Controlled Intestinal Microbiota Colonisation in Broilers under the Industrial Production System
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animal Trial
2.2. Sample Collection
2.3. DNA Extraction, Sequencing and Data Analysis
2.4. Histology
2.5. Statistical Methods
3. Results
3.1. Bird Performance
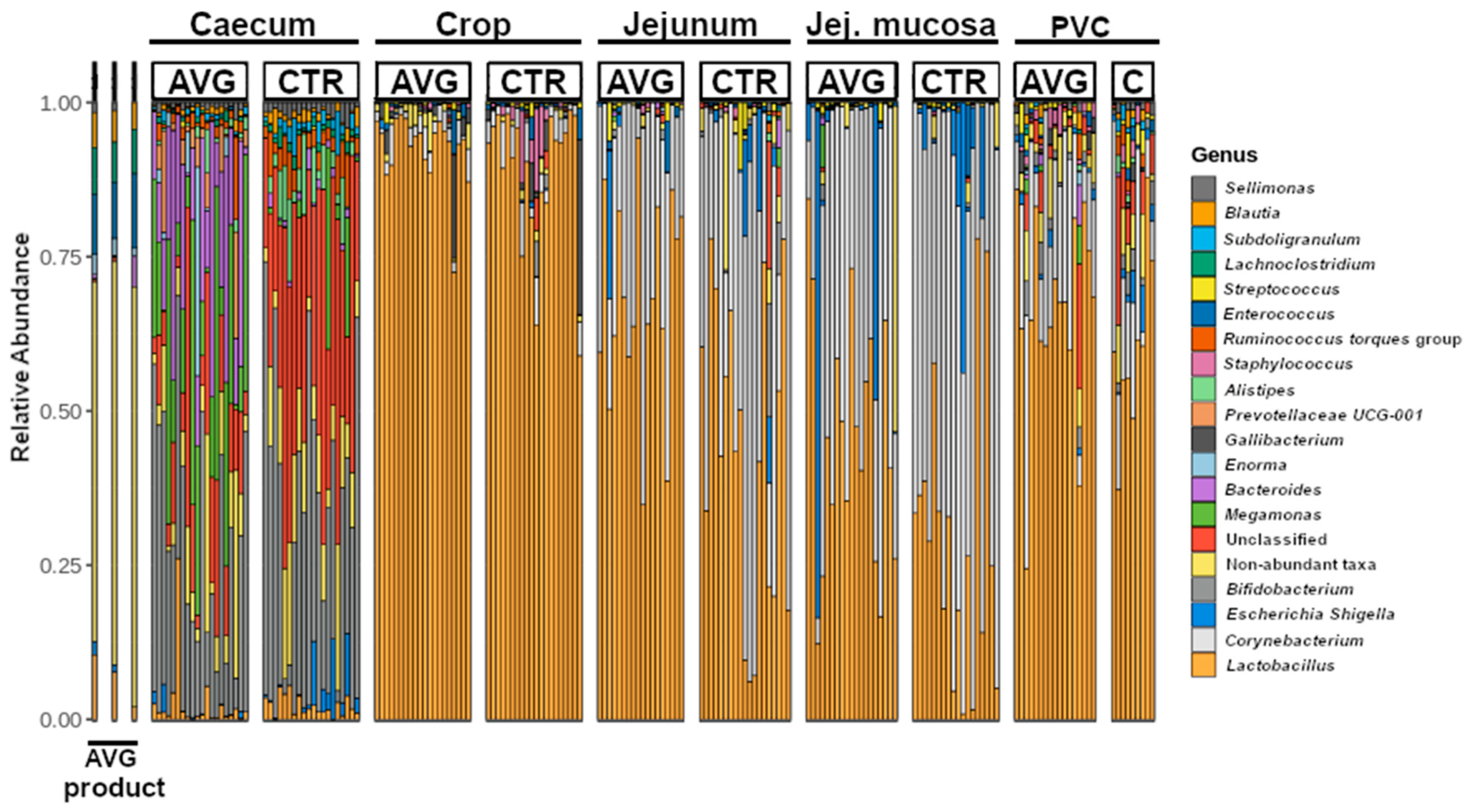
3.2. Community Structure
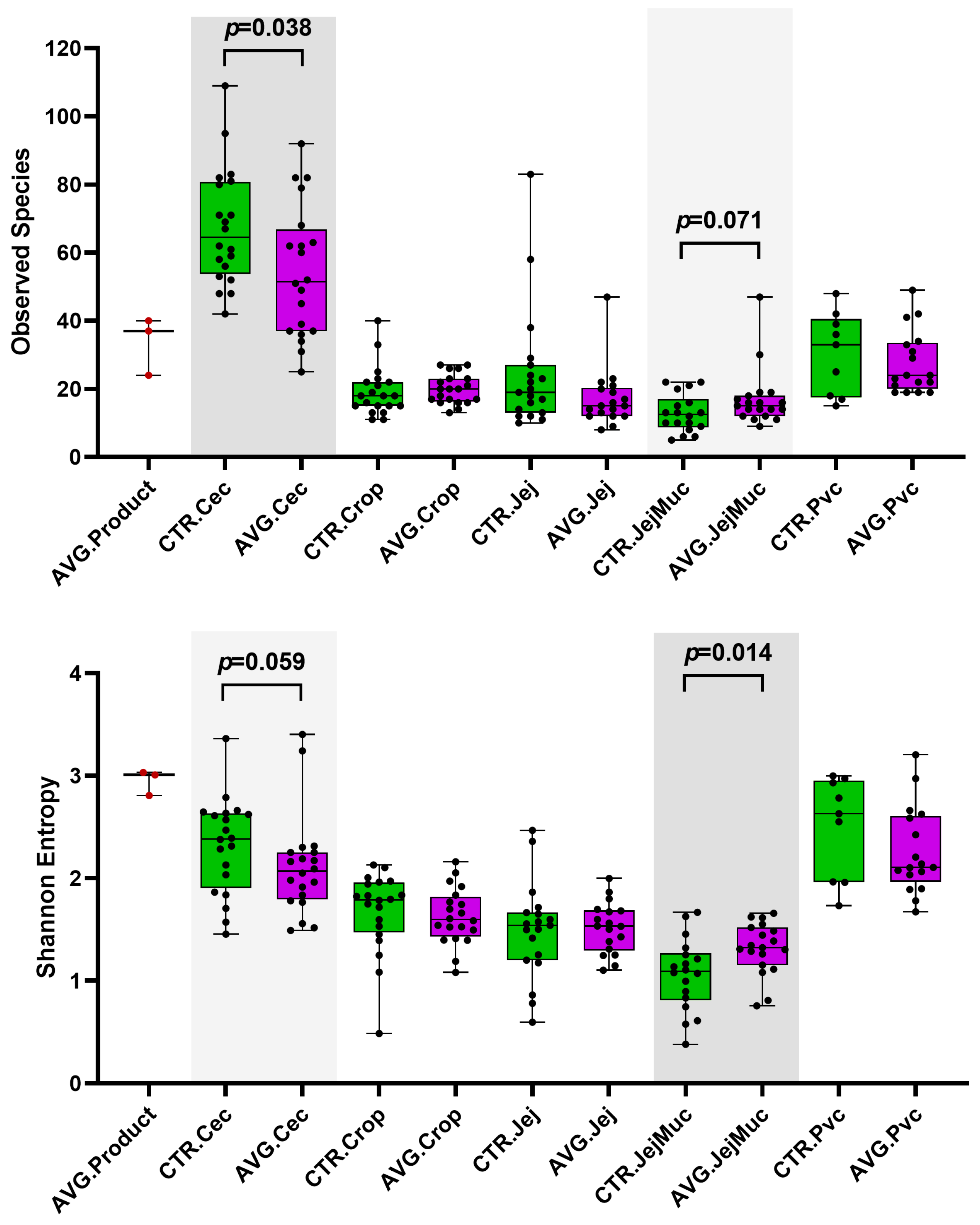
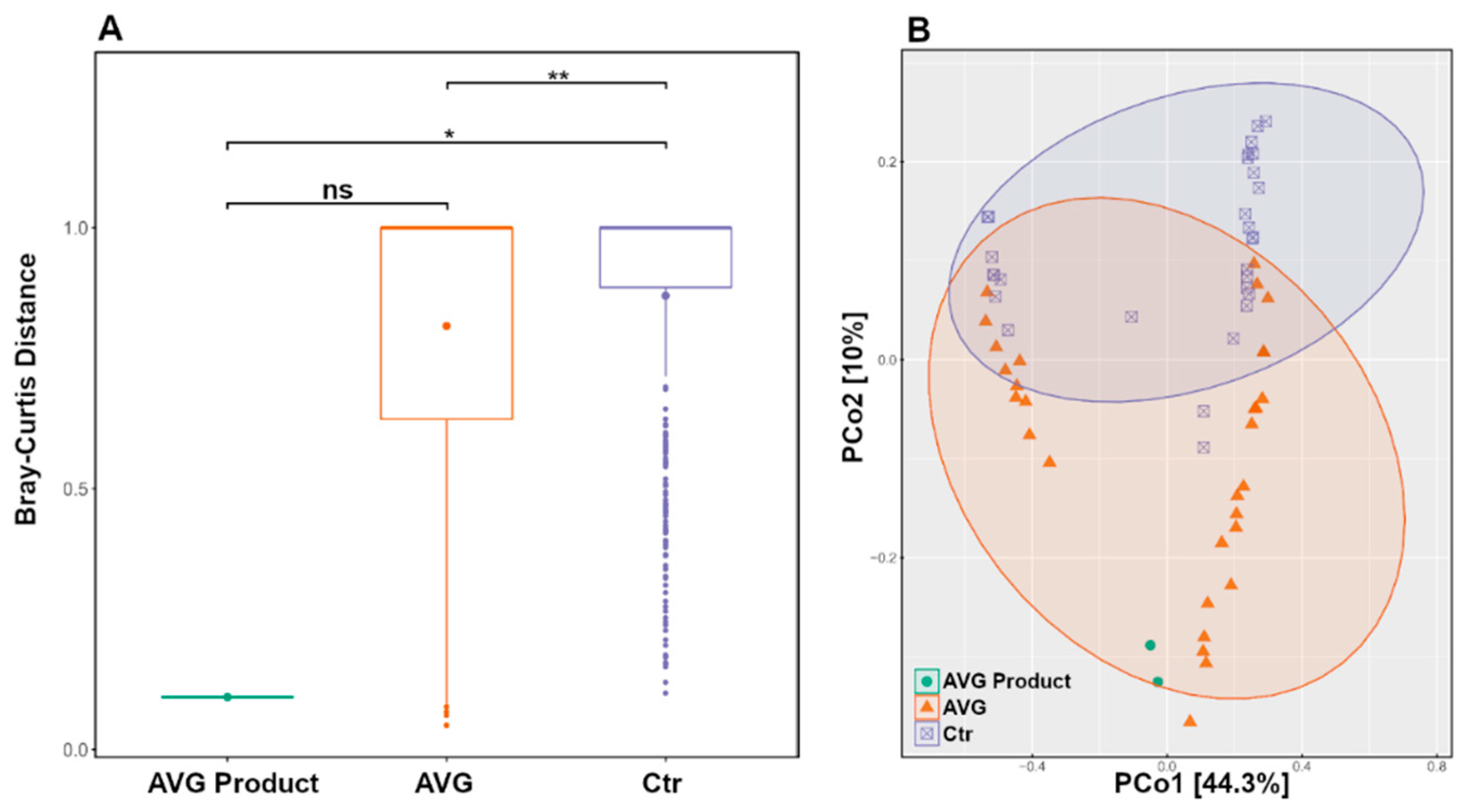
3.3. Alpha and Beta Diversity
3.4. Long Term Colonisation
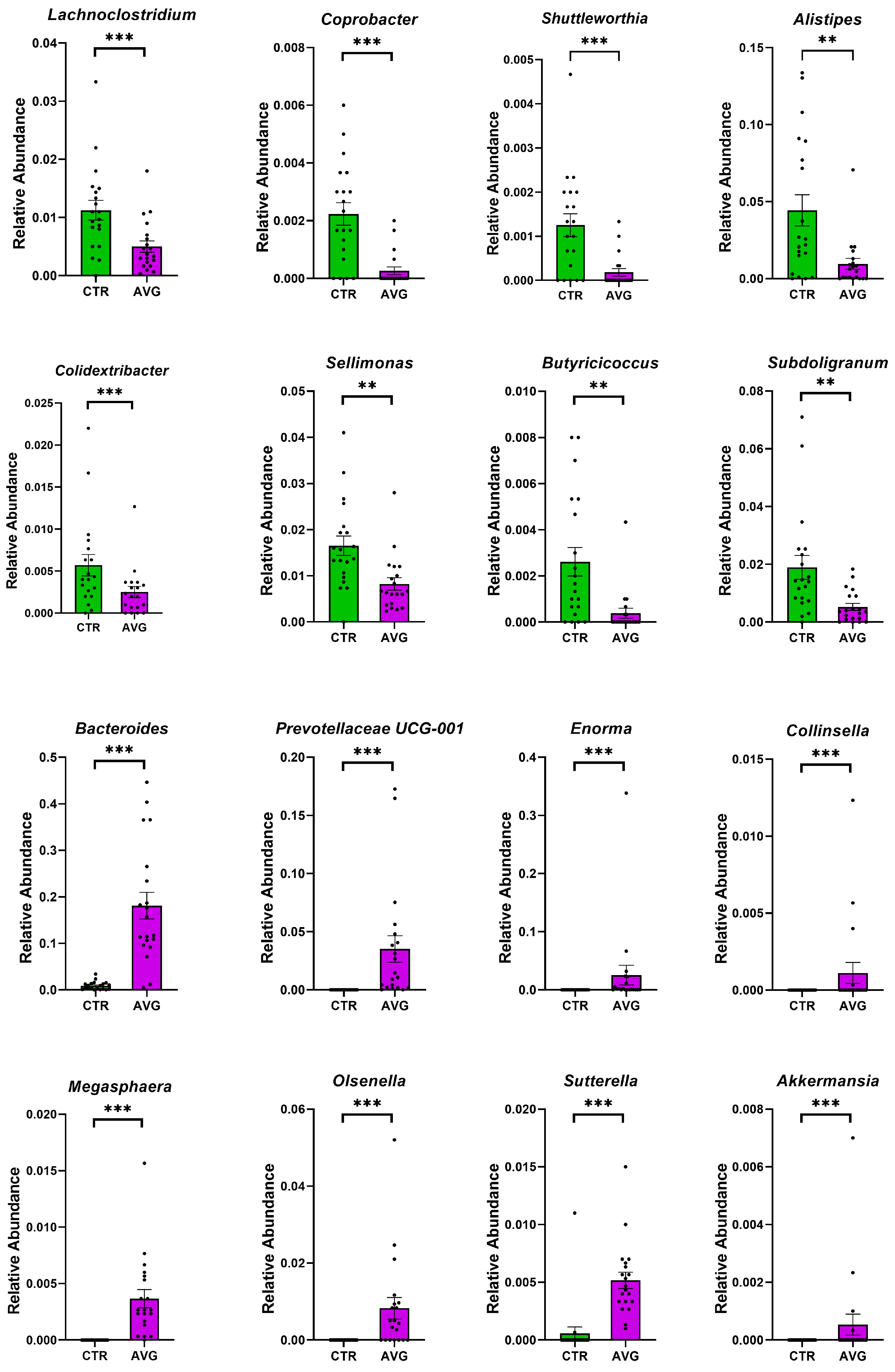
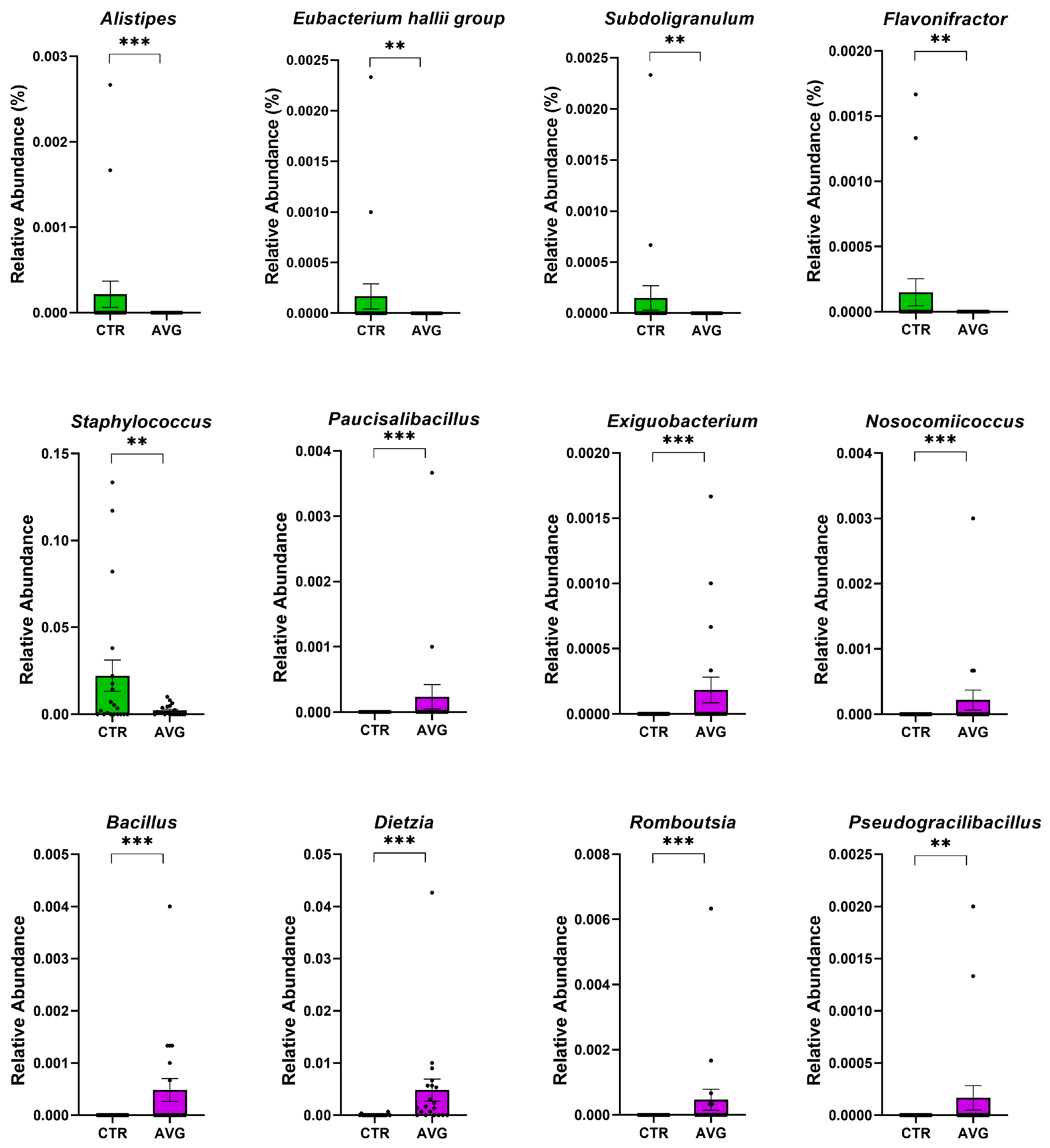
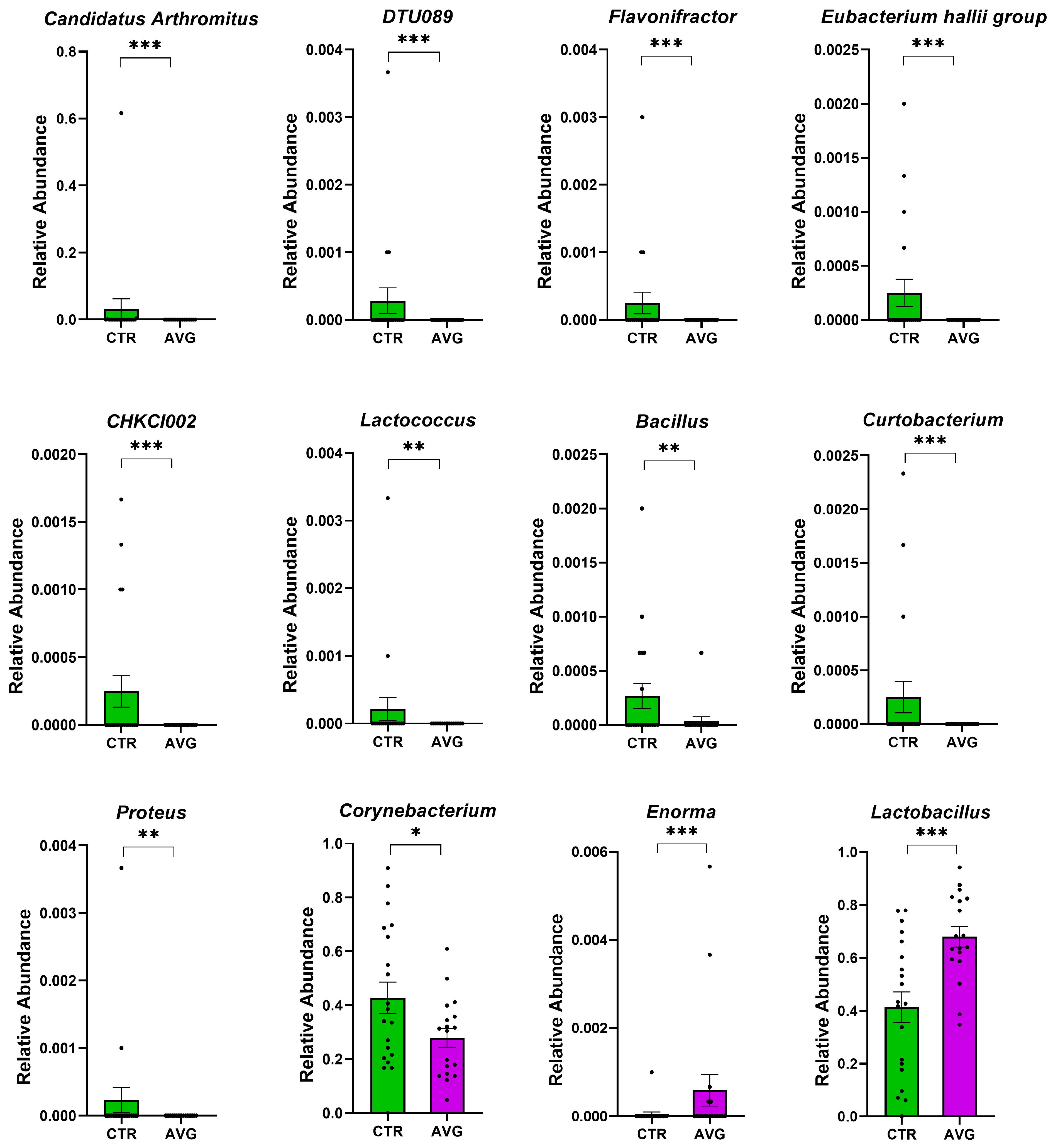
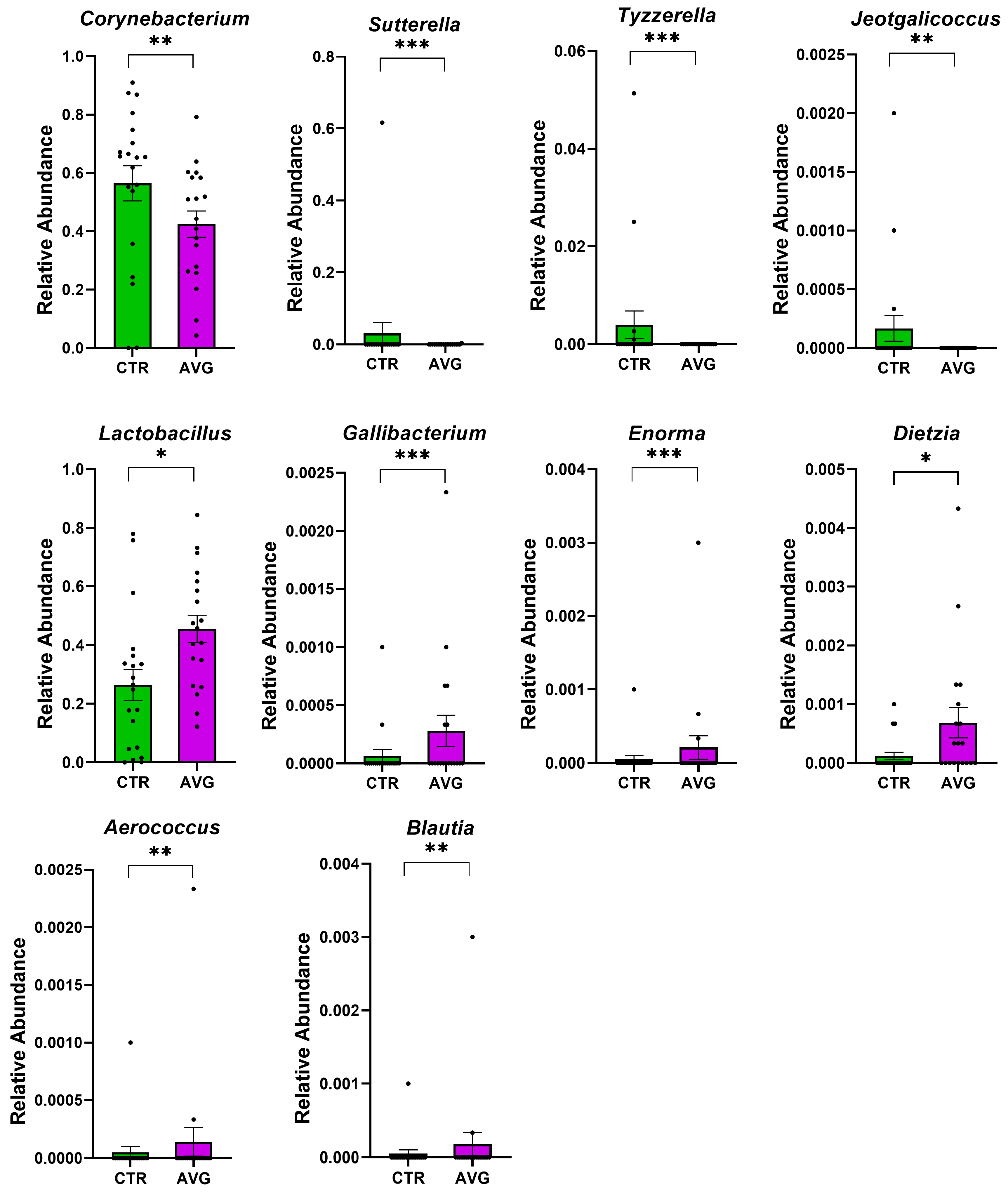
3.5. Univariate Taxa Alterations
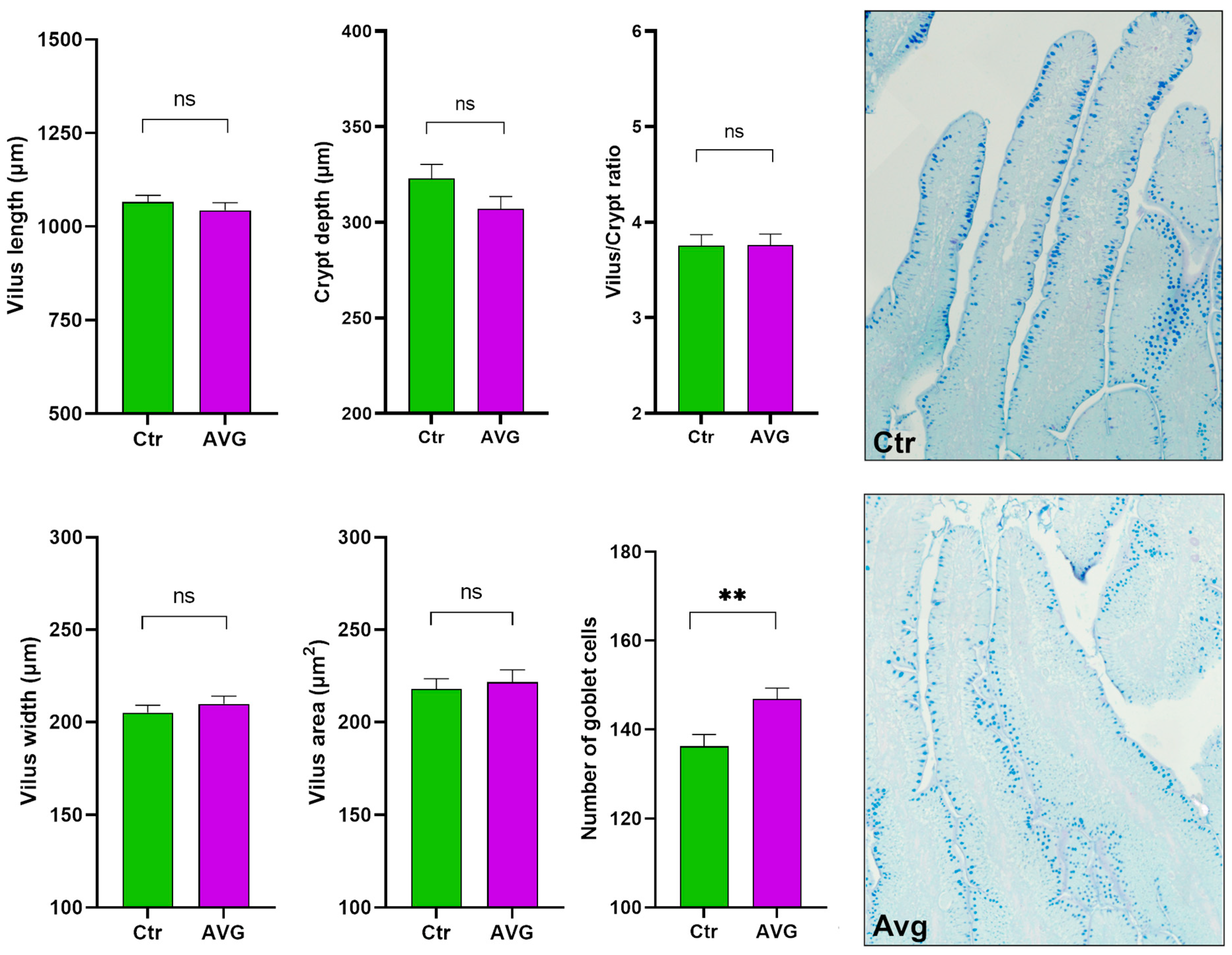
3.6. Histology
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- AgriFutures Australia. AgriFutures Chicken Meat. 2022. Available online: https://agrifutures.com.au/rural-industries/chicken-meat/ (accessed on 9 June 2022).
- Shahbandeh, M. Global Chicken Meat Production 2021 & 2022, by Selected Country. 2022. Available online: https://www.statista.com/statistics/237597/leading-10-countries-worldwide-in-poultry-meat-production-in-2007/#statisticContainer (accessed on 18 August 2022).
