Horse Grimace Scale Does Not Detect Pain in Horses with Equine Gastric Ulcer Syndrome
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Horse Grimace Scale Image Acquisition
2.3. Lameness Assessment
2.4. Serum Amyloid A Testing (SAA)
2.5. Gastroscopy Examination and Equine Gastric Ulcer Syndrome Group Allocation
2.6. Horse Grimace Scale Scoring
2.7. Statistical Analysis
2.7.1. Sample Size Calculation
2.7.2. Horse Grimace Scale Inter-Observer Reliability Scores
2.7.3. Horse Grimace Scale Scores between Groups
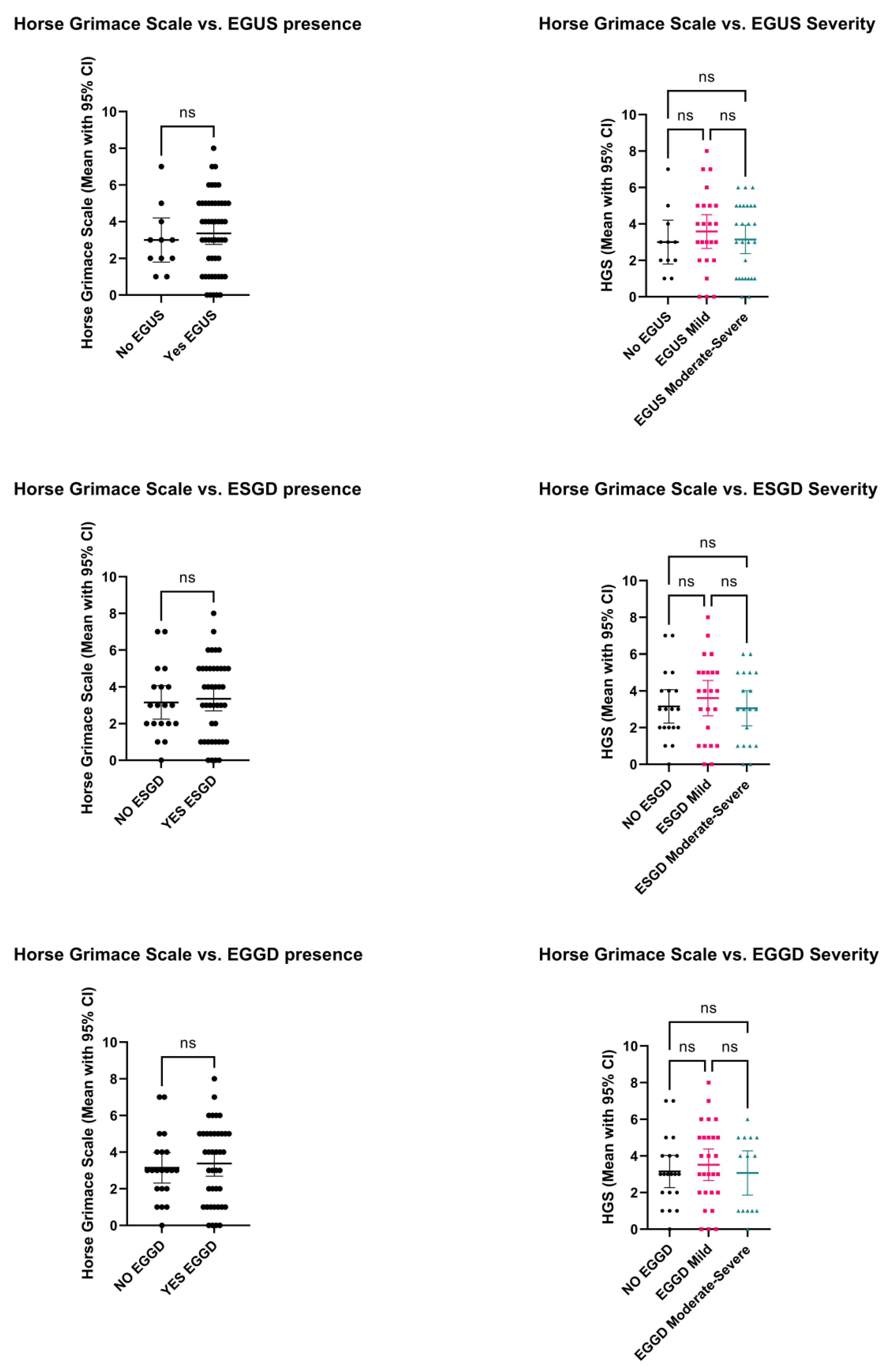
3. Results
3.1. Horse Grimace Scale Inter-Observer Reliability
3.2. Horse Grimace Scale Scores between Horses with and without EGUS
4. Discussion
4.1. The Use of a Pain Scale for Investigating Pain in Horses with EGUS
4.2. Horse Grimace Scale Scores in Horses with and without Gastric Ulcers
4.3. Horse Grimace Scale Inter-Observer Reliability
4.4. Study Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sykes, B.; Hewetson, M.; Hepburn, R.; Luthersson, N.; Tamzali, Y. European College of Equine Internal Medicine Consensus Statement—Equine gastric ulcer syndrome in adult horses. J. Vet. Intern. Med. 2015, 29, 1288. [Google Scholar] [CrossRef]
- Vatistas, N.; Snyder, J.; Carlson, G.; Johnson, B.; Arthu, R.; Thurmond, M.; Zhou, H.; Lloyd, K.L.K. Cross-sectional study of gastric ulcers of the squamous mucosa in Thoroughbred racehorses. Equine Vet. J. 1999, 31 (Suppl. S29), 34–39. [Google Scholar] [CrossRef]
- Begg, L.; O’sullivan, C. The prevalence and distribution of gastric ulceration in 345 racehorses. Aust. Vet. J. 2003, 81, 199–201. [Google Scholar] [CrossRef]
- Murray, M.; Schusser, G.; Pipers, F.; Gross, S.J. Factors associated with gastric lesions in Thoroughbred racehorses. Equine Vet. J. 1996, 28, 368–374. [Google Scholar] [CrossRef]
- Sykes, B.; Sykes, K.; Hallowell, G. A comparison of three doses of omeprazole in the treatment of equine gastric ulcer syndrome: A blinded, randomised, dose–response clinical trial. Equine Vet. J. 2015, 47, 285–290. [Google Scholar] [CrossRef]
- Nieto, J.E.; Snyder, J.R.; Vatistas, N.J.; Jones, J.H. Effect of gastric ulceration on physiologic responses to exercise in horses. Am. J. Vet. Res. 2009, 70, 787–795. [Google Scholar] [CrossRef] [PubMed]
- McClure, S.; Glickman, L.; Glickman, N. Prevalence of gastric ulcers in show horses. J. Am. Vet. Med. Assoc. 1999, 215, 1130–1133. [Google Scholar]
- Millares-Ramirez, E.M.; Le Jeune, S.S. Girthiness: Retrospective study of 37 horses (2004–2016). J. Equine Vet. Sci. 2019, 79, 100–104. [Google Scholar] [CrossRef] [PubMed]
- Dionne, R.M.; Vrins, A.; Doucet, M.Y.; Pare, J. Gastric ulcers in standardbred racehorses: Prevalence, lesion description, and risk factors. J. Vet. Intern. Med. 2003, 17, 218–222. [Google Scholar] [CrossRef] [PubMed]
