Integrated Taxonomy Revealed Genetic Differences in Morphologically Similar and Non-Sympatric Scoliodon macrorhynchos and S. laticaudus
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Collection
2.2. Molecular Analyses
2.3. Phylogenetic Analysis
2.4. Morphological and Meristic Data
2.5. Multivariate Analyses
2.6. Museum Holdings
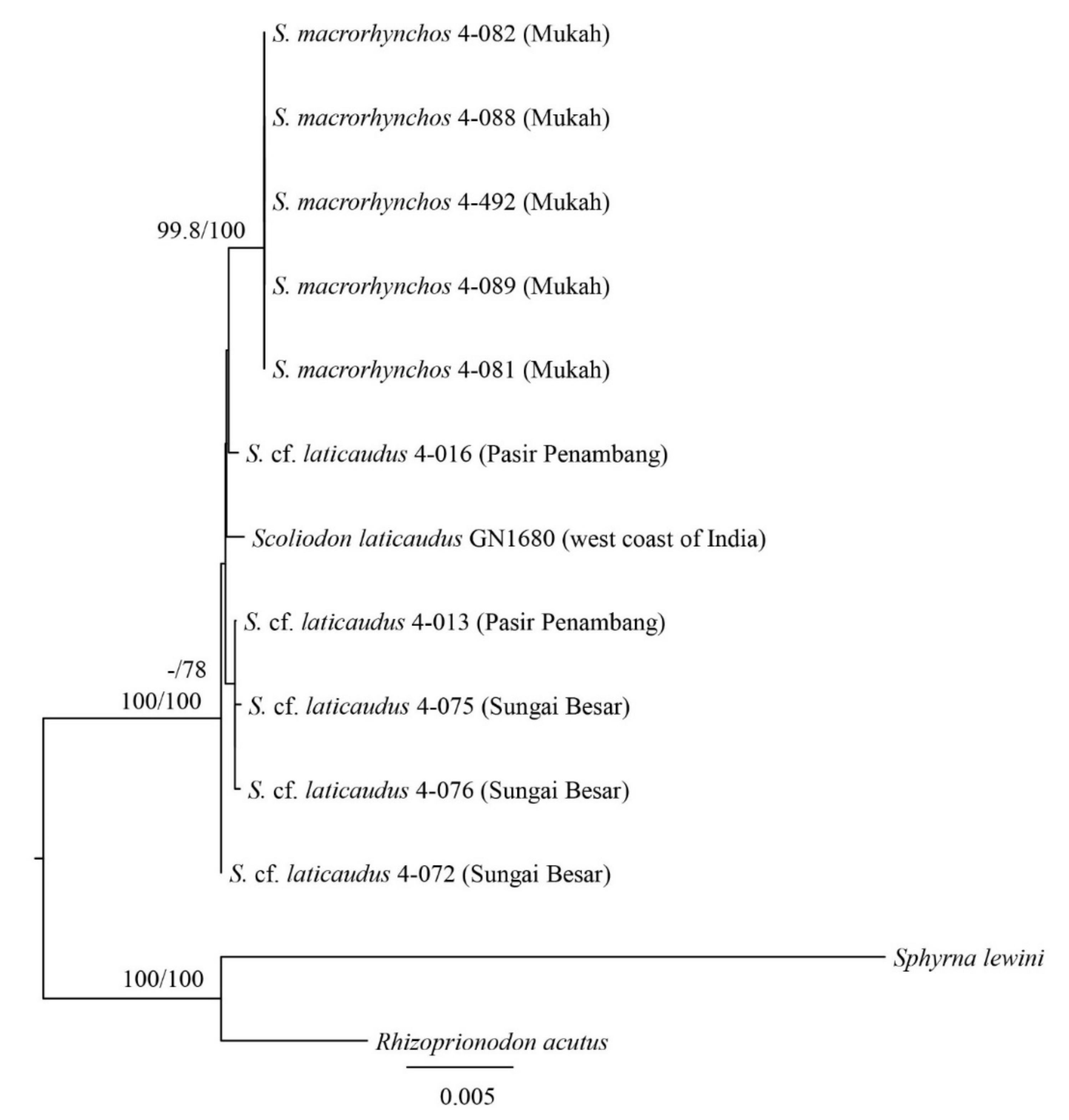
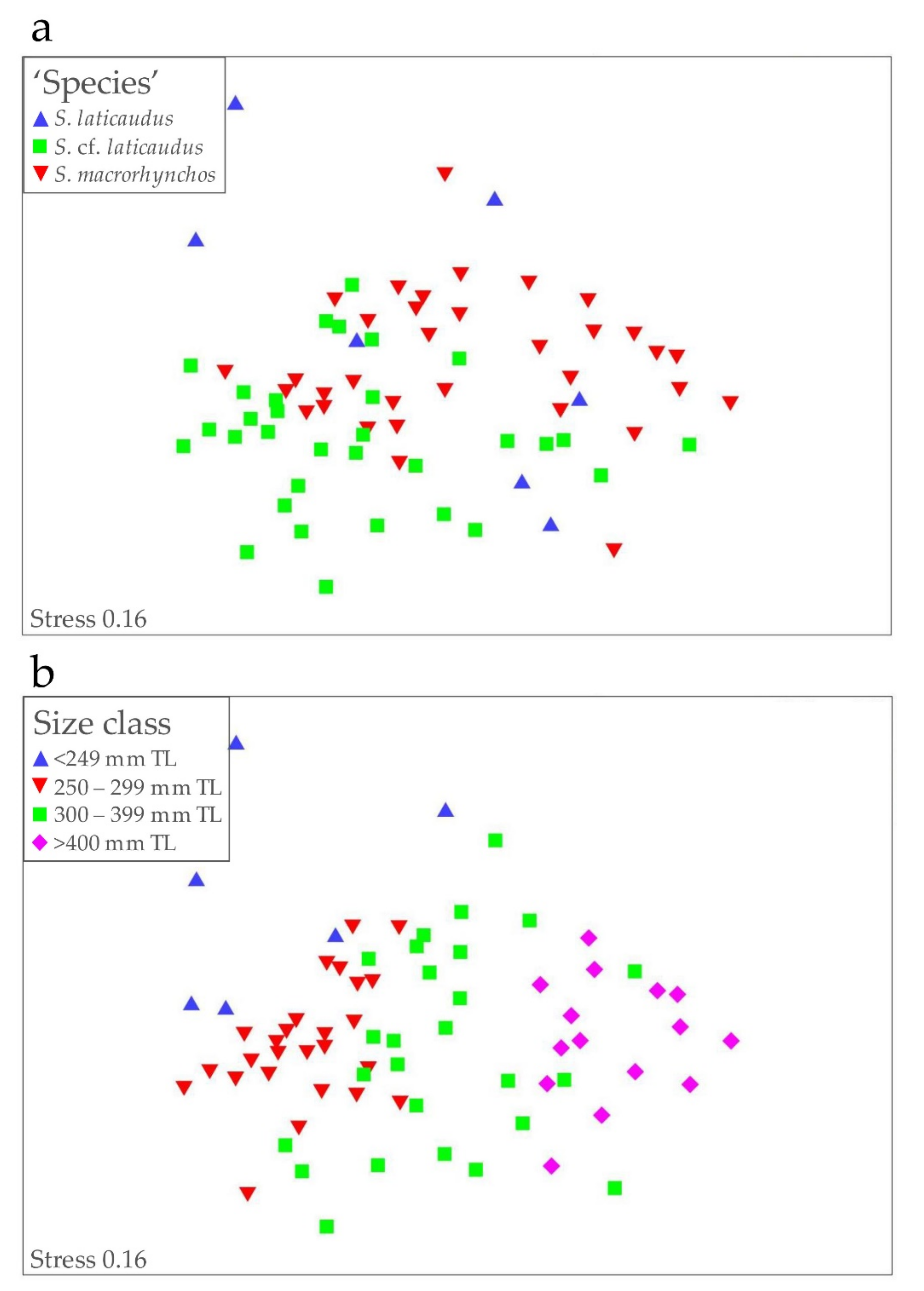
3. Results
3.1. Molecular Analysis
3.2. Morphology and Meristics
4. Discussion
4.1. Taxonomic Conclusions and Recommendations
4.2. Geographic Range
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Müller, J.; Henle, F.G.J. Gattungen der Haifische und Rochen nach einer von ihm mit Hrn. Henle unternommenen gemeinschaftlichen Arbeit über die Naturgeschichte der Knorpelfische. Ber. Königlichen Preuss. Akad. Wiss. Berl. 1837, 1837, 111–118. [Google Scholar]
- Müller, J.; Henle, F.G.J. Systematische Beschreibung der Plagiostomen; Veit und Comp: Berlin, Germany, 1938–1941; (pp. 1–28 published in 1838, 29–102 in 1839, 103–200 in 1841). [Google Scholar]
- Compagno, L.J.V. Sharks of the Order Carcharhiniformes; The Blackburn Press: Caldwell, NJ, USA, 1988. [Google Scholar]
- Compagno, L.J.V. FAO Species Catalogue. Volume 4, Sharks of the World. An Annotated and Illustrated Catalogue of Shark Species Known to Date; FAO Fisheries Synopsis No 125; FAO: Rome, Italy, 1984. [Google Scholar]
- White, W.T.; Last, P.R.; Naylor, G.J.P. Scoliodon macrorhynchos (Bleeker, 1852), a second species of spadenose shark from the Western Pacific (Carcharhiniformes: Carcharhinidae). In Descriptions of New Australian Chondrichthyans, CSIRO Marine and Atmospheric Research Paper 32; Last, P.R., White, W.T., Pogonoski, J.J., Eds.; CSIRO: Hobart, Australia, 2010; pp. 61–76. [Google Scholar]
- Bleeker, P. Bijdrage tot de kennis der Plagiostomen van den Indischen Archipel. Verh. Batav. Genoots. Kuns. 1852, 24, 1–92. [Google Scholar]
- Simpfendorfer, C. Scoliodon laticaudus. The IUCN Red List of Threatened Species. 2009. Available online: https://doi.org/10.2305/IUCN.UK.2009-2.RLTS.T39383A10188364.en (accessed on 14 February 2020).
- Naylor, G.J.P.; Caira, J.N.; Jensen, K.; Rosana, K.A.M.; White, W.T.; Last, P.R. A DNA sequence-based approach to the identification of shark and ray species and its implications for global elasmobranch diversity and parasitology. Bull. Am. Nat. Hist. Mus. 2012, 367, 1–263. [Google Scholar] [CrossRef]
- Ebert, D.A.; Fowler, S.; Compagno, L. Sharks of the World: A Fully Illustrated Guide; Wild Nature Press: Plymouth, UK, 2013. [Google Scholar]
- Dulvy, N.K.; Simpfendorfer, C.; Akhilesh, K.V.; Derrick, D.; Elhassan, I.; Fernando, D.; Haque, A.B.; Jabado, R.W.; Maung, A.; Valinassab, T.; et al. Scoliodon laticaudus. The IUCN Red List of Threatened Species. 2021. Available online: https://doi.org/10.2305/IUCN.UK.2021-2.RLTS.T169234201A173436322.en (accessed on 14 December 2021).
- Lim, K.C.; Then, A.Y.; Wee, A.K.S.; Sade, A.; Rumpet, R.; Loh, K.-H. Brown banded bamboo shark (Chiloscyllium punctatum) shows high genetic diversity and differentiation in Malaysian waters. Sci. Rep. 2021, 11, 14874. [Google Scholar] [CrossRef] [PubMed]
- Puckridge, M.; Last, P.R.; White, W.T.; Andreakis, N. Phylogeography of the Indo-West Pacific maskrays (Dasyatidae, Neotrygon): A complex example of chondrichthyan radiation in the Cenozoic. Ecol. Evol. 2013, 3, 217–232. [Google Scholar] [CrossRef]
- White, W.T.; Corrigan, S.; Yang, L.; Henderson, A.C.; Bazinet, A.L.; Swofford, D.L.; Naylor, G.J. Phylogeny of the manta and devilrays (Chondrichthyes: Mobulidae), with an updated taxonomic arrangement for the family. Zool. J. Linnean. Soc. 2018, 182, 50–75. [Google Scholar] [CrossRef]
- Petean, F.F.; Naylor, G.J.; Lima, S.M. Integrative taxonomy identifies a new stingray species of the genus Hypanus Rafinesque, 1818 (Dasyatidae, Myliobatiformes), from the Tropical Southwestern Atlantic. J. Fish. Biol. 2020, 97, 1120–1142. [Google Scholar] [CrossRef]
- Galtier, N.; Nabholz, B.; Glémin, S.; Hurst, G.D.D. Mitochondrial DNA as a marker of molecular diversity: A reappraisal. Mol. Ecol. 2009, 18, 4541–4550. [Google Scholar] [CrossRef]
- Balloux, F. The worm in the fruit of the mitochondrial DNA tree. Heredity 2010, 104, 419–420. [Google Scholar] [CrossRef]