- Safe Food Queensland. Spotlight on Australia’s Poultry Meat Industry. 2021. Available online: https://www.safefood.qld.gov.au/newsroom/spotlight-on-australias-poultry-meat-industry/ (accessed on 9 June 2022).
- Diaz Carrasco, J.M.; Casanova, N.A.; Fernandez Miyakawa, M.E. Microbiota, gut health and chicken productivity: What is the connection? Microorganisms 2019, 7, 374. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.C.; Huang, M.Y.; Balasubramanian, B.; Jha, R. Heat stress affects jejunal immunity of Yellow-Feathered broilers and is potentially mediated by the microbiome. Front. Physiol. 2022, 13, 913696. [Google Scholar] [CrossRef] [PubMed]
- Rampoldi, F.; Prinz, I. Three layers of intestinal gammadelta T cells talk different languages with the microbiota. Front. Immunol. 2022, 13, 849954. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Johnson, T.A.; Zhang, H.; Cheng, H.W. The microbiota-gut-brain axis: Gut microbiota modulates conspecific aggression in diversely selected laying hens. Microorganisms 2022, 10, 1081. [Google Scholar] [CrossRef] [PubMed]
- Hafez, H.M.; Attia, Y.A. Challenges to the poultry industry: Current perspectives and strategic future after the COVID-19 outbreak. Front. Vet. Sci. 2020, 7, 516. [Google Scholar] [CrossRef] [PubMed]
