The Effect of Early Neurological Stimulation on Puppy Welfare in Commercial Breeding Kennels
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethical Statement
2.2. Subjects and Facilities
2.3. Handling Procedure
2.3.1. ENS Treatment Group
2.3.2. Held Treatment Group
2.3.3. Control Group
2.4. Physical Health Assessments
2.5. Testing Procedures
2.5.1. Stranger-Approach Tests
2.5.2. Isolation Test
2.6. Data Analysis
2.6.1. Physical Health
2.6.2. Stranger Approach Tests
2.6.3. Isolation Test
3. Results
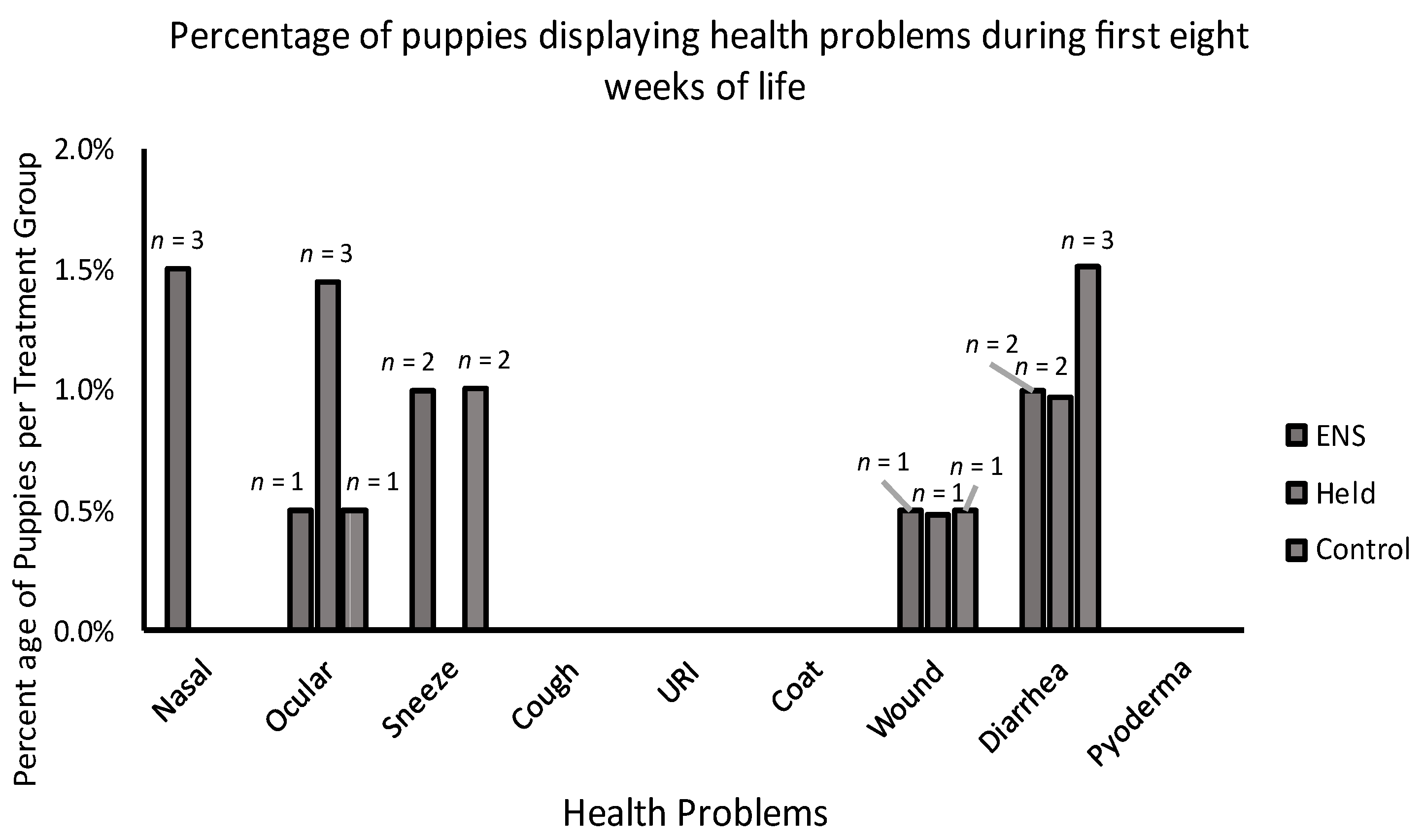
3.1. Physical Health
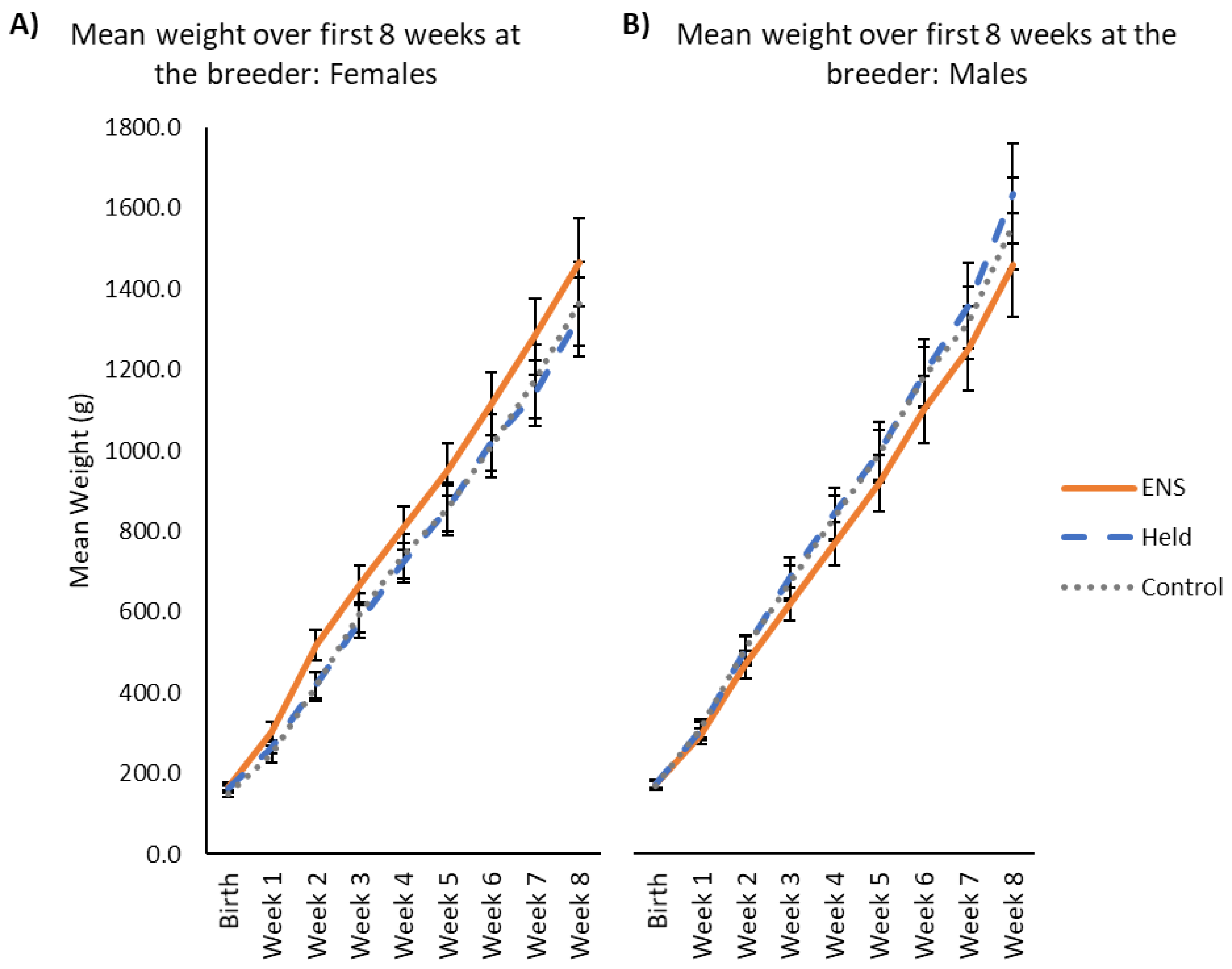
3.2. Weight and Weight Gain
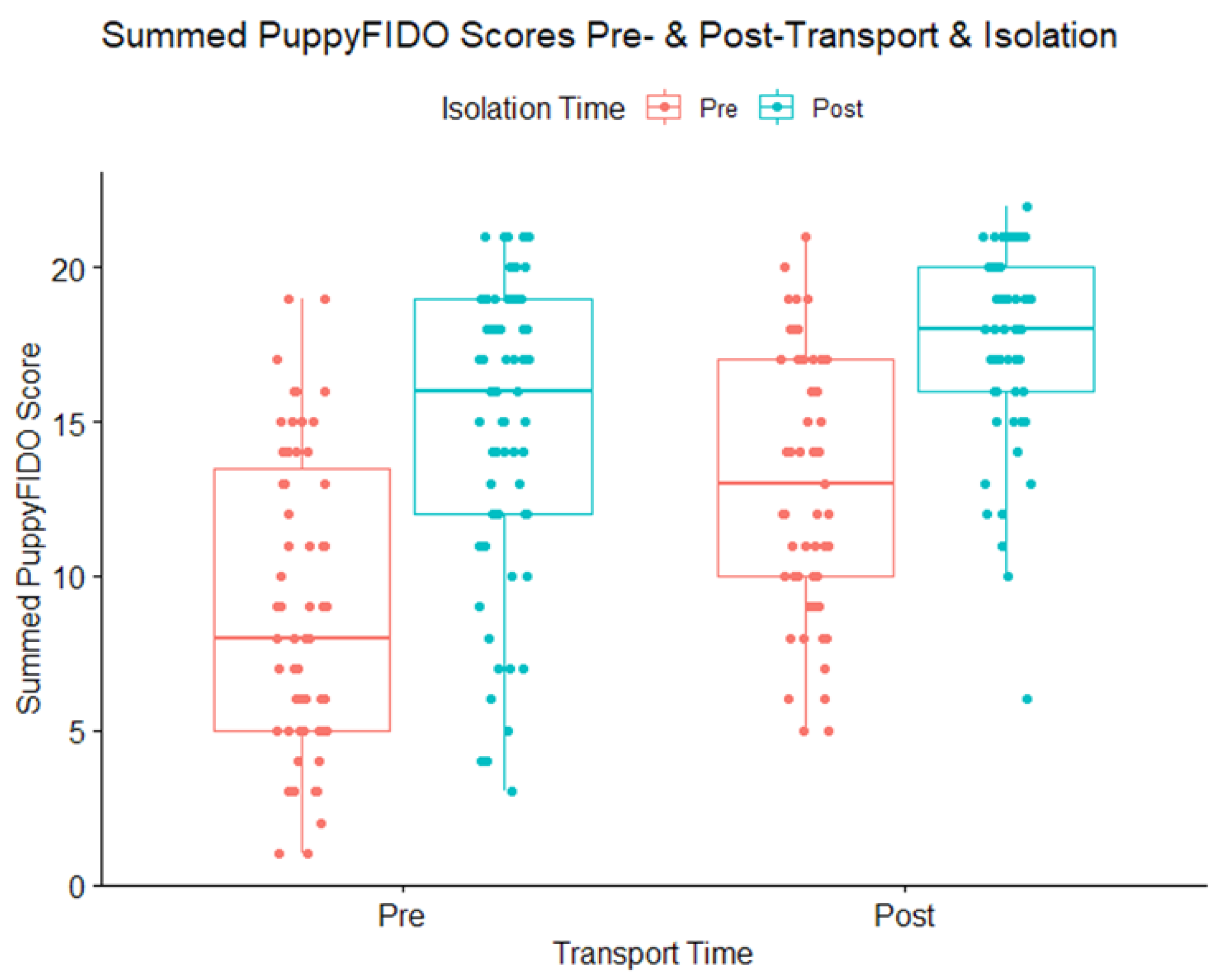
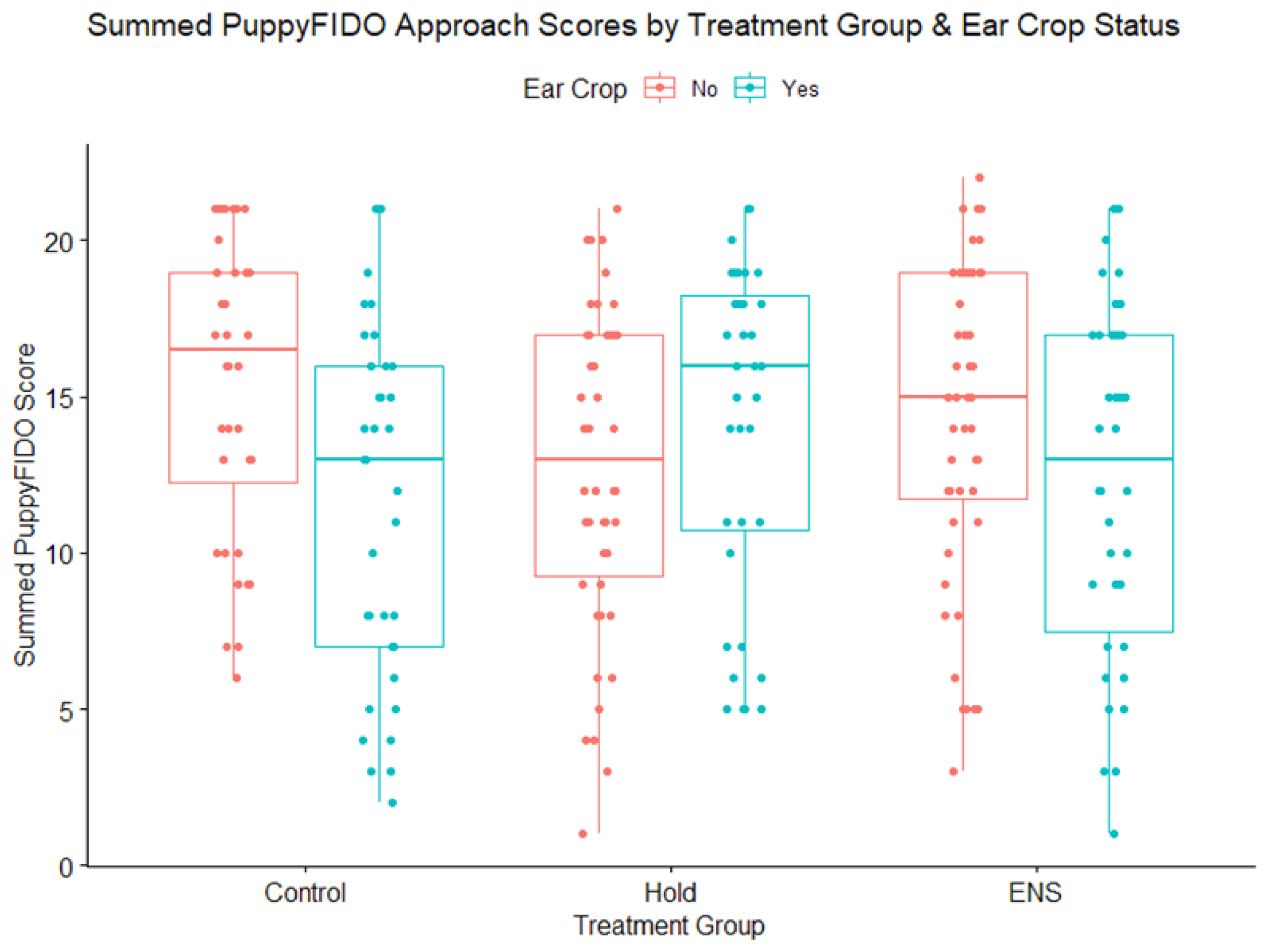
3.3. PuppyFIDO
3.4. Isolation
4. Discussion
4.1. Physical Health
4.2. Weight and Weight Gain
4.3. PuppyFIDO
4.4. Isolation
4.5. Limitations
4.6. Future Directions
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Taylor, K.; Mills, D. The Effect of the Kennel Environment on Canine Welfare: A Critical Review of Experimental Studies. Anim. Welfare 2007, 16, 435–447. [Google Scholar]
- Romaniuk, A.C.; Diana, A.; Barnard, S.; Weller, J.E.; Espinosa, U.B.; Dangoudoubiyam, S.; Shreyer, T.; Arnott, G.; Croney, C. The Effect of Transportation on Puppy Welfare from Commercial Breeding Kennels to a Distributor. Animals 2022, 12, 3379. [Google Scholar] [CrossRef] [PubMed]
- Moberg, G.P. Biological Response to Stress: Implications for Animal Welfare. In The Biology of Animal Stress: Basic Principles and Implications for Animal Welfare; Moberg, G.P., Mench, J.A., Eds.; CABI Publishing: Wallingford, UK, 2000; pp. 1–21. ISBN 0-85199-359-1. [Google Scholar]
- Dhabhar, F.S.; McEwen, B.S. Acute Stress Enhances While Chronic Stress Suppresses Cell-Mediated Immunity in Vivo: A Potential Role for Leukocyte Trafficking. Brain Behav. Immun. 1997, 11, 286–306. [Google Scholar] [CrossRef] [Green Version]
- Lehman, C.D.; Rodin, J.; McEwen, B.S.; Brinton, R. Impact of Environmental Stress on the Expression of Insulin-Dependent Diabetes Mellitus. Behav. Neurosci. 1991, 105, 241–245. [Google Scholar] [CrossRef] [PubMed]
