Exploring the Effect of Age on the Reproductive and Stress Physiology of Octopus bimaculoides Using Dermal Hormones
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethical Statement, Study Site, and Subjects
2.2. Swabbing Method
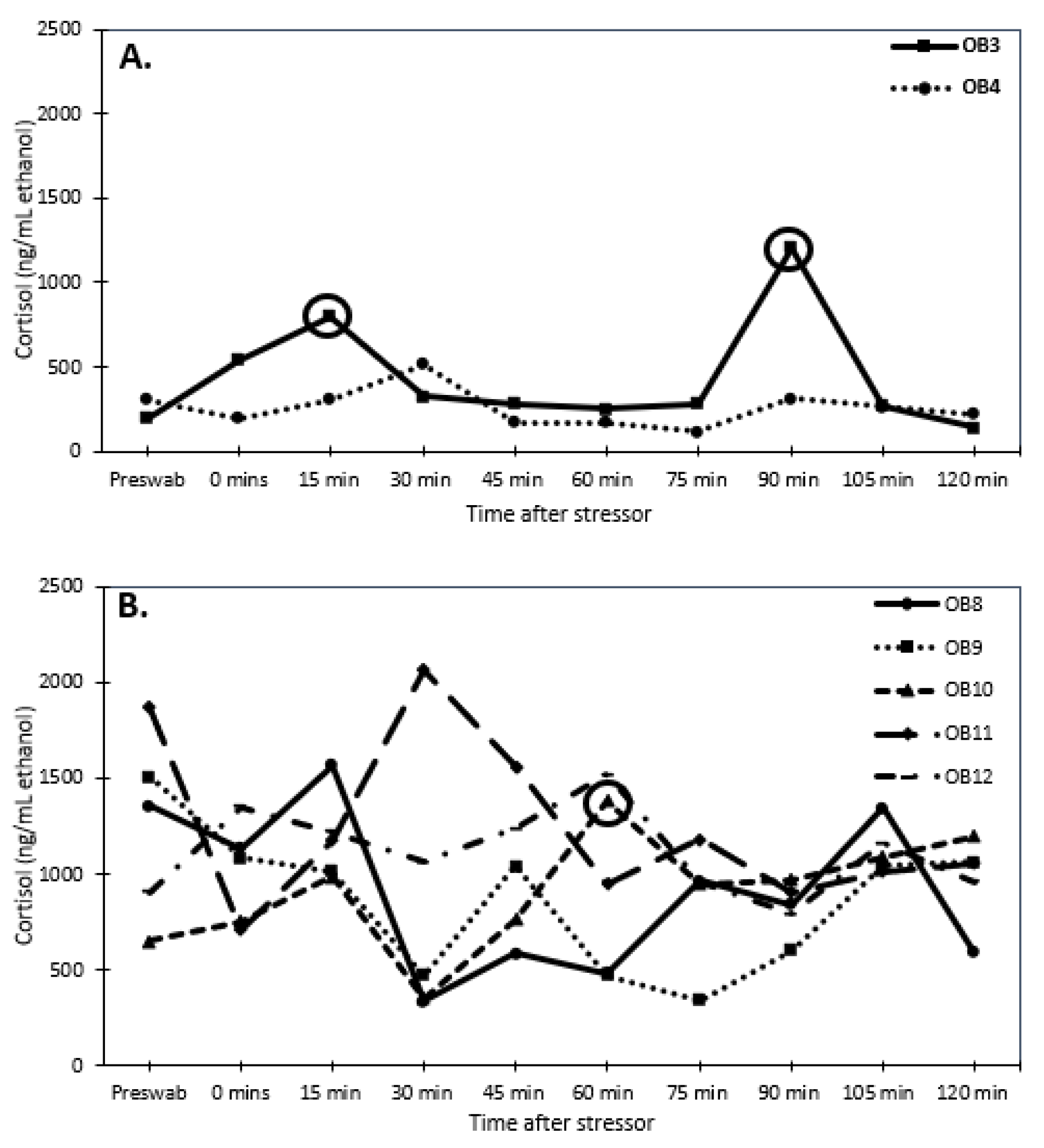
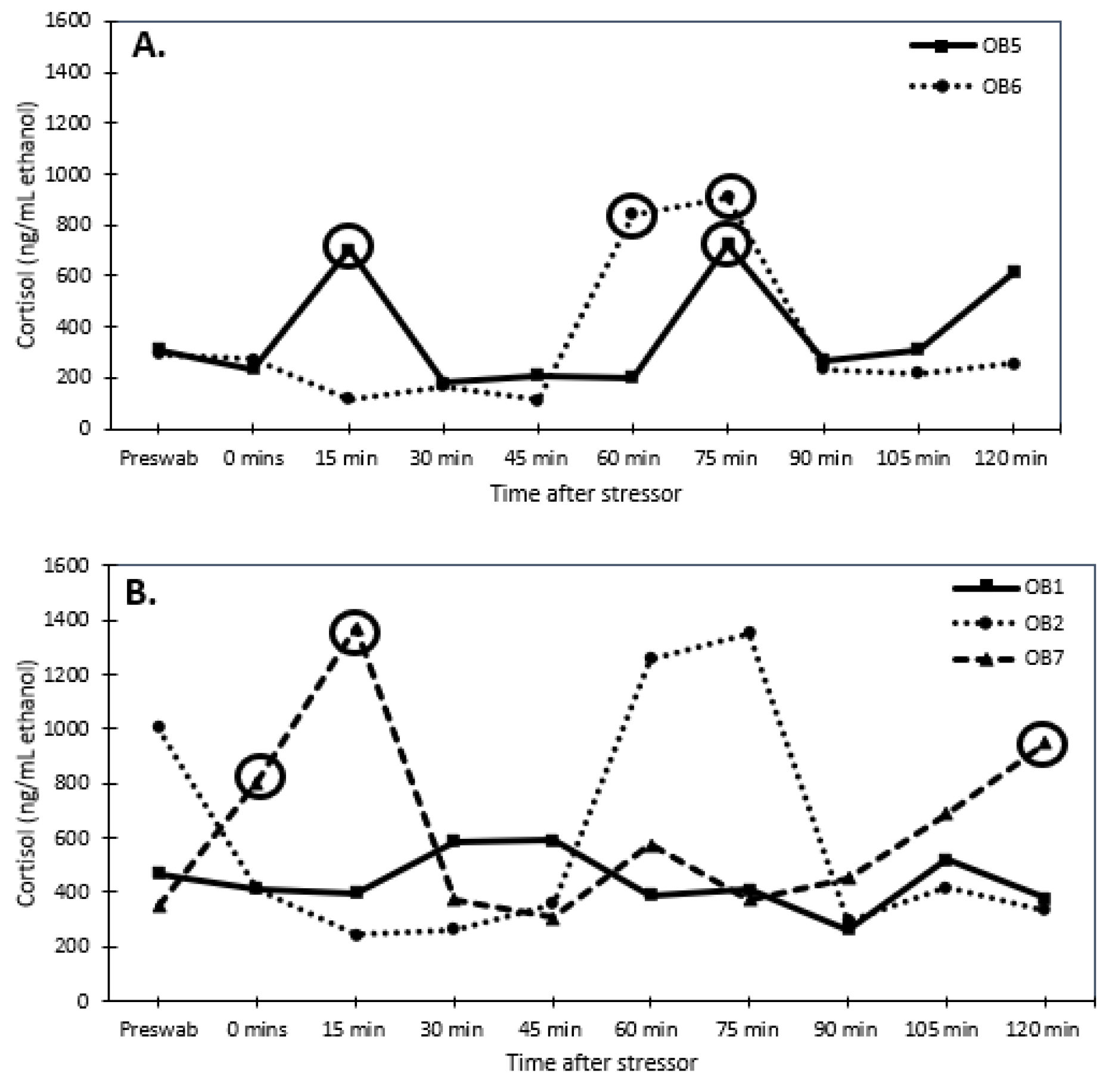
2.3. Validation of Glucocorticoid Analysis Using a Stress Test
2.4. Sample Processing and Hormonal Analysis
2.5. Data Analysis
3. Results
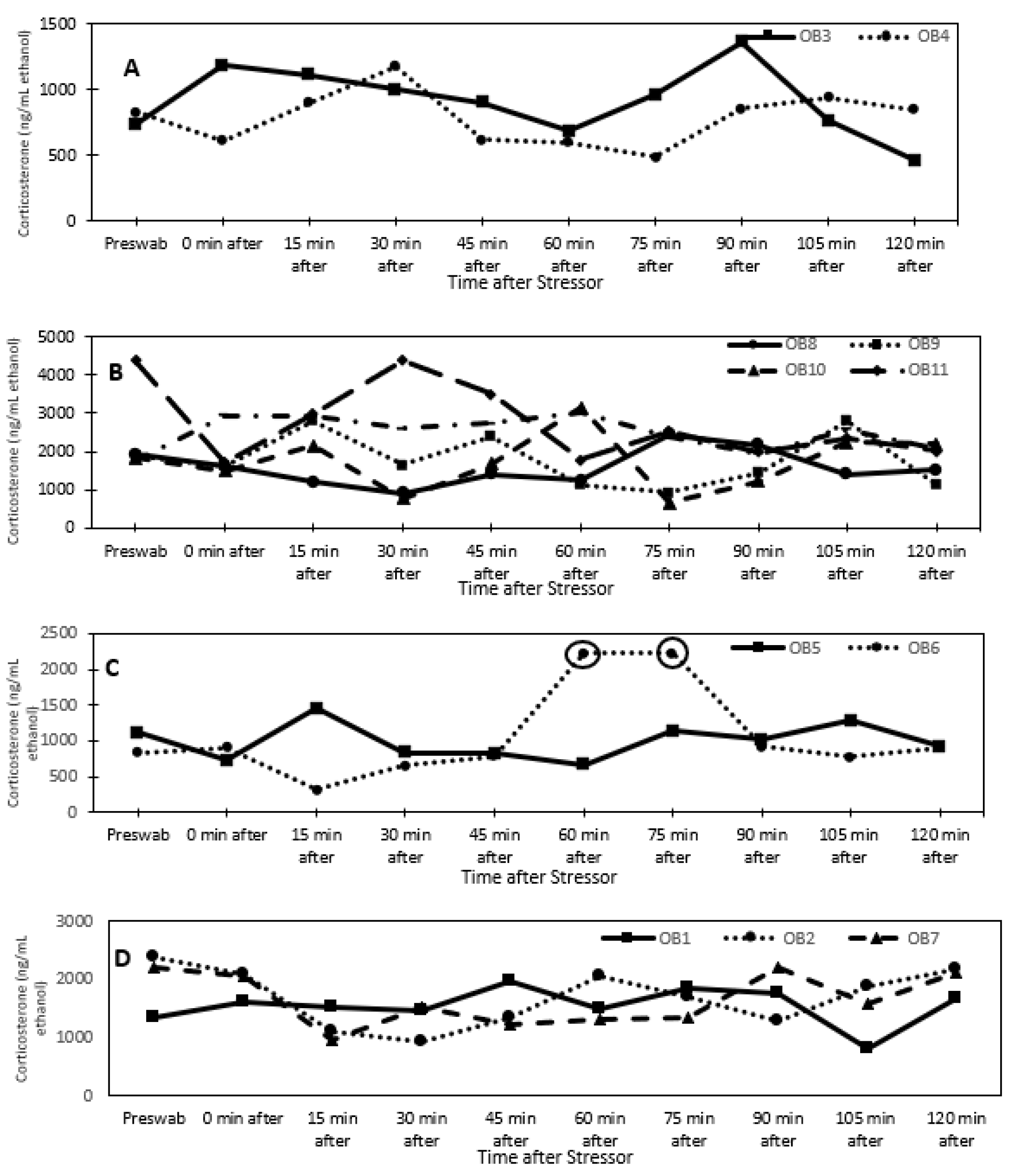
3.1. Glucocorticoid Analysis
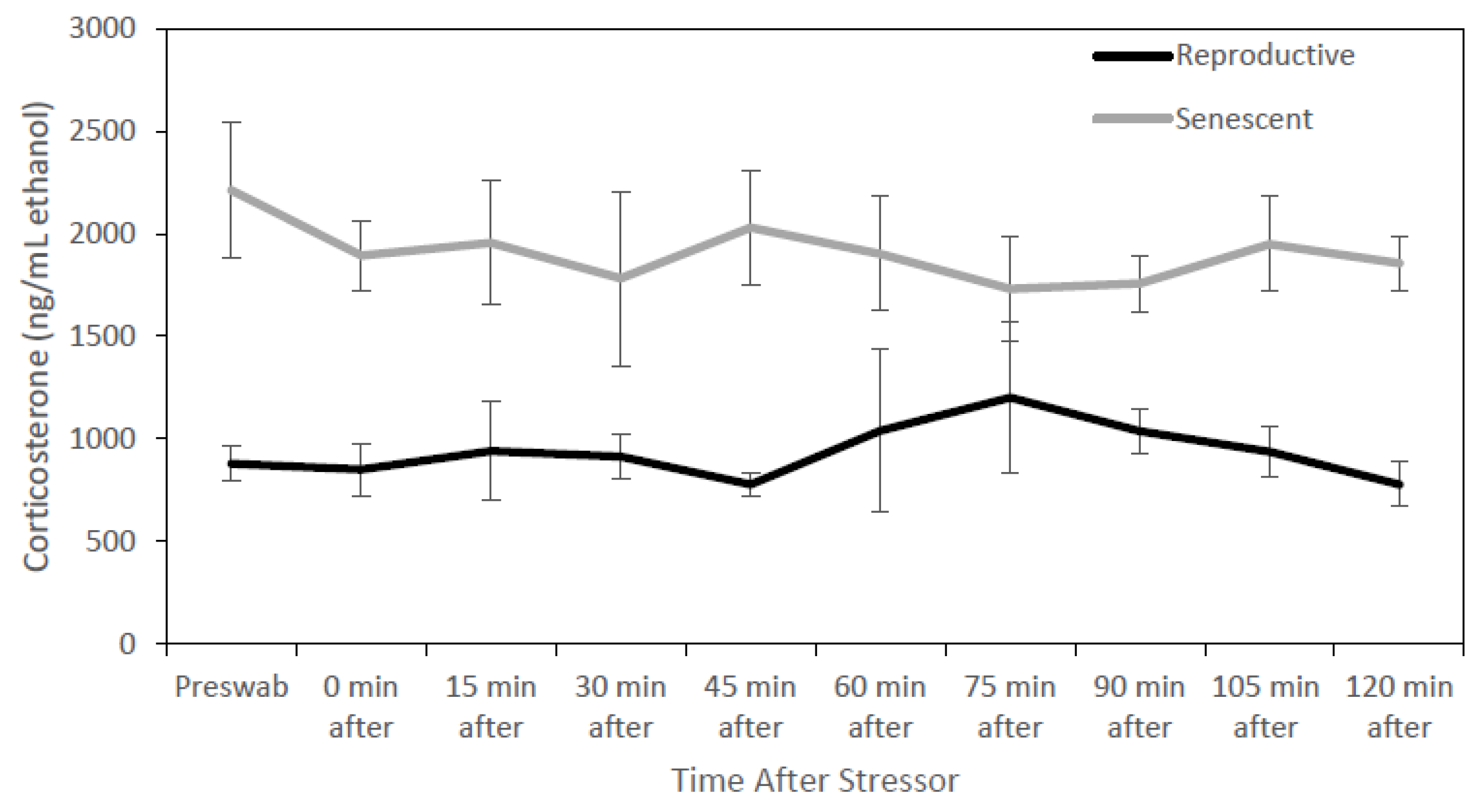
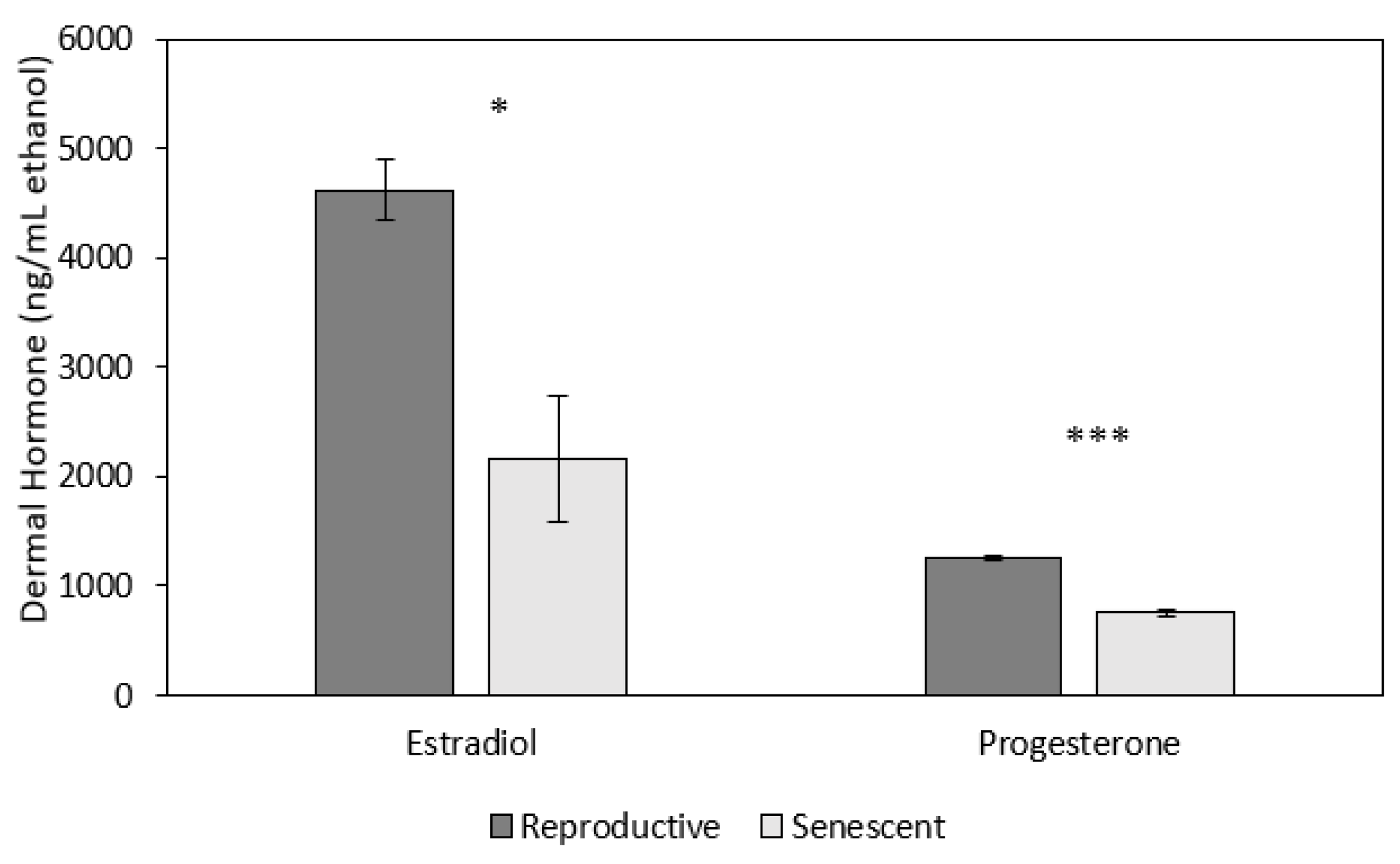
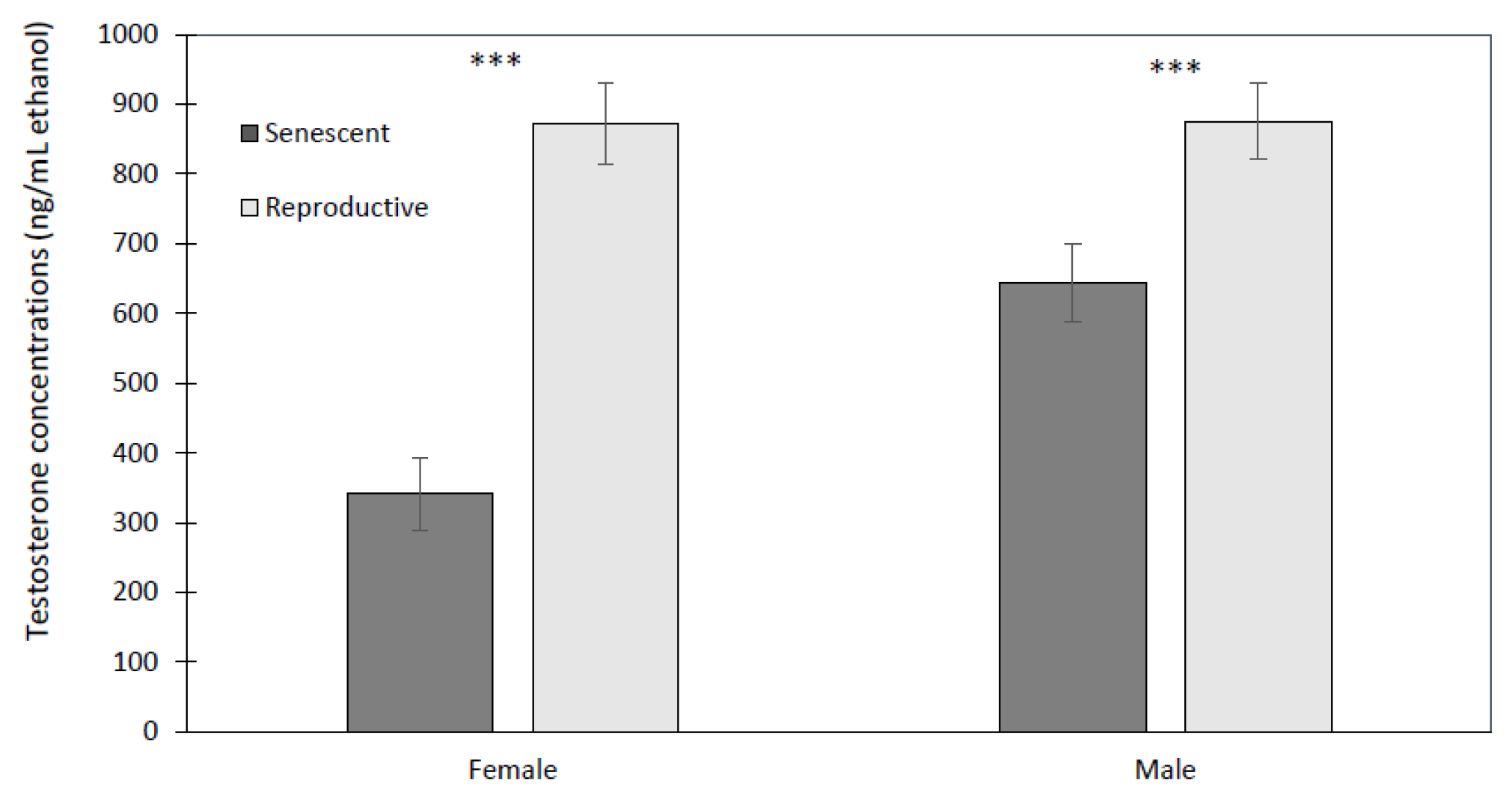
3.2. Effect of Age and Sex on Reproductive Hormones
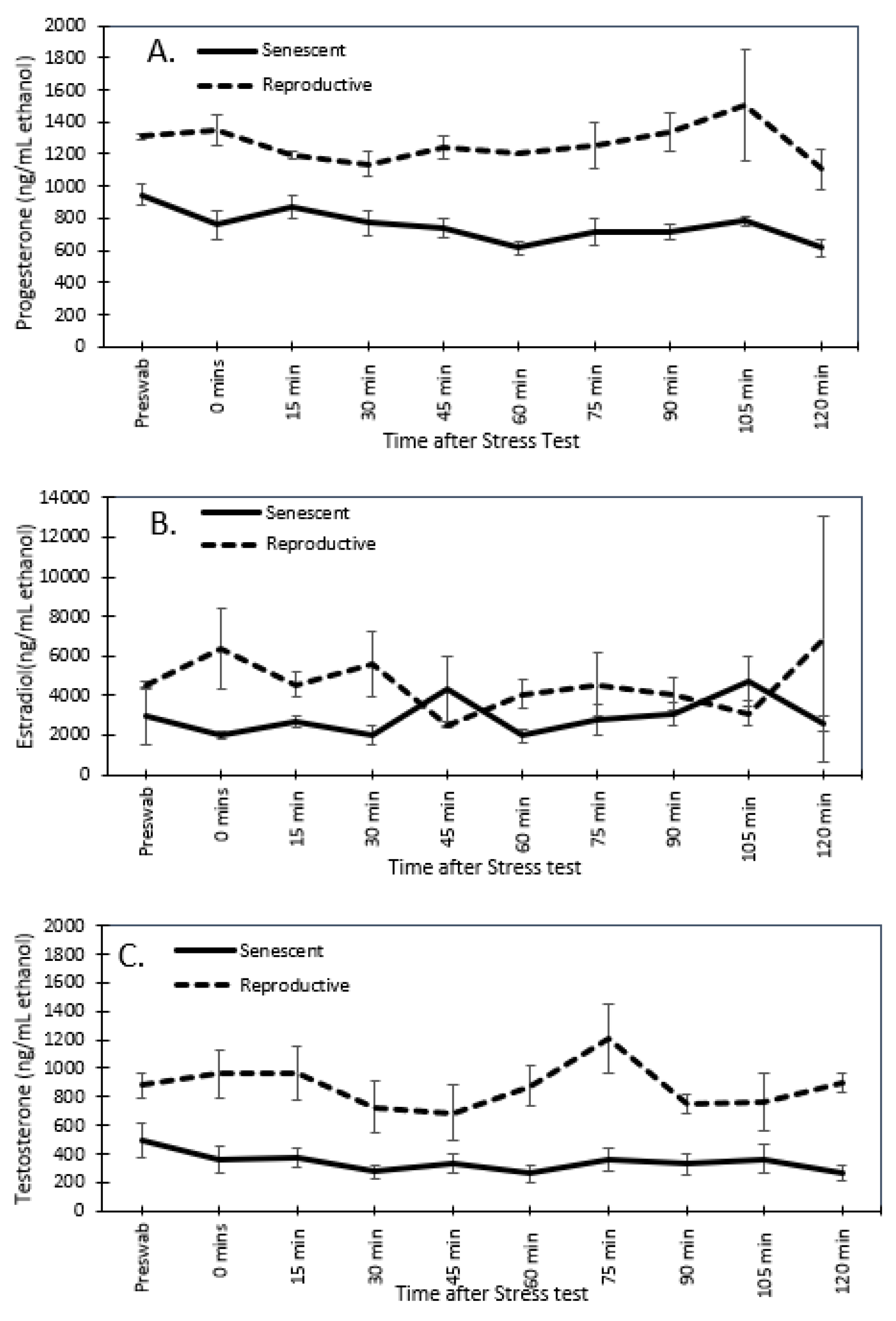
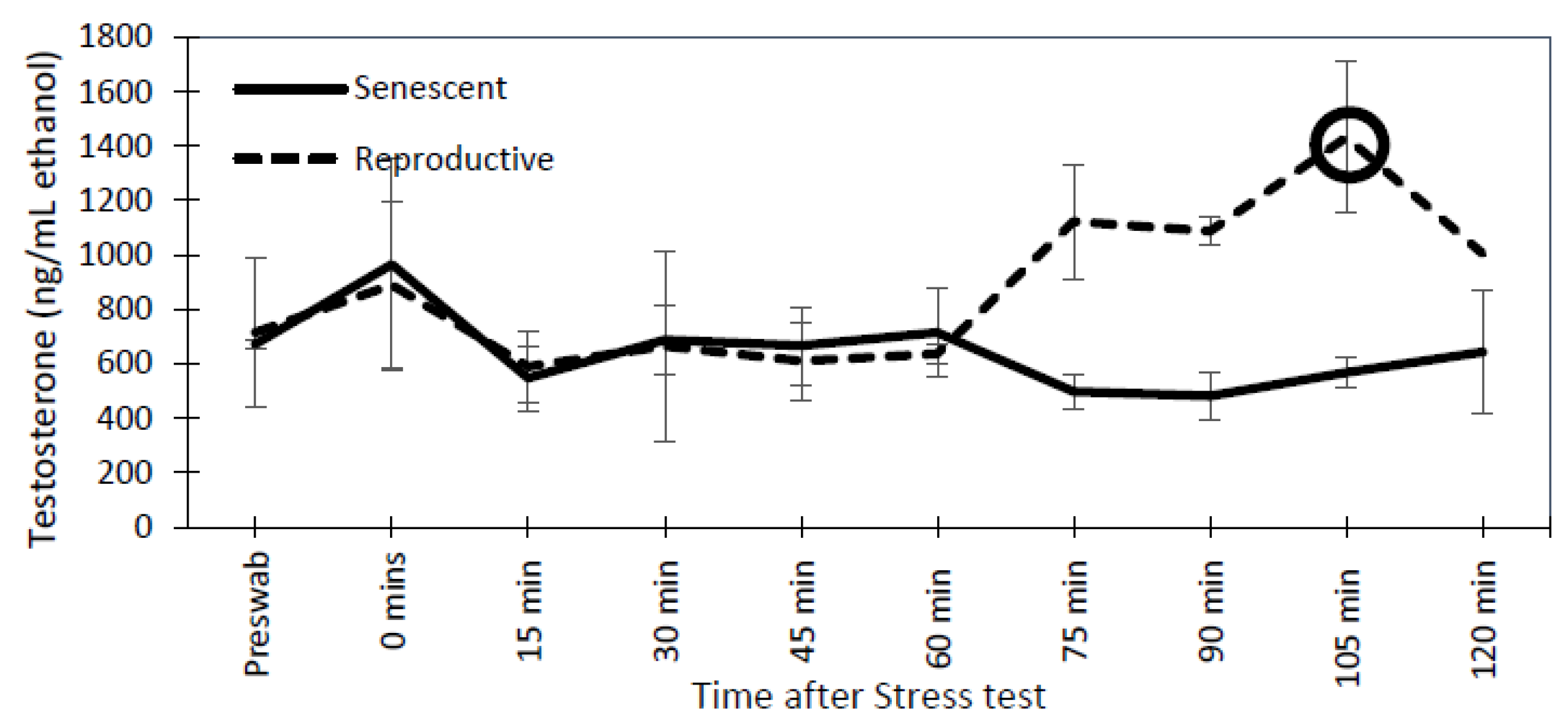
3.3. Effect of the Stress Test on Reproductive Hormones
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zylinski, S.; Johnsen, S. 10—Visual cognition in deep-sea cephalopods: What we don’t know and why we don’t know it. In Cephalopod Cognition; Cambridge University Press: Cambridge, UK, 2014; pp. 223–241. [Google Scholar]
- Chubb, C.; Chiao, C.C.; Ulmer, K.; Buresch, K.; Birk, M.A.; Hanlon, R.T. Dark scene elements strongly influence cuttlefish camouflage responses in visually cluttered environments. Vis. Res. 2018, 149, 86–101. [Google Scholar] [CrossRef] [PubMed]
- Mather, J.A.; Alupay, J.S. An ethogram for Benthic Octopods (Cephalopoda: Octopodidae). J. Comp. Psychol. 2016, 130, 109. [Google Scholar] [CrossRef]
- Hough, A.R.; Case, J.; Boal, J.G. Learned control of body patterning in cuttlefish Sepia officinalis (Cephalopoda). J. Molluscan Stud. 2016, 82, 427–431. [Google Scholar] [CrossRef]
- Mather, J.A. Behaviour Development: A Cephalopod Perspective. Int. J. Comp. Psychol. 2006, 19, 98–115. [Google Scholar] [CrossRef]
- Kuba, M.J.; Byrne, R.A.; Meisel, D.V.; Mather, J.A. When do octopuses play? Effects of repeated testing, object type, age, and food deprivation on object play in Octopus vulgaris. J. Comp. Psychol. 2006, 120, 184. [Google Scholar] [CrossRef]