- Awadalla, P.; Eyre-Walker, A.; Maynard-Smith, J. Linkage disequilibrium and recombination in hominid mitochondrial DNA. Science 1999, 286, 2524–2525. [Google Scholar] [CrossRef]
- Eyre-Walker, A.; Smith, N.H.; Maynard-Smith, J. How clonal are human mitochondria? Proc. R. Soc. B Biol. Sci. 1999, 266, 477–483. [Google Scholar] [CrossRef] [PubMed]
- Hagelberg, E.; Goldman, N.; Lió, P.; Whelan, S.; Schiefenhöel, W.; Clegg, J.B.; Bowden, D.K. Evidence for mitochondrial DNA recombination in a human population of island Melanesia. Proc. R. Soc. B Biol. Sci. 1999, 266, 485–492. [Google Scholar] [CrossRef]
- Rand, D.M. The units of selection on mitochondrial DNA. Annu. Rev. Ecol. Syst. 2001, 32, 415–448. [Google Scholar] [CrossRef]
- Bazin, E.; Glémin, S.; Galtier, N. Population size does not influence mitochondrial genetic diversity in animals. Science 2006, 312, 570–572. [Google Scholar] [CrossRef] [PubMed]
- Castoe, T.A.; de Koning, A.J.; Kim, H.M.; Gu, W.; Noonan, B.P.; Naylor, G.; Jiang, Z.J.; Parkinson, C.L.; Pollock, D.D. Evidence for an ancient adaptive episode of convergent molecular evolution. Proc. Natl. Acad. Sci. USA 2009, 106, 8986–8991. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Jameson, D.; Tang, B.; Higgs, P.G. The relationship between the rate of molecular evolution and the rate of genome rearrangement in animal mitochondrial genomes. J. Mol. Evol. 2006, 63, 375–392. [Google Scholar] [CrossRef] [PubMed]
- Nabholz, B.; Glémin, S.; Galtier, N. Strong variations of mitochondrial mutation rate across mammals—The longevity hypothesis. Mol. Biol. Evol. 2008, 25, 120–130. [Google Scholar] [CrossRef]
- Nabholz, B.; Glémin, S.; Galtier, N. The erratic mitochondrial clock: Variations of mutation rate, not population size, affect mtDNA diversity across mammals and birds. BMC Evol. Biol. 2009, 9, 54. [Google Scholar] [CrossRef]
- Arunrugstichai, S.; True, J.D.; White, W.T. Catch composition and aspects of the biology of sharks caught by Thai commercial fisheries in the Andaman Sea. J. Fish. Biol. 2018, 92, 1487–1504. [Google Scholar] [CrossRef]
- Aschliman, N.C.; Cleason, K.M.; McEachran, J.D. Phylogeny of batoidea. In Biology of Sharks and Their Relatives, 2nd ed.; Carrier, J.C., Musick, J.A., Heithaus, M.R., Eds.; CRC Press: Boca Raton, FL, USA, 2012; pp. 57–95. [Google Scholar]
- Hyde, J.R.; Lynn, E.; Humphreys, R., Jr.; Musyl, M.; West, A.P.; Vetter, R. Shipboard identification of fish eggs and larvae by multiplex PCR, and a description of fertilized eggs of blue marlin, short bill spearfish, and wahoo. Mar. Ecol. Prog. Ser. 2005, 286, 269–277. [Google Scholar] [CrossRef]
- Ward, R.D.; Holmes, B.H.; White, W.T.; Last, P.R. DNA barcoding Australasian chondrichthyans: Results and potential uses in conservation. Mar. Freshwat. Res. 2008, 59, 57–71. [Google Scholar] [CrossRef]
- Naylor, G.J.P.; Ryburn, J.A.; Fedrigo, O.; Lopez, J.A. Phylogenetic relationships among the major lineages of modern elasmobranchs. In Reproductive Biology and Phylogeny: Sharks, Skates, Stingrays, and Chimaeras; Hamlett, W.C., Jamieson, B.G.M., Eds.; Science Publishers Inc.: Plymouth, UK, 2005; pp. 1–25. [Google Scholar]
- Hall, T.A. BioEdit: A user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp. Ser. 1999, 41, 95–98. [Google Scholar]
- Thompson, J.D.; Gibson, T.J.; Plewniak, F.; Jeanmougin, F.; Higgins, D.G. The CLUSTAL_X windows interface: Flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res. 1997, 25, 4876–4882. [Google Scholar] [CrossRef] [PubMed]
- Tanabe, A.S. Kakusan: A computer program to automate the selection of a nucleotide substitution model and the configuration of a mixed model on multilocus data. Mol. Ecol. Notes 2007, 7, 962–964. [Google Scholar] [CrossRef]
- Jobb, G.; von Haeseler, A.; Strimmer, K. Treefinder: A powerful graphical analysis environment for molecular phylogenetics. BMC Evol. Biol. 2004, 4, 18. [Google Scholar] [CrossRef]
- Huelsenbeck, J.P.; Ronquist, F. MrBayes: Bayesian Inference of Phylogenetic Trees. Bioinformatics 2001, 17, 754–755. [Google Scholar] [CrossRef]
- Swofford, D.L. PAUP*: Phylogenetic Analysis Using Parsimony (* and Other Methods), Version 4; Sinauer Associates: Sunderland, MA, USA, 2002. [Google Scholar]
- Zhang, C.; Rabiee, M.; Sayyari, E.; Mirarab, S. ASTRAL-III: Polynomial time species tree reconstruction from partially resolved gene trees. BMC Bioinform. 2018, 19, 153. [Google Scholar] [CrossRef]