- Andrews, F.; Nadeau, J.A. Clinical syndromes of gastric ulceration in foals and mature horses. Equine Vet. J. 1999, 31 (Suppl. S29), 30–33. [Google Scholar] [CrossRef] [PubMed]
- Murray, M.; Grodinsky, C.; Anderson, C.; Radue, P.; Schmidt, G. Gastric ulcers in horses: A comparison of endoscopic findings in horses with and without clinical signs. Equine Vet. J. 1989, 21 (Suppl. S7), 68–72. [Google Scholar] [CrossRef] [PubMed]
- Malmkvist, J.; Poulsen, J.M.; Luthersson, N.; Palme, R.; Christensen, J.W.; Søndergaard, E. Behaviour and stress responses in horses with gastric ulceration. Appl. Anim. Behav. Sci. 2012, 142, 160–167. [Google Scholar] [CrossRef]
- McDonnell, S.M. Practical review of self-mutilation in horses. Anim. Reprod. Sci. 2008, 107, 219–228. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.; Yap, I.; Guan, R.; Tay, H. Acid perfusion of duodenal ulcer craters and ulcer pain: A controlled double blind study. Gut 1986, 27, 942–945. [Google Scholar] [CrossRef] [PubMed]
- Faybush, E.M.; Fass, R. Gastroesophageal reflux disease in noncardiac chest pain. Gastroenterol. Clin. 2004, 33, 41–54. [Google Scholar] [CrossRef]
- Frieling, T. Non-cardiac chest pain. Visc. Med. 2018, 34, 92–96. [Google Scholar] [CrossRef]
- Sutton, G.; Atamna, R.; Steinman, A.; Mair, T. Comparison of three acute colic pain scales: Reliability, validity and usability. Vet. J. 2019, 246, 71–77. [Google Scholar] [CrossRef]
- VanDierendonck, M.C.; van Loon, J.P. Monitoring acute equine visceral pain with the equine Utrecht University scale for composite pain assessment (EQUUS-COMPASS) and the equine Utrecht University scale for facial assessment of pain (EQUUS-FAP): A validation study. Vet. J. 2016, 216, 175–177. [Google Scholar] [CrossRef]
- Bussières, G.; Jacques, C.; Lainay, O.; Beauchamp, G.; Leblond, A.; Cadoré, J.-L.; Desmaizières, L.-M.; Cuvelliez, S.; Troncy, E. Development of a composite orthopaedic pain scale in horses. Res. Vet. Sci. 2008, 85, 294–306. [Google Scholar] [CrossRef]
- Ortolani, F.; Scilimati, N.; Gialletti, R.; Menchetti, L.; Nannarone, S. Development and preliminary validation of a pain scale for ophthalmic pain in horses: The Equine Ophthalmic Pain Scale (EOPS). Vet. J. 2021, 278, 105774. [Google Scholar] [CrossRef]
- Dalla Costa, E.; Minero, M.; Lebelt, D.; Stucke, D.; Canali, E.; Leach, M.C. Development of the Horse Grimace Scale (HGS) as a Pain Assessment Tool in Horses Undergoing Routine Castration. PLoS ONE 2014, 9, e92281. [Google Scholar] [CrossRef]
- Evangelista, M.C.; Watanabe, R.; Leung, V.S.; Monteiro, B.P.; O’Toole, E.; Pang, D.S.; Steagall, P.V. Facial expressions of pain in cats: The development and validation of a Feline Grimace Scale. Sci. Rep. 2019, 9, 19128. [Google Scholar] [CrossRef]
