Haemonchosis in Sheep and Goats, Control Strategies and Development of Vaccines against Haemonchus contortus
Abstract
Simple Summary
Abstract
1. Introduction
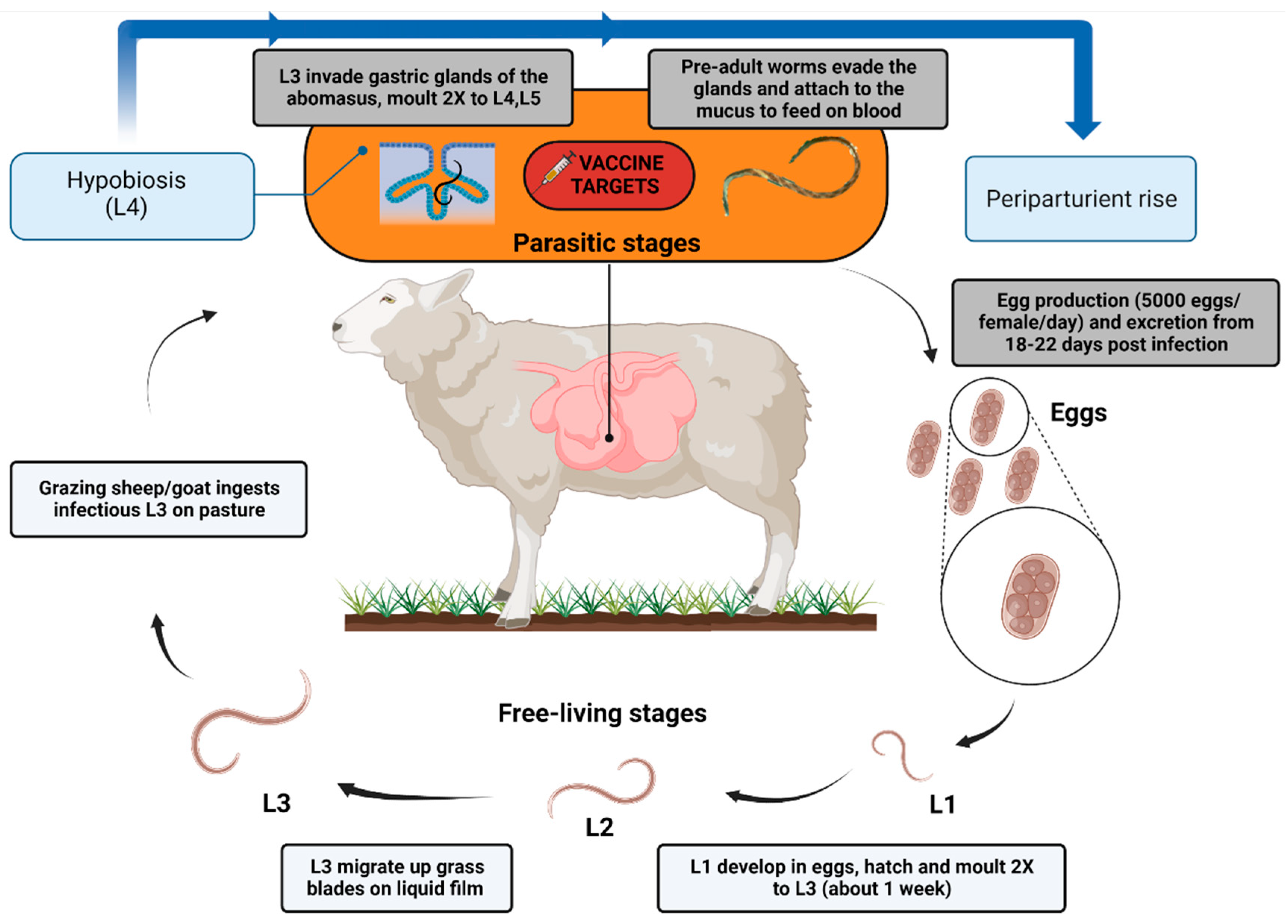
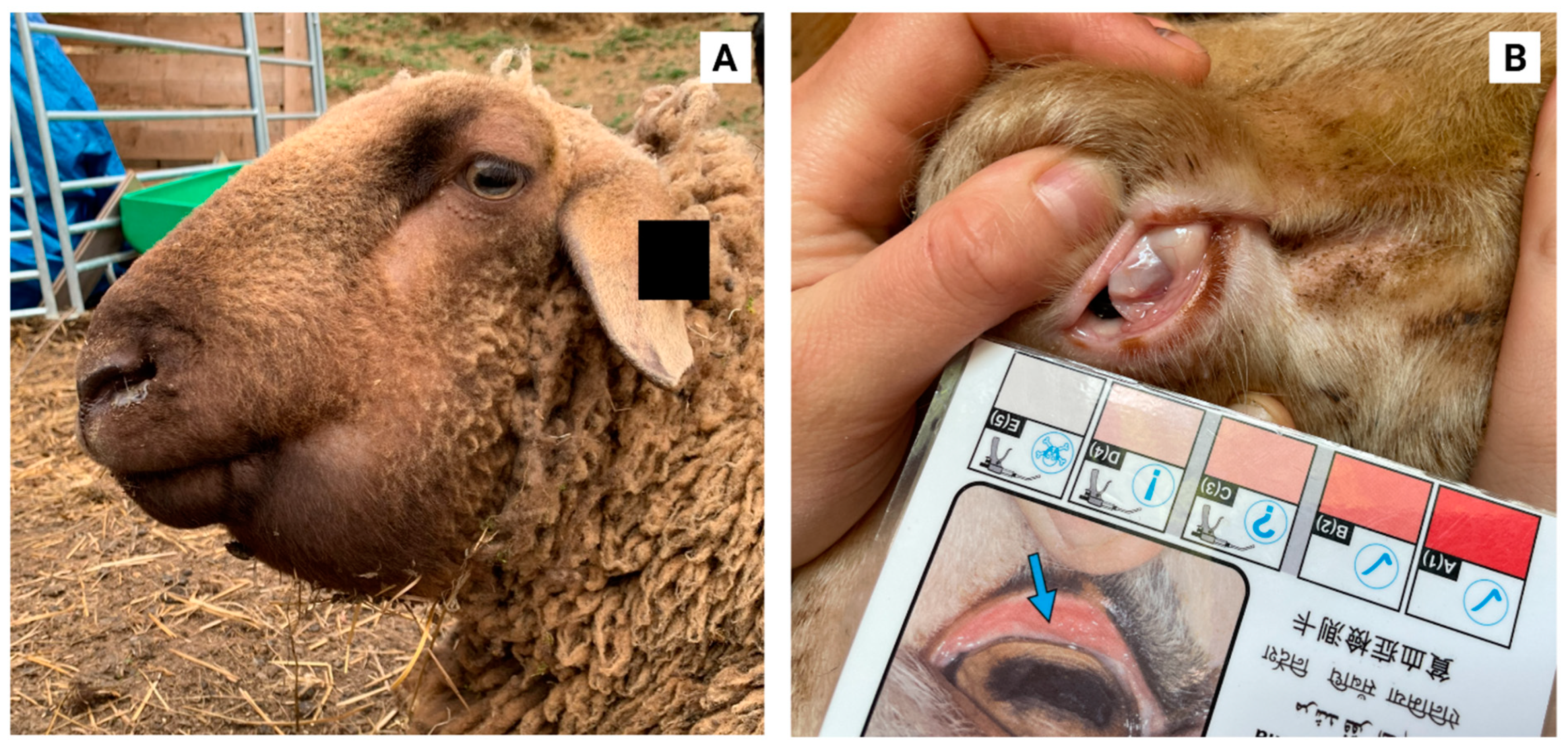
2. Haemonchus contortus
3. Current Control Strategies
4. Vaccination against H. contortus
4.1. Native Antigen Vaccine
4.2. Recombinant Protein-Based Vaccines
4.3. DNA Vaccines
4.4. Efficacy Assessment in Clinical Trials
| Antigens | Adjuvant and Supplement | Animal Breed | Age and Sex | Group Size | Dosage (µg/Dose) | Number of Vaccination | Administration Route * | Infection Dosage L3 Larvae | Efficacy Accessement | |||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Antigen-Specific Antibodies | FEC Reduction | Worm Burden Reduction | References | |||||||||
| native or recombiant antigens | ||||||||||||
| whole ES antigens | Alhydrogel® | Zwartbles lamb | 3-month old | 5 | 75 | 3 | s.c. | 10,000 to 12,000 | IgG↑, IgE↑, IgA↑, IgM↑ | 89.0% | 54.0% | [104] |
| native p26/23 | Freund’s adjuvant | Manchego lamb | 3.5 to 5-month ♀ | 5 | 50 | 3 | s.c., i.m. | 400 | IgG↑ | 64.2% | 61.6% | [77] |
| rHcp26/23 | Freund’s adjuvant | Manchego lamb | 3-month old ♀ | 5 | 100 | 3 | s.c., i.m. | 16,000 | IgG↑ | NS | NS | [63] |
| native Hc23 | aluminium hydroxide | Assaf lamb | 4–5 month old ♀ | 7 | 100 | 3 | s.c., i.m. | 15,000 | IgG↑ | 70.67–85.64% | 67.17% to >86% | [60] |
| rHc23 | aluminium hydroxide | Assaf lamb | 5–6 month old ♀ | 7 | 100 | 3 | NA | 15,000 | IgG↑ | 83.5% | 84.7% | [64] |
| rHc23 | aluminium hydroxide | Manchego lamb | 6-month old ♀ | 7 | 200 | 3 | i.m. | 4000 | IgG↑ | 82.37 ± 5.98% | 71.0 ± 14.3% | [109] |
| rHc23 | Quil A® | Manchego lamb | 6-month old ♀ | 7 | 200 | 3 | i.m. | 4000 | IgG↑ | 74.58 ± 10.94% | 47.3 ± 35.4% | |
| rHc23 | LPS | Entrefino lambs | 5 to 6-month old ♀ | 6 | 100 | 4 | i.m. | 6*1000 | IgG↑ | 43.3% | 45.5% | [108] |
| rHc23 | LPS | Entrefino lambs | 5 to 6-month old ♀ | 7 | 100 | 4 | i.m. | 6*2000 | IgG↑ | 43.5% | 84.3% | |
| native ES15/ES24 | DDA | Texel sheep | 8-month old | 10 | 50–100 | 3 | s.c. | 20,000 | IgG1↑ | 72.9% | 82.2% | [91] |
| native ES15/ES24 | DDA | Texel sheep | 3-month old | 4 | 50–100 | 3 | s.c. | 10,000 | IgG1↑, IgA↑ | ND | −34.4% | [103] |
| native ES15/ES24 | DDA | Texel sheep | 9-month old | 10 | 50–100 | 3 | s.c. | 20,000 | IgG1↑, IgA↑ | ND | 82.2% | |
| rES15/rES24 | DDA/insect cell extract | Texel, Swifter and Zwart Bles | 9-month old | 7 | 100 (cocktail) | 3 | s.c. | 5000 | IgG1↑ | 49.0% | 65.0% | [61] |
| native ES15/ES24 | DDA | Texel, Swifter and Zwart Bles | 9-month old | 7 | 100 | 3 | s.c. | 5000 | IgG1↑ | 57.0% | 70.0% | |
| rH11-1 | insect cell extract | Merino lamb | 5-month old ♀ | 5 | 300 (crude extract) | 2 | i.m | 15,000 | serum antibody↑ | ND | 29.0% | [65] |
| rHcGST-H11-1 | insect cell extract | Merino lamb | 5-month old ♀ | 5 | 300 (crude extract) | 2 | i.m | 15,000 | serum antibody↑ | ND | 20.0% | |
| native H11 | Vax Saponin | Suffolk-cross lamb | 6-month old | 7 | 40 | 3 | s.c. | 5000 | IgG↑, IgE↑, IgM↑ | 99.9% | 93.6% | [66] |
| rH11-1+rH11-4 | Vax Saponin | Suffolk-cross lamb | 6-month old | 7 | 20 (cocktail) | 3 | s.c | 5000 | IgG↑ | NS | NS | |
| rH11-4/5 | Vax Saponin | Suffolk-cross lamb | 6-month old | 7 | 20 (cocktail) | 3 | s.c | 5000 | IgG↑ | NS | NS | |
| rH11 (1-570 aa) | Freund’s adjuvant | Boer goat | 6-month old | 5 | 750 (crude extract) | 3 | i.m. | 5000 | IgG↑ | 37.71% | 24.91% | [67] |