- Kurtz, J.R.; Goggins, J.A.; McLachlan, J.B. Salmonella infection: Interplay between the bacteria and host immune system. Immunol. Lett. 2017, 190, 42–50. [Google Scholar] [CrossRef]
- Rouger, A.; Tresse, O.; Zagorec, M. Bacterial contaminants of poultry meat: Sources, species, and dynamics. Microorganisms 2017, 5, 50. [Google Scholar] [CrossRef]
- Zhao, Y.; Zeng, Y.; Zeng, D.; Wang, H.; Sun, N.; Xin, J.; Zhou, M.; Yang, H.; Lei, L.; Ling, H.; et al. Dietary Probiotic Supplementation Suppresses Subclinical Necrotic Enteritis in Broiler Chickens in a Microbiota-Dependent Manner. Front. Immunol. 2022, 13, 855426. [Google Scholar] [CrossRef]
- Stanley, D.; Geier, M.S.; Hughes, R.J.; Denman, S.E.; Moore, R.J. Highly variable microbiota development in the chicken gastrointestinal tract. PLoS ONE 2013, 8, e84290. [Google Scholar] [CrossRef]
- Chen, S.; Luo, S.; Yan, C. Gut Microbiota Implications for Health and Welfare in Farm Animals: A Review. Animals 2021, 12, 93. [Google Scholar] [CrossRef]
- Markowiak, P.; Slizewska, K. The role of probiotics, prebiotics and synbiotics in animal nutrition. Gut. Pathog. 2018, 10, 21. [Google Scholar] [CrossRef]
