Annual Censuses and Citizen Science Data Show Rapid Population Increases and Range Expansion of Invasive Rose-Ringed and Monk Parakeets in Seville, Spain
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area and Species
2.2. Field Surveys and Population Censuses
2.3. Processing and Validation of Data from Citizen Science
2.4. Statistical Analyses
3. Results
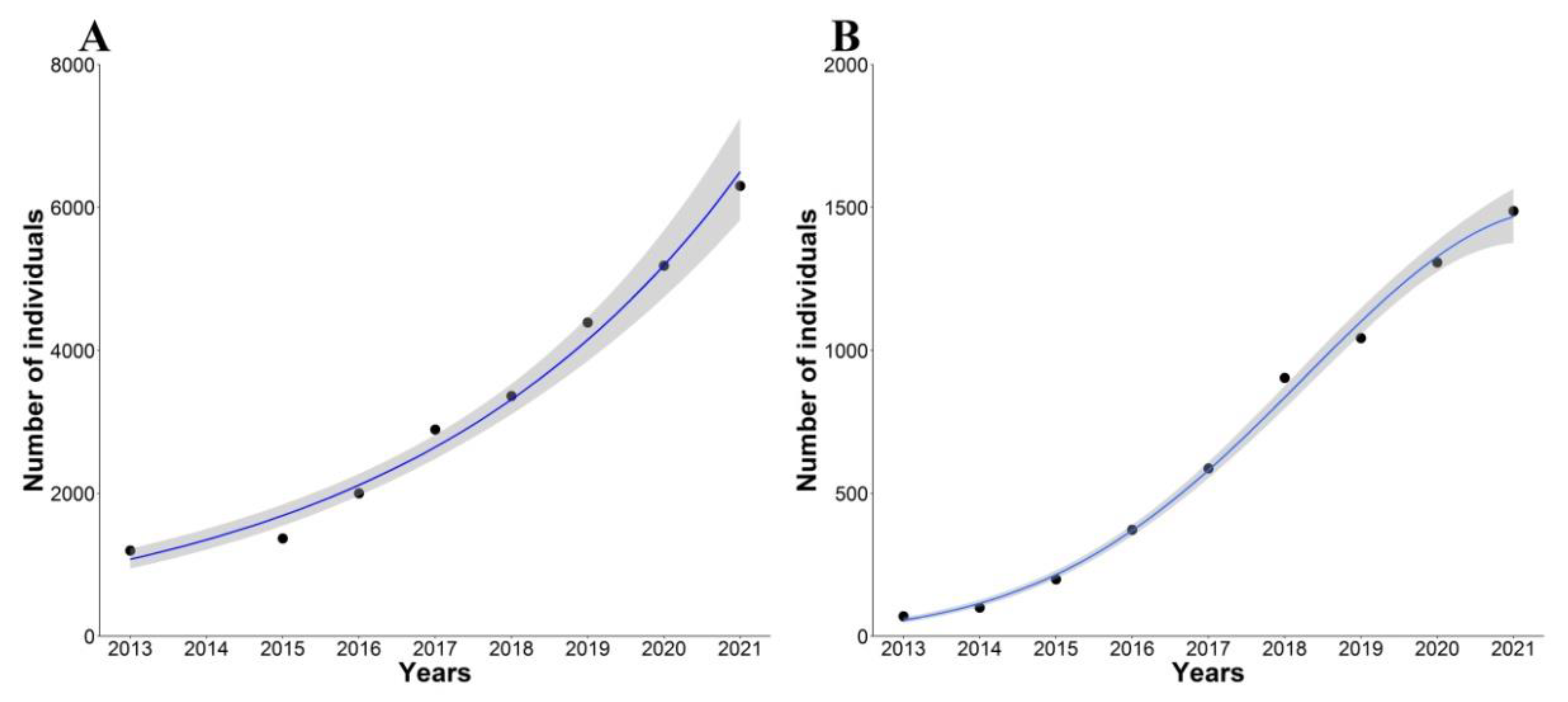
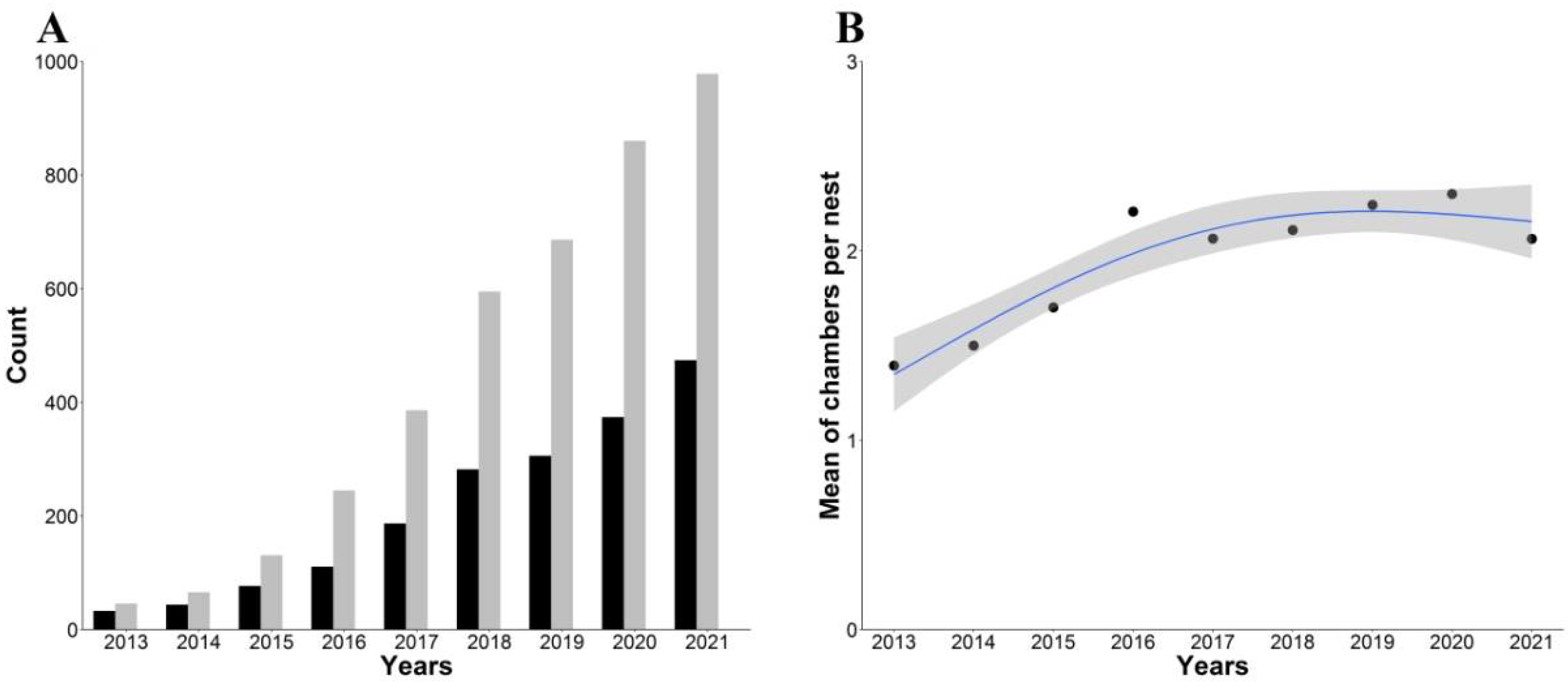
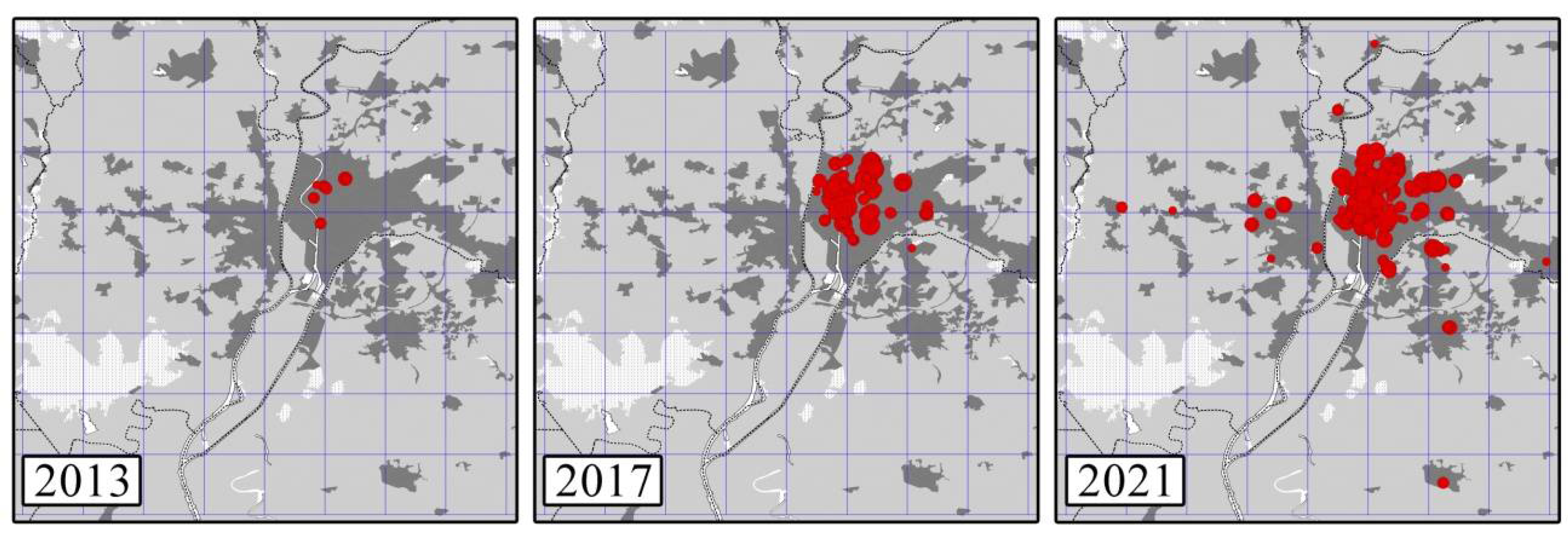
3.1. Censuses and Population Growth Rates
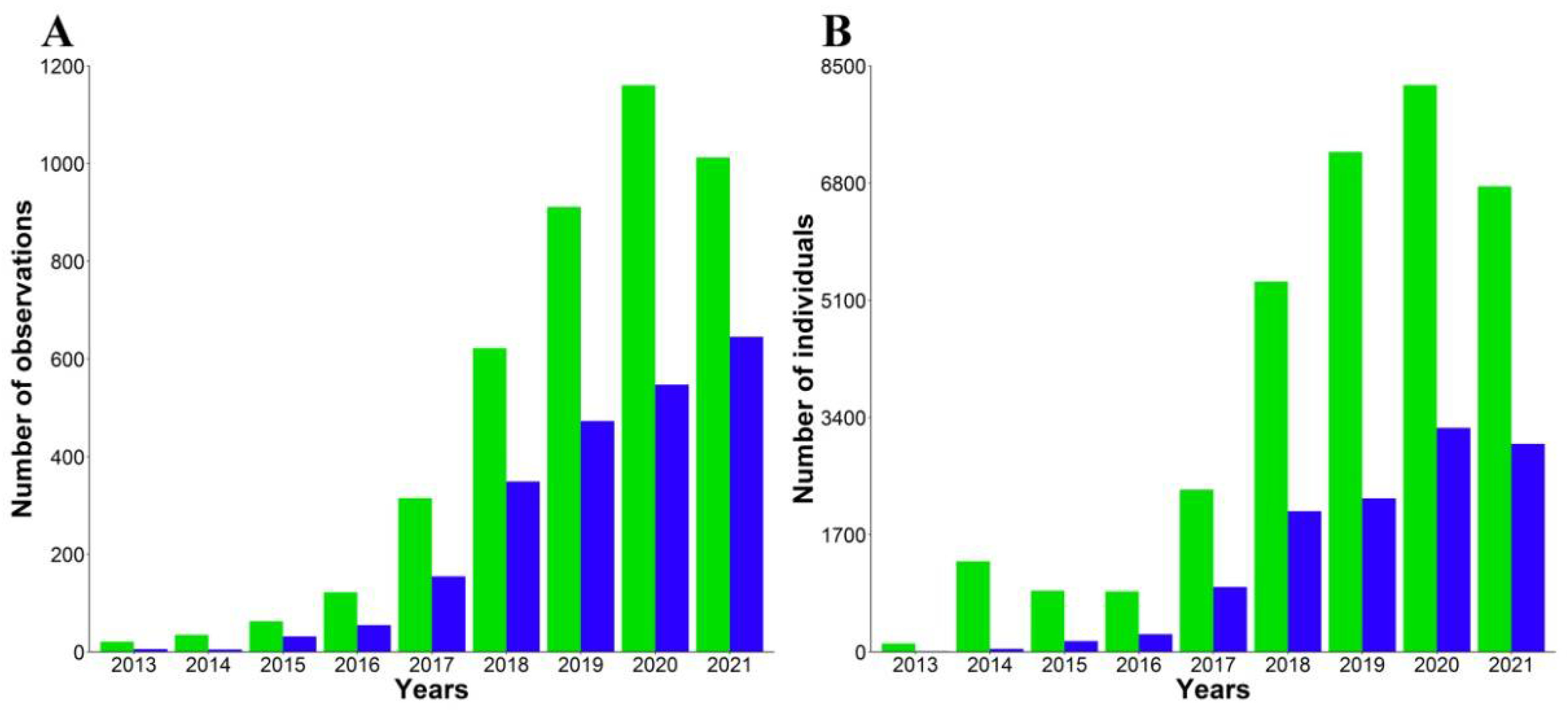
3.2. Citizen Science Observations
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Simberloff, D.; Martin, J.L.; Genovesi, P.; Maris, V.; Wardle, D.A.; Aronson, J.; Courchamp, F.; Galil, B.; García-Berthou, E.; Pascal, M.; et al. Impacts of biological invasions: What’s what and the way forward. Trends Ecol. Evol. 2013, 28, 58–66. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bacher, S.; Blackburn, T.M.; Essl, F.; Genovesi, P.; Heikkilä, J.; Jeschke, J.M.; Jones, J.; Keller, R.; Kenis, M.; Kueffer, C.; et al. Socio-economic impact classification of alien taxa (SEICAT). Methods Ecol. Evol. 2018, 9, 159–168. [Google Scholar] [CrossRef]
- Clavero, M.; García-Berthou, E. Invasive species are a leading cause of animal extinctions. Trends Ecol. Evol. 2005, 20, 110. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bellard, C.; Cassey, P.; Blackburn, T.M. Alien species as a driver of recent extinctions. Biol. Lett. 2016, 12, 20150623. [Google Scholar] [CrossRef]
- Ricciardi, A.; Hoopes, M.F.; Marchetti, M.P.; Lockwood, J.L. Progress toward understanding the ecological impacts of nonnative species. Ecol. Monogr. 2013, 83, 263–282. [Google Scholar] [CrossRef] [Green Version]
- Simberloff, D. How much information on population biology is needed to manage introduced species? Biol. Conserv. 2003, 17, 83–92. [Google Scholar] [CrossRef] [Green Version]
- Hauser, C.E.; Pople, A.R.; Possingham, H.P. Should managed populations be monitored every year? Ecol. Appl. 2006, 16, 807–819. [Google Scholar] [CrossRef] [Green Version]
- Reaser, J.K.; Burgiel, S.W.; Kirkey, J.; Brantley, K.A.; Veatch, S.D.; Burgos-Rodríguez, J. The early detection of and rapid response (EDRR) to invasive species: A conceptual framework and federal capacities assessment. Biol. Invasions 2020, 22, 1–19. [Google Scholar] [CrossRef] [Green Version]
- Gallo, T.; Waitt, D. Creating a successful citizen science model to detect and report invasive species. BioScience 2011, 61, 459–465. [Google Scholar] [CrossRef] [Green Version]
- Tulloch, A.I.; Possingham, H.P.; Joseph, L.N.; Szabo, J.; Martin, T.G. Realising the full potential of citizen science monitoring programs. Biol. Conserv. 2013, 165, 128–138. [Google Scholar] [CrossRef] [Green Version]
- Johnson, B.A.; Mader, A.D.; Dasgupta, R.; Kumar, P. Citizen science and invasive alien species: An analysis of citizen science initiatives using information and communications technology (ICT) to collect invasive alien species observations. Glob. Ecol. Conserv. 2020, 21, e00812. [Google Scholar] [CrossRef]
- Crooks, J.A. Lag times and exotic species: The ecology and management of biological invasions in slow-motion1. Ecoscience 2005, 12, 316–329. [Google Scholar] [CrossRef]