- Cohen, M.M.; Jing, D.; Yang, R.R.; Tottenham, N.; Lee, F.S.; Casey, B.J. Early-Life Stress Has Persistent Effects on Amygdala Function and Development in Mice and Humans. Proc. Natl. Acad. Sci. USA 2013, 110, 18274–18278. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rice, C.J.; Sandman, C.A.; Lenjavi, M.R.; Baram, T.Z. A Novel Mouse Model for Acute and Long-Lasting Consequences of Early Life Stress. Endocrinology 2008, 149, 4892–4900. [Google Scholar] [CrossRef] [Green Version]
- Luine, V.; Villegas, M.; Martinez, C.; McEwen, B.S. Repeated Stress Causes Reversible Impairments of Spatial Memory Performance. Brain Res. 1994, 639, 167–170. [Google Scholar] [CrossRef]
- Boyce, W.T.; Ellis, B.J. Biological Sensitivity to Context: I. An Evolutionary–Developmental Theory of the Origins and Functions of Stress Reactivity. Dev. Psychopathol. 2005, 17, 271–301. [Google Scholar] [CrossRef]
- Maras, P.M.; Baram, T.Z. Early-Life Stress: Rodent Models, Lessons and Challenges. In Neuroendocrinology of Stress; Russell, J.A., Shipston, M.J., Eds.; John Wiley & Sons, Ltd: Chichester, UK, 2015; pp. 265–286. ISBN 9781118921692. [Google Scholar]
- Tarry-Adkins, J.L.; Ozanne, S.E. Mechanisms of Early Life Programming: Current Knowledge and Future Directions. Am. J. Clin. Nutr. 2011, 94, 1765S–1771S. [Google Scholar] [CrossRef] [Green Version]
- Castelli, V.; Lavanco, G.; Brancato, A.; Plescia, F. Targeting the Stress System during Gestation: Is Early Handling a Protective Strategy for the Offspring? Front. Behav. Neurosci. 2020, 14, 12. [Google Scholar] [CrossRef]
- Levine, S.; Haltmeyer, G.C.; Karas, G.G.; Denenberg, V.H. Physiological and Behavioral Effects of Infantile Stimulation. Physiol. Behav. 1967, 2, 55–59. [Google Scholar] [CrossRef]
- Plotsky, P.M.; Meaney, M.J. Early, Postnatal Experience Alters Hypothalamic Corticotropin-Releasing Factor (CRF) MRNA, Median Eminence CRF Content and Stress-Induced Release in Adult Rats. Brain Res. Mol. Brain Res. 1993, 18, 195–200. [Google Scholar] [CrossRef] [PubMed]