- Beck, C.N.; McDaniel, C.D.; Wamsley, K.G.S.; Kiess, A.S. The potential for inoculating Lactobacillus animalis and Enterococcus faecium alone or in combination using commercial in ovo technology without negatively impacting hatch and post-hatch performance. Poult. Sci. 2019, 98, 7050–7062. [Google Scholar] [CrossRef]
- O’Dea, E.E.; Fasenko, G.M.; Allison, G.E.; Korver, D.R.; Tannock, G.W.; Guan, L.L. Investigating the effects of commercial probiotics on broiler chick quality and production efficiency. Poult. Sci. 2006, 85, 1855–1863. [Google Scholar] [CrossRef]
- Luethy, P.M.; Huynh, S.; Ribardo, D.A.; Winter, S.E.; Parker, C.T.; Hendrixson, D.R. Microbiota-derived short-chain fatty acids modulate expression of Campylobacter jejuni determinants required for commensalism and virulence. mBio 2017, 8, e00407-17. [Google Scholar] [CrossRef]
- Lee, J.Y.; Han, G.G.; Kim, E.B.; Choi, Y.J. Comparative genomics of Lactobacillus salivarius strains focusing on their host adaptation. Microbiol. Res. 2017, 205, 48–58. [Google Scholar] [CrossRef]
- La Ragione, R.M.; Woodward, M.J. Competitive exclusion by Bacillus subtilis spores of Salmonella enterica serotype Enteritidis and Clostridium perfringens in young chickens. Vet. Microbiol. 2003, 94, 245–256. [Google Scholar] [CrossRef]
