Exposure to Magnetic Fields Changes the Behavioral Pattern in Honeybees (Apis mellifera L.) under Laboratory Conditions
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Research Material
2.2. Exposure to Magnetic Field (MF)
- 10 min—time responding to a short flight, such as to collect water or for defecation
- 1 h—the mean time of a forage flight
- 3 h—long forage time, necessary to bring a heavy load over a long distance
- 1 mT
- 1.7 mT
2.3. Behavioral Analysis
- Walking—walking on the base surface or walls of the container
- Flight—short episodes of lifting up by wing movement
- Body cleaning—cleaning own body by legs
- Contact between individuals—any kind of near contact between bees, including touching antennas, trophallaxis
- Wings movement—the rapid movement of wings, used for ventilation, does not cause lifting up
- Stillness—staying motionlessly
- Loss of balance—bees from the walls of container fall and land on the bottom of the container upside down.
2.4. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- IPBES. Summary for Policymakers of the Assessment Report of the Intergovernmental Science-Policy Platform on Biodiversity and Ecosystem Services on Pollinators, Pollination and Food Production; Potts, S.G., Imperatriz-Fonseca, V.L., Ngo, H.T., Biesmeijer, J.C., Breeze, T.D., Dicks, L.V., Garibaldi, L.A., Hill, R., Settele, J., Vanbergen, A.J., et al., Eds.; IPBES Secretariat of the Intergovernmental Science-Policy Platform on Biodiversity and Ecosystem Services: Bonn, Germany, 2016. [Google Scholar]
- Gallai, N.; Salles, J.-M.; Settele, J.; Vaissière, B.E. Economic valuation of the vulnerability of world agriculture confronted with pollinator decline. Ecol. Econ. 2009, 68, 810–821. [Google Scholar] [CrossRef]