- Tremml, P.; Lipp, H.-P.; Müller, U.; Wolfer, D.P. Enriched Early Experiences of Mice Underexpressing the Beta-Amyloid Precursor Protein Restore Spatial Learning Capabilities but Not Normal Openfield Behavior of Adult Animals. Genes Brain Behav. 2002, 1, 230–241. [Google Scholar] [CrossRef] [PubMed]
- Parker, K.J.; Buckmaster, C.L.; Justus, K.R.; Schatzberg, A.F.; Lyons, D.M. Mild Early Life Stress Enhances Prefrontal-Dependent Response Inhibition in Monkeys. Biol. Psychiatry 2005, 57, 848–855. [Google Scholar] [CrossRef] [PubMed]
- Parker, K.J.; Buckmaster, C.L.; Schatzberg, A.F.; Lyons, D.M. Prospective Investigation of Stress Inoculation in Young Monkeys. Arch. Gen. Psychiatry 2004, 61, 933–941. [Google Scholar] [CrossRef] [Green Version]
- Parker, K.J.; Rainwater, K.L.; Buckmaster, C.L.; Schatzberg, A.F.; Lindley, S.E.; Lyons, D.M. Early Life Stress and Novelty Seeking Behavior in Adolescent Monkeys. Psychoneuroendocrinology 2007, 32, 785–792. [Google Scholar] [CrossRef] [Green Version]
- McCune, S. The Impact of Paternity and Early Socialisation on the Development of Cats’ Behaviour to People and Novel Objects. Appl. Anim. Behav. Sci. 1995, 45, 109–124. [Google Scholar] [CrossRef]
- Meier, G.W. Infantile Handling and Development in Siamese Kittens. J. Comp. Physiol. Psychol. 1961, 54, 284–286. [Google Scholar] [CrossRef]
- Zupan, M.; Rehn, T.; de Oliveira, D.; Keeling, L.J. Promoting Positive States: The Effect of Early Human Handling on Play and Exploratory Behaviour in Pigs. Animal 2016, 10, 135–141. [Google Scholar] [CrossRef] [Green Version]
- Raineki, C.; Lucion, A.B.; Weinberg, J. Neonatal Handling: An Overview of the Positive and Negative Effects. Dev. Psychobiol. 2014, 56, 1613–1625. [Google Scholar] [CrossRef] [Green Version]
- Núñez, J.F.; Ferré, P.; Escorihuela, R.M.; Tobeña, A.; Fernández-Teruel, A. Effects of Postnatal Handling of Rats on Emotional, HPA-Axis, and Prolactin Reactivity to Novelty and Conflict. Physiol. Behav. 1996, 60, 1355–1359. [Google Scholar] [CrossRef] [PubMed]
