The Intriguing Biogeographic Pattern of the Italian Wall Lizard Podarcis siculus (Squamata: Lacertidae) in the Tuscan Archipelago Reveals the Existence of a New Ancient Insular Clade
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
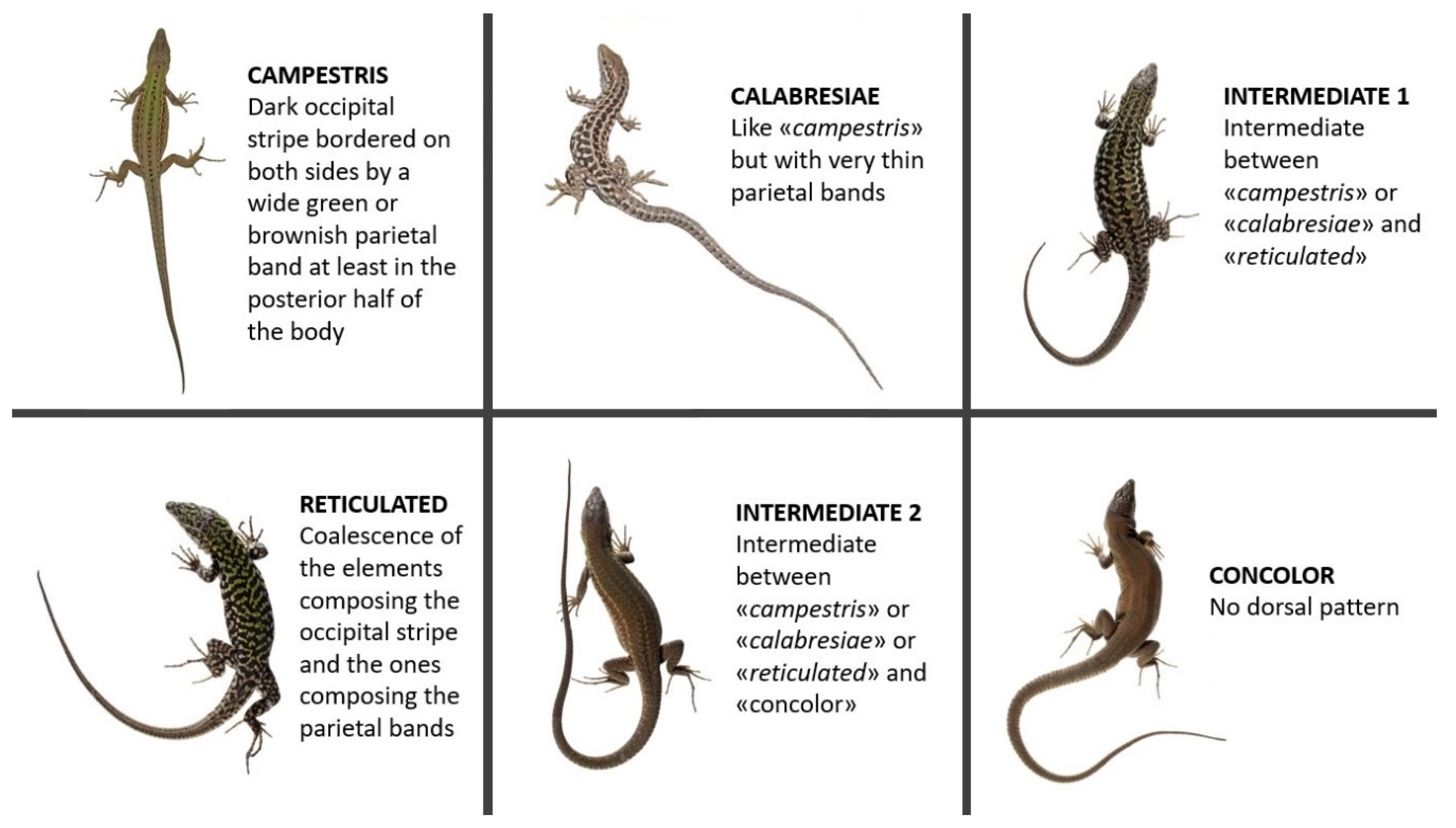
2.1. Dorsal Pattern Diversity
2.2. Morphological Analysis
2.3. Genetic Analysis
3. Results
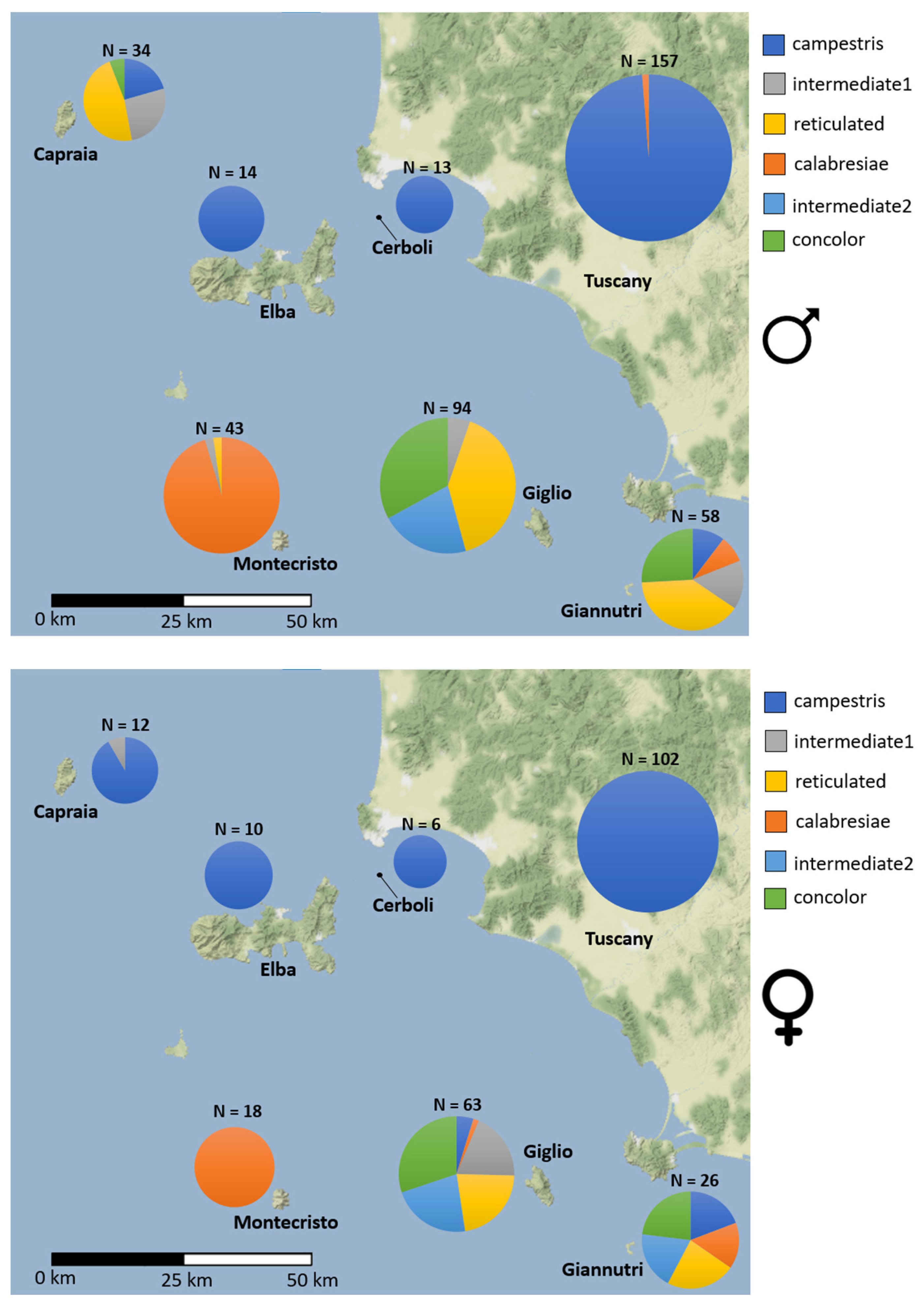
3.1. Dorsal Pattern
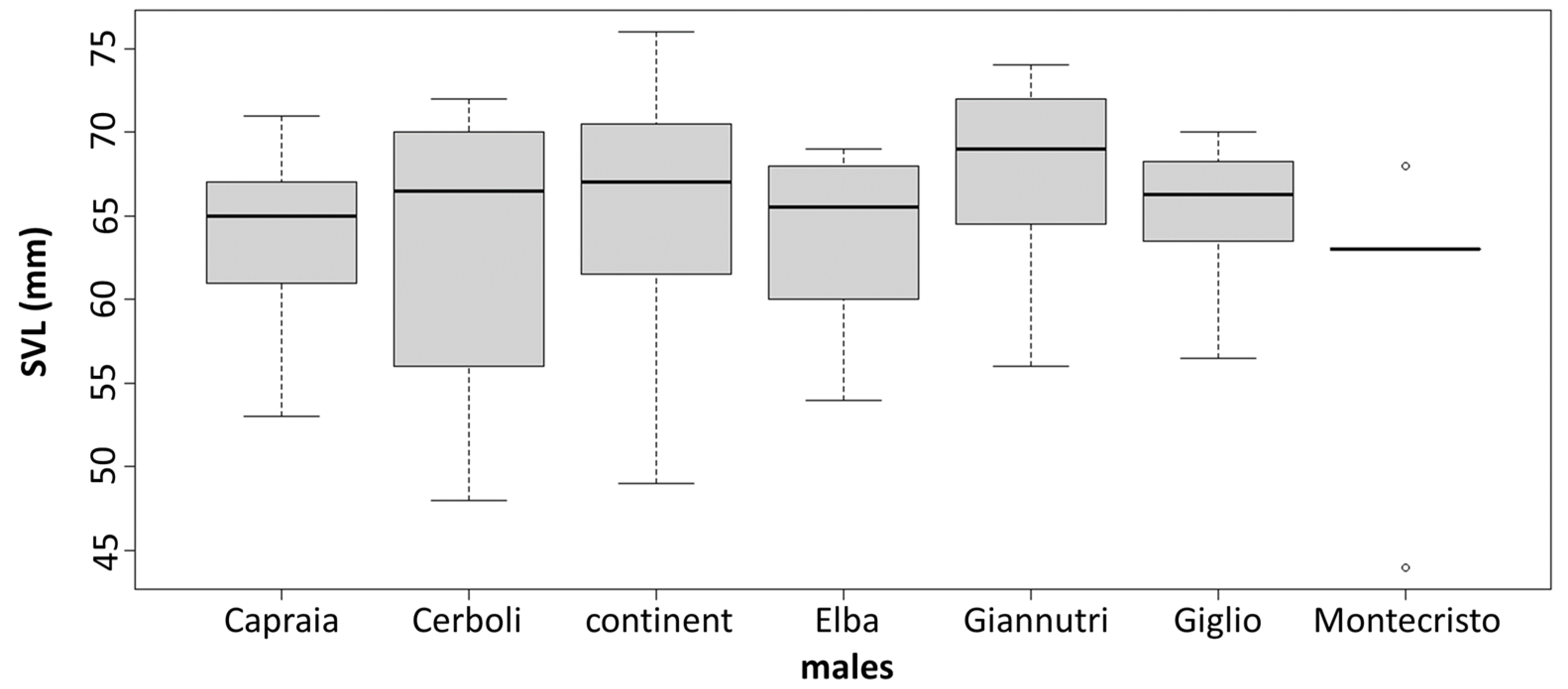
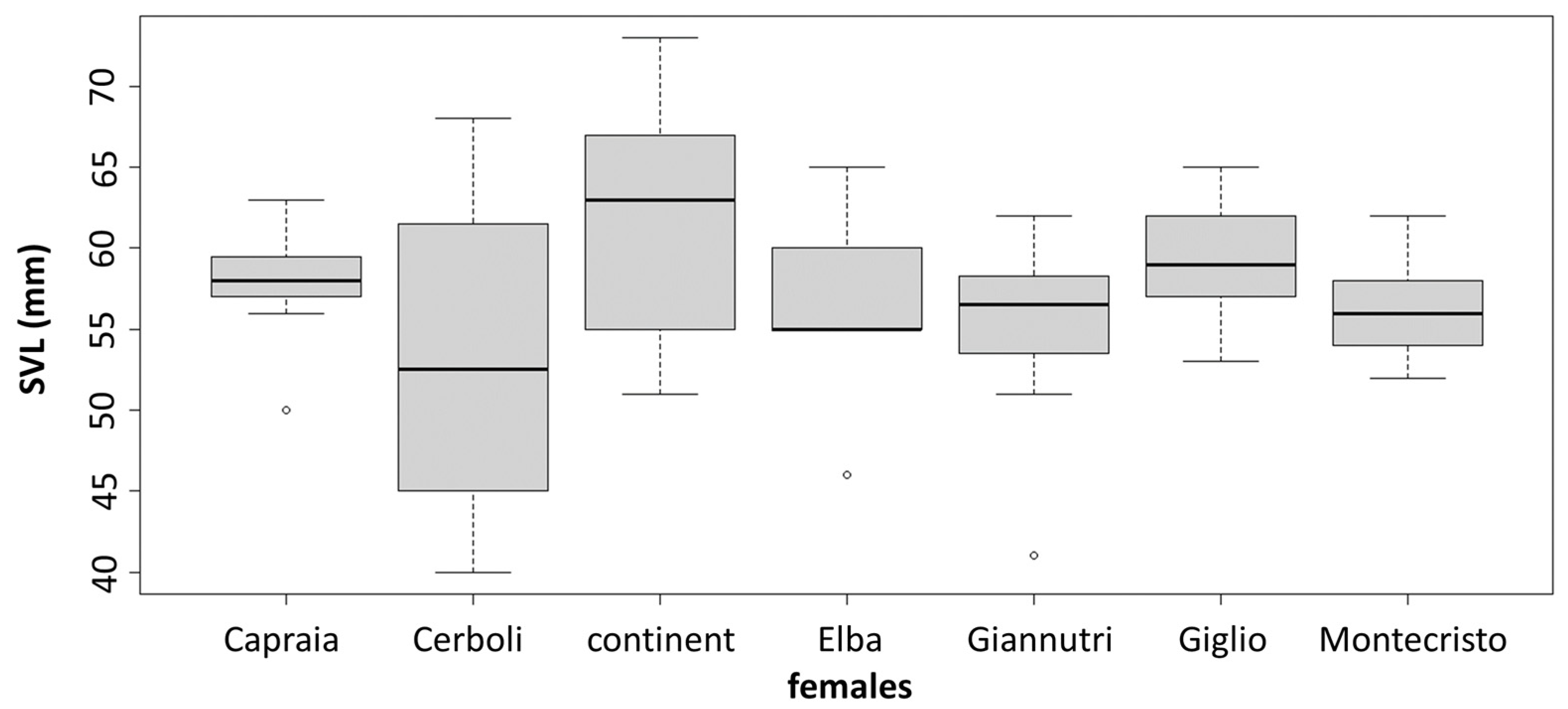
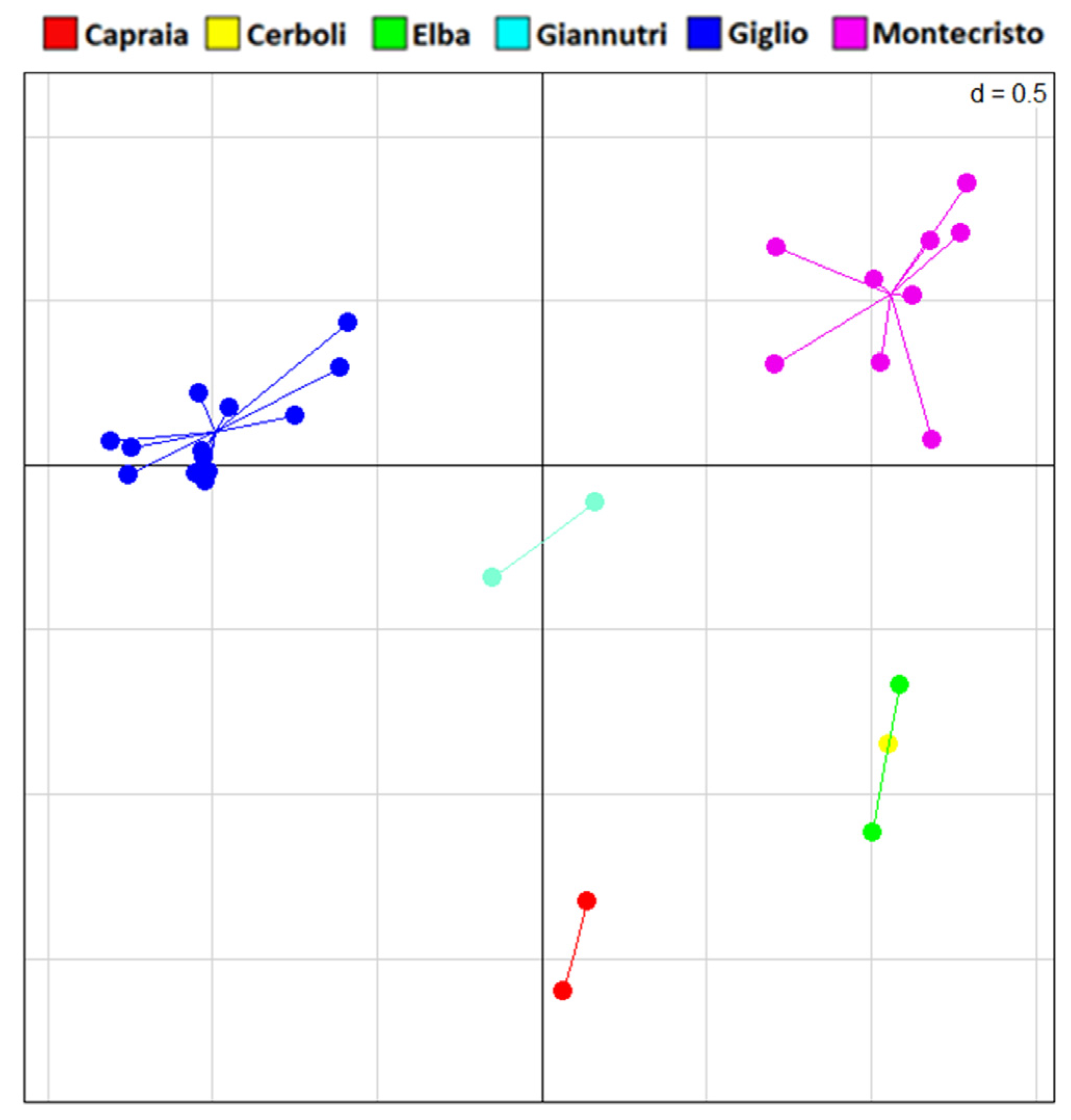
3.2. Morphological Analysis
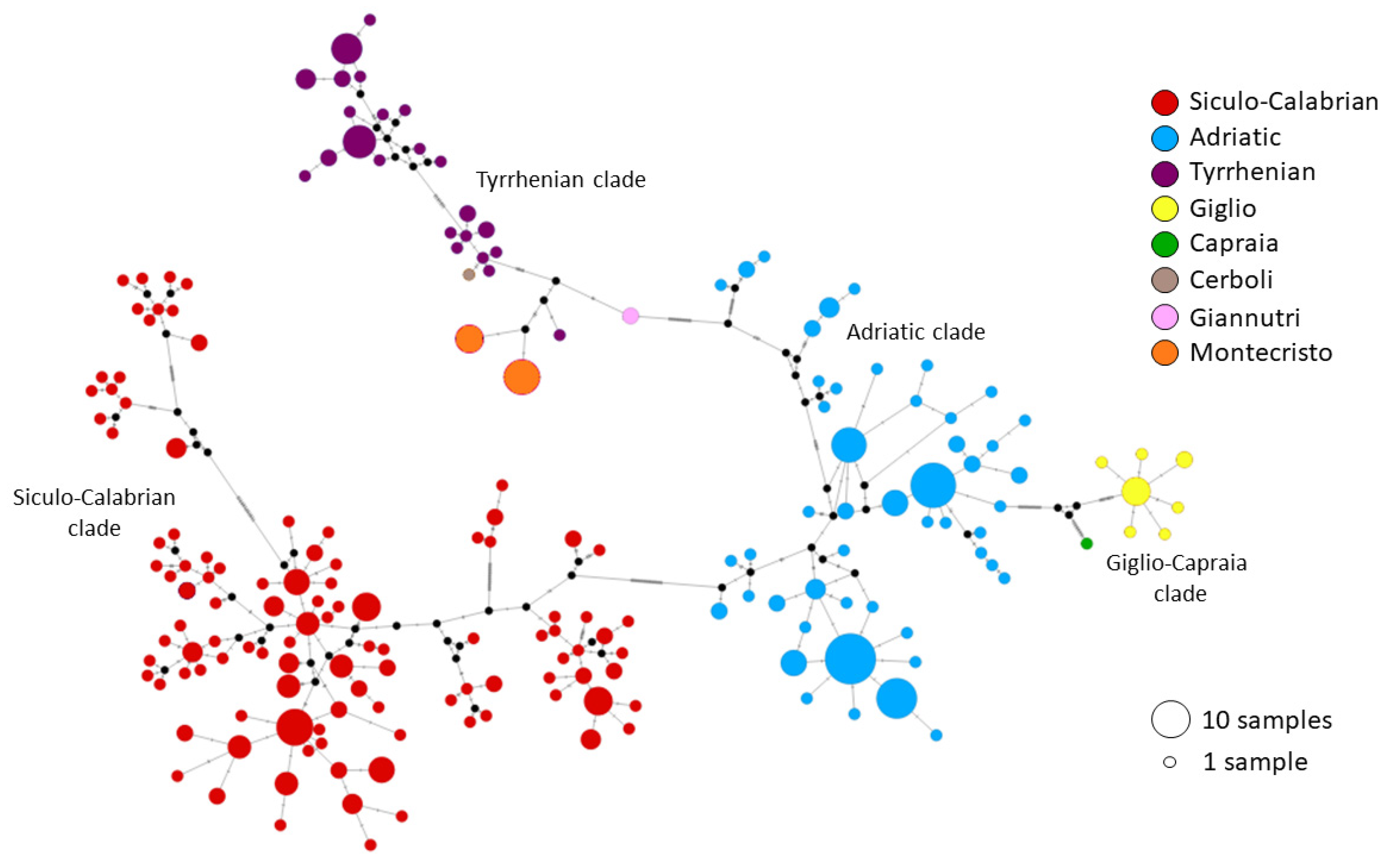
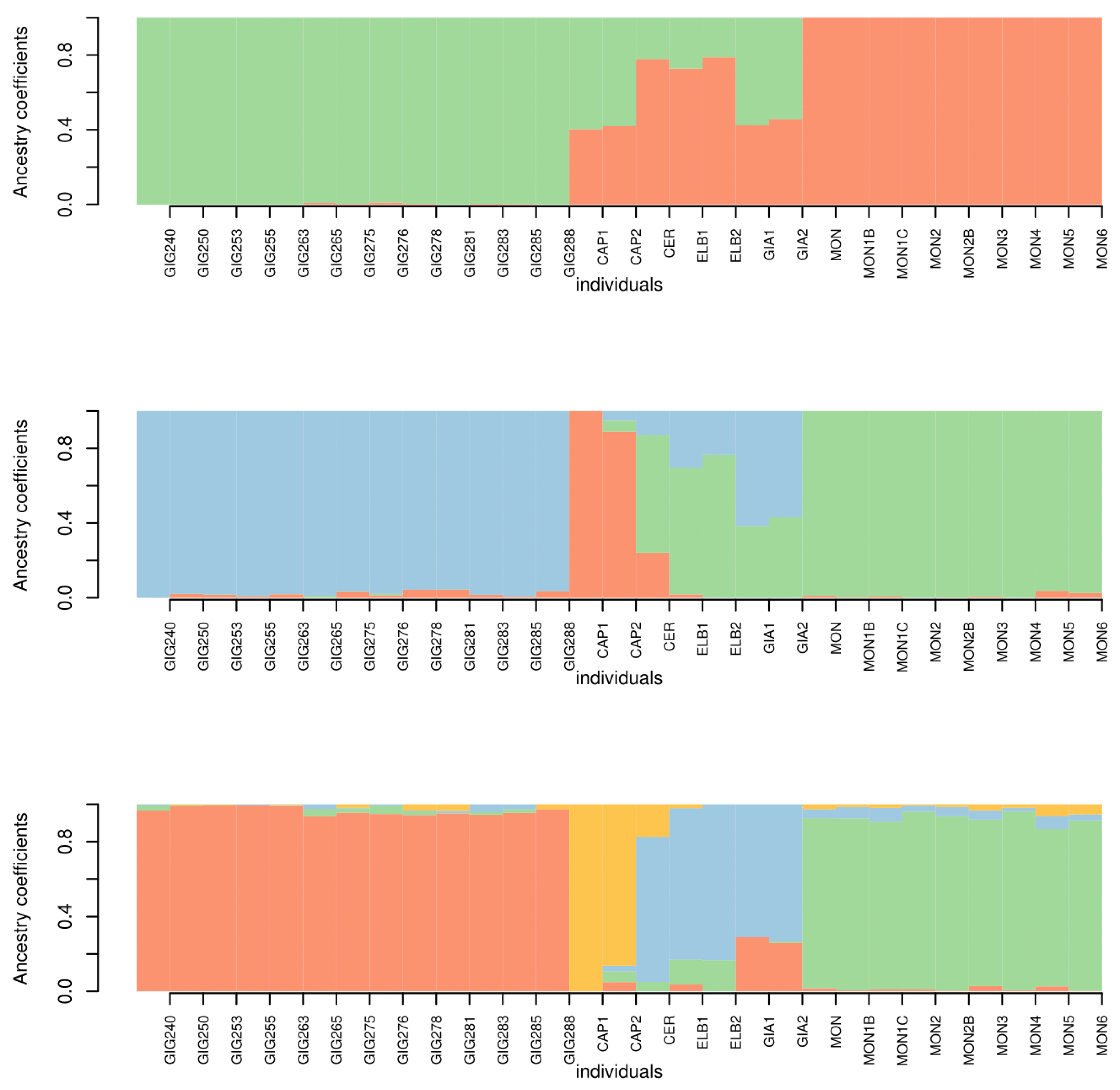
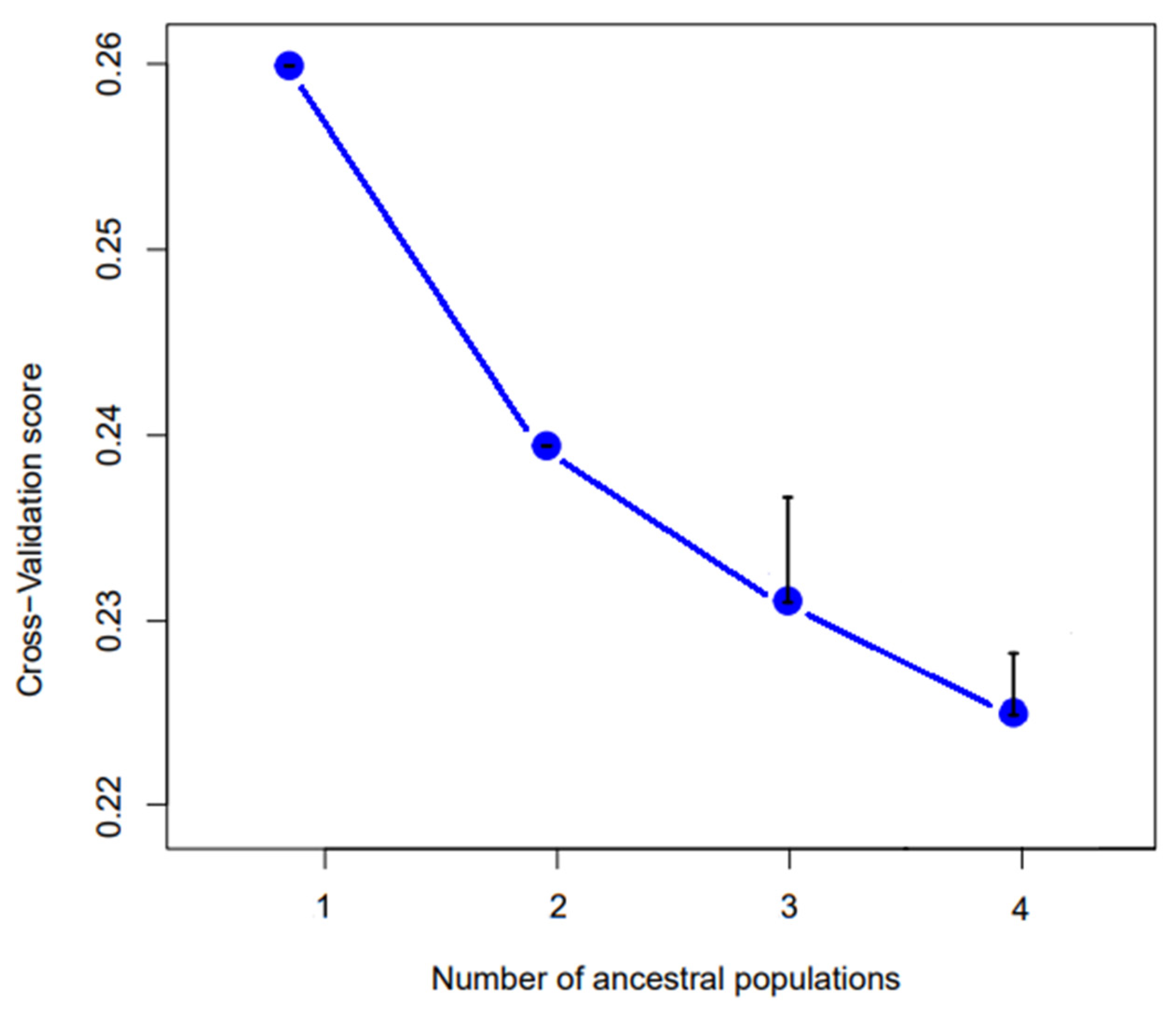
3.3. Genetic Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Brüniche-Olsen, A.; Kellner, K.F.; DeWoody, J.A. Island area, body size and demographic history shape genomic diversity in Darwin’s finches and related tanagers. Mol. Ecol. 2019, 28, 4914–4925. [Google Scholar] [CrossRef] [PubMed]
- Gentile, G.; Argano, R. Island biogeography of the Mediterranean Sea: The species-area relationship for terrestrial isopods. J. Biogeogr. 2005, 32, 1715–1726. [Google Scholar] [CrossRef]
- Vodă, R.; Dapporto, L.; Dincă, V.; Shreeve, T.G.; Khaldi, M.; Barech, G.; Rebbas, K.; Sammut, P.; Scalercio, S.; Hebert, P.D.N.; et al. Historical and contemporary factors generate unique butterfly communities on islands. Sci. Rep. 2016, 6, 28828. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dapporto, L.; Cini, A.; Menchetti, M.; Vodă, R.; Bonelli, S.; Casacci, L.P.; Dincă, V.; Scalercio, S.; Hinojosa, J.C.; Biermann, H.; et al. Rise and fall of island butterfly diversity: Understanding genetic differentiation and extinction in a highly diverse archipelago. Divers. Distrib. 2017, 23, 1169–1181. [Google Scholar] [CrossRef]
- Dapporto, L.; Cini, A. Faunal patterns in Tuscan archipelago butterflies (Lepidoptera): The dominant influence is recent geography not paleogeography. Eur. J. Entomol. 2007, 104, 497–503. [Google Scholar] [CrossRef] [Green Version]
- Fattorini, S. Both Recent and Pleistocene geography determine animal distributional patterns in the Tuscan Archipelago. J. Zool. 2009, 277, 291–301. [Google Scholar] [CrossRef]