- Rabiee, M.; Sayyari, E.; Mirarab, S. Multi-Allele Species Reconstruction Using ASTRAL. Mol. Phylogenet. Evol. 2019, 130, 286–296. [Google Scholar] [CrossRef]
- Yang, Z.; Rannala, B. Unguided species delimitation using DNA sequence data from multiple loci. Mol. Biol. Evol. 2014, 31, 3125–3135. [Google Scholar] [CrossRef]
- Rannala, B.; Yang, Z. Efficient Bayesian species tree inference under the multispecies coalescent. Syst. Biol. 2017, 66, 823–842. [Google Scholar] [CrossRef]
- Flouri, T.; Jiao, X.; Rannala, B.; Yang, Z. Species tree inference with BPP using genomic sequences and the multispecies coalescent. Mol. Biol. Evol. 2018, 35, 2585–2593. [Google Scholar] [CrossRef] [PubMed]
- Silvestro, D.; Michalak, I. raxmlGUI: A graphical front-end for RAxML. Org. Divers. Evol. 2012, 12, 335–337. [Google Scholar] [CrossRef]
- Compagno, L.J.V. Sharks of the World: An Annotated and Illustrated Catalogue of Shark Species Known to Date, Volume 2. Bullhead, Mackerel and Carpet Sharks (Heterodontiformes, Lamniformes and Orectolobiformes); FAO: Rome, Italy, 2001. [Google Scholar]
- Compagno, L.J.V. Carcharhinoid Sharks: Morphology, Systematics and Phylogeny. Ph.D. Thesis, Stanford University, Stanford, CA, USA, 1979. [Google Scholar]
- Springer, V.G.; Garrick, J.A.F. A survey of vertebral numbers in sharks. Proc. U. S. Natl. Mus. 1964, 116, 73–96. [Google Scholar] [CrossRef]
- Clarke, K.R.; Gorley, R.N. PRIMER v6 User Manual/Tutorial; Primer-E Ltd.: Plymouth, UK, 2006. [Google Scholar]
- Finucci, B.; White, W.T.; Kemper, J.M.; Naylor, G.J. Redescription of Chimaera ogilbyi (Chimaeriformes; Chimaeridae) from the Indo-Australian region. Zootaxa 2018, 4375, 191–210. [Google Scholar] [CrossRef] [PubMed]
- Hirano, T.; Saito, T.; Tsunamoto, Y.; Koseki, J.; Ye, B.; Miura, O.; Suyama, Y.; Chiba, S. Enigmatic incongruence between mtDNA and nDNA revealed by multi-locus phylogenomic analyses in freshwater snails. Sci. Rep. 2019, 9, 6223. [Google Scholar] [CrossRef] [PubMed]
- Wiens, J.J.; Kuczynski, C.A.; Stephens, P.R. Discordant mitochondrial and nuclear gene phylogenies in emydid turtles: Implications for speciation and conservation. Biol. J. Linn. Soc. 2010, 99, 445–461. [Google Scholar] [CrossRef]
- Giles, J.L.; Riginos, C.; Naylor, G.J.; Dharmadi; Ovenden, J.R. Genetic and phenotypic diversity in the wedgefish Rhynchobatus australiae, a threatened ray of high value in the shark fin trade. Mar. Ecol. Prog. Ser. 2016, 548, 165–180. [Google Scholar] [CrossRef]
- Ma, K.Y.; van Herverden, L.; Newman, S.J.; Brumen, M.L.; Choat, J.H.; Chu, K.H.; de Mitcheson, Y.S. Contrasting population genetic structure in three aggregating groupers (Percoidei: Epinephelidae) in the Indo-West Pacific: The importance of reproductive mode. BMC Evol. Biol. 2018, 18, 180. [Google Scholar] [CrossRef]
- Reid, D.G.; Lal, K.; Mackenzie-Dodds, J.; Kaligis, F.; Littlewood, D.T.J.; Williams, S.T. Comparative phylogeography and species boundaries in Echinolittorina snails in the central Indo-West Pacific. J. Biogeogr. 2006, 33, 990–1006. [Google Scholar] [CrossRef]
- Mantiquilla, J.A.; Shiao, M.S.; Shih, H.C.; Chen, W.H.; Chiang, Y.C. A review on the genetic structure of ecologically and economically important mangrove species in the Indo-West Pacific. Ecol. Genet. Genom. 2021, 18, 100078. [Google Scholar] [CrossRef]
- Hall, R. Cenozoic geological and plate tectonic evolution of SE Asia and the SW Pacific: Computer-based reconstructions, model and animations. J. Asian Earth Sci. 2002, 20, 353–431. [Google Scholar] [CrossRef]
- Ahmad, A.; Abdul Haris Hilmi, A.A.; Ismail, I. Implementation of the National Plan of Action for Conservation and Management of Shark Resources in Malaysia (Malaysia NPOA-Shark); Terminal Report; SEAFDEC/MFRDMD: Kuala Terengganu, Malaysia, 2015. [Google Scholar]
- Arai, T.; Azri, A. Diversity, occurrence and conservation of sharks in the southern South China Sea. PLoS ONE 2019, 14, e0213864. [Google Scholar] [CrossRef] [PubMed]
- Fahmi; Tibbetts, I.R.; Bennett, M.B.; Ali, A.; Krajangdara, T.; Dudgeon, C.L. Population structure of the brown-banded bamboo shark, Chiloscyllium punctatum and its relation to fisheries management in the Indo-Malay region. Fish. Res. 2021, 240, 105972. [Google Scholar] [CrossRef]
- FAO. FAO Yearbook. Fishery and Aquaculture Statistics 2016; FAO: Rome, Italy, 2018; 104p. [Google Scholar]