- Caldji, C.; Tannenbaum, B.; Sharma, S.; Francis, D.; Plotsky, P.M.; Meaney, M.J. Maternal Care during Infancy Regulates the Development of Neural Systems Mediating the Expression of Fearfulness in the Rat. Proc. Natl. Acad. Sci. USA 1998, 95, 5335–5340. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, D.; Diorio, J.; Tannenbaum, B.; Caldji, C.; Francis, D.; Freedman, A.; Sharma, S.; Pearson, D.; Plotsky, P.M.; Meaney, M.J. Maternal Care, Hippocampal Glucocorticoid Receptors, and Hypothalamic- Pituitary-Adrenal Responses to Stress. Science 1997, 277, 1659–1662. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Macrì, S.; Würbel, H. Developmental Plasticity of HPA and Fear Responses in Rats: A Critical Review of the Maternal Mediation Hypothesis. Horm. Behav. 2006, 50, 667–680. [Google Scholar] [CrossRef]
- Macrì, S.; Würbel, H. Effects of Variation in Postnatal Maternal Environment on Maternal Behaviour and Fear and Stress Responses in Rats. Anim. Behav. 2007, 73, 171–184. [Google Scholar] [CrossRef]
- Schaefer, T., Jr. Early “Experience” and Its Effects on Later Behavioral Processes in Rats: A Critical Factor in the Early Handling Phenomenon. Trans. NY Acad. Sci. 1963, 25, 871–889. [Google Scholar] [CrossRef]
- Durand, M.; Sarrieau, A.; Aguerre, S.; Mormède, P.; Chaouloff, F. Differential Effects of Neonatal Handling on Anxiety, Corticosterone Response to Stress, and Hippocampal Glucocorticoid and Serotonin (5-Ht)2a Receptors in Lewis Rats. Psychoneuroendocrinology 1998, 23, 323–335. [Google Scholar] [CrossRef]
- Karkow, A.R.M.; Lucion, A.B. Mild Environmental Intervention in Mother-Infant Interactions Reduces Social Play Behavior in Rats. Psychol. Neurosci. 2013, 6, 39–44. [Google Scholar] [CrossRef] [Green Version]
- Raineki, C.; Lutz, M.L.; Sebben, V.; Ribeiro, R.A.; Lucion, A.B. Neonatal Handling Induces Deficits in Infant Mother Preference and Adult Partner Preference. Dev. Psychobiol. 2013, 55, 496–507. [Google Scholar] [CrossRef]