- Sotocina, S.G.; Sorge, R.E.; Zaloum, A.; Tuttle, A.H.; Martin, L.J.; Wieskopf, J.S.; Mapplebeck, J.C.; Wei, P.; Zhan, S.; Zhang, S. The Rat Grimace Scale: A partially automated method for quantifying pain in the laboratory rat via facial expressions. Mol. Pain 2011, 7, 55. [Google Scholar] [CrossRef] [PubMed]
- Viscardi, A.V.; Hunniford, M.; Lawlis, P.; Leach, M.; Turner, P.V. Development of a piglet grimace scale to evaluate piglet pain using facial expressions following castration and tail docking: A pilot study. Front. Vet. Sci. 2017, 4, 51. [Google Scholar] [CrossRef]
- Dalla Costa, E.; Stucke, D.; Dai, F.; Minero, M.; Leach, M.; Lebelt, D. Using the Horse Grimace Scale (HGS) to Assess Pain Associated with Acute Laminitis in Horses (Equus caballus). Animals 2016, 6, 47. [Google Scholar] [CrossRef] [PubMed]
- Coneglian, M.M.; Borges, T.D.; Weber, S.H.; Bertagnon, H.G.; Michelotto, P.V. Use of the horse grimace scale to identify and quantify pain due to dental disorders in horses. Appl. Anim. Behav. Sci. 2020, 225, 104970. [Google Scholar] [CrossRef]
- Schwartz, D.; Pusterla, N.; Jacobsen, S.; Christopher, M. Analytical validation of a new point-of-care assay for serum amyloid A in horses. Equine Vet. J. 2018, 50, 678–683. [Google Scholar] [CrossRef] [PubMed]
- Bowlby, C.; Mudge, M.; Schroeder, E.; Godman, J.; Hurcombe, S. Equine inflammatory response to abdominal surgery in the absence of gastrointestinal disease. J. Vet. Emerg. Crit. Care 2021, 31, 601–607. [Google Scholar] [CrossRef]
- Bergstrom, T.; Frey, M.; Rao, S.; Bass, L. Comparison of post-operative inflammatory response in horses undergoing elective castration treated preoperatively with ceftiofur crystalline free acid or procaine penicillin G. Equine Vet. Educ. 2022, 34, 409–416. [Google Scholar] [CrossRef]
- Wise, J.C.; Wilkes, E.J.; Raidal, S.L.; Xie, G.; Crosby, D.E.; Hale, J.N.; Hughes, K.J. Interobserver and intraobserver reliability for 2 grading systems for gastric ulcer syndrome in horses. J. Vet. Intern. Med. 2021, 35, 571–579. [Google Scholar] [CrossRef]
- Sykes, B.; Kathawala, K.; Song, Y.; Garg, S.; Page, S.; Underwood, C.; Mills, P. Preliminary investigations into a novel, long-acting, injectable, intramuscular formulation of omeprazole in the horse. Equine Vet. J. 2017, 49, 795–801. [Google Scholar] [CrossRef]
- Crumpton, S.; Baiker, K.; Hallowell, G.; Habershon-Butcher, J.; Bowen, I. Diagnostic value of gastric mucosal biopsies in horses with glandular disease. Equine Vet. J. 2015, 47, 9. [Google Scholar] [CrossRef] [PubMed]
- Cicchetti, D.V. Guidelines, criteria, and rules of thumb for evaluating normed and standardized assessment instruments in psychology. Psychol. Assess. 1994, 6, 284. [Google Scholar] [CrossRef]