- Lazzaro, L.; Ferretti, G.; Giuliani, C.; Foggi, B. A checklist of the alien flora of the Tuscan Archipelago (Italy). Webbia 2014, 69, 157–176. [Google Scholar] [CrossRef]
- Ruzzier, E.; Menchetti, M.; Bortolotti, L.; Selis, M.; Monterastelli, E.; Forbicioni, L. Updated distribution of the invasive Megachile sculpturalis (Hymenoptera: Megachilidae) in Italy and its first record on a Mediterranean island. Biodivers. Data J. 2020, 8, e57783. [Google Scholar] [CrossRef]
- Fattorini, S. Influence of recent geography and paleogeography on the structure of reptile communities in a land-bridge archipelago. J. Herpetol. 2010, 44, 242–252. [Google Scholar] [CrossRef]
- Olden, J.D.; Poff, N.L. Ecological processes driving biotic homogenization: Testing a mechanistic model using fish faunas. Ecology 2004, 85, 1867–1875. [Google Scholar] [CrossRef] [Green Version]
- Jeschke, J.M.; Bacher, S.; Blackburn, T.M.; Dick, J.T.A.; Essl, F.; Evans, T.; Gaertner, M.; Hulme, P.E.; Kühn, I.; Mrugała, A.; et al. Defining the Impact of Non-Native Species. Conserv. Biol. 2014, 28, 1188–1194. [Google Scholar] [CrossRef] [Green Version]
- Nogué, S.; de Nascimento, L.; Froyd, C.A.; Wilmshurst, J.M.; de Boer, E.J.; Coffey, E.E.D.; Whittaker, R.J.; Fernández-Palacios, J.M.; Willis, K.J. Island biodiversity conservation needs palaeoecology. Nat. Ecol. Evol. 2017, 1, 181. [Google Scholar] [CrossRef] [PubMed]
- Somenzi, E.; Senczuk, G.; Ciampolini, R.; Cortellari, M.; Vajana, E.; Tosser-Klopp, G.; Pilla, F.; Ajmone-Marsan, P.; Crepaldi, P.; Colli, L. The SNP-Based Profiling of Montecristo Feral Goat Populations Reveals a History of Isolation, Bottlenecks, and the Effects of Management. Genes 2022, 13, 213. [Google Scholar] [CrossRef] [PubMed]
- Vanni, S.; Nistri, A. Atlante Degli Anfibi e Dei Rettili Della Toscana; Museo di Storia Naturale dell’Università degli Studi di Firenze Sezione di Zoologia ‘‘La Specola’’: Firenze, Regione Toscana, Italy, 2006. [Google Scholar]
- Vaccaro, A.; Turrisi, G.F. Ritrovamento di Zamenis longissimus (Laurenti, 1768) (Reptilia, Colubridae) sull’Isola d’Elba (Toscana, Italia). Acta Herpetol. 2007, 2, 59–63. [Google Scholar] [CrossRef]