- Silveira, P.P.; Portella, A.K.; Crema, L.; Correa, M.; Nieto, F.B.; Diehl, L.; Lucion, A.B.; Dalmaz, C. Both Infantile Stimulation and Exposure to Sweet Food Lead to an Increased Sweet Food Ingestion in Adult Life. Physiol. Behav. 2008, 93, 877–882. [Google Scholar] [CrossRef]
- Fox, M.W.; Stelzner, D. Behavioural Effects of Differential Early Experience in the Dog. Anim. Behav. 1966, 14, 273–281. [Google Scholar] [CrossRef]
- Battaglia, C.L. Periods of Early Development and the Effects of Stimulation and Social Experiences in the Canine. J. Vet. Behav. 2009, 4, 203–210. [Google Scholar] [CrossRef]
- Schoon, A.; Berntsen, T.G. Evaluating the Effect of Early Neurological Stimulation on the Development and Training of Mine Detection Dogs. J. Vet. Behav. 2011, 6, 150–157. [Google Scholar] [CrossRef]
- Gazzano, A.; Mariti, C.; Notari, L.; Sighieri, C.; McBride, E.A. Effects of Early Gentling and Early Environment on Emotional Development of Puppies. Appl. Anim. Behav. Sci. 2008, 110, 294–304. [Google Scholar] [CrossRef]
- Gazit, I.; Terkel, J.; Goldblatt, A. Are There Long-Term Effects of Early Neurological Stimulation (ENS) on Working Dogs? Appl. Anim. Behav. Sci. 2022, 249, 105588. [Google Scholar] [CrossRef]
- Bauer, A.E.; Jordan, M.; Colon, M.; Shreyer, T.; Croney, C.C. Evaluating FIDO: Developing and Pilot Testing the Field Instantaneous Dog Observation Tool. Pet Behav. Sci. 2017, 4, 1–15. [Google Scholar] [CrossRef] [Green Version]
- Stella, J.; Shreyer, T.; Ha, J.; Croney, C. Improving Canine Welfare in Commercial Breeding (CB) Operations: Evaluating Rehoming Candidates. Appl. Anim. Behav. Sci. 2019, 220, 104861. [Google Scholar] [CrossRef]
- Serpell, J.A. The Domestic Dog; Serpell, J.A., Ed.; Cambridge University Press: Cambridge, UK, 2016; ISBN 9781107024144. [Google Scholar]
- Guardini, G.; Mariti, C.; Bowen, J.; Fatjó, J.; Ruzzante, S.; Martorell, A.; Sighieri, C.; Gazzano, A. Influence of Morning Maternal Care on the Behavioural Responses of 8-Week-Old Beagle Puppies to New Environmental and Social Stimuli. Appl. Anim. Behav. Sci. 2016, 181, 137–144. [Google Scholar] [CrossRef] [Green Version]
