Feed Intake Parameters of Horses Fed Soaked or Steamed Hay and Hygienic Quality of Hay Stored following Treatment
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
3. Results
3.1. Proximate Nutrients
3.2. Feed intake Parameters
3.3. Microbial Counts
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Ellis, A.D. Biological basis of behaviour in relation to nutrient and feed intake in horses. In The Impact of Nutrition on the Health and Welfare of Horses; Ellis, A.D., Longland, A.C., Coenen, M., Miraglia, N., Eds.; Wageningen Academic Publishers: Wageningen, The Netherlands; EAAP Publication: Rome, Italy, 2010; Volume 127, pp. 53–74. [Google Scholar]
- Bonin, S.J.; Clayton, H.M.; Lanovaz, J.L.; Johnston, T. Comparison of mandibular motion in horses chewing hay and pellets. Equine Vet. J. 2007, 39, 258–262. [Google Scholar] [CrossRef]
- Bochnia, M.; Boesel, M.; Bahrenthien, L.; Wensch-Dorendorf, M.; Zeyner, A. Feed intake patterns of sport ponies and warmblood horses following iso-energetic intake of pelleted fibre-rich mixed feed, muesli feed and semicrushed oat grains. J. Anim. Physiol. Anim. Nutr. 2017, 101 (Suppl. 1), 37–42. [Google Scholar] [CrossRef]
- Janis, C.M.; Constable, E.C.; Houpt, K.A.; Streich, W.J.; Clauss, M. Comparative ingestive mastication in domestic horses and cattle: A pilot investigation. J. Anim. Physiol. Anim. Nutr. 2010, 94, e402–e409. [Google Scholar] [CrossRef] [PubMed]
- Bruessow, N.A.S. Effekte Verschiedener Futtermittel und-Bearbeitungsformen auf die Futteraufnahmedauer, Kaufrequenz und die Kauintensität beim Pferd; Stiftung Tierärztliche Hochschule Hannover: Hannover, Germany, 2006. [Google Scholar]
- Bochnia, M.; Goetz, F.; Wensch-Dorendorf, M.; Koelln, M.; Zeyner, A. Chewing patterns in horses during the intake of variable quantities of two pelleted compound feeds differing in their physical characteristics only. Res. Vet. Sci. 2019, 125, 189–194. [Google Scholar] [CrossRef] [PubMed]
- Moore-Colyer, M.J.S.; Payne, V. Palatability and ingestion behaviour of six polo ponies offered a choice of dry, soaked and steamed hay for 1 h on three separate occassions. Adv. Ani. Biosci 2012, 3, 127. [Google Scholar]
- Brown, T.; Tracey, S.; Gowers, I. An investigation to determine palatability of steamed hay, dry hay and haylage. In Proceedings of the British Society of Animal Science Conference, Nottingham, UK, 30 March–1 April 2013. [Google Scholar]