- Klein, A.-M.; Vaissière, B.E.; Cane, J.H.; Steffan-Dewenter, I.; Cunningham, S.A.; Kremen, C.; Tscharntke, T. Importance of pollinators in changing landscapes for world crops. Proc. R. Soc. B Biol. Sci. 2007, 274, 303–313. [Google Scholar] [CrossRef] [PubMed]
- Brown, M.J.F.; Paxton, R.J. The conservation of bees: A global perspective. Apidologie 2009, 40, 410–416. [Google Scholar] [CrossRef]
- Higes, M.; Hernández, R.M.; Meana, A. Nosema ceranaein Europe: An emergent type C nosemosis. Apidologie 2010, 41, 375–392. [Google Scholar] [CrossRef]
- Amdam, G.V.; Hartfelder, K.; Norberg, K.; Hagen, A.; Omholt, S.W. Altered Physiology in Worker Honey Bees (Hymenoptera: Apidae) Infested with the Mite Varroa destructor (Acari: Varroidae): A Factor in Colony Loss During Overwintering? J. Econ. Èntomol. 2004, 97, 741–747. [Google Scholar] [CrossRef]
- Parveen, N.; Miglani, R.; Kumar, A.; Dewali, S.; Kumar, K.; Sharma, N.; Bisht, S.S. Honey bee pathogenesis posing threat to its global population: A short review. Proc. Indian Natl. Sci. Acad. 2022, 88, 11–32. [Google Scholar] [CrossRef]
- Clair, A.L.S.; Zhang, G.; Dolezal, A.G.; O’Neal, E.M.; Toth, A.L. Diversified Farming in a Monoculture Landscape: Effects on Honey Bee Health and Wild Bee Communities. Environ. Èntomol. 2020, 49, 753–764. [Google Scholar] [CrossRef]
- Fairbrother, A.; Purdy, J.; Anderson, T.; Fell, R. Risks of neonicotinoid insecticides to honeybees. Environ. Toxicol. Chem. 2014, 33, 719–731. [Google Scholar] [CrossRef]
- Strachecka, A.; Gryzinska, M.; Krauze, M. Influence of Environmental Pollution on the Protective Proteolytic Barrier of the Honey Bee Apis mellifera mellifera. Pol. J. Environ. Stud. 2010, 19, 855–859. [Google Scholar]
- Le Conte, Y.; Navajas, M. Climate change: Impact on honey bee populations and diseases. Rev. Sci. Tech. 2008, 27, 485–510. [Google Scholar] [CrossRef]
