Prevalence of Shiga Toxin-Producing Escherichia coli O157 in Wild Scottish Deer with High Human Pathogenic Potential
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Sampling Methodology
2.2. Isolation of E. coli O157
2.3. Sequence Analysis of E. coli O157 Isolates
2.4. Prevalence Estimates of STEC O157 in Scottish Wild Deer
3. Results
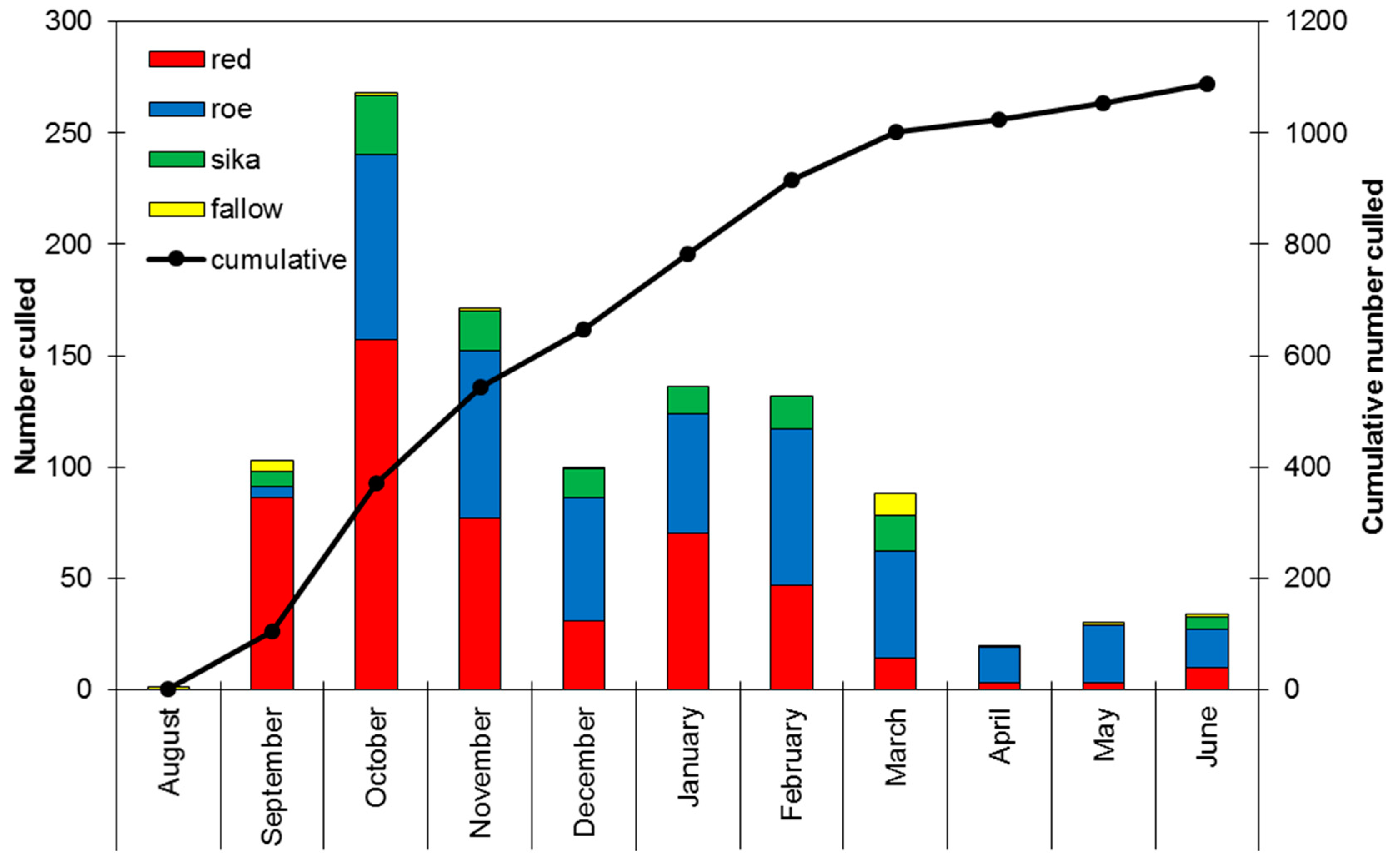
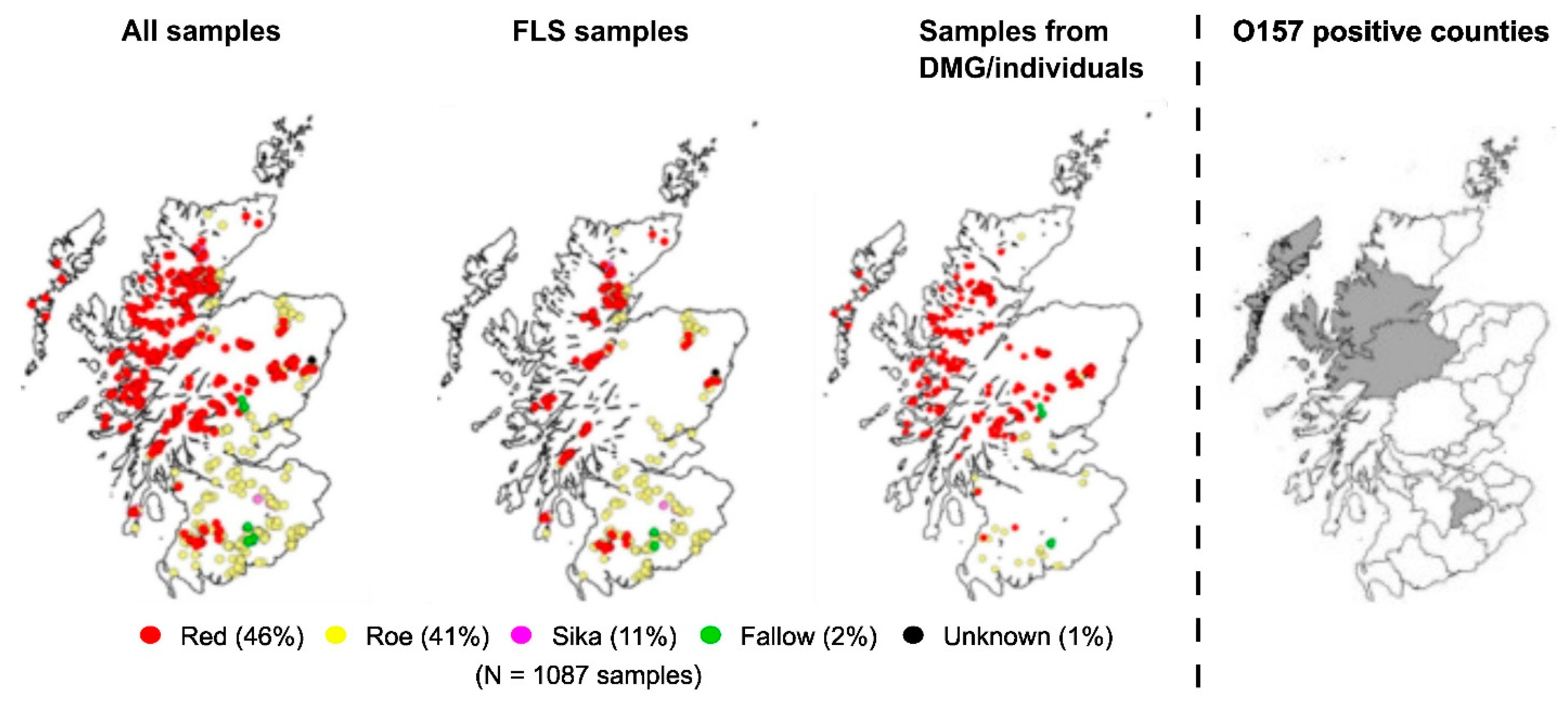
3.1. Deer Sampling
3.2. Prevalence of STEC O157 Strains Isolated from Scottish Wild Deer
3.3. Virulence Potential of STEC O157 Strains Isolated from Scottish Wild Deer
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- García, A.; Fox, J.G.; Besser, T.E. Zoonotic Enterohemorrhagic Escherichia coli: A One Health Perspective. ILAR J. 2010, 51, 221–232. [Google Scholar] [CrossRef] [PubMed]
- Melton-Celsa, A.R. Shiga Toxin (Stx) Classification, Structure, and Function. Microbiol. Spectr. 2014, 2, 2–4. [Google Scholar] [CrossRef]
- Dallman, T.J.; Ashton, P.M.; Byrne, L.; Perry, N.T.; Petrovska, L.; Ellis, R.; Allison, L.; Hanson, M.; Holmes, A.; Gunn, G.J.; et al. Applying phylogenomics to understand the emergence of Shiga-toxin-producing Escherichia coli O157:H7 strains causing severe human disease in the UK. Microb. Genom. 2015, 1, e000029. [Google Scholar] [CrossRef]
- Majowicz, S.E.; Scallan, E.; Jones-Bitton, A.; Sargeant, J.M.; Stapleton, J.; Angulo, F.J.; Yeung, D.H.; Kirk, M.D. Global Incidence of Human Shiga Toxin–Producing Escherichia coli Infections and Deaths: A Systematic Review and Knowledge Synthesis. Foodborne Pathog. Dis. 2014, 11, 447–455. [Google Scholar] [CrossRef] [PubMed]