- Pereira, F.C.; Berry, D. Microbial nutrient niches in the gut. Environ. Microbiol. 2017, 19, 1366–1378. [Google Scholar] [CrossRef]
- Birchenough, G.M.; Johansson, M.E.; Gustafsson, J.K.; Bergstrom, J.H.; Hansson, G.C. New developments in goblet cell mucus secretion and function. Mucosal Immunol. 2015, 8, 712–719. [Google Scholar] [CrossRef]
- Dao, D.P.D.; Le, P.H. Histology, goblet cells. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- McDole, J.R.; Wheeler, L.W.; McDonald, K.G.; Wang, B.; Konjufca, V.; Knoop, K.A.; Newberry, R.D.; Miller, M.J. Goblet cells deliver luminal antigen to CD103+ dendritic cells in the small intestine. Nature 2012, 483, 345–349. [Google Scholar] [CrossRef]
- Wilkinson, N.; Hughes, R.J.; Bajagai, Y.S.; Aspden, W.J.; Hao Van, T.T.; Moore, R.J.; Stanley, D. Reduced environmental bacterial load during early development and gut colonisation has detrimental health consequences in Japanese quail. Heliyon 2020, 6, e03213. [Google Scholar] [CrossRef]
- Inc, A.S.P. Edible Gel Concentrate for Delivering Vaccines and Oral Additives Using Poultry Hatchery Tap Water. 2020. Available online: https://864d07.p3cdn1.secureserver.net/wp-content/uploads/2020/03/Gel-Pac-Booklet-WEB-03-23-20.pdf (accessed on 28 September 2022).