- Earing, J.E.; Hathaway, M.R.; Sheaffer, C.C.; Hetchler, B.P.; Jacobson, L.D.; Paulson, J.C.; Martinson, K.L. Effect of hay steaming on forage nutritive values and dry matter intake by horses. J. Anim. Sci. 2013, 91, 5813–5820. [Google Scholar] [CrossRef]
- Mueller, C.E.; Udén, P. Preference of horses for grass conserved as hay, haylage or silage. Anim. Feed Sci. Technol. 2007, 132, 66–78. [Google Scholar] [CrossRef]
- Argo, C.M.; Dugdale, A.H.A.; McGowan, C.M. Considerations for the use of restricted, soaked grass hay diets to promote weight loss in the management of equine metabolic syndrome and obesity. Vet. J. 2015, 206, 170–177. [Google Scholar] [CrossRef] [PubMed]
- Blackman, M.; Moore-Colyer, M.J. Hay for horses: The effects of three different wetting treatments on dust and nutrient content. Anim. Sci. 1998, 66, 745–750. [Google Scholar] [CrossRef]
- Clements, J.M.; Pirie, R.S. Respirable dust concentrations in equine stables. Part 2. The benefits of soaking hay and optimising the environment in a neighbouring stable. Res. Vet. Sci. 2007, 83, 263–268. [Google Scholar] [CrossRef] [PubMed]
- James, R.; Moore-Colyer, M.J. The effect of steam treatment on the total viable count, mould and yeast numbers in hay using the Haygain hay steamer. In The Impact of Nutrition on the Health and Welfare of Horses; Ellis, A.D., Longland, A.C., Coenen, M., Miraglia, N., Eds.; Wageningen Academic Publishers: Wageningen, The Netherlands, 2010; Volume 128, pp. 128–132. [Google Scholar]
- Wichert, B.; Nater, S.; Wittenbrink, M.M.; Wolf, P.; Meyer, K.; Wanner, M. Judgement of hygienic quality of roughage in horse stables in Switzerland. J. Anim. Physiol. Anim. Nutr. 2008, 92, 432–437. [Google Scholar] [CrossRef]
- Moore-Colyer, M.J.; Fillery, B.G. The effect of three different treatments on the respirable particle content, total vialbe count and mould concentrations in hay for horses. In Forage and Grazing in Horse’s Nutrition; Saastamoinen, M., Fradinho, M.J., Santos, A.S., Miraglia, N., Eds.; Wageningen Academic Publishers: Wageningen, The Netherlands, 2012; Volume 132, pp. 101–106. [Google Scholar]
- Moore-Colyer, M.J.; Taylor, J.L.; James, R. The Effect of Steaming and Soaking on the Respirable Particle, Bacteria, Mould, and Nutrient Content in Hay for Horses. J. Equ. Vet. Sci. 2016, 39, 62–68. [Google Scholar] [CrossRef]