- Richter, J.N.; Hochner, B.; Kuba, M.J. Pull or push? Octopuses solve a puzzle problem. PLoS ONE 2016, 11, e0152048. [Google Scholar] [CrossRef] [PubMed]
- Anderson, R.C.; Wood, J.B.; Byrne, R.A.; Octopus, R.A.B. Octopus Senescence: The Beginning of the End. J. Appl. Anim. Welf. Sci. 2002, 5, 275–283. [Google Scholar] [CrossRef]
- Hanlon, R.T.; Messenger, J.B. Cephalopod Behaviour, 2nd ed.; Cambridge University Press: Cambridge, UK, 2018. [Google Scholar]
- Huffard, C.L.; Caldwell, R.L.; Boneka, F. Male-male and male-female aggression may influence mating associations in wild octopuses (Abdopus aculeatus). J. Comp. Psychol. 2010, 124, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Wells, B.Y.M.J.; Wells, J. Hormonal Control of Sexual Maturity in Octopus. J. Exp. Biol. 1959, 36, 1–33. [Google Scholar] [CrossRef]
- Wang, Z.Y.; Ragsdale, C.W. Multiple optic gland signaling pathways implicated in octopus maternal behaviors and death. J. Exp. Biol. 2018, 221, jeb185751. [Google Scholar] [CrossRef]
- Minakata, H.; Shigeno, S.; Kano, N.; Haraguchi, S.; Osugi, T.; Tsutsui, K. Octopus gonadotrophin-releasing hormone: A multifunctional peptide in the endocrine and nervous systems of the cephalopod. J. Neuroendocrinol. 2009, 21, 322–326. [Google Scholar] [CrossRef] [PubMed]
- D’Aniello, A.; Di Cosmo, A.; Di Cristo, C.; Assisi, L.; Botte, V.; Di Fioe, M.M. Occurrence of Sex Steroid Hormones and Their Binding Protiens in Octopus vulgaris Lam. Biochem. Biophys. Res. Commun. 1996, 227, 782–788. [Google Scholar] [CrossRef] [PubMed]
- O’Dor, R.K.; Wells, M.J. Reproduction versus somatic growth: Hormonal control in Octopus vulgaris. J. Exp. Biol. 1978, 77, 15–31. [Google Scholar]
- Di Cosmo, A.; Di Cristo, C.; Paolucci, M. Sex steroid hormone fluctuations and morphological changes of the reproductive system of the female of Octopus vulgaris throughout the annual cycle. J. Exp. Zool. 2001, 289, 33–47. [Google Scholar] [CrossRef] [PubMed]