- Jarić, J.; Heger, T.; Castro Monzon, F.; Jeschke, J.M.; Kowarik, I.; McConkey, K.R.; Pyšek, P.; Sagouis, A.; Essl, F. Crypticity in biological invasions. Trends Ecol. Evol. 2019, 34, 291–302. [Google Scholar] [CrossRef] [PubMed]
- Shea, K.; Chesson, P. Community ecology theory as a framework for biological invasions. Trends Ecol. Evol. 2002, 17, 170–176. [Google Scholar] [CrossRef]
- Catford, J.A.; Jansson, R.; Nilsson, C. Reducing redundancy in invasion ecology by integrating hypotheses into a single theoretical framework. Divers. Distrib. 2009, 15, 22–40. [Google Scholar] [CrossRef] [Green Version]
- Hulme, P.E. Trade, transport and trouble: Managing invasive species pathways in an era of globalization. J. Appl. Ecol. 2009, 46, 10–18. [Google Scholar] [CrossRef]
- Tella, J.L.; Hiraldo, F. Illegal and legal parrot trade shows a long-term, cross-cultural preference for the most attractive species increasing their risk of extinction. PLoS ONE 2014, 9, e107546. [Google Scholar] [CrossRef] [Green Version]
- Calzada Preston, C.E.; Pruett-Jones, S. The Number and Distribution of Introduced and Naturalized Parrots. Diversity 2021, 13, 412. [Google Scholar] [CrossRef]
- Cardador, L.; Abellán, P.; Anadón, J.D.; Carrete, M.; Tella, J.L. The world parrot trade. In Naturalized Parrots of the World: Distribution, Ecology, and Impacts of the World’s Most Colorful Colonizers; Pruett-Jones, S., Ed.; Princeton University Press: Princeton, NJ, USA, 2021; pp. 13–21. [Google Scholar]
- Abellán, P.; Tella, J.L.; Carrete, M.; Cardador, L.; Anadón, J.D. Climate matching drives spread rate but not establishment success in recent unintentional bird introductions. Proc. Natl. Acad. Sci. USA 2017, 114, 9385–9390. [Google Scholar] [CrossRef] [Green Version]
- Mori, E.; Grandi, G.; Menchetti, M.; Tella, J.L.; Jackson, H.A.; Reino, L.; van Kleunen, A.; Figueira, R.; Ancillotto, L. Worldwide distribution of non–native Amazon parrots and temporal trends of their global trade. Anim. Biodivers. Conserv. 2017, 40, 49–62. [Google Scholar] [CrossRef] [Green Version]
- Royle, K.; Donner, W.B. The distribution of naturalized parrot populations. In Naturalized Parrots of the World: Distribution, Ecology, and Impacts of the World’s Most Colorful Colonizers; Pruett-Jones, S., Ed.; Princeton University Press: Princeton, NJ, USA, 2021; pp. 22–40. [Google Scholar]
- Butler, C.J. Feral parrots in the continental United States and United Kingdom: Past, present, and future. J. Avian Med. Surg. 2005, 19, 142–149. [Google Scholar] [CrossRef]
- Strubbe, D.; Matthysen, E. Establishment success of invasive ring-necked and monk parakeets in Europe. J. Biogeogr. 2009, 36, 2264–2278. [Google Scholar] [CrossRef]
- Pruett-Jones, S.; Newman, J.R.; Newman, C.M.; Lindsay, J.R. Population growth of monk parakeets in Florida. Fla. Field Nat. 2005, 33, 1–14. [Google Scholar]
- Pruett-Jones, S.; Appelt, C.W.; Sarfaty, A.; Van Vossen, B.; Leibold, M.A.; Minor, E.S. Urban parakeets in Northern Illinois: A 40-year perspective. Urban Ecosyst. 2012, 15, 709–719. [Google Scholar] [CrossRef]
- Aagaard, K.; Lockwood, J.L. Severe and rapid population declines in exotic birds. Biol. Invasions 2016, 18, 1667–1678. [Google Scholar] [CrossRef]
- Uehling, J.J.; Tallant, J.; Pruett-Jones, S. Status of naturalized parrots in the United States. J. Ornithol. 2019, 160, 907–921. [Google Scholar] [CrossRef]