- Evangelista, M.C.; Monteiro, B.P.; Steagall, P.V. Measurement properties of grimace scales for pain assessment in nonhuman mammals: A systematic review. Pain 2022, 163, e697–e714. [Google Scholar] [CrossRef] [PubMed]
- Andrews, F.M.; Buchanan, B.R.; Smith, S.H.; Elliott, S.B.; Saxton, A.M. In vitro effects of hydrochloric acid and various concentrations of acetic, propionic, butyric, or valeric acids on bioelectric properties of equine gastric squamous mucosa. Am. J. Vet. Res. 2006, 67, 1873–1882. [Google Scholar] [CrossRef]
- Nadeau, J.A.; Andrews, F.M.; Patton, C.S.; Argenzio, R.A.; Mathew, A.G.; Saxton, A.M. Effects of hydrochloric, valeric, and other volatile fatty acids on pathogenesis of ulcers in the nonglandular portion of the stomach of horses. Am. J. Vet. Res. 2003, 64, 413–417. [Google Scholar] [CrossRef]
- Lorenzo-Figueras, M.; Merritt, A.M. Effects of exercise on gastric volume and pH in the proximal portion of the stomach of horses. Am. J. Vet. Res. 2002, 63, 1481–1487. [Google Scholar] [CrossRef]
- Dalla Costa, E.; Bracci, D.; Dai, F.; Lebelt, D.; Minero, M. Do different emotional states affect the horse grimace scale score? A pilot study. J. Equine Vet. Sci. 2017, 54, 114–117. [Google Scholar] [CrossRef]
- Dai, F.; Leach, M.; Macrae, A.M.; Minero, M.; Dalla Costa, E. Does Thirty-Minute Standardised Training Improve the Inter-Observer Reliability of the Horse Grimace Scale (HGS)? A Case Study. Animals 2020, 10, 781. [Google Scholar] [CrossRef]
- Dalla Costa, E.; Pascuzzo, R.; Leach, M.C.; Dai, F.; Lebelt, D.; Vantini, S.; Minero, M. Can grimace scales estimate the pain status in horses and mice? A statistical approach to identify a classifier. PLoS ONE 2018, 13, e0200339. [Google Scholar] [CrossRef]
| Grade | Severity |
|---|---|
| 0 | No lameness is visible. |
| 0.5 | Lameness difficult to observe or is not consistent and repeatable. |
| 1 | Forelimb lameness involves a discernible head nod; however, full symmetrical limb movement is present. Hindlimb lameness involves a mild hip drop or hip hike only. |
| 2 | Forelimb lameness involves a consistent and moderate head nod and asymmetrical limb placement. Hindlimb lameness involves a discernible and consistent hip drop and hip hike. |
| 3 | Forelimb lameness is consistent and marked; however, lameness is not visible at the walk. Limb placement may be obviously altered and ground contact may not be complete in the affected limb. Hindlimb lameness involves an obvious hip drop and hip hike and may result in a compensatory head nod. |
| 4 | Forelimb and/or hindlimb lameness is visible at the walk. |
| 5 | Lameness produces minimal weight bearing in motion or at rest or a complete inability to move. |
| Grade | Squamous Mucosa | Glandular Mucosa |
|---|---|---|
| 0 | The epithelium is intact and there is no appearance of hyperkeratosis | The epithelium is intact and there is no obvious hyperemia |
