 40 | Male | No | none | UE |
| 19 | German shepherd | 6.5 | 34.5 | Male | No | none | UE |
| 20 | Kurzhaar | 5 | 27.5 | Male | No | none | UE |
| 21 | Bracco italiano | 7.5 | 31.6 | Male | No | none | UE |
| 22 | German shepherd | 4 | 32.5 | Male | No | none | UE |
| 23 | Bernese Mountain Dog | 3 | 51.8 | Male | No | none | UE |
| 24 | Kurzhaar | 6.5 | 28.4 | Male | No | none | UE |
| 25 | Hovawart | 3 | 31 | Male | No | none | UE |
| 26 | Mongrel | 1.5 | 16 | Male | No | none | UE |
| 27 | Belgian shepherd | 8 | 20.7 | Female | No | none | UE |
| 28 | Bernese Mountain Dog | 2 | 47.5 | Female | No | none | UE |
| 29 | Mongrel | 0.5 | 10 | Female | No | none | UE |
| 30 | Bouledogue | 2 | 17.6 | Female | No | none | UE |
| 31 | Mongrel | 0.5 | 11.6 | Female | No | none | UE |
| 32 | Mongrel | 0.5 | 10 | Female | No | none | UE |
| Parameter | Exposed Dogs (EX) | Unexposed Dogs (UE) | ||
|---|---|---|---|---|
| Cotinine (ng/mL) | Cotinine (ng/mL) | |||
| serum | hair | serum | hair | |
| 7.4 ± 3.7 | 4.4 ± 3.9 | 1.5 ± 0.7 | 0.9 ± 0.3 | |
| Intensity + | 4.3 ± 1.3 | 2.2 ± 0.6 | - | - |
| Intensity ++ | 9.4 ± 2.9 | 6.2 ± 5.8 | - | - |
| Intensity +++ | 7.8 ± 3.9 | 4.6 ± 3.9 | - | - |
| Age 1 | 6.5 ± 1.3 | 5.5 ± 5.03 | 0.9 ± 0.6 | 0.7 ± 0.2 |
| Age 2 | 7.5 ± 4.6 | 3.1 ± 1.04 | 2.1 ± 0.4 | 1.2 ± 0.3 |
| Age 3 | 8.3 ± 3.5 | 6.02 ± 6.2 | 1.6 ± 0.3 | 0.9 ± 0.2 |
| BW 1 | 8.6 | 5.1 | 0.9 ± 0.6 | 0.8 ± 0.2 |
| BW 2 | 8.3 ± 3.9 | 4.6 ± 3.2 | 1.7 ± 0.3 | 0.9 ± 0.2 |
| BW 3 | 5.9 ± 3.4 | 4.1 ± 5.3 | 1.8 ± 0.6 | 1.1 ± 0.4 |
| Male | 7.2 ± 3.9 | 2.7 ± 0.9 | 1.9 ± 0.4 | 1.1 ± 0.3 |
| Female | 7.8 ± 3.4 | 7.3 ± 5.2 | 0.8 ± 0.4 | 0.6 ± 0.1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Groppetti, D.; Pizzi, G.; Pecile, A.; Bronzo, V.; Mazzola, S.M. Cotinine as a Sentinel of Canine Exposure to Tobacco Smoke. Animals 2023, 13, 693. https://doi.org/10.3390/ani13040693
Groppetti D, Pizzi G, Pecile A, Bronzo V, Mazzola SM. Cotinine as a Sentinel of Canine Exposure to Tobacco Smoke. Animals. 2023; 13(4):693. https://doi.org/10.3390/ani13040693
Chicago/Turabian StyleGroppetti, Debora, Giulia Pizzi, Alessandro Pecile, Valerio Bronzo, and Silvia Michela Mazzola. 2023. "Cotinine as a Sentinel of Canine Exposure to Tobacco Smoke" Animals 13, no. 4: 693. https://doi.org/10.3390/ani13040693
APA StyleGroppetti, D., Pizzi, G., Pecile, A., Bronzo, V., & Mazzola, S. M. (2023). Cotinine as a Sentinel of Canine Exposure to Tobacco Smoke. Animals, 13(4), 693. https://doi.org/10.3390/ani13040693