- Valilis, E.; Ramsey, A.; Sidiq, S.; DuPont, H.L. Non-O157 Shiga toxin-producing Escherichia coli—A poorly appreciated enteric pathogen: Systematic review. Int. J. Infect. Dis. 2018, 76, 82–87. [Google Scholar] [CrossRef]
- Health Protection Scotland. STEC in Scotland 2018: Enhanced Surveillance and Reference Laboratory Data; Health Protection Scotland: Glasgow, UK, 2019. [Google Scholar]
- Scottish Government, Population HealthDirectorate. VTEC/E. coli O157 Action Plan for Scotland, 2013–2017; The Scottish Government: Edinburgh, UK, 2013.
- Fitzgerald, S.F.; Beckett, A.E.; Palarea-Albaladejo, J.; McAteer, S.; Shaaban, S.; Morgan, J.; Ahmad, N.I.; Young, R.; Mabbott, N.A.; Morrison, L.; et al. Shiga toxin sub-type 2a increases the efficiency of Escherichia coli O157 transmission between animals and restricts epithelial regeneration in bovine enteroids. PLoS Pathog. 2019, 15, e1008003. [Google Scholar] [CrossRef] [PubMed]
- Pearce, M.C.; Chase-Topping, M.E.; McKendrick, I.J.; Mellor, D.J.; Locking, M.E.; Allison, L.; Ternent, H.E.; Matthews, L.; Knight, H.I.; Smith, A.W.; et al. Temporal and spatial patterns of bovine Escherichia coli O157 prevalence and comparison of temporal changes in the patterns of phage types associated with bovine shedding and human E. coli O157 cases in Scotland between 1998–2000 and 2002–2004. BMC Microbiol. 2009, 9, 276. [Google Scholar] [CrossRef]