- Hernández-Brito, D.; Tella, J.L.; Carrete, M.; Blanco, G. Successful hybridization between non-congeneric parrots in a small introduced population. IBIS 2021, 163, 1093–1098. [Google Scholar] [CrossRef]
- Hernández-Brito, D.; Tella, J.L.; Blanco, G.; Carrete, M. Nesting innovations allow population growth in an invasive population of rose-ringed parakeets. Curr. Zool. 2021, in press. [Google Scholar] [CrossRef]
- Forshaw, J.M. Parrots of the World; Princeton University Press: Princeton, NJ, USA, 2010. [Google Scholar]
- Edelaar, P.; Roques, S.; Hobson, E.A.; Gonçalves da Silva, A.; Avery, M.L.; Russello, M.A.; Senar, J.C.; Wright, T.F.; Carrete, M.; Tella, J.L. Shared genetic diversity across the global invasive range of the monk parakeet suggests a common restricted geographic origin and the possibility of convergent selection. Mol. Ecol. 2015, 24, 2164–2176. [Google Scholar] [CrossRef] [Green Version]
- Jackson, H.; Strubbe, D.; Tollington, S.; Prys-Jones, R.; Matthysen, E.; Groombridge, J.J. Ancestral origins and invasion pathways in a globally invasive bird correlate with climate and influences from bird trade. Mol. Ecol. 2015, 24, 4269–4285. [Google Scholar] [CrossRef] [Green Version]
- Pârâu, L.G.; Strubbe, D.; Mori, E.; Menchetti, M.; Ancillotto, L.; Kleunen, A.V.; White, R.L.; Luna, Á.; Hernández-Brito, D.; Louarn, M.L.; et al. Rose-ringed parakeet Psittacula krameri populations and numbers in Europe: A complete overview. Open Ornithol. J. 2016, 9, 1–13. [Google Scholar] [CrossRef] [Green Version]
- Postigo, J.L.; Strubbe, D.; Mori, E.; Ancillotto, L.; Carneiro, I.; Latsoudis, P.; Menchetti, M.; Pârâu, L.G.; Parrott, D.; Reino, L.; et al. Mediterranean versus Atlantic monk parakeets Myiopsitta monachus: Towards differentiated management at the European scale. Pest Manag. Sci. 2019, 75, 915–922. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Brito, D.; Carrete, M.; Popa-Lisseanu, A.G.; Ibáñez, C.; Tella, J.L. Crowding in the city: Losing and winning competitors of an invasive bird. PLoS ONE 2014, 9, e100593. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Brito, D.; Carrete, M.; Ibáñez, C.; Juste, J.; Tella, J.L. Nest-site competition and killing by invasive parakeets cause the decline of a threatened bat population. R. Soc. Open Sci. 2018, 5, 172477. [Google Scholar] [CrossRef] [Green Version]
- Hernández-Brito, D.; Carrete, M.; Blanco, G.; Romero-Vidal, P.; Senar, J.C.; Mori, E.; Luna, Á.; Tella, J.L. The Role of Monk Parakeets as Nest-Site Facilitators in Their Native and Invaded Areas. Biology 2021, 10, 683. [Google Scholar] [CrossRef]
- Morinha, F.; Carrete, M.; Tella, J.L.; Blanco, G. High prevalence of novel beak and feather disease virus in sympatric invasive parakeets introduced to Spain from Asia and South America. Diversity 2020, 12, 192. [Google Scholar] [CrossRef]
- Tella, J.L.; Baños-Villalba, A.; Hernández-Brito, D.; Rojas, A.; Pacífico, E.; Díaz-Luque, J.A.; Carrete, M.; Blanco, G.; Hiraldo, F. Parrots as overlooked seed dispersers. Front. Ecol. Environ. 2015, 13, 338–339. [Google Scholar] [CrossRef] [Green Version]
- Hernández-Brito, D.; Romero-Vidal, P.; Hiraldo, F.; Blanco, G.; Díaz-Luque, J.A.; Barbosa, J.M.; Symes, C.T.; White, T.H.; Pacífico, E.C.; Sebastián-González, E.; et al. Epizoochory in parrots as an overlooked yet widespread plant–animal mutualism. Plants 2021, 10, 760. [Google Scholar] [CrossRef]