- Avila-Poveda, O.H.; Montes-Pérez, R.C.; Koueta, N.; Benítez-Villalobos, F.; Ramírez-Pérez, J.S.; Jimenez-Gutierrez, L.R.; Rosas, C. Seasonal changes of progesterone and testosterone concentrations throughout gonad maturation stages of the Mexican octopus, Octopus maya (Octopodidae: Octopus). Molluscan Res. 2015, 35, 161–172. [Google Scholar] [CrossRef]
- Gestal, C.; Pascual, S.; Guerra, Á.; Fiorito, G.; Vieites, J.M. Handbook of Pathogens and Diseases in Cephalopods; Springer: Berlin/Heidelberg, Germany, 2019. [Google Scholar] [CrossRef]
- Azevedo, A.; Bailey, L.; Bandeira, V.; Dehnhard, M.; Fonseca, C.; de Sousa, L.; Jewgenow, K. Age, sex and storage time influence hair cortisol levels in a wild mammal population. PLoS ONE 2019, 14, e0222963. [Google Scholar] [CrossRef]
- Monfort, S.L.; Mashburn, K.L.; Brewer, B.A.; Creel, S.R. Evaluating Adrenal Activity in African Wild Dogs (Lycaon pictus) by Fecal Corticosteroid Analysis. J. Zoo Wildl. Med. 1988, 29, 129–133. [Google Scholar]
- Romero, L.M. Physiological stress in ecology: Lessons from biomedical research. Trends Ecol. Evol. 2004, 19, 249–255. [Google Scholar] [CrossRef] [PubMed]
- Schell, C.J.; Young, J.K.; Lonsdorf, E.V.; Santymire, R.M. Anthropogenic and physiologically induced stress responses in captive coyotes. J. Mammal. 2013, 94, 1131–1140. [Google Scholar] [CrossRef]
- Touma, C.; Palme, R. Measuring fecal glucocorticoid metabolites in mammals and birds: The importance of validation. Ann. N. Y. Acad. Sci. 2005, 1046, 54–74. [Google Scholar] [CrossRef] [PubMed]
- Moberg, G.P.; Mench, J.A. The Biology of Animal Stress: Basic Principles and Implications for Animal Welfare; CABI: Wallingford, UK, 2000. [Google Scholar]
- Palme, R.; Rettenbacher, S.; Touma, C.; El-Bahr, S.M.; Moestl, E. Stress hormones in mammals and birds: Comparative aspects regarding metabolism, excretion, and noninvasive measurement in fecal samples. Ann. N. Y. Acad. Sci. 2005, 1040, 162–171. [Google Scholar] [CrossRef]