- Greenberg, B.; Bindokas, V.P.; Frazier, M.J.; Gauger, J.R. Response of Honey Bees, Apis mellifera L., to High-Voltage Transmission Lines 1. Environ. Èntomol. 1981, 10, 600–610. [Google Scholar] [CrossRef]
- Migdał, P.; Roman, A.; Strachecka, A.; Murawska, A.; Bieńkowski, P. Changes of selected biochemical parameters of the honeybee under the influence of an electric field at 50 Hz and variable intensities. Apidologie 2020, 51, 956–967. [Google Scholar] [CrossRef]
- Halliday, D.; Resnick, R.; Walker, J. Podstawy Fizyki, Tom 3; Wydawnictwo Naukowe PWN: Warszawa, Poland, 2003. [Google Scholar]
- Fleischmann, P.N.; Grob, R.; Rössler, W. Magnetoreception in Hymenoptera: Importance for navigation. Anim. Cogn. 2020, 23, 1051–1061. [Google Scholar] [CrossRef] [PubMed]
- Moradi, M.; Naghdi, N.; Hemmati, H.; Asadi-Samani, M.; Bahmani, M. Effect of Ultra High Frequency Mobile Phone Radiation on Human Health. Electron. Physician 2016, 8, 2452–2457. [Google Scholar] [CrossRef] [PubMed]
- Shepherd, S.; Lima, M.A.P.; Oliveira, E.E.; Sharkh, S.M.; Jackson, C.W.; Newland, P.L. Extremely Low Frequency Electromagnetic Fields impair the Cognitive and Motor Abilities of Honey Bees. Sci. Rep. 2018, 8, 7932. [Google Scholar] [CrossRef]
- Kremers, D.; Marulanda, J.L.; Hausberger, M.; Lemasson, A. Behavioural evidence of magnetoreception in dolphins: Detection of experimental magnetic fields. Naturwissenschaften 2014, 101, 907–911. [Google Scholar] [CrossRef]
- Lohmann, K.J.; Putman, N.F.; Lohmann, C.M. The magnetic map of hatchling loggerhead sea turtles. Curr. Opin. Neurobiol. 2012, 22, 336–342. [Google Scholar] [CrossRef]