| rH11 (223-570 aa) | Freund’s adjuvant | Boer goat | 6-month old | 5 | 50 | 3 | i.m. | 5000 | IgG↑ | 26.04% | 18.46% | |
| rH11-5/AC1/PEP1 | Quil A/Rehydragel/Covexin | White mountain breed lamb | 6-month old | 6 | 100 | 3 | i.m. | 5000 | IgG↑ | 23.9% | 13.5% | [93] |
| native H-gal-GP | Quil A® | Suffolk–Greyface crosses | 5 to 9-month old | 7 | 100 | 3 | i.m. | 5000 | IgG↑ | 93.0% | 69.0% | [81] |
| native H-gal-GP | Freund’s adjuvant | lamb | 2-month old | 7 | 200 | 3 | i.m. | 5000 | ND | 93.0% | 52.9% | [57] |
| rMEP-1/3/4/rPEP-1 | Quil A® | castrated Blackface×Leicester | 9-month old ♂ | 7 | 200 (cocktail) | 3 | i.m. | 5000 | IgG↑ | 2.5% | 1.0% | [62] |
| native H-gal-GP | Quil A® | castrated Blackface×Leicester | 9-month old ♂ | 7 | 100 | 3 | i.m. | 5000 | IgG↑ | 88.5% | 72.3% | |
| H11 and H-gal-GP | saponin | crossbred Brergamacia | ewes | 39 | 5 and 50 | 3 + 5 | i.m. | natural infection | IgG↑ | NS | ND | [85] |
| H11 and H-gal-GP | saponin | crossbred Brergamacia | lambs | 48 | 5 and 50 | 3 + 3 | i.m. | natural infection | IgG↑ | 72.0% | 68.0% | |
| Barbervax® | Quil A® | crossbred Bergamasco | ewes | 29 | 5 | 3 + 3 | s.c. | natural infection | IgG↑ | NA | ND | [40] |
| Barbervax® | Quil A® | crossbred Bergamasco | lambs | 29 | 5 | 3 | s.c. | natural infection | IgG↑ | 80.0% | ND | |
| Barbervax® | Quil A® | Saanen Nubian dairy goat | 6-month old | 10 | 5 | 3 + 3 | s.c. | 3*6000 | IgG↑ | 57.4 ± 17.6% | ND | [59] |
| Barbervax® | Quil A® | Anglo Nubian dairy goat | 6-month old | 10 | 5 | 3 + 3 | s.c. | 3*6000 | IgG↑ | 69.8 ± 11.7% | ND | |
| Barbervax® | Quil A® | alpacas | 4 to 6-month old | 7 | 5 | 3 | s.c. | 3*1500 | IgG↑ | NS | ND | [86] |
| Barbervax® | Quil A® | crossbred Santa Ines hair sheep | ewes | 45 | 5 | 3 + 5 | s.c. | natural infection | IgG↑ | 90.2 ± 4.03% | ND | [84] |
| Barbervax® | Quil A® | crossbred Santa Ines hair sheep | 1-year old | 12 | 5 | 3 | s.c. | 4*3000 | IgG↑ | 87 ± 5.4% | 79.0% | |
| DNA Vaccine candidates | ||||||||||||
| HC29 | PBS buffer (pH 7.4) | local-bred goat | 8 to 10-month old | 5 | 500 | 2 | i.m | 5000 | IgG↑ | 36.1% | 35.6% | [70] |
| H11 | PBS buffer (pH 7.4) * | local-bred goat | 10-month old | 4 | 300 | 2 | i.m | 5000 | IgG↑ | 56.6% | 46.7% | [71] |
| GAPDH | PBS buffer (pH 7.4) | local-bred goat | 9 to 10-month old | 5 | 500 | 2 | i.m | 5000 | IgG↑ | 34.9% | 37.7% | [72] |
| Dim-1 | PBS buffer (pH 7.4) | local-bred goat | 8 to 10-month old | 5 | 500 | 2 | i.m. | 5000 | IgG↑ | 45.7% | 51.1% | [73] |
| Actin | PBS buffer (pH 7.4) | local-bred goat | 8 to 10-month old | 5 | 100 | 2 | i.m. | 5000 | IgG↑ | 34.4% | 33.1% | [74] |
5. New Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Knox, D.P.; Redmond, D.L.; Jones, D.G. Characterization of proteinases in extracts of adult Haemonchus contortus, the ovine abomasal nematode. Parasitology 1993, 106 Pt 4, 395–404. [Google Scholar] [CrossRef]
- Charlier, J.; Rinaldi, L.; Musella, V.; Ploeger, H.W.; Chartier, C.; Vineer, H.R.; Hinney, B.; von Samson-Himmelstjerna, G.; Băcescu, B.; Mickiewicz, M.; et al. Initial assessment of the economic burden of major parasitic helminth infections to the ruminant livestock industry in Europe. Prev. Vet. Med. 2020, 182, 105103. [Google Scholar] [CrossRef]
- Food and Agriculture Organization. FAOSTAT Dataset: Crops and Livestock Products. Available online: https://www.fao.org/faostat/en/#data/QCL (accessed on 15 June 2022).