- Molina, B.; Postigo, J.L.; Muñoz, A.-R.; del Moral, J.C. La Cotorra Argentina en España: Población Reproductora en 2015 y Método de Censo; SEO/BirdLife: Madrid, Spain, 2016. [Google Scholar]
- del Moral, J.C.; Somoza, A.; Muñoz, A.-R.; Molina, B. La Cotorra de Kramer en España, Población Reproductora en 2015 y Método de Censo; SEO/BirdLife: Madrid, Spain, 2017. [Google Scholar]
- Prados, S. Seguimiento de Aves Exóticas en la Ciudad de Sevilla; Plegadis, SEO/BirdLife: Seville, Spain, 2000. [Google Scholar]
- Muñoz, A.R. Cotorra de Kramer Psittacula krameri. In Atlas de las Aves Reproductoras de España; Martí, R., del Moral, J.C., Eds.; Dirección General de Conservación de la Naturaleza-Sociedad Española de Ornitología: Madrid, Spain, 2003; pp. 76–77. [Google Scholar]
- Dénes, F.V.; Tella, J.L.; Beissinger, S.R. Revisiting methods for estimating parrot abundance and population size. Emu-Austral Ornithol. 2018, 118, 67–79. [Google Scholar] [CrossRef] [Green Version]
- Luna, A.; Franz, D.; Strubbe, D.; Shwartz, A.; Braun, M.P.; Hernández-Brito, D.; Malihi, Y.; Kaplan, A.; Mori, E.; Menchetti, M.; et al. Reproductive timing as a constraint on invasion success in the Ringnecked parakeet (Psittacula krameri). Biol. Invasions 2017, 19, 2247–2259. [Google Scholar] [CrossRef]
- Bucher, E.; Martin, L.; Martella, M.; Navarro, J. Social behaviour and population dynamics of the Monk Parakeet. Proc. Int. Ornithol. Congr. 1991, 20, 681–689. [Google Scholar]
- Domènech, J.; Carrillo, J.; Senar, J.C. Population size of the Monk Parakeet (Myiopsitta monachus) in Catalonia. Rev. Catalana Ornitol. 2003, 20, 1–9. [Google Scholar]
- Postigo, J.L.; Shwartz, A.; Strubbe, D.; Muñoz, A.R. Unrelenting spread of the alien monk parakeet Myiopsitta monachus in Israel. Is it time to sound the alarm? Pest Manag. Sci. 2017, 73, 349–353. [Google Scholar] [CrossRef] [PubMed]
- eBird. eBird: An online Database of Bird Distribution and Abundance [Web Application]. eBird, Cornell Lab of Ornithology, Ithaca, New York. 2021. Available online: https://ebird.org (accessed on 18 January 2022).
- Observation. Observation.org, Stichting Observation International and Local Partners. 2021. Available online: https://observation.org (accessed on 18 January 2022).
- Hartig, F. DHARMa: Residual Diagnostics for Hierarchical (Multi-Level/Mixed) Regression Models. R Package Version 0.2.0. 2018. Available online: https://CRAN.R-project.org/package=DHARMa (accessed on 10 February 2022).
- Wood, S. Mixed GAM Computation Vehicle with Automatic Smoothness Estimation. R Package Version 1.8-38. 2021. Available online: https://cran.r-project.org/package=mgcv (accessed on 10 February 2022).
- Brooks, M.E.; Kristensen, K.; van Benthem, K.J.; Magnusson, A.; Berg, C.W.; Nielsen, A.; Skaug, H.J.; Maechler, M.; Bolker, B.M. glmmTMB Balances Speed and Flexibility among Packages for Zero-Inflated Generalized Linear Mixed Modeling. R J. 2017, 9, 378–400. [Google Scholar] [CrossRef] [Green Version]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2020. [Google Scholar]
- Colléony, A.; Shwartz, A. When the winners are the losers: Invasive alien bird species outcompete the native winners in the biotic homogenization process. Biol. Conserv. 2020, 241, 108314. [Google Scholar] [CrossRef]