- Lohmann, K.J.; Lohmann, C.M.; el Jundi, B.; Kelber, A.; Webb, B. There and back again: Natal homing by magnetic navigation in sea turtles and salmon. J. Exp. Biol. 2019, 222 (Suppl. 1), jeb184077. [Google Scholar] [CrossRef]
- Guerra, A.P.; Gegear, R.J.; Reppert, S.M. A magnetic compass aids monarch butterfly migration. Nat. Commun. 2014, 5, 4164. [Google Scholar] [CrossRef]
- Lohmann, K.J.; Willows, A.O.; Pinter, R.B. An identifiable molluscan neuron responds to changes in earth-strength magnetic fields. J. Exp. Biol. 1991, 161, 1–24. [Google Scholar] [CrossRef]
- Cain, S.D.; Boles, L.C.; Wang, J.H.; Lohmann, K.J. Magnetic Orientation and Navigation in Marine Turtles, Lobsters, and Molluscs: Concepts and Conundrums. Integr. Comp. Biol. 2005, 45, 539–546. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lambinet, V.; Hayden, M.E.; Bieri, M.; Gries, G. Does the Earth’s Magnetic Field Serve as a Reference for Alignment of the Honeybee Waggle Dance? PLoS ONE 2014, 9, e115665. [Google Scholar] [CrossRef] [PubMed]
- Walker, M.M.; Bitterman, M. Short communication: Honeybees can be trained to respond to very small changes in geomagnetic field intensity. J. Exp. Biol. 1989, 145, 489–494. [Google Scholar] [CrossRef]
- Frey, A.H. Electromagnetic field interactions with biological systems 1. FASEB J. 1993, 7, 272–281. [Google Scholar] [CrossRef]
- Geffre, A.C.; Gernat, T.; Harwood, G.P.; Jones, B.M.; Gysi, D.M.; Hamilton, A.R.; Bonning, B.C.; Toth, A.L.; Robinson, G.E.; Dolezal, A.G. Honey bee virus causes context-dependent changes in host social behavior. Proc. Natl. Acad. Sci. USA 2020, 117, 10406–10413. [Google Scholar] [CrossRef]
- Williamson, S.M.; Willis, S.J.; Wright, G.A. Exposure to neonicotinoids influences the motor function of adult worker honeybees. Ecotoxicology 2014, 23, 1409–1418. [Google Scholar] [CrossRef]