- Hoberg, E.P.; Zarlenga, D.S. Evolution and Biogeography of Haemonchus contortus: Linking Faunal Dynamics in Space and Time. Adv. Parasitol. 2016, 93, 1–30. [Google Scholar] [CrossRef]
- Kaplan, R.M.; Vidyashankar, A.N. An inconvenient truth: Global worming and anthelmintic resistance. Vet. Parasitol. 2012, 186, 70–78. [Google Scholar] [CrossRef]
- Besier, R.B.; Kahn, L.P.; Sargison, N.D.; van Wyk, J.A. Diagnosis, Treatment and Management of Haemonchus contortus in Small Ruminants. Adv. Parasitol. 2016, 93, 181–238. [Google Scholar] [CrossRef]
- Rowe, J.B.; Nolan, J.V.; de Chaneet, G.; Teleni, E.; Holmes, P.H. The effect of haemonchosis and blood loss into the abomasum on digestion in sheep. Br. J. Nutr. 1988, 59, 125–139. [Google Scholar] [CrossRef]
- Kotze, A.C.; Hunt, P.W.; Skuce, P.; von Samson-Himmelstjerna, G.; Martin, R.J.; Sager, H.; Krücken, J.; Hodgkinson, J.; Lespine, A.; Jex, A.R.; et al. Recent advances in candidate-gene and whole-genome approaches to the discovery of anthelmintic resistance markers and the description of drug/receptor interactions. Int. J. Parasitol. Drugs Drug Resist. 2014, 4, 164–184. [Google Scholar] [CrossRef]
- Rose Vineer, H.; Morgan, E.R.; Hertzberg, H.; Bartley, D.J.; Bosco, A.; Charlier, J.; Chartier, C.; Claerebout, E.; de Waal, T.; Hendrickx, G.; et al. Increasing importance of anthelmintic resistance in European livestock: Creation and meta-analysis of an open database. Parasite 2020, 27, 69. [Google Scholar] [CrossRef]
- Charlier, J.; Bartley, D.J.; Sotiraki, S.; Martinez-Valladares, M.; Claerebout, E.; von Samson-Himmelstjerna, G.; Thamsborg, S.M.; Hoste, H.; Morgan, E.R.; Rinaldi, L. Anthelmintic resistance in ruminants: Challenges and solutions. Adv. Parasitol. 2022, 115, 171–227. [Google Scholar] [CrossRef]
- Meeusen, E.N.T.; Walker, J.; Peters, A.; Pastoret, P.-P.; Jungersen, G. Current status of veterinary vaccines. Clin. Microbiol. Rev. 2007, 20, 489–510. [Google Scholar] [CrossRef]
- Claerebout, E.; Geldhof, P. Helminth Vaccines in Ruminants: From Development to Application. Vet. Clin. North Am. Food Anim. Pract. 2020, 36, 159–171. [Google Scholar] [CrossRef]
- Knox, D.P.; Smith, W.D. Vaccination against gastrointestinal nematode parasites of ruminants using gut-expressed antigens. Vet. Parasitol. 2001, 100, 21–32. [Google Scholar] [CrossRef]
- Smith, T.S.; Munn, E.A. Strategies for vaccination against gastro-intestinal nematodes. Rev. Sci. Tech. 1990, 9, 577–595. [Google Scholar] [CrossRef]
- Munn, E.A. Development of a vaccine against haemonchus contortus. Parasitol. Today 1993, 9, 338–339. [Google Scholar] [CrossRef]
- Skuce, P.J.; Redmond, D.L.; Liddell, S.; Stewart, E.M.; Newlands, G.F.; Smith, W.D.; Knox, D.P. Molecular cloning and characterization of gut-derived cysteine proteinases associated with a host protective extract from Haemonchus contortus. Parasitology 1999, 119 Pt 4, 405–412. [Google Scholar] [CrossRef]
- Newton, S.E.; Meeusen, E.N.T. Progress and new technologies for developing vaccines against gastrointestinal nematode parasites of sheep. Parasite Immunol. 2003, 25, 283–296. [Google Scholar] [CrossRef]
- Nisbet, A.J.; Meeusen, E.N.; González, J.F.; Piedrafita, D.M. Immunity to Haemonchus contortus and Vaccine Development. Adv. Parasitol. 2016, 93, 353–396. [Google Scholar] [CrossRef]
- Veglia, F. The Anatomy and Life History of the Haemonchus contortus (Rud.); Government Printer and Stationery Office: Pretoria, South Africa, 1916.