- Henry, M.K.; Tongue, S.C.; Evans, J.; Webster, C.; Mc, K.I.; Morgan, M.; Willett, A.; Reeves, A.; Humphry, R.W.; Gally, D.L.; et al. British Escherichia coli O157 in Cattle Study (BECS): To determine the prevalence of E. coli O157 in herds with cattle destined for the food chain. Epidemiol. Infect. 2017, 145, 3168–3179. [Google Scholar] [CrossRef]
- Chase-Topping, M.; Gally, D.; Low, C.; Matthews, L.; Woolhouse, M. Super-shedding and the link between human infection and livestock carriage of Escherichia coli O157. Nat. Rev. Microbiol. 2008, 6, 904–912. [Google Scholar] [CrossRef]
- Dias, D.; Costa, S.; Fonseca, C.; Baraúna, R.; Caetano, T.; Mendo, S. Pathogenicity of Shiga toxin-producing Escherichia coli (STEC) from wildlife: Should we care? Sci. Total Environ. 2022, 812, 152324. [Google Scholar] [CrossRef]
- Smith-Palmer, A.; Hawkins, G.; Browning, L.; Allison, L.; Hanson, M.; Bruce, R.; McElhiney, J.; Horne, J. Outbreak of Escherichia coli O157 Phage Type 32 linked to the consumption of venison products. Epidemiol. Infect. 2018, 146, 1922–1927. [Google Scholar] [CrossRef]
- The Risk of STEC (Shiga Toxin-Producing Escherichia coli) Contamination in Wild Venison. 2020. Available online: https://www.foodstandards.gov.scot/publications-and-research/publications/the-risk-of-stec-contamination-in-wild-venison (accessed on 30 August 2021).
- QGIS Development Team. QGIS Geographic Information System. Available online: http://qgis.osgeo.org (accessed on 1 August 2019).