- Flint, H.E.; Coe, J.B.; Serpell, J.A.; Pearl, D.L.; Niel, L. Identification of Fear Behaviors Shown by Puppies in Response to Nonsocial Stimuli. J. Vet. Behav. 2018, 28, 17–24. [Google Scholar] [CrossRef]
- Friard, O.; Gamba, M. BORIS: A Free, Versatile Open-Source Event-Logging Software for Video/Audio Coding and Live Observations. Methods Ecol. Evol. 2016, 7, 1325–1330. [Google Scholar] [CrossRef]
- Scaglia, E.; Cannas, S.; Minero, M.; Frank, D.; Bassi, A.; Palestrini, C. Video Analysis of Adult Dogs When Left Home Alone. J. Vet. Behav. 2013, 8, 412–417. [Google Scholar] [CrossRef]
- Denham, H.D.C.; Bradshaw, J.W.S.; Rooney, N.J. Repetitive Behaviour in Kennelled Domestic Dog: Stereotypical or Not? Physiol. Behav. 2014, 128, 288–294. [Google Scholar] [CrossRef] [PubMed]
- Lefebvre, D.; Depiereux, E.; Giffroy, J.-M.; Diederich, C. Research Note: Installation of Videorecording Equipment Had Minimal Effect on Behavioral Measurements in 14 Kenneled Military Dogs. J. Vet. Behav. Clin. Appl. Res. 2010, 5, 2–6. [Google Scholar] [CrossRef]
- Overall, K.L. The Ethogram Project. J. Vet. Behav. 2014, 9, 1–5. [Google Scholar] [CrossRef]
- Mugenda, L.W. Refining and Validating an On-Site Canine Welfare Assessment Tool Developed for Use in Commercial Breeding Kennels. MS Thesis, Purdue University, West Lafayette, IN, USA, 2018. [Google Scholar]
- Pinheiro, J.; Bates, D.; R Core Team. nlme: Linear and Nonlinear Mixed Effects Models, R Package Version 3.1-160; R Foundation for Statistical Computing: Vienna, Austria, 2021. [Google Scholar]
- Pinheiro, J.; Bates, D. Mixed-Effects Models in S and S-PLUS; Springer: New York, NY, USA, 2000. [Google Scholar] [CrossRef]
- Fox, J.; Weisburg, S. An R Companion to Applied Regression, 3rd ed.; Sage Publications: Thousand Oaks, CA, USA, 2019. [Google Scholar]
- Length, R. Emmeans: Estimated Marginal Means, AKA Least-Squares Means. R Package Version 1.8.3. 2022. Available online: https://rdrr.io/cran/emmeans/ (accessed on 12 October 2022).