- Migdał, P.; Murawska, A.; Bieńkowski, P.; Berbeć, E.; Roman, A. Changes in Honeybee Behavior Parameters under the Influence of the E-Field at 50 Hz and Variable Intensity. Animals 2021, 11, 247. [Google Scholar] [CrossRef]
- Migdał, P.; Murawska, A.; Strachecka, A.; Bieńkowski, P.; Roman, A. Honey Bee Proteolytic System and Behavior Parameters under the Influence of an Electric Field at 50 Hz and Variable Intensities for a Long Exposure Time. Animals 2021, 11, 863. [Google Scholar] [CrossRef]
- Shepherd, S.; Jackson, C.W.; Sharkh, S.M.; Aonuma, H.; Oliveira, E.E.; Newland, P.L. Extremely Low-Frequency Electromagnetic Fields Entrain Locust Wingbeats. Bioelectromagnetics 2021, 42, 296–308. [Google Scholar] [CrossRef]
- Todorović, D.; Marković, T.; Prolić, Z.; Mihajlović, S.; Rauš, S.; Nikolić, L.; Janać, B. The influence of static magnetic field (50 mT) on development and motor behaviour ofTenebrio (Insecta, Coleoptera). Int. J. Radiat. Biol. 2012, 89, 44–50. [Google Scholar] [CrossRef]
- Suchail, S.; Guez, D.; Belzunces, L.P. Characteristics of imidacloprid toxicity in twoApis melliferasubspecies. Environ. Toxicol. Chem. 2000, 19, 1901–1905. [Google Scholar] [CrossRef]
- Nahar, N.; Ohtani, T. Imidacloprid and Fipronil induced abnormal behavior and disturbed homing of forager honey bees Apis mellifera. J. Entomol. Zool. Stud. 2015, 3, 65–72. [Google Scholar]
- Shepherd, S.; Hollands, G.; Godley, V.C.; Sharkh, S.M.; Jackson, C.W.; Newland, P.L. Increased aggression and reduced aversive learning in honey bees exposed to extremely low frequency electromagnetic fields. PLoS ONE 2019, 14, e0223614. [Google Scholar] [CrossRef] [PubMed]
- Shepherd, S.; Lima, M.A.P.; Oliveira, E.E.; Sharkh, S.M.; Aonuma, H.; Jackson, C.; Newland, P.L. Sublethal neonicotinoid exposure attenuates the effects of electromagnetic fields on honey bee flight and learning. Environ. Adv. 2021, 4, 100051. [Google Scholar] [CrossRef]