- Besier, R.B.; Kahn, L.P.; Sargison, N.D.; van Wyk, J.A. The Pathophysiology, Ecology and Epidemiology of Haemonchus contortus Infection in Small Ruminants. Adv. Parasitol. 2016, 93, 95–143. [Google Scholar] [CrossRef]
- Rose, H.; Caminade, C.; Bolajoko, M.B.; Phelan, P.; van Dijk, J.; Baylis, M.; Williams, D.; Morgan, E.R. Climate-driven changes to the spatio-temporal distribution of the parasitic nematode, Haemonchus contortus, in sheep in Europe. Glob. Chang. Biol. 2016, 22, 1271–1285. [Google Scholar] [CrossRef]
- Gibbs, H.C. Mechanisms of survival of nematode parasites with emphasis on hypobiosis. Vet. Parasitol. 1982, 11, 25–48. [Google Scholar] [CrossRef]
- Gibbs, H.C. Hypobiosis and the periparturient rise in sheep. Vet. Clin. North Am. Food Anim. Pract. 1986, 2, 345–353. [Google Scholar] [CrossRef]
- Amarante, A.F.; Bagnola Júnior, J.; Amarante, M.R.; Barbosa, M.A. Host specificity of sheep and cattle nematodes in São Paulo state, Brazil. Vet. Parasitol. 1997, 73, 89–104. [Google Scholar] [CrossRef]
- Achi, Y.L.; Zinsstag, J.; Yao, K.; Yeo, N.; Dorchies, P.; Jacquiet, P. Host specificity of Haemonchus spp. for domestic ruminants in the savanna in northern Ivory Coast. Vet. Parasitol. 2003, 116, 151–158. [Google Scholar] [CrossRef]
- Charlier, J.; Höglund, J.; Morgan, E.R.; Geldhof, P.; Vercruysse, J.; Claerebout, E. Biology and Epidemiology of Gastrointestinal Nematodes in Cattle. Vet. Clin. North Am. Food Anim. Pract. 2020, 36, 1–15. [Google Scholar] [CrossRef]
- Edwards, E.E.; Garner, B.C.; Williamson, L.H.; Storey, B.E.; Sakamoto, K. Pathology of Haemonchus contortus in New World camelids in the southeastern United States: A retrospective review. J. Vet. Diagn. Invest. 2016, 28, 105–109. [Google Scholar] [CrossRef]
- Gareh, A.; Elhawary, N.M.; Tahoun, A.; Ramez, A.M.; El-Shewehy, D.M.M.; Elbaz, E.; Khalifa, M.I.; Alsharif, K.F.; Khalifa, R.M.A.; Dyab, A.K.; et al. Epidemiological, Morphological, and Morphometric Study on Haemonchus spp. Recovered From Goats in Egypt. Front. Vet. Sci. 2021, 8, 705619. [Google Scholar] [CrossRef]
- Zajac, A.M. Gastrointestinal nematodes of small ruminants: Life cycle, anthelmintics, and diagnosis. Vet. Clin. North Am. Food Anim. Pract. 2006, 22, 529–541. [Google Scholar] [CrossRef]
- Pérez, J.; Garcia, P.M.; Hernandez, S.; Martinez-Moreno, A.; Martin de las Mulas, J.; Camara, S. Pathological and immunohistochemical study of the abomasum and abomasal lymph nodes in goats experimentally infected with Haemonchus contortus. Vet. Res. 2001, 32, 463–473. [Google Scholar] [CrossRef]
- Salman, S.K.; Duncan, J.L. The abomasal histology of worm-free sheep given primary and challenge infections of Haemonchus contortus. Vet. Parasitol. 1984, 16, 43–54. [Google Scholar] [CrossRef]
- van Wyk, J.A. Refugia—Overlooked as perhaps the most potent factor concerning the development of anthelmintic resistance. Onderstepoort J. Vet. Res. 2001, 68, 55–67. [Google Scholar]
- Taylor, M.A. SCOPS and COWS—’Worming it out of UK farmers’. Vet. Parasitol. 2012, 186, 65–69. [Google Scholar] [CrossRef] [PubMed]
- Torres-Acosta, J.; Hoste, H. Alternative or improved methods to limit gastro-intestinal parasitism in grazing sheep and goats. Small Ruminant Research 2008, 77, 159–173. [Google Scholar] [CrossRef]
- van Wyk, J.A.; Malan, F.S.; Randles, J.L. How long before resistance makes it impossible to control some field strains of Haemonchus contortus in South Africa with any of the modern anthelmintics? Vet. Parasitol. 1997, 70, 111–122. [Google Scholar] [CrossRef]
- Woodgate, R.G.; Love, S. WormKill to WormBoss--can we sell sustainable sheep worm control? Vet. Parasitol. 2012, 186, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Kearney, P.E.; Murray, P.J.; Hoy, J.M.; Hohenhaus, M.; Kotze, A. The ’Toolbox’ of strategies for managing Haemonchus contortus in goats: What’s in and what’s out. Vet. Parasitol. 2016, 220, 93–107. [Google Scholar] [CrossRef] [PubMed]
- Greer, A.W.; van Wyk, J.A.; Hamie, J.C.; Byaruhanga, C.; Kenyon, F. Refugia-Based Strategies for Parasite Control in Livestock. Vet. Clin. North Am. Food Anim. Pract. 2020, 36, 31–43. [Google Scholar] [CrossRef] [PubMed]
- Lanusse, C.; Canton, C.; Virkel, G.; Alvarez, L.; Costa-Junior, L.; Lifschitz, A. Strategies to Optimize the Efficacy of Anthelmintic Drugs in Ruminants. Trends Parasitol. 2018, 34, 664–682. [Google Scholar] [CrossRef]
- Bassetto, C.C.; Almeida, F.A.; Newlands, G.F.J.; Smith, W.D.; Castilhos, A.M.; Fernandes, S.; Siqueira, E.R.; Amarante, A.F.T. Trials with the Haemonchus vaccine, Barbervax®, in ewes and lambs in a tropical environment: Nutrient supplementation improves protection in periparturient ewes. Vet. Parasitol. 2018, 264, 52–57. [Google Scholar] [CrossRef]
- Cériac, S.; Archimède, H.; Feuillet, D.; Félicité, Y.; Giorgi, M.; Bambou, J.-C. Supplementation with rumen-protected proteins induces resistance to Haemonchus contortus in goats. Sci. Rep. 2019, 9, 1237. [Google Scholar] [CrossRef]
- Houdijk, J.G.M.; Kyriazakis, I.; Kidane, A.; Athanasiadou, S. Manipulating small ruminant parasite epidemiology through the combination of nutritional strategies. Vet. Parasitol. 2012, 186, 38–50. [Google Scholar] [CrossRef]
- Sweeney, T.; Hanrahan, J.P.; Ryan, M.T.; Good, B. Immunogenomics of gastrointestinal nematode infection in ruminants - breeding for resistance to produce food sustainably and safely. Parasite Immunol. 2016, 38, 569–586. [Google Scholar] [CrossRef] [PubMed]
- Gray, G.D. Genetic resistance to haemonchosis in sheep. Parasitol. Today 1987, 3, 253–255. [Google Scholar] [CrossRef]
- Balconi Marques, C.; Goldberg, V.; Ciappesoni, G. Genetic parameters for production traits, resistance and resilience to Nematode parasites under different worm burden challenges in Corriedale sheep. Vet. Parasitol. 2020, 287, 109272. [Google Scholar] [CrossRef] [PubMed]
- Gasser, R.B.; Schwarz, E.M.; Korhonen, P.K.; Young, N.D. Understanding Haemonchus contortus Better Through Genomics and Transcriptomics. Adv. Parasitol. 2016, 93, 519–567. [Google Scholar] [CrossRef]