- Kamohara, T. Revised catalogue of fishes of Kochi Prefecture, Japan. Rep. USA Mar. Biol. Stat. 1964, 11, 1–99. [Google Scholar]
- Nakaya, K. Carcharhinidae. In The Fishes of the Japanese Archipelago; Masuda, H., Amaoka, K., Araga, C., Uyeno, T., Yoshino, T., Eds.; Tokai University Press: Tokyo, Japan, 1984; pp. 5–6. [Google Scholar]
- Nakabo, T. Fishes of Japan with Pictorial Keys to the Species, 3rd ed.; Tokai University Press: Hadano, Japan, 2013. [Google Scholar]
- Okada, Y.; Mori, K. Descriptions and figures of marine fishes obtained at Mie Prefecture, the middle of Honshu, Japan. J. Fac. Fish. Pref. Univ. Mie 1958, 3, 1–39. [Google Scholar]
- Kamohara, T. List of fishes from Amami-Oshima and adjacent regions, Kagoshima Prefecture, Japan. Rep. USA Mar. Biol. Stat. 1957, 4, 1–65. [Google Scholar]
- Shinohara, G.; Matsuura, K.; Shirai, S. Fishes of Tachibana Bay, Nagasaki, Japan. Mem. Nat. Sci. Mus. Tokyo 1998, 30, 105–138. [Google Scholar]
- Cho, H.G.; Kweon, S.M.; Kim, B.J. New record of the spadenose shark, Scoliodon laticaudus (Carcharhiniformes: Carcharhinidae) from South Sea, Korea. Korean J. Ichthyol. 2014, 26, 336–339. [Google Scholar]
- Wang, K.F. Preliminary notes on the fishes of Chekiang (Elasmobranches). Contr. Biol. Lab. Sci. Soc. China Zool. Ser. 1933, 9, 87–117. [Google Scholar]
- Zhu, J.F.; Dai, X.J.; Li, Y. Preliminary study on biological characteristics of spadenose shark, Scoliodon laticaudus, caught from coastal waters of Zhejiang Province. J. Shanghai Fish. Inst. 2008, 17, 635–639. [Google Scholar]
- Lam, V.Y.Y.; Sadovy de Micheson, Y. The sharks of South East Asia—Unknown, unmonitored and unmanaged. Fish Fish. 2010, 12, 51–74. [Google Scholar] [CrossRef]
- Ebert, D.A.; White, W.T.; Ho, H.C.; Last, P.R.; Nakaya, K.; Séret, B.; Straube, N.; Naylor, G.J.P.; de Carvalho, M.R. An annotated checklist of the chondrichthyans of Taiwan. Zootaxa 2013, 3752, 279–386. [Google Scholar] [CrossRef] [PubMed]
- Orlov, A.M. Brief review of the marine ichthyofauna of Cambodia. J. Ichthyol. 1995, 35, 81–87. [Google Scholar]
- Deechum, W. Species Compositions and Some Biological Aspects of Sharks and Rays from the Gulf of Thailand and Andaman Landing Sites. Ph.D. Thesis, Prince of Songkla University, Songkla, Thailand, 2009. [Google Scholar]
- Springer, V.G. A revision of the carcharhinid shark genera Scoliodon, Loxodon, and Rhizoprionodon. Proc. U. S. Nat. Mus. 1964, 115, 559–632. [Google Scholar] [CrossRef]
- Compagno, L.J.V.; Last, P.R.; Stevens, J.D.; Alava, M.N.R. Checklist of Philippine chondrichthyes. CSIRO Mar. Lab. Rep. 2005, 243, 103. [Google Scholar]
- Alava, M.N.R.; Gaudiano, J.P.A.; Utzurrum, J.T.; Capuli, E.E.; Aquino, M.T.R.; Luchvez-Maypa, M.M.A.; Santos, M.D. Pating Ka Ba? In An Identification Guide to Sharks, Batoids and Chimaeras of the Philippines; Department of Agriculture Bureau of Fisheries and Aquatic Resources—National Fisheries Research and Development Institute and the Marine Wildlife Watch of the Philippines: Metro Manila, Philippines, 2014; 200p. [Google Scholar]
- Last, P.R.; White, W.T.; Caira, J.N.; Jensen, K.; Lim, A.P.K.; Manjaji-Matsumoto, B.M.; Naylor, G.J.P.; Pogonoski, J.J.; Stevens, J.D.; Yearsley, G.K. Sharks and Rays of Borneo; CSIRO Publishing: Melbourne, Australia, 2010. [Google Scholar]
- Emiliya, P.A.; Putra, R.D. Identifikasi jenis hiu Hasil Tangkapan Nelayan di Pulau Bintan Provinsi Kepulauan Riau [Identification of the Type Shark Fishermen Catch in Bintan Island Riau Islands Province]; Project Report; Universitas Maritim Raja Ali Haji: Kota Tanjung Pinang, Indonesia, 2017. [Google Scholar]
- Teshima, K.; Ahmad, M.; Mizue, K. Studies on sharks—XIV. Reproduction in the Telok Anson shark collected from Perak River, Malaysia. Jpn. J. Ichthyol. 1978, 25, 181–189. [Google Scholar]
- Widodo, J.; Pralampita, W.A.; Chodriyah, U. Length-weight relationships and condition factors of sharks landed from the Indian Ocean south of Java, Bali, and Lombok, Indonesia. In Proceedings of the First Annual Meeting on Artisanal Shark and Rays Fisheries in East Indonesia: Their Socio-Economic and Fishery Characteristics and relationship to Australian Resources, Perth, Australia, 4–5 April 2002. [Google Scholar]
- Widodo, A.A.; Mahiswara, M. Sumberdaya ikan cucut (hiu) yang tertangkap nelayan di perairan Laut Jawa [The shark resource caught by fishermen in Java Sea]. J. Iktiol. Indones. 2007, 7, 15–21. [Google Scholar]