- Norbury, G.L.; Pech, R.P.; Byrom, A.E.; Innes, J. Density-impact functions for terrestrial vertebrate pests and indigenous biota: Guidelines for conservation managers. Biol. Conserv. 2015, 191, 409–420. [Google Scholar] [CrossRef]
- Cassey, P.; Blackburn, T.M.; Russell, G.J.; Jones, K.E.; Lockwood, J.L. Influences on the transport and establishment of exotic bird species: An analysis of the parrots (Psittaciformes) of the world. Glob. Chang. Biol. 2004, 10, 417–426. [Google Scholar] [CrossRef]
- Aagaard, K.; Lockwood, J. Exotic birds show lags in population growth. Divers. Distrib. 2014, 20, 547–554. [Google Scholar] [CrossRef]
- Carrete, M.; Abellán, P.; Cardador, L.; Anadón, J.D.; Tella, J.L. The fate of multistage parrot invasions in Spain and Portugal. In Naturalized Parrots of the World: Distribution, Ecology, and Impacts of the World’s Most Colorful Colonizers; Pruett-Jones, S., Ed.; Princeton University Press: Princeton, NJ, USA, 2021; pp. 240–248. [Google Scholar]
- Latombe, G.; Pysek, P.; Jeschke, J.M.; Blackburn, T.M.; Bacher, S.; Capinha, C.; Costello, M.J.; Fernandez, M.; Gregory, R.D.; Hobern, D.; et al. A vision for global monitoring of biological invasions. Biol. Conserv. 2017, 213, 295–308. [Google Scholar] [CrossRef]
- Butler, C.J.; Cresswell, W.; Gosler, A.; Perrins, C. The breeding biology of Rose-ringed Parakeets Psittacula krameri in England during a period of rapid population expansion. Bird Study 2013, 60, 527–532. [Google Scholar] [CrossRef]
- Senar, J.C.; Carrillo-Ortiz, J.G.; Ortega-Segalerva, A.; Dawson Pell, F.S.E.; Pascual, J.; Arroyo, L.; Mazzoni, D.; Montalvo, T.; Hatchwell, B.J. The reproductive capacity of Monk Parakeets Myiopsitta monachus is higher in their invasive range. Bird Study 2019, 66, 136–140. [Google Scholar] [CrossRef]
- Casagrande, D.G.; Beissinger, S.R. Evaluation of four methods for estimating parrot population size. Condor 1997, 99, 445–457. [Google Scholar] [CrossRef]
- Dupin, M.K.; Dahlin, C.R.; Wright, T.F. Range-Wide Population Assessment of the Endangered Yellow-Naped Amazon (Amazona auropalliata). Diversity 2020, 12, 377. [Google Scholar] [CrossRef]
- Zulian, V.; Müller, E.S.; Cockle, K.L.; Lesterhuis, A.; Tomasi Júnior, R.; Prestes, N.P.; Martinez, J.; Kéry, M.; Ferraz, G. AddressingMultiple Sources of Uncertainty in the Estimation of Global Parrot Abundance from Roost Counts: A Case Study with theVinaceous-Breasted Parrot (Amazona vinacea). Biol. Conserv. 2020, 248, 108672. [Google Scholar] [CrossRef]
- Zulian, V.; Miller, D.A.W.; Ferraz, G. Endemic and Threatened Amazona Parrots of the Atlantic Forest: An Overview of Their Geographic Range and Population Size. Diversity 2021, 13, 416. [Google Scholar] [CrossRef]
- Blackburn, T.M.; Lockwood, J.L.; Cassey, P. The influence of numbers on invasion success. Mol. Ecol. 2015, 24, 1942–1953. [Google Scholar] [CrossRef]
- Carrete, M.; Tella, J. Wild-bird trade and exotic invasions: A new link of conservation concern? Front. Ecol. Environ. 2008, 6, 207–211. [Google Scholar] [CrossRef] [Green Version]
- CITES Trade Database. UNEP World Conservation Monitoring Centre: Cambridge, UK. Available online: http://www.unep-wcmc.org/citestrade/trade.cfm (accessed on 1 December 2021).
- Senar, J.C. Cotorreta de pit gris. In Atles Dels Ocells Nidificants a Barcelona; Ajuntament de Barcelona, Institut Català d’Ornitologia i Universitat de Barcelona: Barcelona, Spain, 2017; pp. 136–137. [Google Scholar]