- Blundell, E.L.; Adjei, L.J.; Brigden, C.V. The effect of steaming and soaking treatments on respirable dust content of hay and the potential environmental polluting impact on waste water produced. In Forage and Grazing in Horse´s Nutrition; Saastamoinen, M., Fradinho, M.J., Santos, A.S., Miraglia, N., Eds.; Wageningen Academic Publishers: Wageningen, The Netherlands, 2012; Volume 132, pp. 125–128. [Google Scholar]
- Moore-Colyer, M.J.S.; Lumbis, K.; Longland, A.; Harris, P. The effect of five different wetting treatments on the nutrient content and microbial concentration in hay for horses. PLoS ONE 2014, 9, e114079. [Google Scholar] [CrossRef]
- Mueller, C.E.; Nostell, K.; Bröjer, J. Methods for reduction of water-soluble carbohydrate content in grass forages for horses. Liv. Sci. 2016, 186, 46–52. [Google Scholar] [CrossRef]
- Martinson, K.; Jung, H.; Hathaway, M.; Sheaffer, C. The Effect of Soaking Hay on Dry Matter Loss and Fructan Removal. J. Equ. Vet. Sci. 2011, 31, 278–279. [Google Scholar] [CrossRef]
- Martinson, K.L.; Hathaway, M.; Jung, H.; Sheaffer, C. The Effect of Soaking on Protein and Mineral Loss in Orchardgrass and Alfalfa Hay. J. Equ. Vet. Sci. 2012, 32, 776–782. [Google Scholar] [CrossRef]
- Bochnia, M.; Pietsch, C.; Wensch-Dorendorf, M.; Greef, M.; Zeyner, A. Effect of Hay Soaking Duration on Metabolizable Energy, Total and Prececal Digestible Crude Protein and Amino Acids, Non-Starch Carbohydrates, Macronutrients and Trace Elements. J. Equ. Vet. Sci. 2021, 101, 103452. [Google Scholar] [CrossRef]
- Kienzle, E.; Schramme, S. Beurteilung des Ernährungszustandes mittels Body Condition Scores und Gewichtsschätzung beim adulten Warmblutpferd. Pferdeheilkunde 2004, 20, 517–524. [Google Scholar] [CrossRef]
- Gesellschaft für Ernährungsphysiologie. Empfehlungen zur Energie- und Nährstoffversorgung von Pferden. In Energie-und Nährstoffbedarf Landwirtschaftlicher Nutztiere; DLG-Verlag: Frankfurt, Germany, 2014; Volume 11. [Google Scholar]
- Association of German Agriculture and Research Institutes (VDLUFA, Publisher). The chemical control of feed (supplementary deliveries). In Handbook of the Agricultural Experimental and Research Methodology (VDLUFA-Methodenbuch); Association of German Agriculture and Research Institutes: Darmstadt, Germany, 2012; Volume III. [Google Scholar]
- Kienzle, E.; Zeyner, A. The development of a metabolizable energy system for horses. J. Anim. Physiol. Anim. Nutr. 2010, 94, e231–e240. [Google Scholar] [CrossRef] [PubMed]
- Zeyner, A.; Kirchhof, S.; Susenbeth, A.; Südekum, K.-H.; Kienzle, E. A new protein evaluation system for horse feed from literature data. J. Nutr. Sci. 2015, 4, e4. [Google Scholar] [CrossRef] [PubMed]