- Corti, C. Ricerche sulla variabilità morfologica e genetica della Podarcis sicula Rafinesque, 1810) dell’Arcipelago Toscano. Master’s Thesis, Università degli Studi di Firenze, Florence, Italy, 1987, unpublished. [Google Scholar]
- Corti, C.; Capula, M.; Nascetti, G. Biochemical taxonomy of the Podarcis sicula of the Tuscan Archipelago (Reptilia, Sauria, Lacertidae). In Proceedings of the 1st World Congress of Herpetology, Canterbury, UK, 11–19 September 1989. Abstracts: 75. [Google Scholar]
- Corti, C.; Böhme, W.; Delfino, M.; Masseti, M. Man and Lacertids on the Mediterranean Islands: Conservation perspectives. Nat. Croat. 1999, 8, 287–300. [Google Scholar]
- Harris, D.J.; Corti, C.; Pinho, C.; Carretero, M.; Böhme, W. Determination of genetic diversity within the insular lizard Podarcis tiliguerta using mtDNA sequence data, with a reassessment of the phylogeny of Podarcis. Amphibia-Reptilia 2005, 26, 401–407. [Google Scholar] [CrossRef] [Green Version]
- Lymberakis, P.; Poulakakis, N.; Kaliontzopoulou, A.; Valakos, E.; Mylonas, M. Two new species of Podarcis (Squamata; Lacertidae) from Greece. Syst. Biodivers. 2008, 6, 307–318. [Google Scholar] [CrossRef]
- Kaliontzopoulou, A.; Pinho, C.; Harris, J.D.; Carretero, M.A. When cryptic diversity blurs the picture: A cautionary tale from Iberian and North African Podarcis wall lizards. Biol. J. Linn. Soc. 2011, 103, 779–800. [Google Scholar] [CrossRef] [Green Version]
- Psonis, N.; Antoniou, A.; Kukushkin, O.; Jablonski, D.; Petrov, B. Hidden diversity in the Podarcis tauricus (Sauria, Lacertidae) species subgroup in the light of multilocus phylogeny and species delimitation. Mol. Phylogenetics Evol. 2017, 106, 6–17. [Google Scholar] [CrossRef]
- Rodríguez, V.; Buades, J.M.; Brown, R.P.; Terrasa, B.; Pérez-Mellado, V.; Corti, C.; Delaugerre, M.; Castro, J.A.; Picornell, A.; Ramon, M.M. Evolutionary history of Podarcis tiliguerta on Corsica and Sardinia. BMC Evol. Biol. 2017, 17, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Salvi, D.; Pinho, C.; Harris, D.J. Digging up the roots of an insular hotspot of genetic diversity: Decoupled mito-nuclear histories in the evolution of the Corsican-Sardinian endemic lizard Podarcis tiliguerta. BMC Evol. Biol. 2017, 17, 1–22. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Senczuk, G.; Colangelo, P.; De Simone, E.; Aloise, G.; Castiglia, R. A combination of long term fragmentation and glacial persistence drove the evolutionary history of the Italian wall lizard Podarcis siculus. BMC Evol. Biol. 2017, 17, 6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Senczuk, G.; Colangelo, P.; Avramo, V.; Castiglia, R.; Böhme, W.; Corti, C. A study in scarlet: Incipient speciation, phenotypic differentiation and conservation implications of the Podarcis lizards of the western Pontine Islands, Italy. Biol. J. Linn. Soc. 2018, 125, 50–60. [Google Scholar] [CrossRef]
- Senczuk, G.; Havenstein, K.; Milana, V.; Ripa, C.; De Simone, E.; Tiedemann, R.; Castiglia, R. Spotlight on islands: On the origin and diversification of an ancient lineage of the Italian wall lizard Podarcis siculus in the western Pontine Islands. Sci. Rep. 2018, 8, 15111. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Senczuk, G.; Castiglia, R.; Böhme, W.; Corti, C. Podarcis siculus latastei (Bedriaga, 1879) of the Western Pontine Islands (Italy) raised to the species rank, and a brief taxonomic overview of Podarcis lizards. Acta Herpetol. 2019, 14, 71–80. [Google Scholar] [CrossRef]
- Lanza, B.; Corti, C. Erpetofauna italiana: Acquisizioni ed estinzioni nel corso del Novecento. Suppl. Ric. Biol. Selvaggina 1993, 21, 5–49. [Google Scholar]
- Taddei, A. Le Lacerte (Podarcis) delle Isole dell’Arcipelago Toscano. Monit. Zool. Ital. Firenze 1949, 57, 12–34. [Google Scholar]
- Lunghi, E.; Bacci, F.; Zhao, Y. How can we record reliable information on animal colouration in the wild? Diversity 2021, 13, 356. [Google Scholar] [CrossRef]
- Salah, M.A.; Martinez, I. Universal and rapid salt-extraction of high quality genomic DNA for PCR-based techniques. Nucleic Acids Res. 1997, 25, 4692–4693. [Google Scholar] [CrossRef]
- Podnar, M.; Mayer, W.; Tvrtković, N. Phylogeography of the Italian wall lizard, Podarcis sicula, as revealed by mitochondrial DNA sequences. Mol. Ecol. 2005, 14, 575–588. [Google Scholar] [CrossRef]