- de Oliveira, D.; da Costa, M.J.R.P.; Zupan, M.; Rehn, T.; Keeling, L.J. Early Human Handling in Non-Weaned Piglets: Effects on Behaviour and Body Weight. Appl. Anim. Behav. Sci. 2015, 164, 56–63. [Google Scholar] [CrossRef]
- Barnard, S.; Pedernera, C.; Candeloro, L.; Ferri, N.; Velarde, A.; Villa, P.D. Development of a New Welfare Assessment Protocol for Practical Application in Long-Term Dog Shelters. Vet. Rec. 2016, 178, 18. [Google Scholar] [CrossRef]
- Stella, J.; Hurt, M.; Bauer, A.E.; Gomes, P.; Ruple, A.; Beck, A.; Croney, C.C. Does Flooring Substrate Impact Kennel and Dog Cleanliness in Commercial Breeding Facilities? Animals 2018, 8, 59. [Google Scholar] [CrossRef] [Green Version]
- Tallet, C.; Rakotomahandry, M.; Herlemont, S.; Prunier, A. Evidence of Pain, Stress, and Fear of Humans During Tail Docking and the Next Four Weeks in Piglets (Sus Scrofa Domesticus). Front. Vet. Sci. 2019, 6. [Google Scholar] [CrossRef]
- Elliot, O.; Scott, J.P. The Development of Emotional Distress Reactions to Separation, in Puppies. J. Genet. Psychol. 1961, 99, 3–22. [Google Scholar] [CrossRef]
- Ohl, F.; Arndt, S.S.; van der Staay, F.J. Pathological Anxiety in Animals. Vet. J. 2008, 175, 18–26. [Google Scholar] [CrossRef]
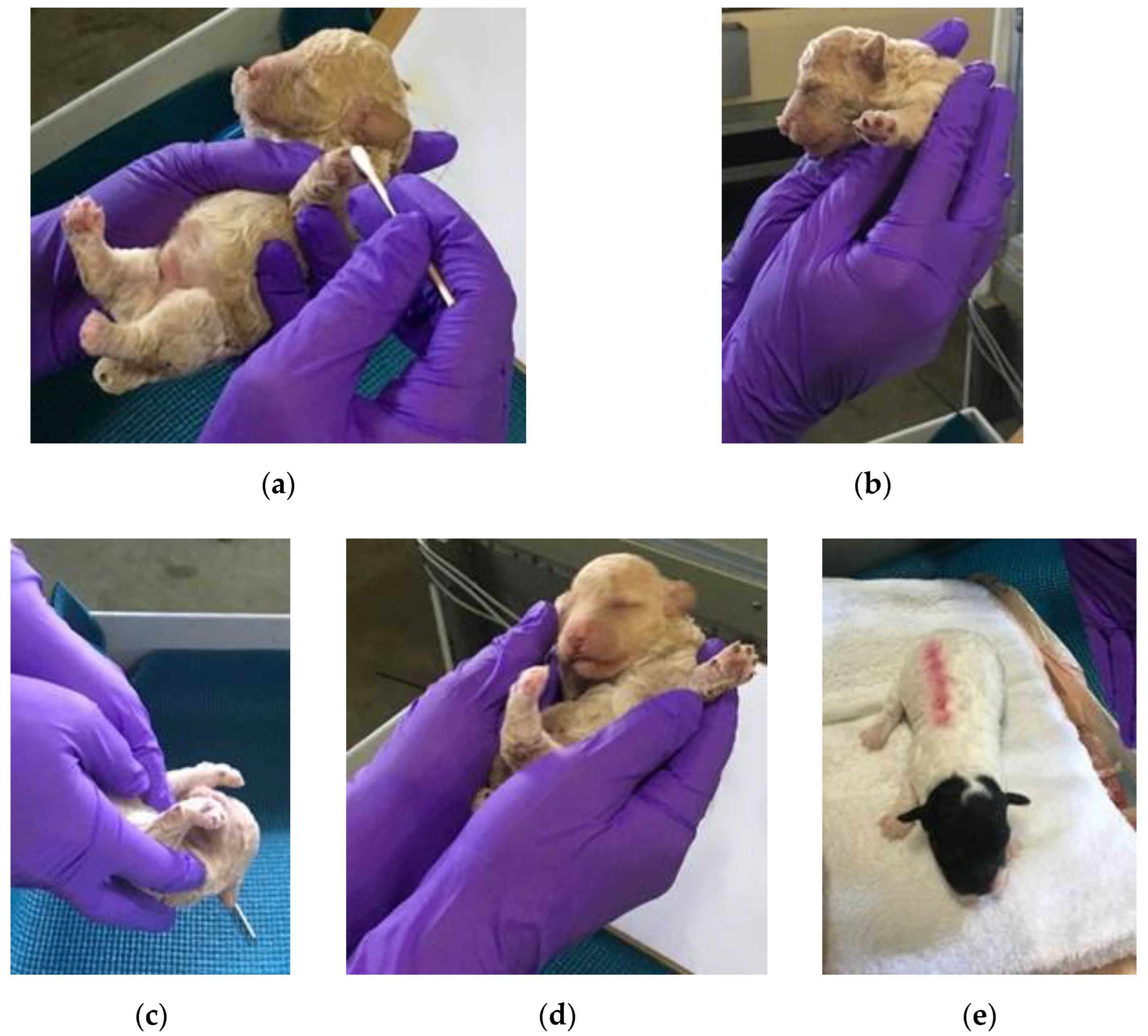
| Breed | Number of Puppies | Control | ENS | Held |
|---|---|---|---|---|
| Bichon/Toy Poodle cross | 8 | 3 | 2 | 3 |
| Miniature Pinscher * | 13 | 4 | 5 | 4 |
| Miniature Schnauzer * | 16 | 6 | 5 | 5 |
| Pomeranian | 15 | 6 | 4 | 5 |
| Bichon/Shih Tzu Cross | 11 | 3 | 4 | 4 |
| Toy Poodle | 13 | 3 | 5 | 5 |
| Total | 76 | 25 | 25 | 26 |
| Stage | Response | Definition |
|---|---|---|
| Orientation | Orientation | The puppy acknowledges the experimenter (i.e., makes eye contact/ is oriented toward) within 7 s. |
| No Orientation | The puppy does not acknowledge the experimenter’s presence within 7 s (or the duration of the step). | |
| Approach | Approach | The puppy moves toward the experimenter (i.e., takes steps toward him/her or leans toward him/her if he/she cannot step any closer). |
| Ambivalent Approach | The puppy approaches and retreats or approaches but then stops before reaching the experimenter [38]. | |
| No Approach | The puppy does not approach (i.e., does not move toward the experimenter) [38]. | |
| Behavior | Affiliative Behavior | Any behaviors exhibited by the puppy that are intended to facilitate the establishment or reinforcement of a social bond. Examples include approaching the experimenter while maintaining eye contact and/or making physical contact (e.g., licking and touching) with the experimenter. Outgoing The puppy jumps up or “scrambles” at the front of cage and/or attempts to cross/crosses the front barrier of cage and/or exhibits repeated physical contact with the experimenter (e.g., repeatedly licking, jumping on hands, etc.) and/or approaches the experimenter while exhibiting intense tail wagging. |
| Undisturbed | The puppy is engaging in an active behavior (e.g., sniffing, eating, etc.) when the step begins, then acknowledges the experimenter’s presence and returns to the same behavior [38]. | |
| Avoid | The puppy avoids the experimenter (i.e., moves away from them and turns its head in the opposite direction) [38]. | |
| Stationary | The dog is in a static posture (i.e., sitting and lying). There may or may not be visual orientation toward the environment. The dog may change posture in place but does not show any displacement [41] | |
| Posture | Normal | “Normal posture under neutral conditions” for specific breed and age [42] |