- Bechert, U.; Hixon, S.; Schmitt, D. Diurnal variation in serum concentrations of cortisol in captive African (Loxodonta africana) and Asian (Elephas maximus) elephants. Zoo Biol. 2021, 40, 458–471. [Google Scholar] [CrossRef]
- Larson, S.E.; Anderson, R.C. Fecal hormones measured within giant pacific octopuses Enteroctopus dofleini. J. Aquat. Anim. Health 2010, 22, 152–157. [Google Scholar] [CrossRef]
- Chancellor, S.; Abbo, L.; Grasse, B.; Sakmar, T.; Brown, J.S.; Scheel, D.; Santymire, R.M. Determining the effectiveness of using dermal swabs to evaluate the stress physiology of laboratory cephalopods: A preliminary investigation. Gen. Comp. Endocrinol. 2021, 314, 113903. [Google Scholar] [CrossRef] [PubMed]
- Vaz-Pires, P.; Seixas, P.; Barbosa, A. Aquaculture potential of the common octopus (Octopus vulgaris Cuvier, 1797): A review. Aquaculture 2004, 238, 221–238. [Google Scholar] [CrossRef]
- Vidal, E.A.; Villanueva, R.; Andrade, J.P.; Gleadall, I.G.; Iglesias, J.; Koueta, N.; Rosas, C.; Segawa, S.; Grasse, B.; Franco-Santos, R.M.; et al. Cephalopod culture: Current status of main biological models and research priorities. Adv. Mar. Biol. 2014, 67, 1–98. [Google Scholar] [PubMed]
- Malham, S.K.; Lacoste, A.; Gélébart, F.; Cueff, A.; Poulet, S.A. A first insight into stress-induced neuroendocrine and immune changes in the octopus Eledone cirrhosa. Aquat. Living Resour. 2002, 15, 187–192. [Google Scholar] [CrossRef]
- De Mercado, E.; Larrán, A.M.; Pinedo, J.; Tomás-Almenar, C. Skin mucous: A new approach to assess stress in rainbow trout. Aquaculture 2018, 484, 90–97. [Google Scholar] [CrossRef]
- Guardiola, F.A.; Cuesta, A.; Esteban, M.Á. Using skin mucus to evaluate stress in gilthead seabream (Sparus aurata L.). Fish Shellfish Immunol. 2016, 59, 323–330. [Google Scholar] [CrossRef] [PubMed]