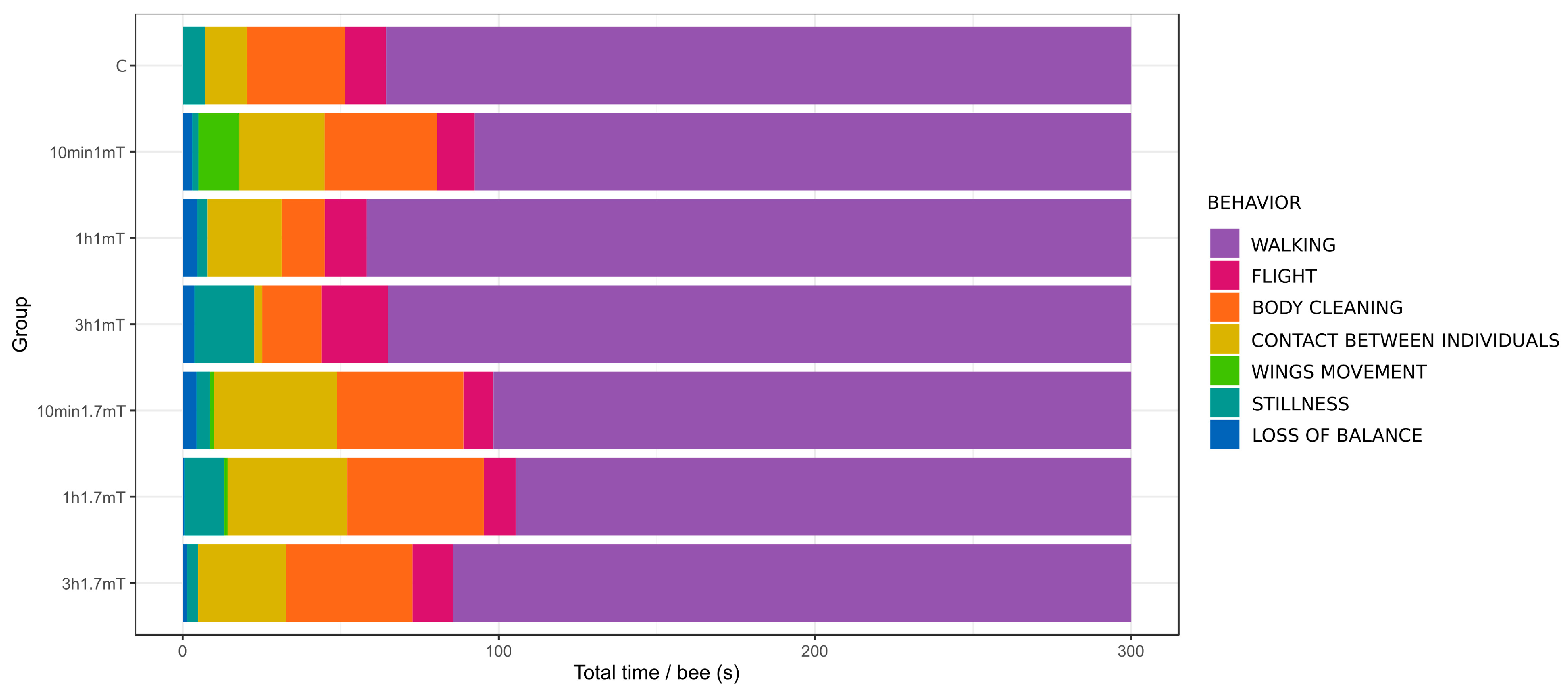
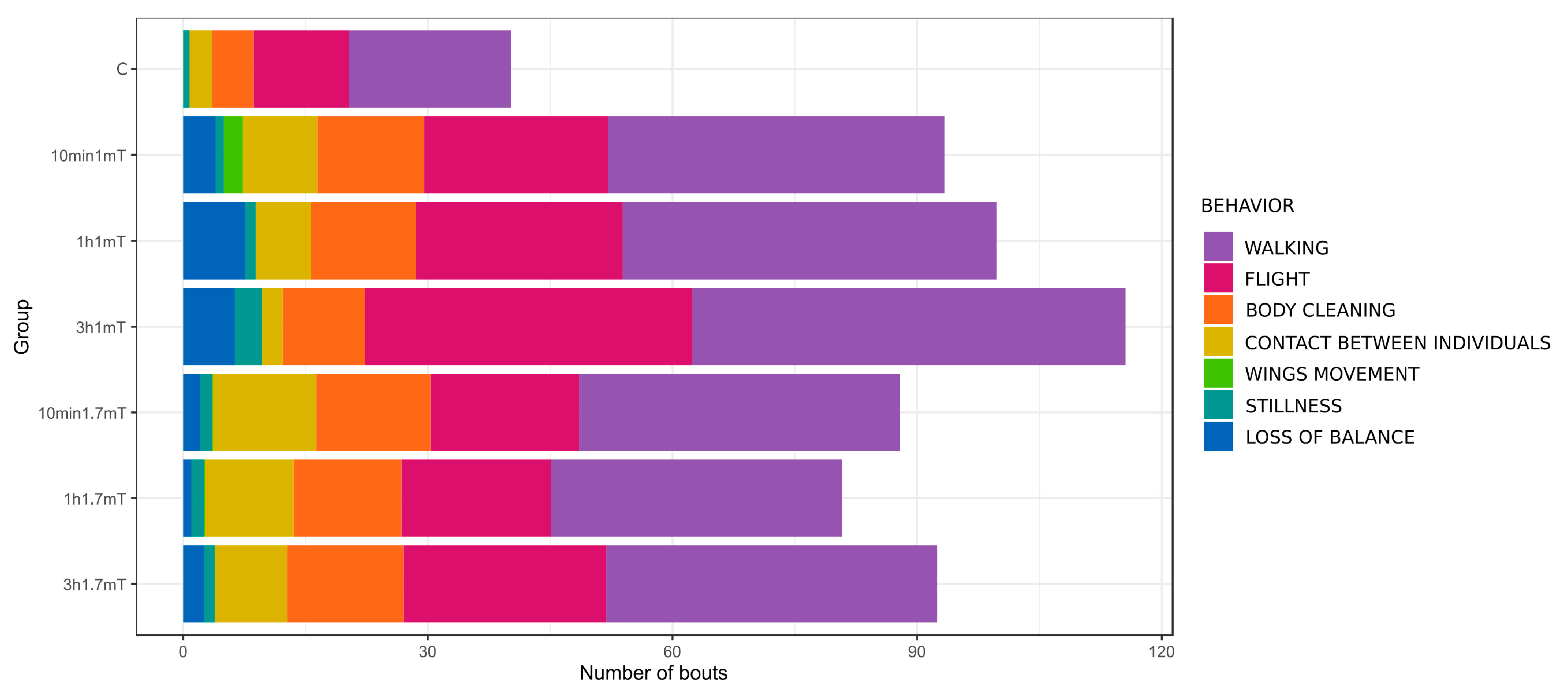
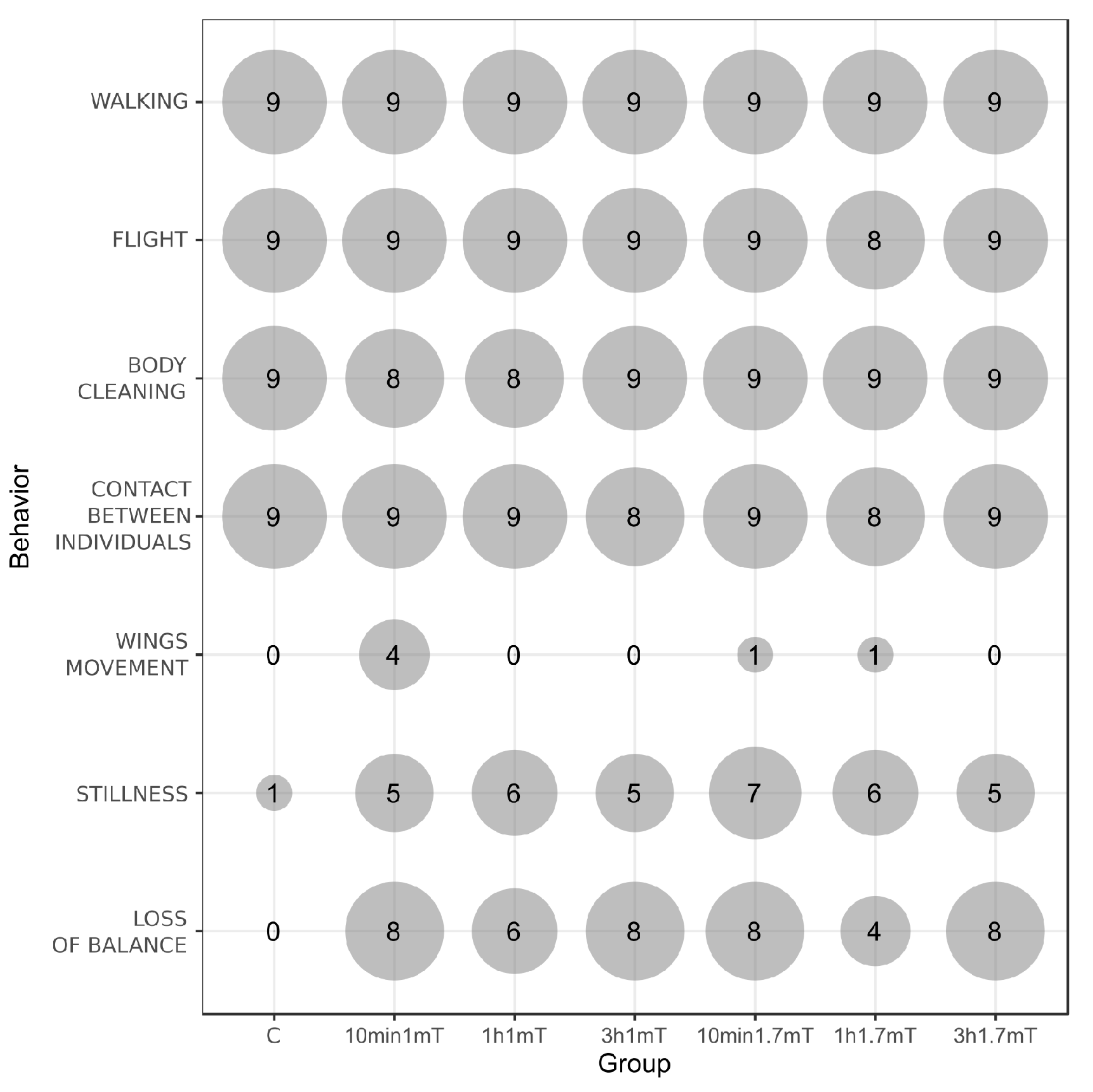
| Name of Behavior | Groups and Time of Exposure | p (chisq) | ||||||
|---|---|---|---|---|---|---|---|---|
| Control | 1 mT | 1.7 mT | ||||||
| 10 min | 1 h | 3 h | 10 min | 1 h | 3 h | |||
| Walking | 235.7 ± 30.7 | 207.7 ± 39.6 | 241.8 ± 24.9 | 235.1 ± 33.0 | 201.7 ± 42.6 | 194.7 ± 60.9 | 214.5 ± 42.0 | 0.094 |
| Flight | 12.9 ± 9.0 ab | 11.9 ± 12.3 ab | 13.2 ± 6.0 ab | 21.1 ± 8.1 a | 9.4 ± 9.1 b | 10.1 ± 9.8 ab | 12.8 ± 7.6 ab | 0.035 |
| Body cleaning | 31.1 ± 37.2 | 35.5 ± 29.0 | 13.7 ± 12.1 | 18.7 ± 29.0 | 40.1 ± 32.5 | 43.2 ± 64.9 | 40.1 ± 49.4 | 0.239 |
| Contact between individuals | 13.2 ± 13.4 ab | 27.0 ± 22.0 b | 23.5 ± 19.5 b | 2.6 ± 2.6 a | 38.8 ± 28.5 b | 37.8 ± 33.3 b | 27.8 ± 20.4 b | 0.0019 |
| Wings movement | NO | 13.0 ± 37.3 | NO | NO | 1.4 ± 4.2 | 1.0 ± 3.0 | NO | 0.558 |
| Stillness | 7.1 ± 21.4 | 1.9 ± 2.8 | 3.17 ± 5.4 | 18.9 ± 29.8 | 4.2 ± 6.0 | 12.6 ± 18.3 | 3.4 ± 4.2 | 0.348 |
| Loss of balance | NO | 3.1 ± 3.6 | 4.7 ± 11.7 | 3.7 ± 4.2 | 4.4 ± 10.9 | 0.6 ± 0.8 | 1.4 ± 1.0 | 0.289 |
| Behavior | Groups and Time of Exposure | p (chisq) | ||||||
|---|---|---|---|---|---|---|---|---|
| Control | 1 mT | 1.7 mT | ||||||
| 10 min | 1 h | 3 h | 10 min | 1 h | 3 h | |||
| Walking | 19.9 ± 6.5 a | 41.2 ± 17.0 b | 45.9 ± 9.3 b | 53.1 ± 12.4 b | 39.3 ± 13.5 b | 35.7 ± 15.2 ab | 40.6 ± 12.7 b | 0.00033 |
| Flight | 11.7 ± 7.4 a | 22.6 ± 22.3 ab | 25.3 ± 11.7 ab | 40.1 ± 14.9 b | 18.2 ± 17.0 a | 18.3 ± 16.3 a | 24.9 ± 14.6 ab | 0.0053 |
| Body cleaning | 5.1 ± 3.6 a | 13.1 ± 9.8 ab | 12.9 ± 7.0 ab | 10.1 ± 6.5 ab | 14.0 ± 5.8 b | 13.2 ± 3.5 b | 14.2 ± 5.8 b | 0.021 |