- Hagel, I.; Giusti, T. Ascaris lumbricoides: An overview of therapeutic targets. Infect. Disord. Drug Targets 2010, 10, 349–367. [Google Scholar] [CrossRef][Green Version]
- Urquhart, G.M.; Jarrett, W.F.; Jennings, F.W.; McIntyre, W.I.; Mulligan, W.; Sharp, N.C. Immunity to Haemonchus contortus infection. Failure of x-irradiated larvae to immunize young lambs. Am. J. Vet. Res. 1966, 27, 1641–1643. [Google Scholar]
- Tavernor, A.S.; Smith, T.S.; Langford, C.F.; Graham, M.; Munn, E.A. Immune response of Clun Forest sheep to vaccination with membrane glycoproteins from Haemonchus contortus. Parasite Immunol. 1992, 14, 671–675. [Google Scholar] [CrossRef]
- Ashman, K.; Mather, J.; Wiltshire, C.; Jacobs, H.J.; Meeusen, E. Isolation of a larval surface glycoprotein from Haemonchus contortus and its possible role in evading host immunity. Molecular and Biochemical Parasitology 1995, 70, 175–179. [Google Scholar] [CrossRef]
- Wang, T.; Ma, G.; Ang, C.-S.; Korhonen, P.K.; Xu, R.; Nie, S.; Koehler, A.V.; Simpson, R.J.; Greening, D.W.; Reid, G.E.; et al. Somatic proteome of Haemonchus contortus. Int. J. Parasitol. 2019, 49, 311–320. [Google Scholar] [CrossRef]
- Wang, T.; Ma, G.; Ang, C.-S.; Korhonen, P.K.; Koehler, A.V.; Young, N.D.; Nie, S.; Williamson, N.A.; Gasser, R.B. High throughput LC-MS/MS-based proteomic analysis of excretory-secretory products from short-term in vitro culture of Haemonchus contortus. J. Proteomics 2019, 204, 103375. [Google Scholar] [CrossRef]
- Newton, S.E.; Munn, E.A. The development of vaccines against gastrointestinal nematode parasites, particularly Haemonchus contortus. Parasitol. Today 1999, 15, 116–122. [Google Scholar] [CrossRef]
- Gupta, R.; Brunak, S. Prediction of glycosylation across the human proteome and the correlation to protein function. Pac. Symp. Biocomput. 2002, 310–322. [Google Scholar]
- Smith, T.S.; Munn, E.A.; Graham, M.; Tavernor, A.S.; Greenwood, C.A. Purification and evaluation of the integral membrane protein H11 as a protective antigen against Haemonchus contortus. Int. J. Parasitol. 1993, 23, 271–280. [Google Scholar] [CrossRef]
- Jacobs, H.J.; Wiltshire, C.; Ashman, K.; Meeusen, E.N. Vaccination against the gastrointestinal nematode, Haemonchus contortus, using a purified larval surface antigen. Vaccine 1999, 17, 362–368. [Google Scholar] [CrossRef]
- Smith, W.D.; Smith, S.K.; Murray, J.M. Protection studies with integral membrane fractions of Haemonchus contortus. Parasite Immunol. 1994, 16, 231–241. [Google Scholar] [CrossRef]
- Knox, D.P.; Smith, S.K.; Smith, W.D. Immunization with an affinity purified protein extract from the adult parasite protects lambs against infection with Haemonchus contortus. Parasite Immunol. 1999, 21, 201–210. [Google Scholar] [CrossRef]
- de Matos, A.F.I.M.; Nobre, C.O.R.; Monteiro, J.P.; Bevilaqua, C.M.L.; Smith, W.D.; Teixeira, M. Attempt to control Haemonchus contortus in dairy goats with Barbervax ®, a vaccine derived from the nematode gut membrane glycoproteins. Small Rumin. Res. 2017, 151, 1–4. [Google Scholar] [CrossRef]
- Fawzi, E.M.; González-Sánchez, M.E.; Corral, M.J.; Cuquerella, M.; Alunda, J.M. Vaccination of lambs against Haemonchus contortus infection with a somatic protein (Hc23) from adult helminths. Int. J. Parasitol. 2014, 44, 429–436. [Google Scholar] [CrossRef]
- Vervelde, L.; Van Leeuwen, M.A.W.; Kruidenier, M.; Kooyman, F.N.J.; Huntley, J.F.; van Die, I.; Cornelissen, A.W.C.A. Protection studies with recombinant excretory/secretory proteins of Haemonchus contortus. Parasite Immunol. 2002, 24, 189–201. [Google Scholar] [CrossRef] [PubMed]
- Cachat, E.; Newlands, G.F.J.; Ekoja, S.E.; McAllister, H.; Smith, W.D. Attempts to immunize sheep against Haemonchus contortus using a cocktail of recombinant proteases derived from the protective antigen, H-gal-GP. Parasite Immunol. 2010, 32, 414–419. [Google Scholar] [CrossRef]
- García-Coiradas, L.; Angulo-Cubillán, F.; Valladares, B.; Martínez, E.; de La Fuente, C.; Alunda, J.M.; Cuquerella, M. Immunization against Lamb Haemonchosis with a Recombinant Somatic Antigen of Haemonchus contortus (rHcp26/23). Vet. Med. Int. 2010, 2010, 852146. [Google Scholar] [CrossRef] [PubMed]
- Fawzi, E.M.; González-Sánchez, M.E.; Corral, M.J.; Alunda, J.M.; Cuquerella, M. Vaccination of lambs with the recombinant protein rHc23 elicits significant protection against Haemonchus contortus challenge. Vet. Parasitol. 2015, 211, 54–59. [Google Scholar] [CrossRef]
- Reszka, N.; Rijsewijk, F.A.M.; Zelnik, V.; Moskwa, B.; Bieńkowska-Szewczyk, K. Haemonchus contortus: Characterization of the baculovirus expressed form of aminopeptidase H11. Exp. Parasitol. 2007, 117, 208–213. [Google Scholar] [CrossRef] [PubMed]
- Roberts, B.; Antonopoulos, A.; Haslam, S.M.; Dicker, A.J.; McNeilly, T.N.; Johnston, S.L.; Dell, A.; Knox, D.P.; Britton, C. Novel expression of Haemonchus contortus vaccine candidate aminopeptidase H11 using the free-living nematode Caenorhabditis elegans. Vet. Res. 2013, 44, 111. [Google Scholar] [CrossRef]
- Zhou, Q.-J.; Yang, Y.; Guo, X.-L.; Duan, L.-J.; Chen, X.-Q.; Yan, B.-L.; Zhang, H.-L.; Du, A.-F. Expression of Caenorhabditis elegans-expressed Trans-HPS, partial aminopeptidase H11 from Haemonchus contortus. Exp. Parasitol. 2014, 145, 87–98. [Google Scholar] [CrossRef]
- Lu, M.; Tian, X.; Zhang, Y.; Aimulajiang, K.; Wang, W.; Ehsan, M.; Li, C.; Yan, R.; Xu, L.; Song, X.; et al. Unveiling the immunomodulatory properties of Haemonchus contortus adhesion regulating molecule 1 interacting with goat T cells. Parasit. Vectors 2020, 13, 424. [Google Scholar] [CrossRef] [PubMed]
- Lu, M.; Tian, X.; Zhang, Y.; Wang, W.; Tian, A.-L.; Aimulajiang, K.; Liu, L.; Li, C.; Yan, R.; Xu, L.; et al. Protection studies of an excretory-secretory protein HcABHD against Haemonchus contortus infection. Vet. Res. 2021, 52, 3. [Google Scholar] [CrossRef]