- Pinho, C.; Sequeira, F.; Harris, D.J.; Godinho, R.; Ferrand, N. Isolation and characterization of nine microsatellite loci in Podarcis bocagei (Squamata: Lacertidae). Mol. Ecol. Notes 2004, 4, 286–288. [Google Scholar] [CrossRef]
- Nembrini, M.; Oppliger, A. Characterization of microsatellite loci in the wall lizard Podarcis muralis (Sauria: Lacer-tidae). Mol. Ecol. Resour. 2003, 3, 123–124. [Google Scholar] [CrossRef]
- Bloor, P.; Rodríguez, V.; Terrasa, B.; Brown, R.P.; Perez-Mellado, V.; Castro, J.A.; Ramon, M.M. Polymorphic microsatellite loci for the Balearic Island Lizard Podarcis lilfordi (Squamata: Lacertidae). Conserv. Genet. Resour. 2010, 3, 323–325. [Google Scholar] [CrossRef]
- Boudjemadi, K.; Martin, O.; Simon, J.C.; Estoup, A. Development and cross-species comparison of microsatellite markers in two lizard species, Lacerta vivipara and Podarcis muralis. Mol. Ecol. 1999, 8, 518–520. [Google Scholar]
- Edgar, R.C. MUSCLE: Multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 2004, 32, 1792–1797. [Google Scholar] [CrossRef] [Green Version]
- Tamura, K.; Stecher, G.; Kumar, S. MEGA11: Molecular Evolutionary Genetics Analysis Version 11. Mol. Biol. Evol. 2021, 38, 3022–3027. [Google Scholar] [CrossRef]
- Bandelt, H.J.; Forster, P.; Rohl, A. Median-joining networks for inferring intraspecific phylogenies. Mol. Biol. Evol. 1999, 16, 37–48. [Google Scholar] [CrossRef]
- French, N.; Yu, S.; Biggs, P.; Holland, B.; Fearnhead, P.; Binney, B.; Fox, A.; Grove-White, D.; Leigh, J.W.; Miller, W.; et al. Evolution of Campylobacter species in New Zealand. In Campylobacter Ecology and Evolution; Sheppard, S.K., Méric, G., Eds.; Caister Academic Press: Norfolk, UK, 2014; pp. 221–240. [Google Scholar] [CrossRef]
- Jombart, T.; Ahmed, I. adegenet 1.3-1: New tools for the analysis of genome-wide SNP data. Bioinformatics 2011, 27, 3070–3071. [Google Scholar] [CrossRef] [Green Version]
- Caye, K.; Deist, T.M.; Martins, H.; Michel, O.; François, O. TESS3: Fast inference of spatial population structure and genome scans for selection. Mol. Ecol. Resour. 2015, 16, 540–548. [Google Scholar] [CrossRef]
- Mertens, R. Zur Verbreitung uns Systematik einiger Lacerta-Formen der Apenninischen Halbinsel und der Tyrrhenischen Inserlwelt. Senckerbergiana 1932, 14, 235–259. [Google Scholar]