- Menke, S.; Gillingham, M.A.; Wilhelm, K.; Sommer, S. Home-Made Cost Effective Preservation Buffer Is a Better Alternative to Commercial Preservation Methods for Microbiome Research. Front. Microbiol. 2017, 8, 102. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Morrison, M. Improved extraction of PCR-quality community DNA from digesta and fecal samples. Biotechniques 2004, 36, 808–812. [Google Scholar] [CrossRef] [PubMed]
- Fadrosh, D.W.; Ma, B.; Gajer, P.; Sengamalay, N.; Ott, S.; Brotman, R.M.; Ravel, J. An improved dual-indexing approach for multiplexed 16S rRNA gene sequencing on the Illumina MiSeq platform. Microbiome 2014, 2, 6. [Google Scholar] [CrossRef] [PubMed]
- Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet J. 2011, 17, 10–12. [Google Scholar] [CrossRef]
- Bolyen, E.; Rideout, J.R.; Dillon, M.R.; Bokulich, N.A.; Abnet, C.C.; Al-Ghalith, G.A.; Alexander, H.; Alm, E.J.; Arumugam, M.; Asnicar, F.; et al. Reproducible, interactive, scalable and extensible microbiome data science using QIIME 2. Nat. Biotechnol. 2019, 37, 852–857. [Google Scholar] [CrossRef] [PubMed]
- Callahan, B.J.; McMurdie, P.J.; Rosen, M.J.; Han, A.W.; Johnson, A.J.; Holmes, S.P. DADA2: High-resolution sample inference from Illumina amplicon data. Nat. Methods 2016, 13, 581–583. [Google Scholar] [CrossRef]
- Quast, C.; Pruesse, E.; Yilmaz, P.; Gerken, J.; Schweer, T.; Yarza, P.; Peplies, J.; Glockner, F.O. The SILVA ribosomal RNA gene database project: Improved data processing and web-based tools. Nucleic Acids Res. 2013, 41, D590–D596. [Google Scholar] [CrossRef]
- Pfeiffer, S.; Pastar, M.; Mitter, B.; Lippert, K.; Hackl, E.; Lojan, P.; Oswald, A.; Sessitsch, A. Improved group-specific primers based on the full SILVA 16S rRNA gene reference database. Environ. Microbiol. 2014, 16, 2389–2407. [Google Scholar] [CrossRef]
- White, J.R.; Nagarajan, N.; Pop, M. Statistical methods for detecting differentially abundant features in clinical metagenomic samples. PLoS Comp. Biol. 2009, 5, e1000352. [Google Scholar] [CrossRef]
- Metzler-Zebeli, B.U.; Siegerstetter, S.C.; Magowan, E.; Lawlor, P.G.; Ne, O.C.; Zebeli, Q. Fecal microbiota transplant from highly feed efficient donors affects cecal physiology and microbiota in low- and high-feed efficient chickens. Front. Microbiol. 2019, 10, 1576. [Google Scholar] [CrossRef]
- Yin, Y.; Lei, F.; Zhu, L.; Li, S.; Wu, Z.; Zhang, R.; Gao, G.F.; Zhu, B.; Wang, X. Exposure of different bacterial inocula to newborn chicken affects gut microbiota development and ileum gene expression. ISME J. 2010, 4, 367–376. [Google Scholar] [CrossRef]
- Kubasova, T.; Kollarcikova, M.; Crhanova, M.; Karasova, D.; Cejkova, D.; Sebkova, A.; Matiasovicova, J.; Faldynova, M.; Sisak, F.; Babak, V.; et al. Gut anaerobes capable of chicken caecum colonisation. Microorganisms 2019, 7, 597. [Google Scholar] [CrossRef]
- Gong, Y.; Yang, H.; Wang, X.; Xia, W.; Lv, W.; Xiao, Y.; Zou, X. Early intervention with cecal fermentation broth regulates the colonization and development of gut microbiota in broiler chickens. Front. Microbiol. 2019, 10, 1422. [Google Scholar] [CrossRef]
- Fu, Y.; Hu, J.; Erasmus, M.A.; Johnson, T.A.; Cheng, H.W. Effects of early-life cecal microbiota transplantation from divergently selected inbred chicken lines on growth, gut serotonin, and immune parameters in recipient chickens. Poult. Sci. 2022, 101, 101925. [Google Scholar] [CrossRef]
- Glendinning, L.; Chintoan-Uta, C.; Stevens, M.P.; Watson, M. Effect of cecal microbiota transplantation between different broiler breeds on the chick flora in the first week of life. Poult. Sci. 2022, 101, 101624. [Google Scholar] [CrossRef]
- Johansson, M.E.; Jakobsson, H.E.; Holmen-Larsson, J.; Schutte, A.; Ermund, A.; Rodriguez-Pineiro, A.M.; Arike, L.; Wising, C.; Svensson, F.; Backhed, F.; et al. Normalization of Host Intestinal Mucus Layers Requires Long-Term Microbial Colonization. Cell Host Microbe 2015, 18, 582–592. [Google Scholar] [CrossRef]