| Low | “Back rounded and/or legs bent…, head lowered” [42]. | |
| Additional | Fight/ Aggression | The puppy exhibits aggression (e.g., lunging, growling, teeth baring, etc.) [38]. |
| Bark | Negative affect Barking associated with avoidance, aggression, frustration, etc. Positive affect Barking associated with play, greeting, excitement, etc. | |
| Stereotypic Behavior | The puppy performs a pattern of behavior repeatedly (e.g., pacing, circling, etc.) [38]. |
| Category | Behavior | Description |
|---|---|---|
| Locomotory behaviors | Exploratory locomotion (duration) | Puppy engaged in motor activity (e.g., walking, trotting, and running) involved in targeted investigation of the environment (sniffing, pawing, licking, etc.) (adapted from [36,44]). |
| Non-exploratory locomotion (duration) | Puppy engaged in motor activity (e.g., walking, trotting, and running) without exploration of the environment (adapted from [44]). | |
| Repetitive behaviors * (duration) | Movement repeated along the same path (imaginary line, along the fence, or in a circle) or any stereotypic behaviors (repeated bouncing off pen walls, jumping up and down on the same spot with 2 or 4 legs in the air, pivoting on hind legs) (adapted from [45,46]). | |
| Stationary behaviors | Sit (duration) | Front legs straight, rear end lowered, and resting on “hocks” and “perineum” or floor (adapted from [42,47]). |
| Stand (duration) | Puppy is in an upright position supported by 3 or 4 legs (adapted from [46,47]). | |
| Lie down * (duration) | Puppy’s body is in contact with the ground, not supported by its legs. Head may be lifted or resting on the ground, and the eyes may be open or closed (adapted from [42,46]). | |
| Stationary exploration (duration) | Targeted investigation of the environment (sniffing, pawing, licking, etc.) while in a stationary position (sit, stand, or lie). | |
| Self-grooming * (duration) | “Action of cleaning of the body surface by licking, nibbling, picking, rubbing, scratching, and so on directed toward the animal’s body” [44]. | |
| Shaking head/body * (frequency) | Rotation of the head or the entire body, beginning with the head and moving toward the tail [47]. | |
| Escape attempt (duration) | Puppy attempts to leave test pen by pawing at pen walls, sticking nose through pen bars, or biting pen bars (adapted from [48]). | |
| Vocalizations | Any vocalization, e.g., bark, growl, howl, or whine (frequency) | Bark: “Sharp vocalization, often loud and repetitive” Growl: “Low-pitched grumble, with or without exposed teeth” Howl: “Low pitched, long duration vocalization” Whine: “High-pitched vocalization” [42]. |
| Elimination * | Urination (frequency) | Puppy expels urine from the body. |
| Defecation (frequency) | Puppy expels feces from the body. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Boone, G.; Romaniuk, A.C.; Barnard, S.; Shreyer, T.; Croney, C. The Effect of Early Neurological Stimulation on Puppy Welfare in Commercial Breeding Kennels. Animals 2023, 13, 71. https://doi.org/10.3390/ani13010071
Boone G, Romaniuk AC, Barnard S, Shreyer T, Croney C. The Effect of Early Neurological Stimulation on Puppy Welfare in Commercial Breeding Kennels. Animals. 2023; 13(1):71. https://doi.org/10.3390/ani13010071
Chicago/Turabian StyleBoone, Grace, Aynsley C. Romaniuk, Shanis Barnard, Traci Shreyer, and Candace Croney. 2023. "The Effect of Early Neurological Stimulation on Puppy Welfare in Commercial Breeding Kennels" Animals 13, no. 1: 71. https://doi.org/10.3390/ani13010071