- Pearce, M.C.; Jenkins, C.; Vali, L.; Smith, A.W.; Knight, H.I.; Cheasty, T.; Smith, H.R.; Gunn, G.J.; Woolhouse, M.E.J.; Amyes, S.G.B.; et al. Temporal Shedding Patterns and Virulence Factors of Escherichia coli Serogroups O26, O103, O111, O145, and O157 in a Cohort of Beef Calves and Their Dams. Appl. Environ. Microbiol. 2004, 70, 1708–1716. [Google Scholar] [CrossRef] [PubMed]
- Holmes, A.; Dallman, T.J.; Shabaan, S.; Hanson, M.; Allison, L. Validation of Whole-Genome Sequencing for Identification and Characterization of Shiga Toxin-Producing Escherichia coli To Produce Standardized Data to Enable Data Sharing. J. Clin. Microbiol. 2018, 56, e01388-17. [Google Scholar] [CrossRef] [PubMed]
- Chattaway, M.A.; Schaefer, U.; Tewolde, R.; Dallman, T.J.; Jenkins, C. Identification of Escherichia coli and Shigella Species from Whole-Genome Sequences. J. Clin. Microbiol. 2017, 55, 616–623. [Google Scholar] [CrossRef]
- Langmead, B.; Salzberg, S.L. Fast gapped-read alignment with Bowtie 2. Nat. Methods 2012, 9, 357–359. [Google Scholar] [CrossRef]
- Tewolde, R.; Dallman, T.; Schaefer, U.; Sheppard, C.L.; Ashton, P.; Pichon, B.; Ellington, M.; Swift, C.; Green, J.; Underwood, A. MOST: A modified MLST typing tool based on short read sequencing. PeerJ 2016, 4, e2308. [Google Scholar] [CrossRef]
- Ashton, P.M.; Perry, N.; Ellis, R.; Petrovska, L.; Wain, J.; Grant, K.A.; Jenkins, C.; Dallman, T.J. Insight into Shiga toxin genes encoded by Escherichia coli O157 from whole genome sequencing. PeerJ 2015, 3, e739. [Google Scholar] [CrossRef]
- Dallman, T.; Ashton, P.; Schafer, U.; Jironkin, A.; Painset, A.; Shaaban, S.; Hartman, H.; Myers, R.; Underwood, A.; Jenkins, C.; et al. SnapperDB: A database solution for routine sequencing analysis of bacterial isolates. Bioinformatics 2018, 34, 3028–3029. [Google Scholar] [CrossRef] [PubMed]
- Mainda, G.; Lupolova, N.; Sikakwa, L.; Bessell, P.R.; Muma, J.B.; Hoyle, D.V.; McAteer, S.P.; Gibbs, K.; Williams, N.J.; Sheppard, S.K.; et al. Phylogenomic approaches to determine the zoonotic potential of Shiga toxin-producing Escherichia coli (STEC) isolated from Zambian dairy cattle. Sci. Rep. 2016, 6, 26589. [Google Scholar] [CrossRef][Green Version]
- Price, M.N.; Dehal, P.S.; Arkin, A.P. FastTree 2—Approximately maximum-likelihood trees for large alignments. PLoS ONE 2010, 5, e9490. [Google Scholar] [CrossRef]
- Letunic, I.; Bork, P. Interactive Tree of Life (iTOL) v4: Recent updates and new developments. Nucleic Acids Res. 2019, 47, W256–W259. [Google Scholar] [CrossRef] [PubMed]
- Cascales, E.; Buchanan, S.K.; Duché, D.; Kleanthous, C.; Lloubès, R.; Postle, K.; Riley, M.; Slatin, S.; Cavard, D. Colicin Biology. Microbiol. Mol. Biol. Rev. 2007, 71, 158–229. [Google Scholar] [CrossRef] [PubMed]
- Ferens, W.A.; Hovde, C.J. Escherichia coli O157:H7: Animal Reservoir and Sources of Human Infection. Foodborne Pathog. Dis. 2011, 8, 465–487. [Google Scholar] [CrossRef]
- Kabeya, H.; Sato, S.; Oda, S.; Kawamura, M.; Nagasaka, M.; Kuranaga, M.; Yokoyama, E.; Hirai, S.; Iguchi, A.; Ishihara, T.; et al. Characterization of Shiga toxin-producing Escherichia coli from feces of sika deer (Cervus nippon) in Japan using PCR binary typing analysis to evaluate their potential human pathogenicity. J. Vet. Med Sci. 2017, 79, 834–841. [Google Scholar] [CrossRef]