- Bean, B.A.; Weed, A.C. Notes on a collection of fishes from Java, made by Owen Bryant & William Palmer in 1909. Proc. U. S. Nat. Mus. 1912, 42, 587–611. [Google Scholar]
- White, W.T. Catch composition and reproductive biology of whaler sharks (Carcharhiniformes: Carcharhinidae) caught by fisheries in Indonesia. J. Fish Biol. 2007, 71, 1512–1540. [Google Scholar] [CrossRef]
- Psomadakis, P.N.; Thein, H.; Russell, B.C.; Tun, M.T. Field Identification Guide to the Living Marine Resources of Myanmar, FAO Species Identification Guide for Fishery Purposes; FAO: Rome, Italy, 2019. [Google Scholar]
- Jit, R.B.; Alam, M.F.; Rhaman, M.G.; Singha, N.K.; Akhtar, A. Landing trends, species composition and percentage composition of sharks and rays in Chittagong and Cox’s Bazar, Bangladesh. Int. J. Adv. Res. Biol. Sci. 2014, 1, 81–93. [Google Scholar]
- Kumar, R.R.; Venu, S.; Akhilesh, K.V.; Bineesh, K.K.; Rajan, P.T. First report of four deep-sea chondrichthyans (Elasmobranchii and Holocephali) from Andaman waters, India with an updated checklist from the region. Acta Ichthyol. Piscat. 2018, 48, 289–301. [Google Scholar] [CrossRef]
- Tyabi, Z.; Jabado, R.W.; Sutaria, D. New records of sharks (Elasmobranchii) from the Andaman and Nicobar Archipelago in India with notes on current checklists. Biodivers. Data J. 2018, 6, e28593. [Google Scholar] [CrossRef] [PubMed]
- Sen, S.; Dash, G.; Mukherjee, I. An overview of elasmobranch fisheries of West Bengal in 2018. Mar. Fish Infor. Serv. Tech. Ext. Ser. 2018, 238, 18–22. [Google Scholar]
- Barman, R.P.; Mishra, S.S.; Kar, S.; Mukherjee, P.; Saren, S.C. Marine and estuarine fish fauna of Orissa. Rec. Zool. Surv. India Occas. Pap. 2007, 260, 1–186. [Google Scholar]
- Talwar, P.K. A contribution to the taxonomy of Rhizoprionodon oligolinx Springer 1964: An important component of the shark fishery of Orissa, India. Indian J. Fish. 1974, 21, 604–607. [Google Scholar]
- Rao, S.C.V.S. Scientific, common and local names of commercially important edible marine fin and shell fishes of Andhra Pradesh. Mar. Fish. Infor. Serv. Tech. Ext. Ser. 1991, 108, 1–10. [Google Scholar]
- Joshi, K.K.; Sreeram, M.P.; Zacharia, P.U.; Abdussamad, E.M.; Varghese, M.; Habeeb Mohammed, O.M.M.J.; Jayabalan, K.; Kanthan, K.P.; Kannan, K.; Sreekumar, K.M.; et al. Check list of fishes of the Gulf of Mannar ecosystem, Tamil Nadu, India. J. Mar. Biol. Assoc. India 2016, 58, 34–54. [Google Scholar] [CrossRef]
- Bineesh, K.K.; Gopalakrishnan, A.; Akhilesh, K.V.; Sajeela, K.A.; Abdussamad, E.M.; Pillai, N.G.K.; Basheer, V.S.; Jena, J.K.; Ward, R.D. DNA barcoding reveals species composition of sharks and rays in the Indian commercial fishery. Mitochondrial DNA A 2016, 28, 458–472. [Google Scholar] [CrossRef]
- Kulkarni, G.N.; Shanbhogue, S.L.; Udupa, K.S. Length-weight relationship of Scoliodon laticaudus Muller and Henle and Carcharhinus limbatus (Muller and Henle), from Dakshina Kannada coast. Indian J. Fish. 1988, 35, 300–301. [Google Scholar]
- Veena, S.; Thomas, S.; Raje, S.G.; Durgekar, R. Case of leucism in the spadenose shark, Scoliodon laticaudus (Muller & Henle, 1838) from Mangalore, Karnataka. Indian J. Fish. 2011, 58, 109–112. [Google Scholar]
- Pillai, P.P.; Parakkal, B. Pelagic sharks in the Indian seas their exploitation, trade, management and conservation. CMFRI Spec. Publ. 2000, 70, 1–95. [Google Scholar]
- Nair, R.V.; Appukuttan, K.K.; Rajapandian, M.E. On the systematics and identity of four pelagic sharks of the family Carcharhinidae from Indian region. Indian J. Fish. 1974, 21, 220–232. [Google Scholar]
- Mathew, C.J.; Devaraj, M. The biology and population dynamics of the spadenose shark Scoliodon laticaudus in the coastal waters of Maharastra State, India. Indian J. Fish. 1997, 44, 11–27. [Google Scholar]
- Fofandi, M.D.; Zala, M.; Koya, M. Observations on selected biological aspects of the spadenose shark (Scoliodon laticaudus Müller & Henle, 1838), landed along Saurashtra coast. Indian J. Fish. 2013, 60, 51–54. [Google Scholar]
- Misra, K.S. A check list of the fishes of India, Burma & Ceylon. Part I. Elasmobranchii and Holocephali. Rec. Indian Mus. 1947, 45, 1–46. [Google Scholar]
- Mendis, A.S. Fishes of Ceylon: A catalogue, key & bibliography. Fish. Res. Stat. Bull. 1954, 2, 1–222. [Google Scholar]