- Avramo, V.; Senczuk, G.; Corti, C.; Böhme, W.; Capula, M.; Castiglia, R.; Colangelo, P. Evaluating the island effect on phenotypic evolution in the Italian wall lizard, Podarcis siculus (Reptilia: Lacertidae). Biol. J. Linn. Soc. 2021, 132, 655–665. [Google Scholar] [CrossRef]
- Storniolo, F.; Zuffi, M.A.L.; Coladonato, A.J.; Di Vozzo, L.; Giglio, G.; Gini, A.E.; Leonetti, F.L.; Luccini, S.; Mangiacotti, M.; Scali, S.; et al. Patterns of variations in dorsal colouration of the Italian wall lizard Podarcis siculus. Biol. Open 2021, 10, bio058793. [Google Scholar] [CrossRef] [PubMed]
- Kaliontzopoulou, A.; Carretero, M.; Llorente, G.A. Morphology of the Podarcis wall lizards (Squamata: Lacertidae) from the Iberian Peninsula and North Africa: Patterns of variation in a putative cryptic species complex. Zool. J. Linn. Soc. 2011, 164, 173–193. [Google Scholar] [CrossRef] [Green Version]
- Gallozzi, F.; Colangelo, P.; Senczuk, G.; Castiglia, R. Phylogeographic and Bioclimatic Determinants of the Dorsal Pattern Polymorphism in the Italian Wall Lizard, Podarcis siculus. Diversity 2022, 14, 519. [Google Scholar] [CrossRef]
- Sacchi, R.; Mangiacotti, M.; Scali, S.; Sannolo, M.; Zuffi, M.A.L.; Pellitteri-Rosa, D.; Bellati, A.; Galeotti, P.; Fasola, M. Context-dependent expression of sexual dimorphism in island populations of the common wall lizard (Podarcis muralis). Biol. J. Linn. Soc. 2015, 114, 552–565. [Google Scholar] [CrossRef] [Green Version]
- Sacchi, R.; Ghitti, M.; Scali, S.; Mangiacotti, M.; Zuffi, M.A.; Sannolo, M.; Coladonato, A.J.; Pasquesi, G.; Bovo, M.; Pellitteri-Rosa, D. Common wall lizard females (Podarcis muralis) do not actively choose males based on their colour morph. Ethology 2015, 121, 1145–1153. [Google Scholar] [CrossRef]
- Senczuk, G.; García, A.; Colangelo, P.; Annesi, F.; Castiglia, R. Morphometric and genetic divergence in island and mainland populations of Anolis nebulosus (Squamata: Polychrotidae) from Jalisco (Mexico): An instance of insular gigantism. Ital. J. Zool. 2014, 81, 204–214. [Google Scholar] [CrossRef]
- Avella, I.; Castiglia, R.; Senczuk, G. Who are you? The genetic identity of some insular populations of Hierophis viridiflavus sl from the Tyrrhenian Sea. Acta Herpetol. 2017, 12, 209–214. [Google Scholar] [CrossRef]