| Contact between individuals | 2.8 ± 1.7 a | 9.1 ± 6.7 b | 6.8 ± 3.5 ab | 2.6 ± 2.0 a | 12.7 ± 5.9 b | 10.9 ± 5.6 b | 8.9 ± 4.2 b | 7.8 × 10−5 |
| Wings movement | NO | 2.3 ± 4.3 | NO | NO | 0.1 ± 0.3 | 0.1 ± 0.3 | NO | 0.56 |
| Stillness | 0.8 ± 2.3 | 1.0 ± 1.3 | 1.3 ± 1.7 | 3.3 ± 5.2 | 1.4 ± 1.2 | 1.6 ± 1.5 | 1.3 ± 1.7 | 0.49 |
| Loss of balance | NO | 4.0 ± 5.4 | 7.6 ± 20.1 | 6.3 ± 6.6 | 2.1 ± 3.7 | 1.0 ± 1.7 | 2.6 ± 2.0 | 0.054 |
| TOTAL | 40.2 ± 12.4 a | 93.3 ± 36.7 b | 99.8 ± 19.3 b | 116.0 ± 27.0 b | 87.9 ± 25.8 b | 80.8 ± 31.8 ab | 92.4 ± 26.5 b | 0.00021 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Migdał, P.; Berbeć, E.; Bieńkowski, P.; Plotnik, M.; Murawska, A.; Latarowski, K. Exposure to Magnetic Fields Changes the Behavioral Pattern in Honeybees (Apis mellifera L.) under Laboratory Conditions. Animals 2022, 12, 855. https://doi.org/10.3390/ani12070855
Migdał P, Berbeć E, Bieńkowski P, Plotnik M, Murawska A, Latarowski K. Exposure to Magnetic Fields Changes the Behavioral Pattern in Honeybees (Apis mellifera L.) under Laboratory Conditions. Animals. 2022; 12(7):855. https://doi.org/10.3390/ani12070855
Chicago/Turabian StyleMigdał, Paweł, Ewelina Berbeć, Paweł Bieńkowski, Mateusz Plotnik, Agnieszka Murawska, and Krzysztof Latarowski. 2022. "Exposure to Magnetic Fields Changes the Behavioral Pattern in Honeybees (Apis mellifera L.) under Laboratory Conditions" Animals 12, no. 7: 855. https://doi.org/10.3390/ani12070855
APA StyleMigdał, P., Berbeć, E., Bieńkowski, P., Plotnik, M., Murawska, A., & Latarowski, K. (2022). Exposure to Magnetic Fields Changes the Behavioral Pattern in Honeybees (Apis mellifera L.) under Laboratory Conditions. Animals, 12(7), 855. https://doi.org/10.3390/ani12070855