- Calenge, F.; Beaumont, C. Toward integrative genomics study of genetic resistance to Salmonella and Campylobacter intestinal colonization in fowl. Front. Genet. 2012, 3, 261. [Google Scholar] [CrossRef]
- Wigley, P. Genetic resistance to Salmonella infection in domestic animals. Res. Vet. Sci. 2004, 76, 165–169. [Google Scholar] [CrossRef]
- Connell, S.; Meade, K.G.; Allan, B.; Lloyd, A.T.; Kenny, E.; Cormican, P.; Morris, D.W.; Bradley, D.G.; O’Farrelly, C. Avian resistance to Campylobacter jejuni colonization is associated with an intestinal immunogene expression signature identified by mRNA sequencing. PLoS ONE 2012, 7, e40409. [Google Scholar] [CrossRef]
- Amir Ebrahimi, N.; Salehi Jouzani, G.; Ebrahimi, M.A. Native chicken-derived Lactobacillus spp. strains with high probiotic, cholesterol-assimilation and aflatoxin-degradation capabilities. Iran. J. Microbiol. 2022, 14, 227–237. [Google Scholar] [CrossRef]
- Mindus, C.; van Staaveren, N.; Fuchs, D.; Gostner, J.M.; Kjaer, J.B.; Kunze, W.; Mian, M.F.; Shoveller, A.K.; Forsythe, P.; Harlander-Matauschek, A. Regulatory T cell modulation by Lactobacillus rhamnosus improves feather damage in chickens. Front. Vet. Sci. 2022, 9, 855261. [Google Scholar] [CrossRef]
- Wu, Z.; Yang, K.; Zhang, A.; Chang, W.; Zheng, A.; Chen, Z.; Cai, H.; Liu, G. Effects of Lactobacillus acidophilus on the growth performance, immune response, and intestinal barrier function of broiler chickens challenged with Escherichia coli O157. Poult. Sci. 2021, 100, 101323. [Google Scholar] [CrossRef]
- Zhang, S.; Zhong, G.; Shao, D.; Wang, Q.; Hu, Y.; Wu, T.; Ji, C.; Shi, S. Dietary supplementation with Bacillus subtilis promotes growth performance of broilers by altering the dominant microbial community. Poult. Sci. 2021, 100, 100935. [Google Scholar] [CrossRef] [PubMed]
- Maki, J.J.; Looft, T. Megasphaera stantonii sp. nov., a butyrate-producing bacterium isolated from the cecum of a healthy chicken. Int. J. Syst. Evol. Microbiol. 2018, 68, 3409–3415. [Google Scholar] [CrossRef]
- Hou, Q.; Kwok, L.Y.; Zheng, Y.; Wang, L.; Guo, Z.; Zhang, J.; Huang, W.; Wang, Y.; Leng, L.; Li, H.; et al. Differential fecal microbiota are retained in broiler chicken lines divergently selected for fatness traits. Sci. Rep. 2016, 6, 37376. [Google Scholar] [CrossRef] [PubMed]
- Ogbuewu, I.P.; Mabelebele, M.; Sebola, N.A.; Mbajiorgu, C. Bacillus probiotics as alternatives to in-feed antibiotics and its influence on growth, serum chemistry, antioxidant status, intestinal histomorphology, and lesion scores in disease-challenged broiler chickens. Front. Vet. Sci. 2022, 9, 876725. [Google Scholar] [CrossRef] [PubMed]
- Abudabos, A.M.; Alyemni, A.H.; Dafalla, Y.M.; Khan, R.U. Effect of organic acid blend and Bacillus subtilis alone or in combination on growth traits, blood biochemical and antioxidant status in broilers exposed to Salmonella typhimurium challenge during the starter phase. J. Appl. Anim. Res. 2017, 45, 538–542. [Google Scholar] [CrossRef]
- Bai, K.; Feng, C.; Jiang, L.; Zhang, L.; Zhang, J.; Zhang, L.; Wang, T. Dietary effects of Bacillus subtilis fmbj on growth performance, small intestinal morphology, and its antioxidant capacity of broilers. Poult. Sci. 2018, 97, 2312–2321. [Google Scholar] [CrossRef]
- Dong, Y.; Li, R.; Liu, Y.; Ma, L.; Zha, J.; Qiao, X.; Chai, T.; Wu, B. Benefit of dietary supplementation with Bacillus subtilis BYS2 on growth performance, immune response, and disease resistance of broilers. Probiotics Antimicrob. Proteins 2020, 12, 1385–1397. [Google Scholar] [CrossRef]
- Bindari, Y.R.; Moore, R.J.; Van, T.T.H.; Walkden-Brown, S.W.; Gerber, P.F. Microbial taxa in dust and excreta associated with the productive performance of commercial meat chicken flocks. Anim. Microbiome 2021, 3, 66. [Google Scholar] [CrossRef]
- Zhou, H.; Guo, Y.; Liu, Z.; Wu, H.; Zhao, J.; Cao, Z.; Zhang, H.; Shang, H. Comfrey polysaccharides modulate the gut microbiota and its metabolites SCFAs and affect the production performance of laying hens. Int. J. Biol. Macromol. 2022, 215, 45–56. [Google Scholar] [CrossRef]