- Kuchler, M.; Zeyner, A.; Susenbeth, A.; Kienzle, E. The effect of crude protein content of the diet on renal energy losses in horses. J. Anim. Physiol. Anim. Nutr. 2020, 104, 1494–1500. [Google Scholar] [CrossRef]
- Pavis, N.; Chatterton, N.J.; Harrison, P.A.; Baumgartner, S.; Praznik, W.; Boucard, J.; Prud´homme, M.P. Structure of Fructans in Roots and Leaf tissues of Lolium perenne. New Phyt. 2001, 150, 83–95. [Google Scholar] [CrossRef]
- Meyer, H.; Ahlswede, R.H.J. Untersuchungen über Fressdauer, Kaufrequenz und Futterzerkleinerung beim Pferd. Deut Tierä Wochent 1975, 82, 54–58. [Google Scholar]
- Shingu, Y.; Kondo, S.; Hata, H.; Okubo, M. Digestibility and Number of Bites and Chews on Hay at Fixed Level in Hokkaido Native Horses and Light Half-bred Horses. JES 2001, 12, 145–147. [Google Scholar] [CrossRef]
- Brøkner, C.; Nørgaard, P.; Hansen, H.H. Effect of feed type and essential oil product on equine chewing activity. J. Anim. Physiol. Anim. Nutr. 2008, 92, 621–630. [Google Scholar] [CrossRef] [PubMed]
- Ellis, A.D.; Thomas, S.; Arkell, K.; Harris, P.A. Adding chopped straw to concentrate feed: The effect of inclusion rate and particle length on intake behaviour of horses. Pferdeheilkunde 2005, 21, 35–37. [Google Scholar] [CrossRef]
- Staszyck, C. Lehrbuch der Zahnheilkunde Beim Pferd; Schattauer: Stuttgart, Germany, 2011. [Google Scholar]
- Toussaint, B.; Excoffier, G.; Vignon, M.R. Effect of steam explosion treatment on the physico-chemical characteristics and enzymic hydrolysis of poplar cell wall components. Anim. Feed Sci. Technol. 1991, 32, 235–242. [Google Scholar] [CrossRef]
- Goelema, J.O.; Spreeuwenberg, M.; Hof, G.; van der Poel, A.; Tamminga, S. Effect of pressure toasting on the rumen degradability and intestinal digestibility of whole and broken peas, lupins and faba beans and a mixture of these feedstuffs. Anim. Feed Sci. Technol. 1998, 76, 35–50. [Google Scholar] [CrossRef]
- Goelema, J.; Smits, A.; Vaessen, L.; Wemmers, A. Effects of pressure toasting, expander treatment and pelleting on in vitro and in situ parameters of protein and starch in a mixture of broken peas, lupins and faba beans. Anim. Feed Sci. Technol. 1999, 78, 109–126. [Google Scholar] [CrossRef]
- Theurer, C.B.; Huber, J.T.; Delgado-Elorduy, A.; Wanderley, R. Invited Review: Summary of Steam-Flaking Corn or Sorghum Grain for Lactating Dairy Cows. J. Dair. Sci. 1999, 82, 1950–1959. [Google Scholar] [CrossRef]