- Sun, W.; Song, X.; Yan, R.; Xu, L.; Li, X. Vaccination of goats with a glutathione peroxidase DNA vaccine induced partial protection against Haemonchus contortus infection. Vet. Parasitol. 2011, 182, 239–247. [Google Scholar] [CrossRef]
- Zhao, G.; Yan, R.; Muleke, C.I.; Sun, Y.; Xu, L.; Li, X. Vaccination of goats with DNA vaccines encoding H11 and IL-2 induces partial protection against Haemonchus contortus infection. Vet. J. 2012, 191, 94–100. [Google Scholar] [CrossRef]
- Han, K.; Xu, L.; Yan, R.; Song, X.; Li, X. Vaccination of goats with glyceraldehyde-3-phosphate dehydrogenase DNA vaccine induced partial protection against Haemonchus contortus. Vet. Immunol. Immunopathol. 2012, 149, 177–185. [Google Scholar] [CrossRef]
- Yan, R.; Sun, W.; Song, X.; Xu, L.; Li, X. Vaccination of goats with DNA vaccine encoding Dim-1 induced partial protection against Haemonchus contortus: A preliminary experimental study. Res. Vet. Sci. 2013, 95, 189–199. [Google Scholar] [CrossRef] [PubMed]
- Yan, R.; Wang, J.; Xu, L.; Song, X.; Li, X. DNA vaccine encoding Haemonchus contortus actin induces partial protection in goats. Acta Parasitol. 2014, 59, 698–709. [Google Scholar] [CrossRef] [PubMed]
- Munn, E.A. Rational design of nematode vaccines: Hidden antigens. Int. J. Parasitol. 1997, 27, 359–366. [Google Scholar] [CrossRef]
- Bowles, V.M.; Brandon, M.R.; Meeusen, E. Characterization of local antibody responses to the gastrointestinal parasite Haemonchus contortus. Immunology 1995, 84, 669–674. [Google Scholar] [PubMed]
- Domínguez-Toraño, I.A.; Cuquerella, M.; Gómez-Muñoz, M.; Mendez, S.; Fernández-Pérez, F.J.; Alunda, J.M. Vaccination of Manchego lambs against Haemonchus contortus with a somatic fraction (p26/23) of adult parasites. Parasite Immunol. 2000, 22, 131–138. [Google Scholar] [CrossRef]
- García-Coiradas, L.; Angulo-Cubillán, F.; Méndez, S.; Larraga, V.; de La Fuente, C.; Cuquerella, M.; Alunda, J.M. Isolation and immunolocalization of a putative protective antigen (p26/23) from adult Haemonchus contortus. Parasitol. Res. 2009, 104, 363–369. [Google Scholar] [CrossRef]
- Munn, E.A.; Smith, T.S.; Graham, M.; Greenwood, C.A.; Tavernor, A.S.; Coetzee, G. Vaccination of merino lambs against haemonchosis with membrane-associated proteins from the adult parasite. Parasitology 1993, 106 Pt 1, 63–66. [Google Scholar] [CrossRef]
- Smith, T.S.; Graham, M.; Munn, E.A.; Newton, S.E.; Knox, D.P.; Coadwell, W.J.; McMichael-Phillips, D.; Smith, H.; Smith, W.D.; Oliver, J.J. Cloning and characterization of a microsomal aminopeptidase from the intestine of the nematode Haemonchus contortus. Biochim. Biophys. Acta 1997, 1338, 295–306. [Google Scholar] [CrossRef]
- Smith, W.D.; Newlands, G.F.J.; Smith, S.K.; Pettit, D.; Skuce, P.J. Metalloendopeptidases from the intestinal brush border of Haemonchus contortus as protective antigens for sheep. Parasite Immunol. 2003, 25, 313–323. [Google Scholar] [CrossRef]
- Knox, D.P.; Redmond, D.L.; Newlands, G.F.; Skuce, P.J.; Pettit, D.; Smith, W.D. The nature and prospects for gut membrane proteins as vaccine candidates for Haemonchus contortus and other ruminant trichostrongyloids. Int. J. Parasitol. 2003, 33, 1129–1137. [Google Scholar] [CrossRef]
- Scarff, C.A.; Thompson, R.F.; Newlands, G.F.J.; Jamson, A.H.; Kennaway, C.; Da Silva, V.J.; Rabelo, E.M.; Song, C.-F.; Trinick, J.; Smith, W.D.; et al. Structure of the protective nematode protease complex H-gal-GP and its conservation across roundworm parasites. PLoS Pathog. 2020, 16, e1008465. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, M.; Matos, A.F.I.M.; Albuquerque, F.H.M.A.; Bassetto, C.C.; Smith, W.D.; Monteiro, J.P. Strategic vaccination of hair sheep against Haemonchus contortus. Parasitol. Res. 2019, 118, 2383–2388. [Google Scholar] [CrossRef]
- Bassetto, C.C.; Picharillo, M.É.; Newlands, G.F.J.; Smith, W.D.; Fernandes, S.; Siqueira, E.R.; Amarante, A.F.T. Attempts to vaccinate ewes and their lambs against natural infection with Haemonchus contortus in a tropical environment. Int. J. Parasitol. 2014, 44, 1049–1054. [Google Scholar] [CrossRef]
- VanHoy, G.; Carman, M.; Habing, G.; Lakritz, J.; Hinds, C.A.; Niehaus, A.; Kaplan, R.M.; Marsh, A.E. Safety and serologic response to a Haemonchus contortus vaccine in alpacas. Vet. Parasitol. 2018, 252, 180–186. [Google Scholar] [CrossRef] [PubMed]
- Saylor, K.; Gillam, F.; Lohneis, T.; Zhang, C. Designs of Antigen Structure and Composition for Improved Protein-Based Vaccine Efficacy. Front. Immunol. 2020, 11, 283. [Google Scholar] [CrossRef]
- de La Fuente, J.; Almazán, C.; Canales, M.; Pérez de la Lastra, J.M.; Kocan, K.M.; Willadsen, P. A ten-year review of commercial vaccine performance for control of tick infestations on cattle. Anim. Health Res. Rev. 2007, 8, 23–28. [Google Scholar] [CrossRef]
- Nisbet, A.J.; McNeilly, T.N.; Wildblood, L.A.; Morrison, A.A.; Bartley, D.J.; Bartley, Y.; Longhi, C.; McKendrick, I.J.; Palarea-Albaladejo, J.; Matthews, J.B. Successful immunization against a parasitic nematode by vaccination with recombinant proteins. Vaccine 2013, 31, 4017–4023. [Google Scholar] [CrossRef]
- Nisbet, A.J.; McNeilly, T.N.; Price, D.R.G.; Oliver, E.M.; Bartley, Y.; Mitchell, M.; Palarea-Albaladejo, J.; Matthews, J.B. The rational simplification of a recombinant cocktail vaccine to control the parasitic nematode Teladorsagia circumcincta. Int. J. Parasitol. 2019, 49, 257–265. [Google Scholar] [CrossRef]
- Schallig, H.D.; van Leeuwen, M.A.; Cornelissen, A.W. Protective immunity induced by vaccination with two Haemonchus contortus excretory secretory proteins in sheep. Parasite Immunol. 1997, 19, 447–453. [Google Scholar] [CrossRef]
- Newlands, G.F.J.; Skuce, P.J.; Nisbet, A.J.; Redmond, D.L.; Smith, S.K.; Pettit, D.; Smith, W.D. Molecular characterization of a family of metalloendopeptidases from the intestinal brush border of Haemonchus contortus. Parasitology 2006, 133, 357–368. [Google Scholar] [CrossRef]
- Fleurkens, S.M. Carbohydrate-based vaccines against the sheep parasite Haemonchus contortus; ETH Zurich.