- Postigo, J.L.; Senar, J.C. 2017 Informe Diagnóstico Sobre Las Cotorras Invasoras en el Municipio de Málaga. Ayuntamiento de Málaga. Available online: https://www.researchgate.net/publication/325284551_Informe_diagnostico_sobre_las_cotorras_invasoras_en_el_municipio_de_Malaga (accessed on 31 January 2022).
- Senar, J.C. Cotorra de Kramer. In Atles Dels Ocells Nidificants a Barcelona; Ajuntament de Barcelona, Institut català d’Ornitologia i Universitat de Barcelona: Barcelona, Spain, 2017; pp. 128–129. [Google Scholar]
- Shwartz, A.; Strubbe, D.; Butler, C.J.; Matthysen, E.; Kark, S. The effect of enemy-release and climate conditions on invasive birds: A regional test using the rose-ringed parakeet (Psittacula krameri) as a case study. Divers. Distrib. 2009, 15, 310–318. [Google Scholar] [CrossRef]
- Hernández-Brito, D.; Blanco, G.; Tella, J.L.; Carrete, M. A protective nesting association with native species counteracts biotic resistance for the spread of an invasive parakeet from urban into rural habitats. Front. Zool. 2020, 17, 13. [Google Scholar] [CrossRef]
- Mori, E.; Malfatti, L.; Le Louarn, M.; Brito, D.H.; Ten Cate, B.; Ricci, M.; Menchetti, M. ‘Some like it alien’: Predation on invasive ring–necked parakeets by the long–eared owl in an urban area. Anim. Biodiv. Conserv. 2020, 43, 151–158. [Google Scholar]
- Pruett-Jones, S.; Newman, J.R.; Newman, C.M.; Avery, M.L.; Lindsay, J.R. Population viability analysis of monk parakeets in the United States and examination of alternative management strategies. Hum. Wildl. Confl. 2007, 1, 35–44. [Google Scholar]
- Barlow, K.E.; Briggs, P.A.; Haysom, K.A.; Hutson, A.M.; Lechiara, N.L.; Racey, P.A.; Walsh, A.L.; Langton, S.D. Citizen science reveals trends in bat populations: The National Bat Monitoring Programme in Great Britain. Biol. Conserv. 2015, 182, 14–26. [Google Scholar] [CrossRef]
- Walker, J.; Taylor, P.D. Using eBird data to model population change of migratory bird species. Avian Conserv. Ecol. 2017, 12, 4. [Google Scholar] [CrossRef] [Green Version]
- Clark, C.J. eBird records show substantial growth of the Allen’s Hummingbird (Selasphorus sasin sedentarius) population in urban Southern California. Condor Ornithol. Appl. 2017, 119, 122–130. [Google Scholar] [CrossRef]
- Horns, J.J.; Adler, F.R.; Sekercioglu, C.H. Using opportunistic citizen science data to estimate avian population trends. Biol. Conserv. 2018, 221, 151–159. [Google Scholar] [CrossRef]
- Isaac, N.J.B.; van Strien, A.J.; August, T.A.; de Zeeuw, M.P.; Roy, D.B. Statistics for citizen science: Extracting signals of change from noisy ecological data. Methods Ecol. Evol. 2014, 5, 1052–1060. [Google Scholar] [CrossRef] [Green Version]
- Johnston, A.; Moran, N.; Musgrove, A.; Fink, D.; Baillie, S.R. Estimating species distributions from spatially biased citizen science data. Ecol. Model. 2020, 422, 108927. [Google Scholar] [CrossRef]
- Butler, C.J.; King, C.; Reinking, D.L. Do Citizen Science Methods Identify Regions of High Avian Biodiversity? Diversity 2021, 13, 656. [Google Scholar] [CrossRef]
- Tella, J.L.; Romero-Vidal, P.; Dénes, F.V.; Hiraldo, F.; Toledo, B.; Rossetto, F.; Blanco, G.; Hernández-Brito, D.; Pacífico, E.; Díaz-Luque, J.A.; et al. Roadside Car Surveys: Methodological Constraints and Solutions for Estimating Parrot Abundances across the World. Diversity 2021, 13, 300. [Google Scholar] [CrossRef]