- Schultz, D.R.; Perez, N.; Tan, C.K.; Mendez, A.J.; Capo, T.R.; Snodgrass, D.; Prince, E.D.; Serafy, J.E. Concurrent levels of 11-ketotestosterone in fish surface mucus, muscle tissue and blood. J. Appl. Ichthyol. 2005, 21, 394–398. [Google Scholar] [CrossRef]
- Santymire, R.M.; Young, M.; Lenihan, E.; Murray, M.J. Preliminary Investigation into Developing the Use of Swabs for Skin Cortisol Analysis for the Ocean Sunfish (Mola mola). Animals 2022, 12, 2868. [Google Scholar] [CrossRef]
- Santymire, R.M.; Manjerovic, M.B.; Sacerdote-Velat, A. A novel method for the measurement of glucocorticoids in dermal secretions of amphibians. Conserv. Physiol. 2018, 6, coy008. [Google Scholar] [CrossRef] [PubMed]
- Sykes, A.V.; Baptista, F.D.; Gonçalves, R.A.; Andrade, J.P. Directive 2010/63/EU on animal welfare: A review on the existing scientific knowledge and implications in cephalopod aquaculture research. Rev. Aquac. 2012, 4, 142–162. [Google Scholar] [CrossRef]
- Albertin, C.B.; Simakov, O.; Mitros, T.; Wang, Z.Y.; Pungor, J.R.; Edsinger-Gonzales, E.; Brenner, S.; Ragsdale, C.W.; Rokhsar, D.S. The octopus genome and the evolution of cephalopod neural and morphological novelties. Nature 2015, 524, 220–224. [Google Scholar] [CrossRef]
- Albertin, C.B.; Simakov, O. Cephalopod Biology: At the Intersection Between Genomic and Organismal Novelties. Annu. Rev. Anim. Biosci. 2020, 8, 71–90. [Google Scholar] [CrossRef]
- Boyle, P.R. Cephalopod Life Cycles. In Species Accounts; Academic Press: London, UK, 1983; Volume 1. [Google Scholar]
- Robin, J.P.; Roberts, M.; Zeidberg, L.; Bloor, I.; Rodriguez, A.; Briceño, F.; Downey, N.; Mascaro, M.; Navarro, M.; Guerra, A.; et al. Transitions during cephalopod life history: The role of habitat, environment, functional morphology and behaviour. Adv. Mar. Biol. 2014, 67, 361–437. [Google Scholar] [CrossRef] [PubMed]
- Forsythe, J.W.; Hanlon, R.T. Behavior, body patterning and reproductive biology of Octopus bimaculoides from California. Malacologia 1988, 29, 41–55. [Google Scholar]