- Haslam, S.M.; Coles, G.C.; Munn, E.A.; Smith, T.S.; Smith, H.F.; Morris, H.R.; Dell, A. Haemonchus contortus glycoproteins contain N-linked oligosaccharides with novel highly fucosylated core structures. J. Biol. Chem. 1996, 271, 30561–30570. [Google Scholar] [CrossRef]
- Lu, M.; Tian, X.; Tian, A.-L.; Li, C.; Yan, R.; Xu, L.; Song, X.; Li, X. A Novel α/β Hydrolase Domain Protein Derived From Haemonchus contortus Acts at the Parasite-Host Interface. Front. Immunol. 2020, 11, 1388. [Google Scholar] [CrossRef]
- Lu, M.; Tian, X.; Wang, W.; Zhang, Y.; Aimulajiang, K.; Tian, A.-L.; Li, C.; Yan, R.; Xu, L.; Song, X.; et al. The excretory-secretory antigen HcADRM1 to generate protective immunity against Haemonchus contortus. Parasitology 2021, 148, 1497–1508. [Google Scholar] [CrossRef]
- Robinson, H.L.; Lu, S.; Feltquate, D.M.; Torres, C.T.; Richmond, J.; Boyle, C.M.; Morin, M.J.; Santoro, J.C.; Webster, R.G.; Montefiori, D.; et al. DNA vaccines. AIDS Res. Hum. Retroviruses 1996, 12, 455–457. [Google Scholar] [CrossRef]
- Tang, D.C.; DeVit, M.; Johnston, S.A. Genetic immunization is a simple method for eliciting an immune response. Nature 1992, 356, 152–154. [Google Scholar] [CrossRef]
- Egan, M.A.; Israel, Z.R. The use of cytokines and chemokines as genetic adjuvants for plasmid DNA vaccines. Clin. Appl. Immuno. Rev. 2002, 2, 255–287. [Google Scholar] [CrossRef]
- Scheerlinck, J.-P.Y.; Karlis, J.; Tjelle, T.E.; Presidente, P.J.A.; Mathiesen, I.; Newton, S.E. In vivo electroporation improves immune responses to DNA vaccination in sheep. Vaccine 2004, 22, 1820–1825. [Google Scholar] [CrossRef]
- Henkle-Dührsen, K.; Kampkötter, A. Antioxidant enzyme families in parasitic nematodes. Mol. Biochem. Parasitol. 2001, 114, 129–142. [Google Scholar] [CrossRef]
- Yu, R.; Ono, S. Dual roles of tropomyosin as an F-actin stabilizer and a regulator of muscle contraction in Caenorhabditis elegans body wall muscle. Cell Motil. Cytoskeleton 2006, 63, 659–672. [Google Scholar] [CrossRef]
- Vervelde, L.; Kooyman, F.N.; van Leeuwen, M.A.; Schallig, H.D.; MacKellar, A.; Huntley, J.F.; Cornelissen, A.W. Age-related protective immunity after vaccination with Haemonchus contortus excretory/secretory proteins. Parasite Immunol. 2001, 23, 419–426. [Google Scholar] [CrossRef]
- Vervelde, L.; Bakker, N.; Kooyman, F.N.; Cornelissen, A.W.; Bank, C.M.; Nyame, A.K.; Cummings, R.D.; van Die, I. Vaccination-induced protection of lambs against the parasitic nematode Haemonchus contortus correlates with high IgG antibody responses to the LDNF glycan antigen. Glycobiology 2003, 13, 795–804. [Google Scholar] [CrossRef]
- Hoste, H.; Sotiraki, S.; Landau, S.Y.; Jackson, F.; Beveridge, I. Goat-nematode interactions: Think differently. Trends Parasitol. 2010, 26, 376–381. [Google Scholar] [CrossRef]
- Albuquerque, A.C.A.; Bassetto, C.C.; Almeida, F.A.; Hildersley, K.A.; McNeilly, T.N.; Britton, C.; Amarante, A.F.T. Differences in immune responses to Haemonchus contortus infection in the susceptible Ile de France and the resistant Santa Ines sheep under different anthelmintic treatments regimens. Vet. Res. 2019, 50, 104. [Google Scholar] [CrossRef]
- Shakya, K.P.; Miller, J.E.; Horohov, D.W. A Th2 type of immune response is associated with increased resistance to Haemonchus contortus in naturally infected Gulf Coast Native lambs. Vet. Parasitol. 2009, 163, 57–66. [Google Scholar] [CrossRef]
- González-Sánchez, M.E.; Ndombasi-Bokuy, M.; Cuquerella, M.; Alunda, J.M. Immunization with recombinant rHc23 partially protects lambs against trickle infections by Haemonchus contortus. BMC Vet. Res. 2019, 15, 333. [Google Scholar] [CrossRef]
- González-Sánchez, M.E.; Cuquerella, M.; Alunda, J.M. Vaccination of lambs against Haemonchus contortus with the recombinant rHc23. Effect of adjuvant and antigen dose. PLoS ONE 2018, 13, e0193118. [Google Scholar] [CrossRef]
- Smith, W.D. A Commercial Vaccine for Barber’s Pole Worm—Further Development; Meat and Livestock Australia: North Sydney, Australia, 2014; ISBN 9781925045826. [Google Scholar]
- Gill, H.S.; Gray, G.D.; Watson, D.L.; Husband, A.J. Isotype-specific antibody responses to Haemonchus contortus in genetically resistant sheep. Parasite Immunol. 1993, 15, 61–67. [Google Scholar] [CrossRef]
- Schallig, H.D. Immunological responses of sheep to Haemonchus contortus. Parasitology 2000, 120, S63–S72. [Google Scholar] [CrossRef]
- Lacroux, C.; Nguyen, T.H.C.; Andreoletti, O.; Prevot, F.; Grisez, C.; Bergeaud, J.-P.; Gruner, L.; Brunel, J.-C.; Francois, D.; Dorchies, P.; et al. Haemonchus contortus (Nematoda: Trichostrongylidae) infection in lambs elicits an unequivocal Th2 immune response. Vet. Res. 2006, 37, 607–622. [Google Scholar] [CrossRef]
- Salgado, J.A.; Dos Santos, S.K.; Biz, J.d.F.F.; de Carvalho, M.B.; Rigo, F.; Beirão, B.C.B.; Amarante, A.F.T.d.; Costa, L.B.; Sotomaior, C.S. Salivary, serum, and abomasal mucus IgA as an immune correlate of protection against Haemonchus contortus infection in naturally infected lambs. Res. Vet. Sci. 2022, 144, 82–91. [Google Scholar] [CrossRef]
- Escribano, C.; Saravia, A.; Costa, M.; Castells, D.; Ciappesoni, G.; Riet-Correa, F.; Freire, T. Resistance to Haemonchus contortus in Corriedale sheep is associated to high parasite-specific IgA titer and a systemic Th2 immune response. Sci. Rep. 2019, 9, 19579. [Google Scholar] [CrossRef]
- Kooyman, F.N.; van Kooten, P.J.; Huntley, J.F.; MacKellar, A.; Cornelissen, A.W.; Schallig, H.D. Production of a monoclonal antibody specific for ovine immunoglobulin E and its application to monitor serum IgE responses to Haemonchus contortus infection. Parasitology 1997, 114 Pt 4, 395–406. [Google Scholar] [CrossRef]
- Kooyman, F.N.; Schallig, H.D.; van Leeuwen, M.A.; MacKellar, A.; Huntley, J.F.; Cornelissen, A.W.; Vervelde, L. Protection in lambs vaccinated with Haemonchus contortus antigens is age related, and correlates with IgE rather than IgG1 antibody. Parasite Immunol. 2000, 22, 13–20. [Google Scholar] [CrossRef] [PubMed]