- Capula, M. Population genetics of a colonizing lizard: Loss of variability in introduced populations of Podarcis sicula. Cell. Mol. Life Sci. 1994, 50, 691–696. [Google Scholar] [CrossRef]
- Poulakakis, N.; Lymberakis, P.; Valakos, E.; Pafilis, P.; Zouros, E.; Mylonas, M. Phylogeography of Balkan wall lizard (Podarcis taurica) and its relatives inferred from mitochondrial DNA sequences. Mol. Ecol. 2005, 14, 2433–2443. [Google Scholar] [CrossRef]
- Poulakakis, N.; Lymberakis, P.; Antoniou, A.; Chalkia, D.; Zouros, E.; Mylonas, M.; Valakos, E. Molecular phylogeny and biogeography of the wall-lizard Podarcis erhardii (Squamata: Lacertidae). Mol. Phylogenetics Evol. 2003, 28, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, R.G.; Strickland, T.R.; Kolbe, J.J.; Falk, B.G.; Perry, G.; Revell, L.J.; Losos, J.B. Archipelagic genetics in a widespread Caribbean anole. J. Biogeogr. 2017, 44, 2631–2647. [Google Scholar] [CrossRef]
- Bellati, A.; Pellitteri-Rosa, D.; Sacchi, R.; Nistri, A.; Galimberti, A.; Casiraghi, M.; Fasola, M.; Galeotti, P. Molecular survey of morphological subspecies reveals new mitochondrial lineages in Podarcis muralis (Squamata: Lacertidae) from the Tuscan Archipelago (Italy). J. Zool. Syst. Evol. Res. 2011, 49, 240–250. [Google Scholar] [CrossRef]
- Capula, M. Competitive exclusion between Podarcis lizards from Tyrrhenian islands: Inference from comparative species distributions. In Proceedings of the Sixth Ordinary General Meeting Societas Europaea Herpetologica, Budapest, Hungary, 19–23 August 1991; pp. 89–93. [Google Scholar]


| Island | mtDNA | Microsatellites | Meristic Data/SVL | Dorsal Pattern |
|---|---|---|---|---|
| Capraia | 1 | 2 | 43 M–9 F | 34 M–12 F |
| Cerboli | 1 | 1 | 16 M–5 F | 13 M–6 F |
| Elba | 0 | 2 | 17 M–8 F | 14 M–10 F |
| Giannutri | 1 | 2 | 31 M–19 F | 58 M–26 F |
| Giglio | 13 | 13 | 26 M–21 F | 94 M–63 F |
| Montecristo | 8 | 9 | 57 M–11 F | 43 M–18 F |
| mainland | 0 | 0 | 32 M–19 F | 157 M–102 F |
| MANOVA (Meristic Data) | df | Pillai’s Trace | Approx F | Num df | Den df | p-Value |
|---|---|---|---|---|---|---|
| SEX | 1 | 0.68117 | 47.003 | 9 | 198 | <0.05 |