- Fiorito, G.; Affuso, A.; Basil, J.; Cole, A.; de Girolamo, P.; D’angelo, L.; Dickel, L.; Gestal, C.; Grasso, F.; Kuba, M.; et al. Guidelines for the Care and Welfare of Cephalopods in Research–A consensus based on an initiative by CephRes, FELASA and the Boyd Group. Lab. Anim. 2015, 49 (Suppl. S2), 1–90. [Google Scholar] [CrossRef]
- Smith, J.A.; Andrews, P.L.; Hawkins, P.; Louhimies, S.; Ponte, G.; Dickel, L. Cephalopod research and EU Directive 2010/63/EU: Requirements, impacts and ethical review. J. Exp. Mar. Biol. Ecol. 2013, 447, 31–45. [Google Scholar] [CrossRef]
- Loeding, E.; Thomas, J.; Bernier, D.; Santymire, R. Using fecal hormonal and behavioral analyses to evaluate the introduction of two sable antelope at Lincoln Park Zoo. J. Appl. Anim. Welf. Sci. 2011, 14, 220–246. [Google Scholar] [CrossRef]
- Young, K.M.; Walker, S.L.; Lanthier, C.; Waddell, W.T.; Monfort, S.L.; Brown, J.L. Noninvasive monitoring of adrenocortical activity in carnivores by fecal glucocorticoid analyses. Gen. Comp. Endocrinol. 2004, 137, 148–165. [Google Scholar] [CrossRef]
- Glaeser, S.S.; Edwards, K.L.; Wielebnowski, N.; Brown, J.L. Effects of physiological changes and social life events on adrenal glucocorticoid activity in female zoo-housed Asian elephants (Elephas maximus). PLoS ONE 2020, 15, e0241910. [Google Scholar] [CrossRef]
- Graham, L.; Schwarzenberger, F.; Möstl, E.; Galama, W.; Savage, A. A versatile enzyme immunoassay for the determination of progestogens in feces and serum. Zoo Biol. 2001, 20, 227–236. [Google Scholar] [CrossRef]
- Amaral, R.S.; Rosas, F.C.W.; da Silva, V.M.F.; Nichi, M.; Oliveira, C.A. Endocrine monitoring of the ovarian cycle in captive female Amazonian manatees (Trichechus inunguis). Anim. Reprod. Sci. 2013, 142, 84–88. [Google Scholar] [CrossRef]
- Dloniak, S.M.; French, J.A.; Place, N.J.; Weldele, M.L.; Glickman, S.E.; Holekamp, K.E. Non-invasive monitoring of fecal androgens in spotted hyenas (Crocuta crocuta). Gen. Comp. Endocrinol. 2004, 135, 51–61. [Google Scholar] [CrossRef] [PubMed]