- De Silva, R.I. Taxonomy and status of the sharks and rays of Sri Lanka. Fauna Sri Lanka 2006, 2006, 294–301. [Google Scholar]
- Fernando, D.; Bown, R.M.K.; Tanna, A.; Gobiraj, R.; Ralicki, H.; Jockusch, E.L.; Ebert, D.A.; Jensen, K.; Caira, J.N. New insights into the identities of the elasmobranch fauna of Sri Lanka. Zootaxa 2019, 4585, 201–238. [Google Scholar] [CrossRef]
- Moron, J.; Bertrand, B.; Last, P. A check-list of sharks and rays of western Sri Lanka. J. Mar. Biol. Assoc. India 1998, 40, 142–157. [Google Scholar]
- Misra, K.S. An aid to the identification of the common commercial fishes of India and Pakistan. Rec. Indian Mus. 1962, 57, 1–320. [Google Scholar]
- Gore, M.; Waqas, U.; Khan, M.M.; Ahmad, E.; Baloch, A.S.; Baloch, A.R. A first account of the elasmobranch fishery of Balochistan, south-west Pakistan. West. Indian Ocean. J. Mar. Sci. 2019, 18, 95–105. [Google Scholar] [CrossRef][Green Version]
- Bishop, J.M. History and current checklist of Kuwait’s ichthyofauna. J. Arid. Environ. 2003, 54, 237–256. [Google Scholar] [CrossRef]
- Sivasubramanian, K.; Ibrahim, M.A. Common Fishes of Qatar. Scientific Atlas of Qatar 1; Doha Modern Printing Press: Doha, Qatar, 1982. [Google Scholar]
- Moore, A.B.M.; McCarthy, I.D.; Carvalho, G.R.; Peirce, R. Species, sex, size and male maturity composition of previously unreported elasmobranch landings in Kuwait, Qatar and Abu Dhabi Emirate. J. Fish Biol. 2012, 80, 1619–1642. [Google Scholar] [CrossRef] [PubMed]
- Moore, A.B.M.; Peirce, R. Composition of elasmobranch landings in Bahrain. Afr. J. Mar. Sci. 2013, 35, 593–596. [Google Scholar] [CrossRef]
- Jabado, R.W.; Al Ghais, S.M.; Hamza, W.; Shivji, M.S.; Henderson, A.C. Shark diversity in the Arabian/Persian Gulf higher than previously thought: Insights based on species composition of shark landings in the United Arab Emirates. Mar. Biodiv. 2015, 45, 719–731. [Google Scholar] [CrossRef]
- Almojil, D.K.; Moore, A.B.M.; White, W.T. Sharks and Rays of the Arabian/Persian Gulf; MBG (INT) Ltd.: London, UK, 2015. [Google Scholar]
- Henderson, A.C.; McIlwain, J.L.; Al-Oufi, H.S.; Al-Sheili, S. The Sultanate of Oman shark fishery: Species composition, seasonality and diversity. Fish. Res. 2007, 86, 159–168. [Google Scholar] [CrossRef]
- Henderson, A.C.; McIlwain, J.L.; Al-Oufi, H.S.; Al-Sheile, S.; Al-Abri, N. Size distributions and sex ratios of sharks caught by Oman’s artisanal fishery. Afr. J. Mar. Sci. 2009, 31, 233–239. [Google Scholar] [CrossRef]
- Al Sakaff, H.; Esseen, M. Occurrence and distribution of fish species off Yemen (Gulf of Aden and Arabian Sea). Naga ICLARM Q. 1999, 22, 43–47. [Google Scholar]
- Zajonz, U.; Lavergne, E.; Bogorodsky, S.V.; Saeed, F.N.; Aideed, M.S.; Krupp, F. Coastal fish diversity of the Socotra Archipelago, Yemen. Zootaxa 2019, 4636, 1–108. [Google Scholar] [CrossRef]
- Sommer, C.; Schneider, W.; Poutiers, J.M. Living Marine Resources of Somalia. FAO Species Identification Field Guide for Fishery Purposes; FAO: Rome, Italy, 1996. [Google Scholar]
- Marshall, N.T. The Somali shark fishery in the Gulf of Aden and the Western Indian Ocean. In Trade in Sharks and Shark Products in the Western Indian and Southeast Atlantic Oceans; Marshall, N.T., Barnes, R., Eds.; TRAFFIC East/Southern Africa: Nairobi, Kenya, 1997; pp. 24–30. [Google Scholar]
- Anam, R.; Mostarda, E. Field Identification Guide to the Living Marine Resources of Kenya. FAO Species Identification Field Guide for Fishery Purposes; FAO: Rome, Italy, 2012. [Google Scholar]
- Kiilu, B.K.; Ndegwa, S. Shark Bycatch—Small Scale Tuna Fishery Interactions along the Kenyan Coast; IOTC-2013-WPEB09-13; Indian Ocean Tuna Commission (IOTC): Victoria, Seychelles, 2013. [Google Scholar]
- Fischer, W.; Sousa, I.; Silva, C.; de Freitas, A.; Poutiers, J.M.; Schneider, W.; Borges, T.C.; Feral, J.P.; Massinga, A. Fichas FAO de Identificaçao de Espécies para Actividades de Pesca. Guia de Campo Das Espécies Comerciais Marinhas e de águas Salobras de Moçambique; FAO: Rome, Italy, 1990. [Google Scholar]
- Rigby, C.L.; Bin Ali, A.; Bineesh, K.K.; Chen, X.; Derrick, D.; Dharmadi Ebert, D.A.; Fahmi Fernando, D.; Gautama, D.A.; Haque, A.B.; Herman, K.; et al. Scoliodon macrorhynchos. The IUCN Red List of Threatened Species. 2020, p. e.T169233669A169233911. Available online: https://doi.org/10.2305/IUCN.UK.2020-3.RLTS.T169233669A169233911.en (accessed on 13 December 2021).