- van Die, I.; Gomord, V.; Kooyman, F.N.; van den Berg, T.K.; Cummings, R.D.; Vervelde, L. Core α1→3-fucose is a common modification of N-glycans in parasitic helminths and constitutes an important epitope for IgE from Haemonchus contortus infected sheep. FEBS Letters 1999, 463, 189–193. [Google Scholar] [CrossRef]
- Balic, A.; Cunningham, C.P.; Meeusen, E.N.T. Eosinophil interactions with Haemonchus contortus larvae in the ovine gastrointestinal tract. Parasite Immunol. 2006, 28, 107–115. [Google Scholar] [CrossRef]
- Laing, R.; Kikuchi, T.; Martinelli, A.; Tsai, I.J.; Beech, R.N.; Redman, E.; Holroyd, N.; Bartley, D.J.; Beasley, H.; Britton, C.; et al. The genome and transcriptome of Haemonchus contortus, a key model parasite for drug and vaccine discovery. Genome Biol. 2013, 14, R88. [Google Scholar] [CrossRef]
- Wang, C.; Gao, W.; Yan, S.; Zhu, X.-Q.; Suo, X.; Liu, X.; Gupta, N.; Hu, M. N-glycome and N-glycoproteome of a hematophagous parasitic nematode Haemonchus. Comput. Struct. Biotechnol. J. 2021, 19, 2486–2496. [Google Scholar] [CrossRef]
- van der Kaaij, A.; van Noort, K.; Nibbering, P.; Wilbers, R.H.P.; Schots, A. Glyco-Engineering Plants to Produce Helminth Glycoproteins as Prospective Biopharmaceuticals: Recent Advances, Challenges and Future Prospects. Front. Plant Sci. 2022, 13, 882835. [Google Scholar] [CrossRef]
- Wilbers, R.H.P.; Westerhof, L.B.; van Noort, K.; Obieglo, K.; Driessen, N.N.; Everts, B.; Gringhuis, S.I.; Schramm, G.; Goverse, A.; Smant, G.; et al. Production and glyco-engineering of immunomodulatory helminth glycoproteins in plants. Sci. Rep. 2017, 7, 45910. [Google Scholar] [CrossRef]
- Bangert, C.; Brunner, P.M.; Stingl, G. Immune functions of the skin. Clin. Dermatol. 2011, 29, 360–376. [Google Scholar] [CrossRef]
- Kenney, R.T.; Frech, S.A.; Muenz, L.R.; Villar, C.P.; Glenn, G.M. Dose sparing with intradermal injection of influenza vaccine. N. Engl. J. Med. 2004, 351, 2295–2301. [Google Scholar] [CrossRef] [PubMed]
- Benisek, Z.; Suli, J.; Svrcek, S.; Ondrejkova, A.; Mojzisova, J.; Ondrejka, R. Intradermal anti-rabies immunization - Possibilities of needleless rabies vaccine administration. Bull. Vet. Inst. Pul. 2006, 50, 137–142. [Google Scholar]
- Wong, S.-S.; Webby, R.J. An mRNA vaccine for influenza. Nat. Biotechnol. 2012, 30, 1202–1204. [Google Scholar] [CrossRef] [PubMed]
- Jackson, L.A.; Anderson, E.J.; Rouphael, N.G.; Roberts, P.C.; Makhene, M.; Coler, R.N.; McCullough, M.P.; Chappell, J.D.; Denison, M.R.; Stevens, L.J.; et al. An mRNA Vaccine against SARS-CoV-2—Preliminary Report. N. Engl. J. Med. 2020, 383, 1920–1931. [Google Scholar] [CrossRef] [PubMed]
| Antigens | Molecular Properties | |||||
|---|---|---|---|---|---|---|
| Type (H/N) | Accession Number | N-Glycosylation | Expression Host & Solubility | Efficacy in Challenge Studies * | References | |
| Native antigens | ||||||
| native H11 | H | NA | yes | adult worms | high | [55] |
| Hc-sL3 | N | NA | yes | third-stage larvae | moderate or low | [56] |
| H-gal-GP | H | NA | yes | adult worms | moderate or high | [57] |
| TSBP | H | NA | yes | adult worms | moderate | [58] |
| Barbervax® or Wirevax | H | NA | yes | adult worms | moderate or high | [59] |
| Hc23 | N (somatic) | GenBank: CDJ92660.1 | no | adult worms | high | [60] |
| Recombinant parasite proteins | ||||||
| ES15 | N (E/S) | UniProt: O18518 | no | E. coli, refolded | low | [61] |
| ES24 | N (E/S) | UniProt: O18519 | yes | E. coli, refolded | low | |
| MEP1 | H | UniProt: O45131 | yes | Sf9 insect cell, soluble | NS | [62] |
| MEP3 | H | UniProt: O76751 | yes | Sf9 insect cell, soluble | NS | |
| MEP4 | H | UniProt: Q9Y1I4 | yes | Sf9 insect cell, soluble | NS | |
| PEP1 | H | UniProt: Q25037 | yes | E. coli, refolded | NS | |
| Hcp26/23 | N (somatic) | GenBank: CDJ92660.1 | no | E. coli M15, refolded | NS | [63] |
| Hc23 | N (somatic) | GenBank: CDJ92660.1 | no | E. coli BL21(DE3), NA | high | [64] |
| H11-1 | H | GenBank: CAB57357.1 | yes | Sf9 insect cell, soluble | NS | [17] |
| H11-2 | H | GenBank: CAB57358.1 | yes | Sf9 insect cell, soluble | NS | |
| H11 | H | UniProt: Q10737.2 | yes | Sf9 insect cell, soluble | NS | |
| H11-1 | H | GenBank: CAB57357.1 | yes | Sf21 insect cell, soluble | ND | [65] |
| H11-1 | H | GenBank: CAB57357.1 | yes | C. elegans, soluble | NS | [66] |
| H11-4 | H | UniProt: Q967C6 | yes | C. elegans, soluble | NS | |
| H11-5 | H | UniProt: V5K5H8 | yes | C. elegans, soluble | NS | |
| H11 (partial, 1-570 aa) | H | UniProt: Q10737.2 | yes | C. elegans, soluble | low | [67] |
| HcADRM1 | N (E/S) | UniProt: W6NKS2 | no | E. coli BL21 (DE3), NA | moderate or low | [68] |
| HcABHD | N (E/S) | GenBank: CDJ88804.1 | no | E. coli BL21 (DE3), NA | moderate | [69] |
| DNA Vaccine targets | ||||||
| HC29 | H | UniProt: D0F095 | yes | goats | low | [70] |
| H11 ** | H | GenBank: Q10737.2 | yes | goats | low | [71] |
| GAPDH | N (somatic) | UniProt: D9IL10 | yes | goats | low | [72] |
| Dim-1 | N (somatic) | GenBank: ADZ24723.1 | yes | goats | low | [73] |
| Actin | N (somatic) | GenBank: CDJ80138.1 or CDJ93106.1 | yes | goats | low | [74] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Adduci, I.; Sajovitz, F.; Hinney, B.; Lichtmannsperger, K.; Joachim, A.; Wittek, T.; Yan, S. Haemonchosis in Sheep and Goats, Control Strategies and Development of Vaccines against Haemonchus contortus. Animals 2022, 12, 2339. https://doi.org/10.3390/ani12182339
Adduci I, Sajovitz F, Hinney B, Lichtmannsperger K, Joachim A, Wittek T, Yan S. Haemonchosis in Sheep and Goats, Control Strategies and Development of Vaccines against Haemonchus contortus. Animals. 2022; 12(18):2339. https://doi.org/10.3390/ani12182339
Chicago/Turabian StyleAdduci, Isabella, Floriana Sajovitz, Barbara Hinney, Katharina Lichtmannsperger, Anja Joachim, Thomas Wittek, and Shi Yan. 2022. "Haemonchosis in Sheep and Goats, Control Strategies and Development of Vaccines against Haemonchus contortus" Animals 12, no. 18: 2339. https://doi.org/10.3390/ani12182339
APA StyleAdduci, I., Sajovitz, F., Hinney, B., Lichtmannsperger, K., Joachim, A., Wittek, T., & Yan, S. (2022). Haemonchosis in Sheep and Goats, Control Strategies and Development of Vaccines against Haemonchus contortus. Animals, 12(18), 2339. https://doi.org/10.3390/ani12182339