- Dickinson, J.L.; Zuckerberg, B.; Bonter, D. Citizen science as an ecological research tool: Challenges and benefits. Annu. Rev. Ecol. Syst. 2010, 41, 149–172. [Google Scholar] [CrossRef] [Green Version]
- Kamp, J.; Oppel, S.; Heldbjerg, H.; Nyegaard, T.; Donald, P.F. Unstructured citizen science data fail to detect long-term population declines of common birds in Denmark. Divers. Distrib. 2016, 22, 1024–1035. [Google Scholar] [CrossRef]
- Burgess, H.; DeBey, L.; Froehlich, H.; Schmidt, N.; Theobald, E.; Ettinger, A.; HilleRisLambers, J.; Tewksbury, J.; Parrish, J. The science of citizen science: Exploring barriers to use as a primary research tool. Biol. Conserv. 2017, 208, 113–120. [Google Scholar] [CrossRef] [Green Version]
- Delaney, D.G.; Sperling, C.D.; Adams, C.S.; Leung, B. Marine invasive species: Validation of citizen science and implications for national monitoring networks. Biol. Invasions 2007, 10, 117–128. [Google Scholar] [CrossRef]
- Thiele, J.; Kollmann, J.; Markussen, B.; Otte, A. Impact assessment revisited: Improving the theoretical basis for management of invasive alien species. Biol. Invasions 2010, 12, 2025–2035. [Google Scholar] [CrossRef]
- Jackson, M.C.; Ruiz-Navarro, A.; Britton, J.R. Population density modifies the ecological impacts of invasive species. Oikos 2015, 124, 880–887. [Google Scholar] [CrossRef] [Green Version]
- Mori, E.; Ancillotto, L.; Groombridge, J.; Howard, T.; Smith, V.S.; Menchetti, M. Macroparasites of introduced parakeets in Italy: A possible role for parasite-mediated competition. Parasitol. Res. 2015, 114, 3277–3281. [Google Scholar] [CrossRef]
- Ancillotto, L.; Studer, V.; Howard, T.; Smith, V.S.; McAlister, E.; Beccaloni, J.; Manzia, F.; Renzopaoli, F.; Bosso, L.; Russo, D.; et al. Environmental drivers of parasite load and species richness in introduced parakeets in an urban landscape. Parasitol. Res. 2018, 117, 3591–3599. [Google Scholar] [CrossRef]
- Senar, J.C.; Domènech, J.; Arroyo, L.; Torre, I.; Gordo, O. An evaluation of monk parakeet damage to crops in the metropolitan area of Barcelona. Anim. Biodivers. Conserv. 2016, 39, 141–145. [Google Scholar] [CrossRef]
- Castro, J.; Sáez, C.; Molina-Morales, M. The monk parakeet (Myiopsitta monachus) as a potential pest for agriculture in the Mediterranean basin. Biol. Invasions 2021, 1–9. [Google Scholar] [CrossRef]
- Conroy and Senar 2009 Conroy, M.J.; Senar, J.C. Integration of demographic analyses and decision modeling in support of management of invasive monk parakeets, an urban and agricultural pest. Environ. Ecol. Stat. 2009, 3, 491–510. [Google Scholar]
- Senar, J.C.; Conroy, M.J.; Montalvo, T. Decision-making Models and Management of the Monk Parakeet. In Naturalized Parrots of the World; Pruett-Jones, S., Ed.; Princeton University Press: Princeton, NJ, USA, 2021; pp. 102–122. [Google Scholar]
- Hernández-Brito, D.; Tella, J.L.; Carrete, M. Evaluation of the Effectiveness of Control and Eradication Actions for Invasive Rose-Ringed Parakeet Populations. In Preparation.
- Esteban, A. Control de la Especie Cotorra Argentina Myiopsitta Monachus en Zaragoza; Ayuntamiento de Zaragoza: Zaragoza, Spain, 2016; Available online: https://www.zaragoza.es/contenidos/medioambiente/InformeCotorraArgentina.pdf (accessed on 1 September 2021).