- Szczerba-Turek, A.; Siemionek, J.; Socha, P.; Bancerz-Kisiel, A.; Platt-Samoraj, A.; Lipczynska-Ilczuk, K.; Szweda, W. Shiga toxin-producing Escherichia coli isolates from red deer (Cervus elaphus), roe deer (Capreolus capreolus) and fallow deer (Dama dama) in Poland. Food Microbiol. 2020, 86, 103352. [Google Scholar] [CrossRef]
- Lauzi, S.; Luzzago, C.; Chiani, P.; Michelacci, V.; Knijn, A.; Pedrotti, L.; Corlatti, L.; Pederzoli, C.B.; Scavia, G.; Morabito, S.; et al. Free-ranging red deer (Cervus elaphus) as carriers of potentially zoonotic Shiga toxin-producing Escherichia coli. Transbound. Emerg. Dis. 2021, 69, 1902–1911. [Google Scholar] [CrossRef]
- Singh, P.; Sha, Q.; Lacher, D.W.; Del Valle, J.; Mosci, R.E.; Moore, J.A.; Scribner, K.T.; Manning, S.D. Characterization of enteropathogenic and Shiga toxin-producing Escherichia coli in cattle and deer in a shared agroecosystem. Front. Cell. Infect. Microbiol. 2015, 5, 29. [Google Scholar] [CrossRef] [PubMed]
- Frank, E.; Bonke, R.; Drees, N.; Heurich, M.; Martlbauer, E.; Gareis, M. Shiga toxin-producing Escherichia coli (STEC) shedding in a wild roe deer population. Vet. Microbiol. 2019, 239, 108479. [Google Scholar] [CrossRef]
- Laidler, M.R.; Tourdjman, M.; Buser, G.L.; Hostetler, T.; Repp, K.K.; Leman, R.; Samadpour, M.; Keene, W.E. Escherichia coli O157:H7 Infections Associated with Consumption of Locally Grown Strawberries Contaminated by Deer. Clin. Infect. Dis. 2013, 57, 1129–1134. [Google Scholar] [CrossRef] [PubMed]
- Nagano, H.; Hirochi, T.; Fujita, K.; Wakamori, Y.; Takeshi, K.; Yano, S. Phenotypic and genotypic characterization of β-d-glucuronidase-positive Shiga toxin-producing Escherichia coli O157: H7 isolates from deer. J. Med Microbiol. 2004, 53, 1037–1043. [Google Scholar] [CrossRef]
- Brandal, L.T.; Wester, A.L.; Lange, H.; Løbersli, I.; Lindstedt, B.-A.; Vold, L.; Kapperud, G. Shiga toxin-producing Escherichia coli infections in Norway, 1992–2012: Characterization of isolates and identification of risk factors for haemolytic uremic syndrome. BMC Infect. Dis. 2015, 15, 324. [Google Scholar] [CrossRef]
- Louise, C.B.; Obrig, T.G. Specific interaction of Escherichia coli O157:H7-derived Shiga-like toxin II with human renal endothelial cells. J. Infect. Dis. 1995, 172, 1397–1401. [Google Scholar] [CrossRef]
- Lynn, R.M.; O’Brien, S.J.; Taylor, C.M.; Adak, G.K.; Chart, H.; Cheasty, T.; Coia, J.E.; Gillespie, I.A.; Locking, M.E.; Reilly, W.J.; et al. Childhood hemolytic uremic syndrome, United Kingdom and Ireland. Emerg. Infect. Dis. 2005, 11, 590–596. [Google Scholar] [CrossRef] [PubMed]
- Matthews, L.; Reeve, R.; Gally, D.L.; Low, J.C.; Woolhouse, M.E.J.; McAteer, S.P.; Locking, M.E.; Chase-Topping, M.E.; Haydon, D.T.; Allison, L.J.; et al. Predicting the public health benefit of vaccinating cattle against Escherichia coli O157. Proc. Natl. Acad. Sci. USA 2013, 110, 16265–16270. [Google Scholar] [CrossRef] [PubMed]
- Omisakin, F.; MacRae, M.; Ogden, I.D.; Strachan, N.J.C. Concentration and Prevalence of Escherichia coli O157 in Cattle Feces at Slaughter. Appl. Environ. Microbiol. 2003, 69, 2444–2447. [Google Scholar] [CrossRef]