- Aufrère, J.; Graviou, D.; Melcion, J.P.; Demarquilly, C. Degradation in the rumen of lupin (Lupinus albus L.) and pea (Pisum sativum L.) seed proteins Effect of heat treatment. Anim. Feed Sci. Technol. 2001, 92, 215–236. [Google Scholar] [CrossRef]
- Azarfar, A.; Tamminga, S.; Pellikaan, W.F.; van der Poel, A.F.B. In vitro gas production profiles and fermentation end-products in processed peas, lupins and faba beans. J. Sci. Food Agric. 2008, 88, 1997–2010. [Google Scholar] [CrossRef]
- van Soest, P.J.; Mason, V.C. The influence of the Maillard reaction upon the nutritive value of fibrous feeds. Anim. Feed Sci. Technol. 1991, 32, 45–53. [Google Scholar] [CrossRef]
- Hurrell, R.E.; Finot, R.A. Effect of food processing on protein digestibility and amino acid availability. In Digestibility and Amino Acid Availability in Cereals and Oilseeds; Finley, J.W., Hopkins, D.T., Eds.; American Association of Cereal Chemists: St. Paul, MN, USA, 1985; pp. 516–527. [Google Scholar]
- Yu, P.; Goelema, J.O.; Leury, B.J.; Tamminga, S.; Egan, A.R. An analysis of the nutritive value of heat processed legume seeds for animal production using the DVE/OEB model: A review. Anim. Feed Sci. Technol. 2002, 99, 141–176. [Google Scholar] [CrossRef]
- Hellwig, M.; Henle, T. Baking, ageing, diabetes: A short history of the Maillard reaction. Angew. Chem. Int. Ed. Engl. 2014, 53, 10316–10329. [Google Scholar] [CrossRef]
- Kuhnitzsch, C.; Hofmann, T.; Bachmann, M.; Martens, S.D.; Henle, T.; Zeyner, A.; Steinhöfel, O. Effect of ensiling and toasting of field pea grains on formation of Maillard polymers from lysine and arginine. Adv. Anim. Biosci. 2019, 10, 540. [Google Scholar]
- Harris, P.A.; Ellis, A.D.; Fradinho, M.J.; Jansson, A.; Julliand, V.; Luthersson, N.; Santos, A.S.; Vervuert, I. Review. Feeding conserved forage to horses: Recent advances and recommendations. Animal 2017, 11, 958–967. [Google Scholar] [CrossRef] [PubMed]
- Serrano, L.; Dodd, L.; Wood, B.; D’Antoni, J.; Mehlhorn, J. Time Matters. Effectiveness of Steaming Times on Forage for Animal Consumption. JAS 2015, 3, 219. [Google Scholar] [CrossRef]
- Kaya, G.; Sommerfeld-Stur, I.; Iben, C. Risk factors of colic in horses in Austria. J. Anim. Physiol. Anim. Nutr. 2009, 93, 339–349. [Google Scholar] [CrossRef]
- Longland, A.C.; Barfoot, C.; Harris, P.A. The Loss of Water-Soluble Carbohydrate and Soluble Protein from Nine Different Hays Soaked in Water for up to 16 Hours. J. Equine Vet. Sci. 2009, 29, 383–384. [Google Scholar] [CrossRef]