| Locality | 6 | 2.04875 | 11.695 | 54 | 1218 | <0.05 |
| Residuals | 206 |
| ANOVA (SVL) | df | SS | MS | F Value | p-Value |
|---|---|---|---|---|---|
| Sex | 1 | 2828 | 2827.9 | 86.357 | <0.05 |
| Locality | 6 | 324 | 54.1 | 1.652 | >0.05 |
| Residuals | 206 | 6746 | 32.7 |
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | |
|---|---|---|---|---|---|---|---|---|
| - | - | - | - | - | - | - | - |
| 5.4% | - | - | - | - | - | - | - |
| 8.4% | 9.1% | - | - | - | - | - | - |
| 8.2% | 8.9% | 2.2% | - | - | - | - | - |
| 8.8% | 8.9% | 2.4% | 1.4% | - | - | - | - |
| 8.7% | 9.1% | 0.6% | 1.7% | 2.3% | - | - | - |
| 8.1% | 8.0% | 7.5% | 7.5% | 7.2% | 7.7% | - | - |
| 7.3% | 7.5% | 5.8% | 5.3% | 6.5% | 5.5% | 6.9% | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gallozzi, F.; Corti, C.; Castiglia, R.; Avramo, V.; Senczuk, G.; Mattioni, C.; Colangelo, P. The Intriguing Biogeographic Pattern of the Italian Wall Lizard Podarcis siculus (Squamata: Lacertidae) in the Tuscan Archipelago Reveals the Existence of a New Ancient Insular Clade. Animals 2023, 13, 386. https://doi.org/10.3390/ani13030386
Gallozzi F, Corti C, Castiglia R, Avramo V, Senczuk G, Mattioni C, Colangelo P. The Intriguing Biogeographic Pattern of the Italian Wall Lizard Podarcis siculus (Squamata: Lacertidae) in the Tuscan Archipelago Reveals the Existence of a New Ancient Insular Clade. Animals. 2023; 13(3):386. https://doi.org/10.3390/ani13030386
Chicago/Turabian StyleGallozzi, Francesco, Claudia Corti, Riccardo Castiglia, Vasco Avramo, Gabriele Senczuk, Claudia Mattioni, and Paolo Colangelo. 2023. "The Intriguing Biogeographic Pattern of the Italian Wall Lizard Podarcis siculus (Squamata: Lacertidae) in the Tuscan Archipelago Reveals the Existence of a New Ancient Insular Clade" Animals 13, no. 3: 386. https://doi.org/10.3390/ani13030386
APA StyleGallozzi, F., Corti, C., Castiglia, R., Avramo, V., Senczuk, G., Mattioni, C., & Colangelo, P. (2023). The Intriguing Biogeographic Pattern of the Italian Wall Lizard Podarcis siculus (Squamata: Lacertidae) in the Tuscan Archipelago Reveals the Existence of a New Ancient Insular Clade. Animals, 13(3), 386. https://doi.org/10.3390/ani13030386