- Mason, G.J. Species differences in responses to captivity: Stress, welfare and the comparative method. Trends Ecol. Evol. 2010, 25, 713–721. [Google Scholar] [CrossRef] [PubMed]
- Devenport, L.; Knehans, A.; Sundstrom, A.; Thomas, T. Corticosterone’s dual metabolic actions. Life Sci. 1989, 45, 1389–1396. [Google Scholar] [CrossRef] [PubMed]
- Palme, R. Measuring fecal steroids: Guidelines for practical application. Ann. N. Y. Acad. Sci. 2005, 1046, 75–80. [Google Scholar] [CrossRef]
- Rich, E.L.; Romero, L.M. Exposure to chronic stress downregulates corticosterone responses to acute stressors. Am. J. Physiol. Integr. Comp. Physiol. 2005, 288, R1628–R1636. [Google Scholar] [CrossRef]
- Lafont, R.; Mathieu, M. Steroids in aquatic invertebrates. Ecotoxicology 2007, 16, 109–130. [Google Scholar] [CrossRef] [PubMed]
- Cameron, J.L. Interrelationships between Hormones, Behavior, and Affect during adolescence. Ann. N. Y. Acad. Sci. 2004, 1021, 110–123. [Google Scholar] [CrossRef] [PubMed]
- Fischer, C.P.; Romero, L.M. Chronic captivity stress in wild animals is highly species-specific. Conserv. Physiol. 2018, 7, coz093. [Google Scholar] [CrossRef] [PubMed]
- Moore, I.T.; Jessop, T.S. Stress, reproduction, and adrenocortical modulation in amphibians and reptiles. Horm. Behav. 2003, 43, 39–47. [Google Scholar] [CrossRef]
- Davies, S.; Gao, S.; Valle, S.; Bittner, S.; Hutton, P.; Meddle, S.L.; Deviche, P. Negative energy balance in a male songbird, the Abert’s towhee, constrains the testicular endocrine response to luteinizing hormone stimulation. J. Exp. Biol. 2015, 218, 2685–2693. [Google Scholar] [CrossRef] [PubMed]
- Marler, C.A.; Moore, M.C. Time and energy costs of aggression in testosterone-implanted free living male mountain spiny lizards Sceloporus-jarrovi. Physiol. Zool. 1989, 62, 1334–1350. [Google Scholar] [CrossRef]
- Scheel, D.; Godfrey-Smith, P.; Lawrence, M. Signal use by octopuses in agonistic interactions. Curr. Biol. 2016, 26, 377–382. [Google Scholar] [CrossRef]