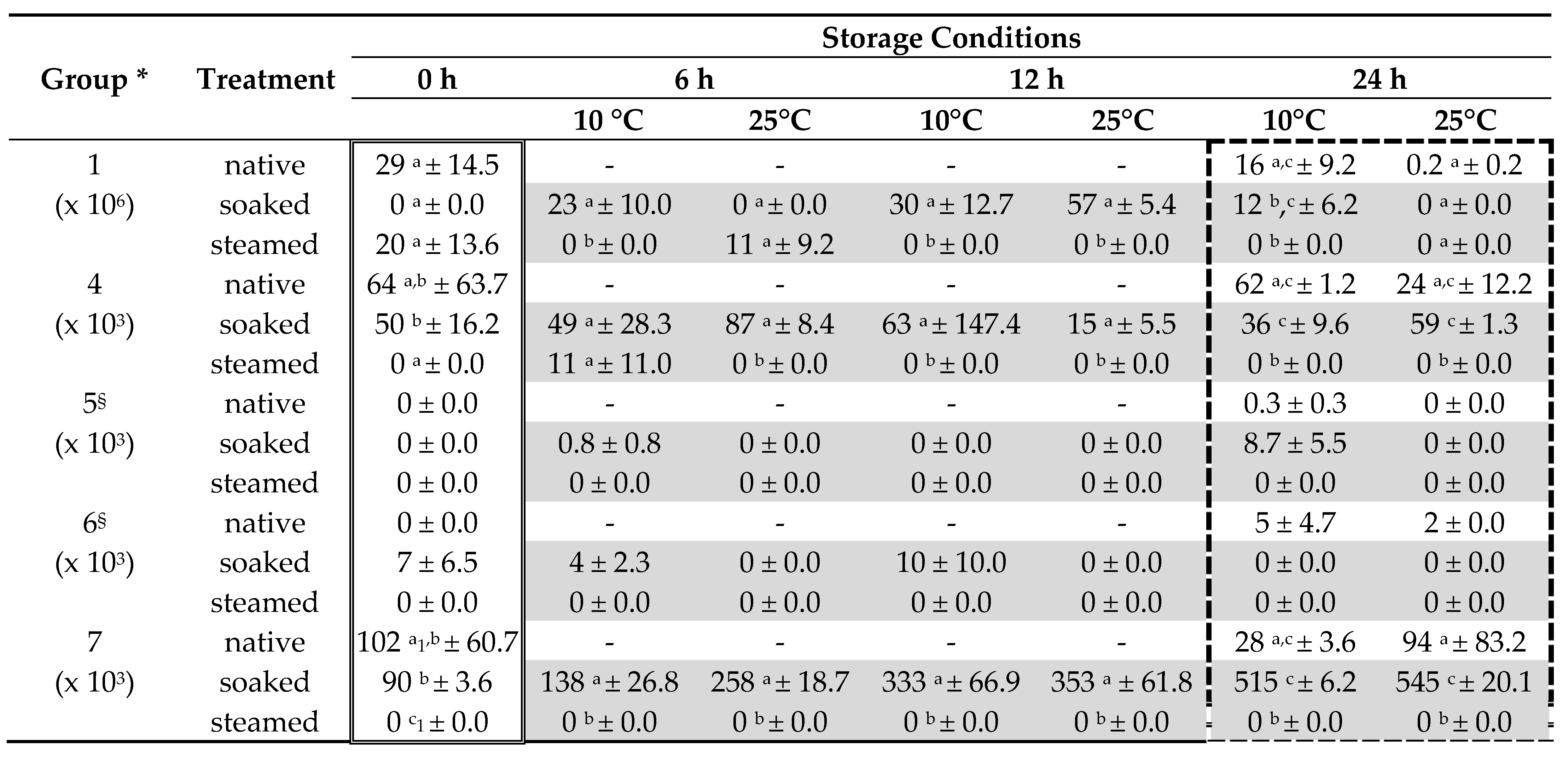
| Group | Indicator Species | Classification | Benchmark Native Hay (CFU/g) |
|---|---|---|---|
| 1 | yellow bacteria germs | typical microbial species | 30 × 106 |
| Pseudomonas spp. Enterobacteriaceae | |||
| other typical bacteria | |||
| 2 | Bacillus spp. Staphylococcus spp. | spoilage-indicating microbial species | 2 × 106 |
| Micrococcus spp. | |||
| 3 | Streptomycetes | spoilage-indicating microbial species | 0.15 × 106 |
| 4 | sooty mold fungi Verticillum spp. | typical microbial species | 200 × 103 |
| Acremonium spp. Fusarium spp. Aureobasidium spp. other typical fungi | |||
| 5 | Aspergillus spp. Penicillum spp. Scopulariopsis spp. Wallemia spp. | spoilage-indicating microbial species | 100 × 103 |
| other spoilage fungi | |||
| 6 | Mucorales | spoilage-indicating microbial species | 5 × 103 |
| 7 | all yeast species | typical or spoilage species | 150 × 103 |
| Treatment | Storage Condition | DM | CA | CP | pcdCP | pcd Lys/Met + Cys/Thre | AEE | CF | aNDFom | ADFom | ADL | ME | MEmod 1 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Time | °C | g/kg | g/kg DM | MJ/kg DM | ||||||||||
| native | 10 | 920 | 92 | 89 | 50 | 1.77/1.06/1.67 | 11 | 356 | 618 | 361 | 62 | 5.8 | 5.8 | |
| soaked | 0 h | 310 | 55 | 73 | 42 | 1.79/0.96/1.75 | 7 | 418 | 707 | 471 | 72 | 5.2 | 5.2 | |
| 6 h | 10 | 320 | 70 | 99 | 10 | 385 | 679 | 444 | 73 | |||||
| 12 h | 10 | 330 | 69 | 100 | 10 | 421 | 683 | 497 | 80 | |||||
| 24 h | 10 | 450 | 65 | 86 | 7 | 420 | 678 | 447 | 76 | |||||
| 6 h | 25 | 350 | 65 | 85 | 9 | 389 | 683 | 439 | 69 | |||||
| 12 h | 25 | 360 | 61 | 88 | 12 | 469 | 704 | 506 | 86 | |||||
| 24 h | 25 | 610 | 103 | 96 | 9 | 408 | 660 | 442 | 81 | |||||
| steamed | 0 h | 680 | 71 | 70 | 33 | 1.31/0.73/1.32 | 6 | 394 | 702 | 465 | 64 | 5.4 | 5.4 | |
| 6 h | 10 | 840 | 73 | 83 | 6 | 386 | 692 | 467 | 75 | |||||
| 12 h | 10 | 840 | 95 | 80 | 9 | 386 | 666 | 460 | 71 | |||||
| 24 h | 10 | 920 | 81 | 86 | 6 | 389 | 672 | 448 | 77 | |||||
| 6 h | 25 | 830 | 122 | 106 | 7 | 395 | 581 | 393 | 70 | |||||
| 12 h | 25 | 680 | 76 | 96 | 10 | 389 | 668 | 436 | 70 | |||||
| 24 h | 25 | 940 | 91 | 101 | 7 | 400 | 653 | 429 | 80 | |||||
| Treatment | Storage Condition | Fructan | Saccharose | Glucose | Fructose | WSC | |
|---|---|---|---|---|---|---|---|
| Time | Temperature | g/kg DM | |||||
| native | (h) | (°C) | 37 | 6 | 11 | 21 | 76 |