- Bunbury, N.; Haverson, P.; Page, N.; Agricole, J.; Angell, G.; Banville, P.; Constance, A.; Friedlander, J.; Leite, L.; Mahoune, T.; et al. Five eradications, three species, three islands: Overview, insights and recommendations from invasive bird eradications in the Seychelles. Island invasives: Scaling up to meet the challenge. Occas. Pap. Ser. 2019, 62, 282–288. [Google Scholar]
- Saavedra, S.; Medina, F.M. Control of invasive ring-necked parakeet (Psittacula krameri) in an island Biosphere Reserve (La Palma, Canary Islands): Combining methods and social engagement. Biol. Invasions 2020, 22, 3653–3667. [Google Scholar] [CrossRef]

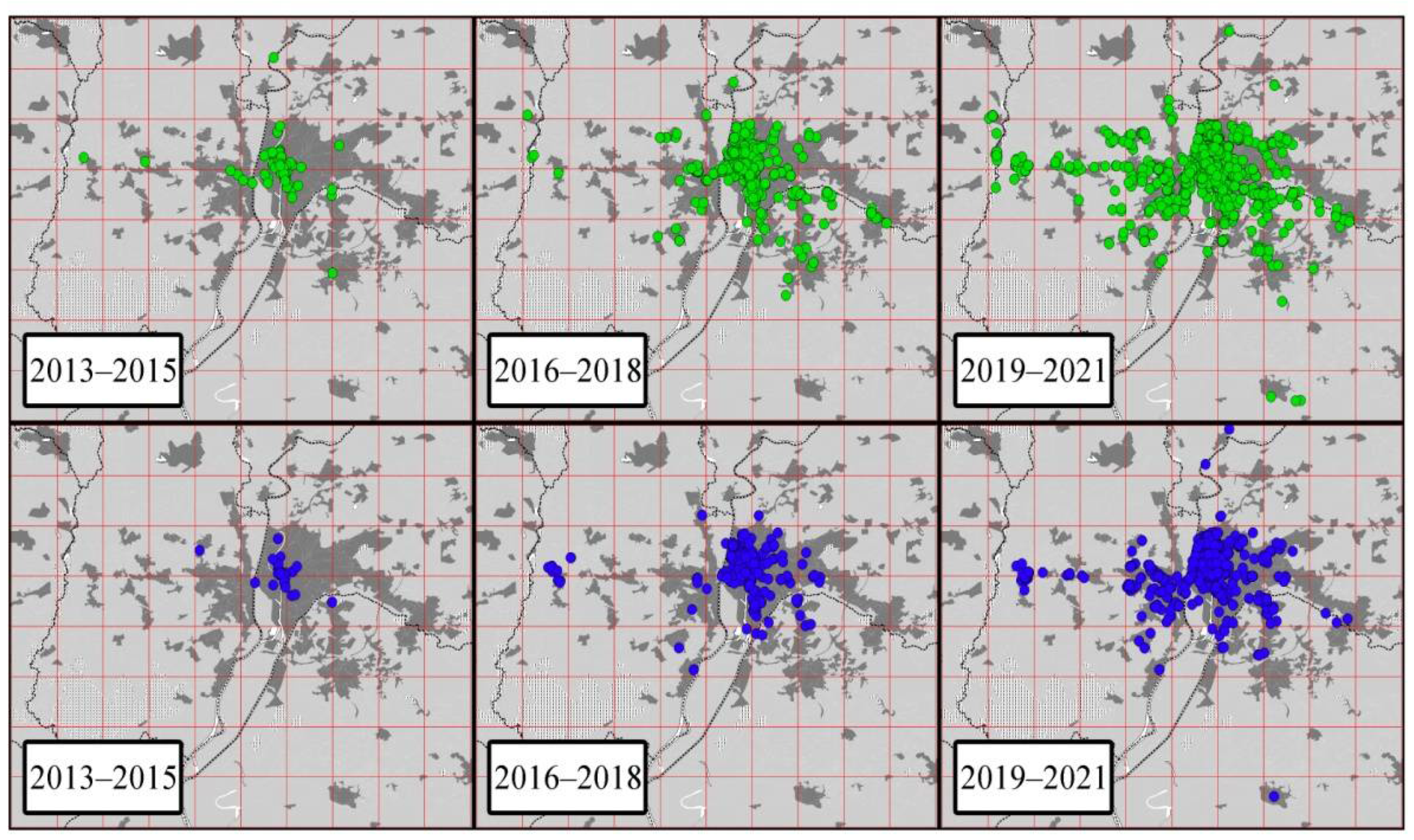
| Rose-Ringed Parakeets | ||||||
| Dependent Variables | Independent Variable | Estimate | SE | z | p | Dev. Expl. |
| Number of observations | Intercept | −3.43 | 0.04 | −82.4 | <0.0001 | 46.33% |
| Survey | 0.16 | 0.12 | 1.29 | 0.19 | ||
| Survey2 | −0.3 | 0.11 | −2.8 | 0.005 | ||
| Number of individuals | Intercept | −1.19 | 2.8 | −4.23 | <0.0001 | 8.51% |
| Survey | <−0.0001 | <0.0001 | −0.62 | 0.54 | ||
| Number of cells | Intercept | −1 | 0.06 | −16.97 | <0.0001 | 62.26% |
| Survey | 0.98 | 0.17 | 5.28 | <0.0001 | ||
| Survey2 | −0.41 | 0.15 | −2.69 | 0.008 | ||
| Monk Parakeets | ||||||
| Dependent Variables | Independent Variable | Estimate | SE | z | p | Dev. Expl. |
| Number of observations | Intercept | −4.2 | 0.06 | −6.6 | <0.0001 | 52.32% |
| Survey | 0.63 | 0.2 | 3.2 | <0.002 | ||
| Survey2 | −0.35 | 0.15 | −2.41 | 0.02 | ||
| Number of individuals | Intercept | −2.85 | 0.26 | −18.98 | <0.0001 | 16.22% |
| Survey | 0.0005 | 0.0003 | 1.48 | 0.137 | ||
| Number of cells | Intercept | −2.12 | 0.14 | −14.54 | <0.0001 | 58.12% |
| Survey | 0.0009 | 0.0001 | 6.63 | <0.0001 | ||
| Number of cells | Intercept | −2.24 | 0.23 | −9.78 | <0.0001 | 68.1% |
| Nests | 0.09 | 0.02 | 4.57 | <0.0001 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hernández-Brito, D.; Carrete, M.; Tella, J.L. Annual Censuses and Citizen Science Data Show Rapid Population Increases and Range Expansion of Invasive Rose-Ringed and Monk Parakeets in Seville, Spain. Animals 2022, 12, 677. https://doi.org/10.3390/ani12060677
Hernández-Brito D, Carrete M, Tella JL. Annual Censuses and Citizen Science Data Show Rapid Population Increases and Range Expansion of Invasive Rose-Ringed and Monk Parakeets in Seville, Spain. Animals. 2022; 12(6):677. https://doi.org/10.3390/ani12060677
Chicago/Turabian StyleHernández-Brito, Dailos, Martina Carrete, and José L. Tella. 2022. "Annual Censuses and Citizen Science Data Show Rapid Population Increases and Range Expansion of Invasive Rose-Ringed and Monk Parakeets in Seville, Spain" Animals 12, no. 6: 677. https://doi.org/10.3390/ani12060677
APA StyleHernández-Brito, D., Carrete, M., & Tella, J. L. (2022). Annual Censuses and Citizen Science Data Show Rapid Population Increases and Range Expansion of Invasive Rose-Ringed and Monk Parakeets in Seville, Spain. Animals, 12(6), 677. https://doi.org/10.3390/ani12060677