- Sapolsky, R.M.; Sapolsky, R.M. Stress-induced elevation of testosterone concentrations in high ranking baboons: Role of catecholamines. Endocrinology 1986, 118, 1630–1635. [Google Scholar] [CrossRef]
- Huffard, C.L.; Bartick, M. Wild Wunderpus photogenicus and Octopus cyanea employ asphyxiating ‘constricting’ in interactions with other octopuses. Molluscan Res. 2015, 35, 12–16. [Google Scholar] [CrossRef]
- Cummins, S.F.; Boal, J.G.; Buresch, K.C.; Kuanpradit, C.; Sobhon, P.; Degnan, B.M.; Nagle, G.T.; Hanlon, R.T. Extreme aggression in male squid induced by a β-MSP-like pheromone. Curr. Biol. 2011, 21, 322–327. [Google Scholar] [CrossRef]
- Goymann, W. On the use of non-invasive hormone research in uncontrolled, natural environments: The problem with sex, diet, metabolic rate and the individual. Methods Ecol. Evol. 2012, 3, 757–765. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chancellor, S.; Grasse, B.; Sakmar, T.; Scheel, D.; Brown, J.S.; Santymire, R.M. Exploring the Effect of Age on the Reproductive and Stress Physiology of Octopus bimaculoides Using Dermal Hormones. Animals 2023, 13, 3115. https://doi.org/10.3390/ani13193115
Chancellor S, Grasse B, Sakmar T, Scheel D, Brown JS, Santymire RM. Exploring the Effect of Age on the Reproductive and Stress Physiology of Octopus bimaculoides Using Dermal Hormones. Animals. 2023; 13(19):3115. https://doi.org/10.3390/ani13193115
Chicago/Turabian StyleChancellor, Stephanie, Bret Grasse, Taylor Sakmar, David Scheel, Joel S. Brown, and Rachel M. Santymire. 2023. "Exploring the Effect of Age on the Reproductive and Stress Physiology of Octopus bimaculoides Using Dermal Hormones" Animals 13, no. 19: 3115. https://doi.org/10.3390/ani13193115
APA StyleChancellor, S., Grasse, B., Sakmar, T., Scheel, D., Brown, J. S., & Santymire, R. M. (2023). Exploring the Effect of Age on the Reproductive and Stress Physiology of Octopus bimaculoides Using Dermal Hormones. Animals, 13(19), 3115. https://doi.org/10.3390/ani13193115