- Stephens, T.P.; McAllister, T.A.; Stanford, K. Perineal swabs reveal effect of super shedders on the transmission of Escherichia coli O157:H7 in commercial feedlots. J. Anim. Sci. 2009, 87, 4151–4160. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Matthews, L.; Low, J.; Gally, D.L.; Pearce, M.C.; Mellor, D.J.; Heesterbeek, J.A.P.; Chase-Topping, M.; Naylor, S.W.; Shaw, D.J.; Reid, S.W.J.; et al. Heterogeneous shedding of Escherichia coli O157 in cattle and its implications for control. Proc. Natl. Acad. Sci. USA 2006, 103, 547–552. [Google Scholar] [CrossRef]
- Richards, P.J.; Wu, S.; Tinker, D.B.; Howell, M.V.; Dodd, C.E.R. Microbial Quality of Venison Meat at Retail in the UK in Relation to Production Practices and Processes. In Game meat Hygiene in Focus: Microbiology, Epidemiology, RISK Analysis and Quality Assurance; Paulsen, P., Bauer, A., Vodnansky, M., Winkelmayer, R., Smulders, F.J.M., Eds.; Wageningen Academic Publishers: Wageningen, The Netherlands, 2011; pp. 113–117. [Google Scholar]
- Soare, C.; Mazeri, S.; McAteer, S.; McNeilly, T.N.; Seguino, A.; Chase-Topping, M. The microbial condition of Scottish wild deer carcasses collected for human consumption and the hygiene risk factors associated with Escherichia coli and total coliforms contamination. Food Microbiol. 2022, 108, 104102. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-S.; Lee, M.-S.; Kim, J.H. Recent Updates on Outbreaks of Shiga Toxin-Producing Escherichia coli and Its Potential Reservoirs. Front. Cell. Infect. Microbiol. 2020, 10, 273. [Google Scholar] [CrossRef] [PubMed]
- Espinosa, L.; Gray, A.; Duffy, G.; Fanning, S.; McMahon, B.J. A scoping review on the prevalence of Shiga-toxigenic Escherichia coli in wild animal species. Zoonoses Public Health 2018, 65, 911–920. [Google Scholar] [CrossRef]

| Strain ID | County | Species | Sex | Age (Years) * | Co-Grazing History | PT | H-Type | stx Subtype | eae | Count (CFU/g Faeces) |
|---|---|---|---|---|---|---|---|---|---|---|
| SME170024 | Inverness-shire | Red | F | 4 | None reported | 21/28 | 7 | stx2a and stx2c | + | 1.0 × 104 |
| SME170025 | Peebles-shire | Sika | F | 5 | None reported | 8 | 7 | stx2a | + | 5.0 × 106 |
| SME170026 | Ross and Cromarty | Red | M | 1.5 | Cattle/sheep | 21/28 | 7 | stx2a | + | 7.7 × 107 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fitzgerald, S.F.; Mitchell, M.C.; Holmes, A.; Allison, L.; Chase-Topping, M.; Lupolova, N.; Wells, B.; Gally, D.L.; McNeilly, T.N. Prevalence of Shiga Toxin-Producing Escherichia coli O157 in Wild Scottish Deer with High Human Pathogenic Potential. Animals 2023, 13, 2795. https://doi.org/10.3390/ani13172795
Fitzgerald SF, Mitchell MC, Holmes A, Allison L, Chase-Topping M, Lupolova N, Wells B, Gally DL, McNeilly TN. Prevalence of Shiga Toxin-Producing Escherichia coli O157 in Wild Scottish Deer with High Human Pathogenic Potential. Animals. 2023; 13(17):2795. https://doi.org/10.3390/ani13172795
Chicago/Turabian StyleFitzgerald, Stephen F., Mairi C. Mitchell, Anne Holmes, Lesley Allison, Margo Chase-Topping, Nadejda Lupolova, Beth Wells, David L. Gally, and Tom N. McNeilly. 2023. "Prevalence of Shiga Toxin-Producing Escherichia coli O157 in Wild Scottish Deer with High Human Pathogenic Potential" Animals 13, no. 17: 2795. https://doi.org/10.3390/ani13172795
APA StyleFitzgerald, S. F., Mitchell, M. C., Holmes, A., Allison, L., Chase-Topping, M., Lupolova, N., Wells, B., Gally, D. L., & McNeilly, T. N. (2023). Prevalence of Shiga Toxin-Producing Escherichia coli O157 in Wild Scottish Deer with High Human Pathogenic Potential. Animals, 13(17), 2795. https://doi.org/10.3390/ani13172795