- Zhang, L.; Hong, Y.; Liao, Y.; Tian, K.; Sun, H.; Liu, X.; Tang, Y.; Hassanin, A.A.; Abdelnour, S.A.; Suthikrai, W.; et al. Dietary Lasia spinosa Thw. improves growth performance in broilers. Front. Nutr. 2021, 8, 775223. [Google Scholar] [CrossRef]
- Gorain, C.; Singh, A.; Bhattacharyya, S.; Kundu, A.; Lahiri, A.; Gupta, S.; Mallick, A.I. Mucosal delivery of live Lactococcus lactis expressing functionally active JlpA antigen induces potent local immune response and prevent enteric colonization of Campylobacter jejuni in chickens. Vaccine 2020, 38, 1630–1642. [Google Scholar] [CrossRef]
- Ren, Y.; Lu, X.; Yang, Z.; Lei, H. Protective immunity induced by oral vaccination with a recombinant Lactococcus lactis vaccine against H5Nx in chickens. BMC Vet. Res. 2022, 18, 3. [Google Scholar] [CrossRef]
- Xiang, H.; Gan, J.; Zeng, D.; Li, J.; Yu, H.; Zhao, H.; Yang, Y.; Tan, S.; Li, G.; Luo, C.; et al. Specific microbial taxa and functional capacity contribute to chicken abdominal fat deposition. Front. Microbiol. 2021, 12, 643025. [Google Scholar] [CrossRef]
- Li, Z.; Peng, C.; Zhang, G.; Shen, Y.; Zhang, Y.; Liu, C.; Liu, M.; Wang, F. Prevalence and characteristics of multidrug-resistant Proteus mirabilis from broiler farms in Shandong Province, China. Poult. Sci. 2022, 101, 101710. [Google Scholar] [CrossRef]
- Sanches, M.S.; Baptista, A.A.S.; de Souza, M.; Menck-Costa, M.F.; Justino, L.; Nishio, E.K.; Oba, A.; Bracarense, A.; Rocha, S.P.D. Proteus mirabilis causing cellulitis in broiler chickens. Braz. J. Microbiol. 2020, 51, 1353–1362. [Google Scholar] [CrossRef]
- Elbestawy, A.R.; Abd-Ellatieff, H.A.; Ellakany, H.F.; Abd El-Hamid, H.S.; Abou Rawash, A.A.; Gado, A.R.; Abd El-Aziz, A.H.; Eid, A.A.M.; El-Shall, N.A. Respiratory and reproductive impairment of commercial layer chickens after experimental infection with Gallibacterium anatis biovar haemolytica. Avian Dis. 2020, 64, 536–541. [Google Scholar] [CrossRef]
- McKenna, A.; Ijaz, U.Z.; Kelly, C.; Linton, M.; Sloan, W.T.; Green, B.D.; Lavery, U.; Dorrell, N.; Wren, B.W.; Richmond, A.; et al. Impact of industrial production system parameters on chicken microbiomes: Mechanisms to improve performance and reduce Campylobacter. Microbiome 2020, 8, 128. [Google Scholar] [CrossRef]
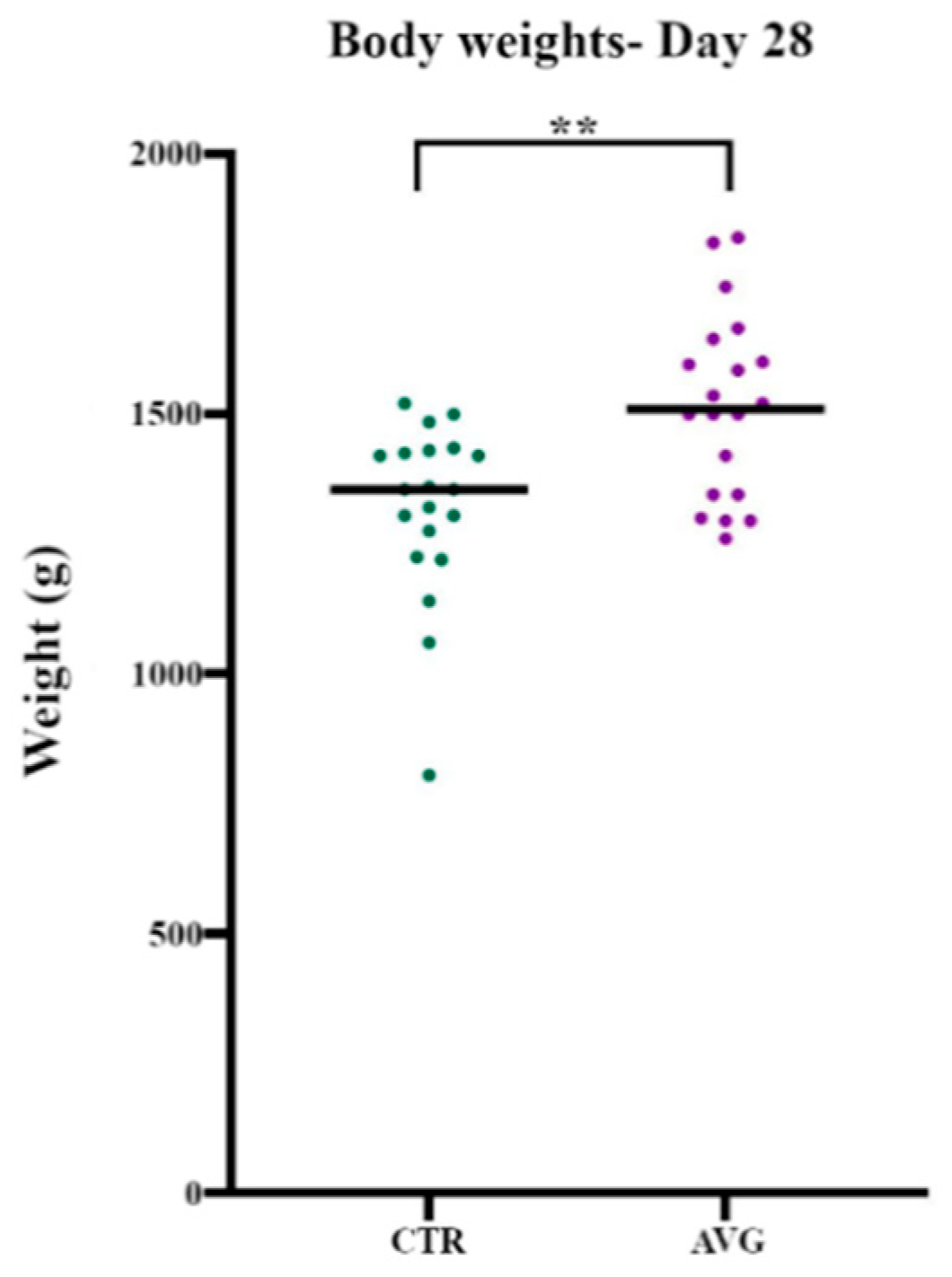

| Group | Average Body Weight (g) | SEM |
|---|---|---|
| Control | 1318 | 37.9 |
| AVG | 1516 | 39.5 |
| Measures | Groups | R2 | p Value | Significance |
|---|---|---|---|---|
| Weighted UniFrac | Cec.AVG vs. Cec.CTR | 0.237666 | <0.001 | *** |
| Jej.AVG vs. Jej.CTR | 0.150488 | 0.007 | ** | |
| JejMuc.AVG vs. JejMuc.CTR | 0.101046 | 0.023 | * | |
| Crop.AVG vs. Crop.CTR | 0.029126 | 0.32 | ||
| PVC.AVG vs. PVC.CTR | 0.043081 | 0.353 | ||
| Unweighted UniFrac | Cec.AVG vs. Cec.CTR | 0.188473 | <0.001 | *** |
| PVC.AVG vs. PVC.CTR | 0.11597 | 0.005 | ** | |
| Jej.AVG vs. Jej.CTR | 0.068658 | 0.01 | ** | |
| Crop.AVG vs. Crop.CTR | 0.062496 | 0.014 | * | |
| JejMuc.AVG vs. JejMuc.CTR | 0.038949 | 0.163 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kayal, A.; Stanley, D.; Radovanovic, A.; Horyanto, D.; Van, T.T.H.; Bajagai, Y.S. Controlled Intestinal Microbiota Colonisation in Broilers under the Industrial Production System. Animals 2022, 12, 3296. https://doi.org/10.3390/ani12233296
Kayal A, Stanley D, Radovanovic A, Horyanto D, Van TTH, Bajagai YS. Controlled Intestinal Microbiota Colonisation in Broilers under the Industrial Production System. Animals. 2022; 12(23):3296. https://doi.org/10.3390/ani12233296
Chicago/Turabian StyleKayal, Advait, Dragana Stanley, Anita Radovanovic, Darwin Horyanto, Thi Thu Hao Van, and Yadav S. Bajagai. 2022. "Controlled Intestinal Microbiota Colonisation in Broilers under the Industrial Production System" Animals 12, no. 23: 3296. https://doi.org/10.3390/ani12233296
APA StyleKayal, A., Stanley, D., Radovanovic, A., Horyanto, D., Van, T. T. H., & Bajagai, Y. S. (2022). Controlled Intestinal Microbiota Colonisation in Broilers under the Industrial Production System. Animals, 12(23), 3296. https://doi.org/10.3390/ani12233296