| 1 | The mucosa is intact, but there are areas of hyperkeratosis | The epithelium is intact and there are areas of hyperemia |
| 2 | Small, single or multifocal lesions | Small, single or multifocal lesions |
| 3 | Large single or extensive superficial lesions | Large single or extensive superficial lesions |
| 4 | Extensive lesions with areas of apparent deep ulceration | Extensive lesions with areas of apparent deep ulceration |
| Breed | Gender | Age (Years) | Weight (kg) |
|---|---|---|---|
| Appaloosa 1 Arabian 2 Brumby 1 Clydesdale 1 Riding pony 4 Standardbred 39 Thoroughbred 8 Warmblood 3 Unknown 2 | Geldings 29 Mares 32 | 8.3 ± (2–18) | 476.6 (296–595) |
| ICC * | All Observers (n = 7) | Veterinary Specialists (n = 4) | Veterinarians with HGS Experience (n = 3) |
|---|---|---|---|
| Overall HGS | 0.75 (0.65–0.82) | 0.64 (0.49–0.75) | 0.61 (0.43–0.74) |
| Ears stiffly backwards | 0.89 (0.85–0.92) | 0.82 (0.75–0.88) | 0.84 (0.75–0.89) |
| Orbital tightening | 0.71 (0.60–0.80) | 0.58 (0.40–0.72) | 0.68 (0.54–0.79) |
| Tension above the eye | 0.59 (0.43–0.71) | 0.52 (0.33–0.67) | 0.26 (0.05–0.49) |
| Prominent and strained chewing muscles | 0.65 (0.52–0.76) | 0.66 (0.52–0.77) | 0.24 (0.09–0.48) |
| Mouth strained and pronounced chin | 0.20 (0.07–0.43) | 0.23 (0.06–0.46) | 0.12 (0.23–0.39) |
| Strained nostrils and flattening of profile | 0.73 (0.63–0.81) | 0.60 (0.44–0.73) | 0.59 (0.40–0.72) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ferlini Agne, G.; May, B.E.; Lovett, A.; Simon, O.; Steel, C.; Santos, L.; Guedes do Carmo, L.; Barbosa, B.; Werner, L.C.; Daros, R.R.; et al. Horse Grimace Scale Does Not Detect Pain in Horses with Equine Gastric Ulcer Syndrome. Animals 2023, 13, 1623. https://doi.org/10.3390/ani13101623
Ferlini Agne G, May BE, Lovett A, Simon O, Steel C, Santos L, Guedes do Carmo L, Barbosa B, Werner LC, Daros RR, et al. Horse Grimace Scale Does Not Detect Pain in Horses with Equine Gastric Ulcer Syndrome. Animals. 2023; 13(10):1623. https://doi.org/10.3390/ani13101623
Chicago/Turabian StyleFerlini Agne, Gustavo, Bridget Eileen May, Amy Lovett, Olivier Simon, Catherine Steel, Luiz Santos, Laize Guedes do Carmo, Bianca Barbosa, Laís Cristine Werner, Ruan R. Daros, and et al. 2023. "Horse Grimace Scale Does Not Detect Pain in Horses with Equine Gastric Ulcer Syndrome" Animals 13, no. 10: 1623. https://doi.org/10.3390/ani13101623
APA StyleFerlini Agne, G., May, B. E., Lovett, A., Simon, O., Steel, C., Santos, L., Guedes do Carmo, L., Barbosa, B., Werner, L. C., Daros, R. R., Somogyi, A. A., Sykes, B., & Franklin, S. (2023). Horse Grimace Scale Does Not Detect Pain in Horses with Equine Gastric Ulcer Syndrome. Animals, 13(10), 1623. https://doi.org/10.3390/ani13101623