| soaked | 0 | 21 | 8 | 16 | 23 | 67 | |
| 6 | 10 | 15 | 3 | 7 | 18 | 42 | |
| 12 | 10 | 11 | 4 | 10 | 14 | 39 | |
| 24 | 10 | 14 | 2 | 6 | 15 | 37 | |
| 6 | 25 | 22 | 3 | 9 | 21 | 54 | |
| 12 | 25 | 15 | 4 | 4 | 11 | 35 | |
| 24 | 25 | 16 | 6 | 4 | 8 | 34 | |
| steamed | 0 | 31 | 18 | 13 | 19 | 81 | |
| 6 | 10 | 21 | 19 | 9 | 14 | 64 | |
| 12 | 10 | 17 | 15 | 11 | 16 | 59 | |
| 24 | 10 | 21 | 20 | 10 | 15 | 67 | |
| 6 | 25 | 19 | 12 | 6 | 11 | 49 | |
| 12 | 25 | 17 | 13 | 10 | 15 | 54 | |
| 24 | 25 | 17 | 16 | 7 | 12 | 52 | |
| p | Ca | K | Na | Mg | Zn | Mn | Cu | Fe | |
|---|---|---|---|---|---|---|---|---|---|
| Variety | g/kg DM | mg/kg DM | |||||||
| native | 2.1 | 5.7 | 18.7 | 0.2 | 0.7 | 16.6 | 19.2 | 5.3 | 139.1 |
| soaked | 1.8 | 5.2 | 10.3 | 0.9 | 1.0 | 15.9 | 32.0 | 7.3 | 157.4 |
| steamed | 2.0 | 5.2 | 17.6 | 1.1 | 1.1 | 16.4 | 31.5 | 9.2 | 88.0 |
| Treatment | |||
|---|---|---|---|
| Item | Native | Soaked | Steamed |
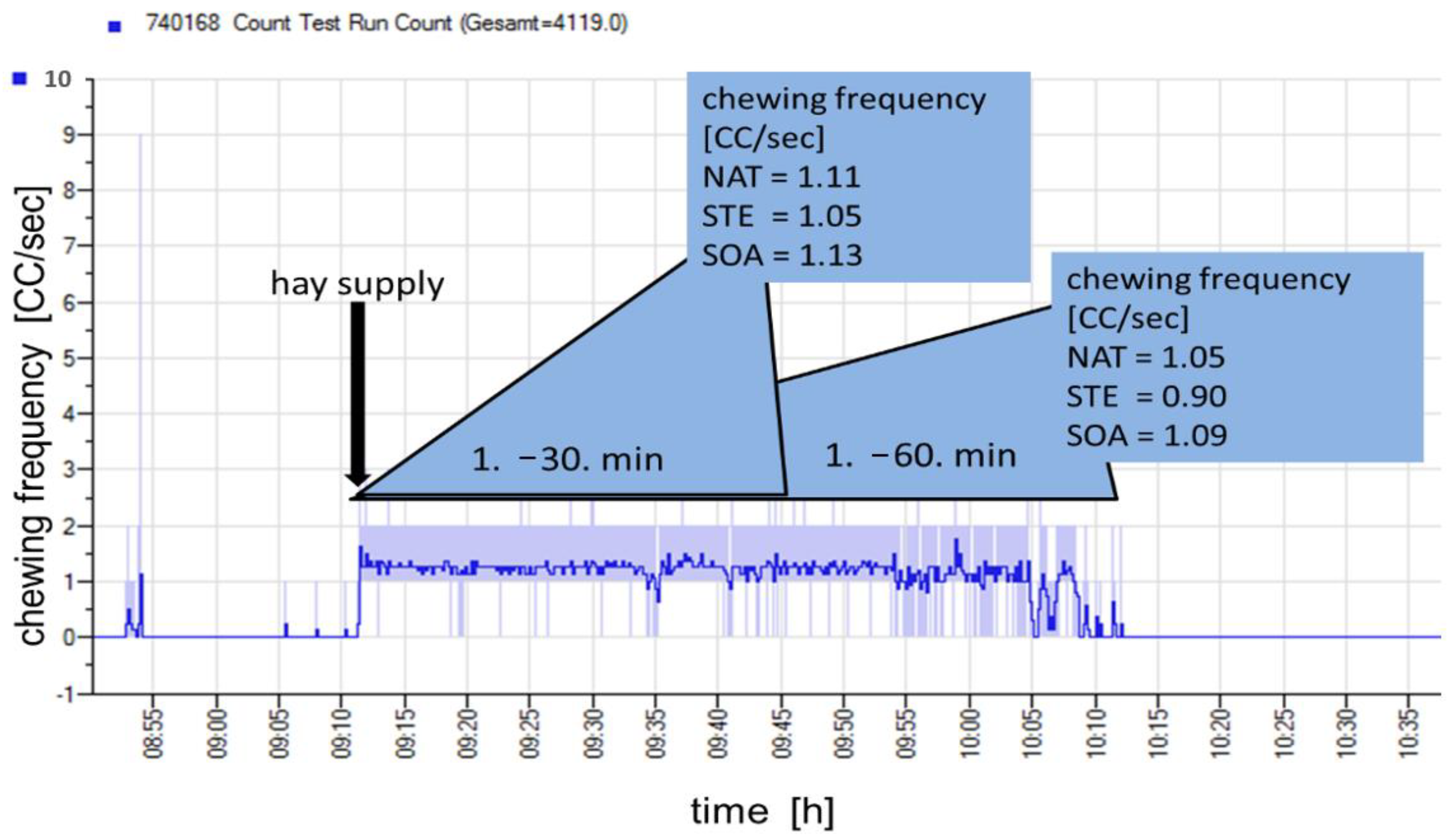
| chewing frequency (CC/s) | 1.05 ±0.078 | 1.09 # ±0.078 | 0.90 # ±0.078 |
| chewing frequency 30 (CC/s) | 1.11 ±0.053 | 1.13 ±0.053 | 1.05 ±0.053 |
| chewing intensity (CC/kg DM) | 2622 b ±217.3 | 3537 a ±217.3 | 2521 b ±217.3 |
| consumption rate (g DM/min) | 24.4 a ±2.89 | 19.5 b ±4.94 | 21.5 a ±2.04 |
| water intake (L/d) | 32.3 A ±2.13 | 21.6 B ±2.13 | 30.3 A ±2.13 |
 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Glatter, M.; Bochnia, M.; Wensch-Dorendorf, M.; Greef, J.M.; Zeyner, A. Feed Intake Parameters of Horses Fed Soaked or Steamed Hay and Hygienic Quality of Hay Stored following Treatment. Animals 2021, 11, 2729. https://doi.org/10.3390/ani11092729
Glatter M, Bochnia M, Wensch-Dorendorf M, Greef JM, Zeyner A. Feed Intake Parameters of Horses Fed Soaked or Steamed Hay and Hygienic Quality of Hay Stored following Treatment. Animals. 2021; 11(9):2729. https://doi.org/10.3390/ani11092729
Chicago/Turabian StyleGlatter, Maren, Mandy Bochnia, Monika Wensch-Dorendorf, Jörg Michael Greef, and Annette Zeyner. 2021. "Feed Intake Parameters of Horses Fed Soaked or Steamed Hay and Hygienic Quality of Hay Stored following Treatment" Animals 11, no. 9: 2729. https://doi.org/10.3390/ani11092729
APA StyleGlatter, M., Bochnia, M., Wensch-Dorendorf, M., Greef, J. M., & Zeyner, A. (2021). Feed Intake Parameters of Horses Fed Soaked or Steamed Hay and Hygienic Quality of Hay Stored following Treatment. Animals, 11(9), 2729. https://doi.org/10.3390/ani11092729