| Marker | Forward Primer (5′–3′) | Reverse Primer (5′–3′) | References |
|---|---|---|---|
| COI | FishF2—TCG ACT AAT CAT AAA GAT ATC GGC AC | FishR2—ACT TCA GGG TGA CCG AAG AAT CAG AA | Ward et al. [29] |
| NADH2 | ILEM—AAG GAG CAG TTT GAT AGA GT | ASNM—AAC GCT TAG CTG TTA ATT AA | Naylor et al. [30] |
| ACT | ACT-F—GCT TTC ATC TCC TTC GGC AGT TTG | ACT-R—CCA CTG GTA ATT GGG ATA CTT GGC | Design based on GN’s sequence of sample GN1680 |
| KBTBD2 | KBT-F—CTC AGT ATC TAT CTT CAG TCC TTG GC | KBT-R—GCT CTT ACA CAG GGA TCA GAG TAG C | Design based on GN’s sequence of sample GN1680 |
| PROX1 | PRO1-F—AAT TCT TCA AGG GAA AGT GCC CAA G | PRO1-R—CAG ACT GCT CCG ACG AGT TTT TG | Design based on GN’s sequence of sample GN1680 |
| RAG1 | RAG1-F—CTT ATT CAA ACC ATC AAC AAC ACA ACA | RAG1-R—CTG CAT GAC TGC TTC CAA CTC ATC | Design based on GN’s sequence of sample GN1680 |
| RAG2 | RAG2-F—TCA GAA TCA AAC AGC CTC ATT TAC C | RAG2-R—TTA ATT TCA TTG GAC CAT TCT GGG G | Design based on GN’s sequence of sample GN1680 |
| SCFD2 | SCFD-F—AGG TGA AAG CGG TAT TTG TGG TG | SCFD-R—TGA GCT GCA GAA CTT CAA ACA TAG | Design based on GN’s sequence of sample GN1680 |
| TOB1 | TOB1-F—ATA TGA AGG TCA CTG GTA TCC AGA C | TOB1-R—GAA AAC AAA CTC CTT GGC ATT GGG A | Design based on GN’s sequence of sample GN1680 |
| S. laticaudus | S. cf. laticaudus | S. macrorhynchos | ||||
|---|---|---|---|---|---|---|
| n = 8 | n = 32 | n = 34 | ||||
| Min. | Max. | Min. | Max. | Min. | Max. | |
| Total length (mm) | 169 | 524 | 239 | 490 | 227 | 562 |
| Precaudal length | 75.6 | 78.0 | 75.3 | 78.0 | 73.6 | 78.0 |
| Pre-second dorsal length | 62.6 | 65.4 | 62.9 | 66.7 | 61.5 | 66.5 |
| Pre-first dorsal length | 35.1 | 38.8 | 33.0 | 37.7 | 33.0 | 38.1 |
| Head length | 21.5 | 29.1 | 21.5 | 26.3 | 21.3 | 25.6 |
| Head length (horiz) | 21.0 | 28.6 | 21.0 | 25.1 | 20.9 | 25.0 |
| Pre-branchial length | 17.1 | 23.5 | 17.1 | 20.4 | 16.5 | 20.7 |
| Pre-branchial length (horiz) | 16.6 | 22.6 | 16.5 | 19.8 | 16.0 | 19.5 |
| Preorbital length | 8.9 | 12.6 | 8.9 | 11.7 | 8.5 | 11.6 |
| Preorbital length (horiz) | 8.1 | 11.3 | 7.9 | 10.8 | 7.0 | 10.7 |
| Preoral length | 7.1 | 11.1 | 7.1 | 10.4 | 7.2 | 10.4 |
| Pre-narial length | 6.6 | 9.1 | 6.6 | 8.7 | 6.2 | 8.4 |
| Pre-narial length (horiz) | 5.9 | 8.2 | 5.6 | 8.1 | 4.8 | 7.8 |
| Pre-pectoral length | 22.1 | 26.4 | 21.5 | 26.6 | 20.1 | 26.2 |
| Pre-pelvic length | 43.9 | 48.4 | 43.9 | 50.2 | 43.8 | 49.2 |
| Snout–vent length | 45.9 | 49.2 | 45.9 | 51.4 | 45.4 | 50.6 |
| Preanal length | 56.7 | 59.9 | 56.7 | 62.0 | 54.8 | 60.4 |
| Interdorsal space | 16.1 | 21.7 | 17.9 | 21.7 | 17.9 | 22.2 |
| Dorsal-caudal space | 7.2 | 9.3 | 7.2 | 9.9 | 7.2 | 9.4 |
| Pectoral–pelvic space | 16.7 | 19.7 | 16.9 | 20.7 | 16.8 | 21.6 |
| Pelvic–anal space | 5.2 | 9.0 | 5.6 | 11.1 | 4.8 | 8.7 |
| Anal–caudal space | 6.4 | 9.1 | 6.4 | 8.8 | 6.4 | 9.1 |
| Eye length | 1.5 | 2.2 | 1.6 | 2.5 | 1.3 | 2.4 |
| Eye height | 1.3 | 2.5 | 1.3 | 2.5 | 1.5 | 2.2 |
| Interorbital space | 7.4 | 11.2 | 7.4 | 9.8 | 7.5 | 10.3 |
| Nostril width | 1.4 | 2.0 | 1.5 | 2.3 | 1.4 | 2.3 |
| Internarial space | 4.9 | 6.9 | 4.9 | 6.7 | 4.9 | 6.5 |
| Anterior nasal flap length | 0.2 | 0.6 | 0.2 | 0.5 | 0.2 | 0.6 |
| Mouth length | 4.5 | 5.6 | 4.1 | 4.9 | 3.5 | 5.2 |
| Mouth width | 6.0 | 7.6 | 5.3 | 7.6 | 5.7 | 7.6 |
| Upper labial furrow length | 0.2 | 0.6 | 0.1 | 0.5 | 0.1 | 0.5 |
| Lower labial furrow length | 0.8 | 1.2 | 0.2 | 1.5 | 0.3 | 1.4 |
| First gill slit height | 2.3 | 3.1 | 2.3 | 4.1 | 2.2 | 4.0 |
| Second gill slit height | 2.3 | 3.6 | 2.1 | 2.6 | 2.2 | 3.2 |
| Third gill slit height | 2.4 | 3.8 | 2.2 | 4.7 | 2.3 | 4.4 |
| Fourth gill slit height | 2.4 | 3.7 | 2.0 | 2.8 | 2.4 | 3.3 |
| Fifth gill slit height | 2.2 | 3.2 | 2.1 | 3.3 | 2.3 | 3.1 |
| Intergill length | 4.6 | 5.9 | 4.6 | 5.4 | 4.5 | 6.4 |
| Head height | 6.1 | 10.2 | 7.7 | 9.9 | 7.0 | 10.6 |
| Trunk height | 7.9 | 10.8 | 8.3 | 10.8 | 7.8 | 13.1 |
| Abdomen height | 7.5 | 11.2 | 10.0 | 11.4 | 9.4 | 13.9 |
| Tail height | 6.3 | 10.2 | 7.0 | 9.4 | 7.5 | 11.3 |
| Caudal peduncle height | 3.9 | 4.5 | 3.8 | 4.5 | 4.0 | 5.0 |
| Head width | 7.3 | 9.4 | 6.9 | 9.9 | 7.9 | 10.8 |
| Trunk width | 6.4 | 8.5 | 6.5 | 8.8 | 6.2 | 11.8 |
| Abdomen width | 5.2 | 7.1 | 4.9 | 6.9 | 5.2 | 8.9 |
| Tail width | 4.1 | 5.6 | 4.2 | 5.6 | 4.6 | 6.5 |
| Caudal peduncle width | 1.9 | 2.7 | 2.3 | 3.5 | 2.2 | 3.7 |
| Pectoral length | 10.2 | 12.1 | 9.8 | 11.6 | 9.9 | 11.7 |
| Pectoral anterior margin | 9.5 | 12.1 | 9.4 | 11.5 | 9.2 | 11.9 |
| Pectoral base | 4.5 | 6.6 | 5.2 | 6.4 | 4.8 | 6.6 |
| Pectoral height | 7.8 | 10.3 | 7.4 | 10.3 | 7.5 | 10.1 |
| Pectoral inner margin | 5.2 | 6.2 | 4.6 | 6.4 | 4.3 | 6.2 |
| Pectoral posterior margin | 6.3 | 10.6 | 6.8 | 12.5 | 6.8 | 9.8 |
| Pelvic length | 7.3 | 8.7 | 7.1 | 8.9 | 6.9 | 8.3 |
| Pelvic anterior margin | 4.7 | 5.4 | 4.3 | 6.0 | 4.3 | 5.6 |
| Pelvic base | 4.7 | 5.6 | 4.3 | 6.3 | 4.3 | 6.1 |
| Pelvic height | 3.2 | 4.3 | 2.3 | 4.4 | 2.7 | 4.2 |
| Pelvic inner margin length | 2.2 | 3.7 | 2.1 | 3.9 | 2.2 | 3.5 |
| Pelvic posterior margin length | 3.4 | 5.3 | 3.4 | 5.3 | 3.8 | 5.1 |
| Clasper outer length | 6.0 | 9.0 | 4.5 | 10.2 | 4.0 | 10.0 |
| Clasper inner length | 8.4 | 11.8 | 6.4 | 12.4 | 6.5 | 12.1 |
| Clasper base width | 1.0 | 1.4 | 0.6 | 1.7 | 0.6 | 1.4 |
| First dorsal length | 13.3 | 15.6 | 13.3 | 15.7 | 12.9 | 15.5 |
| First dorsal anterior margin | 11.1 | 13.5 | 11.8 | 14.3 | 11.2 | 14.6 |
| First dorsal base | 8.9 | 10.9 | 8.9 | 11.4 | 8.8 | 11.0 |
| First dorsal height | 6.6 | 8.6 | 5.8 | 8.8 | 6.5 | 9.0 |
| First dorsal inner margin | 3.8 | 5.1 | 3.9 | 5.3 | 3.5 | 4.9 |
| First dorsal posterior margin | 6.7 | 9.2 | 5.7 | 9.0 | 6.2 | 8.9 |
| Second dorsal length | 7.5 | 9.3 | 7.3 | 9.1 | 6.9 | 8.6 |
| Second dorsal Anterior margin | 4.1 | 5.5 | 3.4 | 5.5 | 3.4 | 5.0 |
| Second dorsal base | 4.0 | 4.8 | 3.2 | 4.9 | 3.2 | 4.8 |
| Second dorsal height | 1.7 | 2.2 | 1.2 | 2.4 | 1.3 | 2.0 |
| Second dorsal inner margin | 3.2 | 4.7 | 3.8 | 5.1 | 3.3 | 4.8 |
| Second dorsal posterior margin | 3.8 | 5.3 | 3.9 | 4.9 | 3.6 | 4.7 |
| Anal length | 11.4 | 13.5 | 9.6 | 13.7 | 10.8 | 14.1 |
| Anal anterior margin | 5.1 | 6.7 | 4.1 | 7.0 | 4.9 | 7.8 |
| Anal base | 8.0 | 10.3 | 6.1 | 10.3 | 7.2 | 11.2 |
| Anal height | 2.8 | 3.7 | 2.2 | 3.6 | 2.6 | 3.8 |
| Anal Inner margin | 3.0 | 3.9 | 3.0 | 3.9 | 2.8 | 4.1 |
| Anal posterior margin | 6.6 | 8.4 | 5.8 | 8.8 | 6.5 | 8.9 |
| Dorsal caudal margin | 22.0 | 24.9 | 21.6 | 24.6 | 21.9 | 25.6 |
| Pre-ventral caudal margin | 8.5 | 10.2 | 7.8 | 10.7 | 8.0 | 10.5 |
| Lower post-ventral caudal margin | 3.4 | 4.7 | 2.9 | 5.0 | 2.9 | 4.8 |
| Upper post-ventral caudal margin | 9.5 | 11.5 | 8.9 | 11.0 | 9.1 | 12.3 |
| Caudal fork width | 5.4 | 7.5 | 5.4 | 6.8 | 5.9 | 7.1 |
| Caudal fork length | 7.8 | 9.7 | 8.0 | 9.8 | 7.8 | 9.8 |
| Subterminal caudal margin | 3.9 | 5.6 | 3.1 | 4.7 | 3.1 | 5.3 |
| Subterminal caudal width | 2.6 | 3.4 | 2.6 | 3.5 | 2.7 | 3.4 |
| Terminal caudal margin | 4.5 | 7.4 | 4.8 | 6.8 | 4.9 | 7.3 |
| Terminal caudal lobe | 7.6 | 8.9 | 6.8 | 8.6 | 7.2 | 9.3 |
| Second dorsal origin | 4.6 | 6.9 | 3.0 | 6.9 | 5.2 | 9.1 |
| Second dorsal insertion | 0.5 | 2.2 | 0.6 | 2.0 | 0.6 | 2.7 |
| Mid-base first dorsal fin to pectoral insertion | 10.9 | 12.7 | 10.5 | 13.4 | 11.0 | 14.6 |
| Mid-base first dorsal fin to pelvic origin | 4.4 | 6.2 | 4.6 | 7.9 | 4.4 | 7.6 |
| First dorsal insertion to pelvic mid-base | 2.8 | 3.9 | 2.8 | 5.4 | 1.9 | 5.1 |
| Pelvic mid-base to second dorsal origin | 12.9 | 18.1 | 13.6 | 18.1 | 13.5 | 19.0 |
| Slat-Scflat | Slat-Smac | Scflat-Smac | |
|---|---|---|---|
| COI | 0.82 (0.61–1.53) | 2.35 (1.99–2.75) | 2.29 (2.14–3.06) |
| NADH2 | 3.05 (2.98–3.27) | 3.06 (2.98–3.26) | 3.64 (3.46–4.23) |
| Mitochondrial | 2.16 (2.12–2.18) | 2.82 (2.71–2.89) | 3.05 (2.95–3.18) |
| ACT | 0.10 (0.00–0.25) | 0.50 (0.50–0.50) | 0.50 (0.25–0.74) |
| KBTBD2 | 0.00 (0.00–0.00) | 0.22 (0.22–0.22) | 0.22 (0.22–0.22) |
| PROX1 | 0.00 (0.00–0.00) | 0.02 (0.00–0.11) | 0.02 (0.00–0.11) |
| RAG1 | 0.12 (0.12–0.12) | 0.12 (0.12–0.12) | 0.02 (0.00–0.12) |
| RAG2 | 0.54 (0.45–0.61) | 0.91 (0.91–0.91) | 0.58 (0.45–0.61) |
| SCFD2 | 0.13 (0.00–0.21) | 0.21 (0.21–0.21) | 0.17 (0.00–0.42) |
| TOB1 | 0.00 (0.00–0.00) | 0.00 (0.00–0.00) | 0.00 (0.00–0.00) |
| Nuclear | 0.12 (0.10–0.14) | 0.25 (0.25–0.25) | 0.19 (0.16–0.21) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lim, K.C.; White, W.T.; Then, A.Y.H.; Naylor, G.J.P.; Arunrugstichai, S.; Loh, K.-H. Integrated Taxonomy Revealed Genetic Differences in Morphologically Similar and Non-Sympatric Scoliodon macrorhynchos and S. laticaudus. Animals 2022, 12, 681. https://doi.org/10.3390/ani12060681
Lim KC, White WT, Then AYH, Naylor GJP, Arunrugstichai S, Loh K-H. Integrated Taxonomy Revealed Genetic Differences in Morphologically Similar and Non-Sympatric Scoliodon macrorhynchos and S. laticaudus. Animals. 2022; 12(6):681. https://doi.org/10.3390/ani12060681
Chicago/Turabian StyleLim, Kean Chong, William T. White, Amy Y. H. Then, Gavin J. P. Naylor, Sirachai Arunrugstichai, and Kar-Hoe Loh. 2022. "Integrated Taxonomy Revealed Genetic Differences in Morphologically Similar and Non-Sympatric Scoliodon macrorhynchos and S. laticaudus" Animals 12, no. 6: 681. https://doi.org/10.3390/ani12060681
APA StyleLim, K. C., White, W. T., Then, A. Y. H., Naylor, G. J. P., Arunrugstichai, S., & Loh, K.-H. (2022). Integrated Taxonomy Revealed Genetic Differences in Morphologically Similar and Non-Sympatric Scoliodon macrorhynchos and S. laticaudus. Animals, 12(6), 681. https://doi.org/10.3390/ani12060681






