Interaction Patterns between Wildlife and Cattle Reveal Opportunities for Mycobacteria Transmission in Farms from North-Eastern Atlantic Iberian Peninsula
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Camera Trap Survey
2.3. Variables Definition
2.4. Statistical Analysis
3. Results
3.1. Data Collected from the Field Samplings
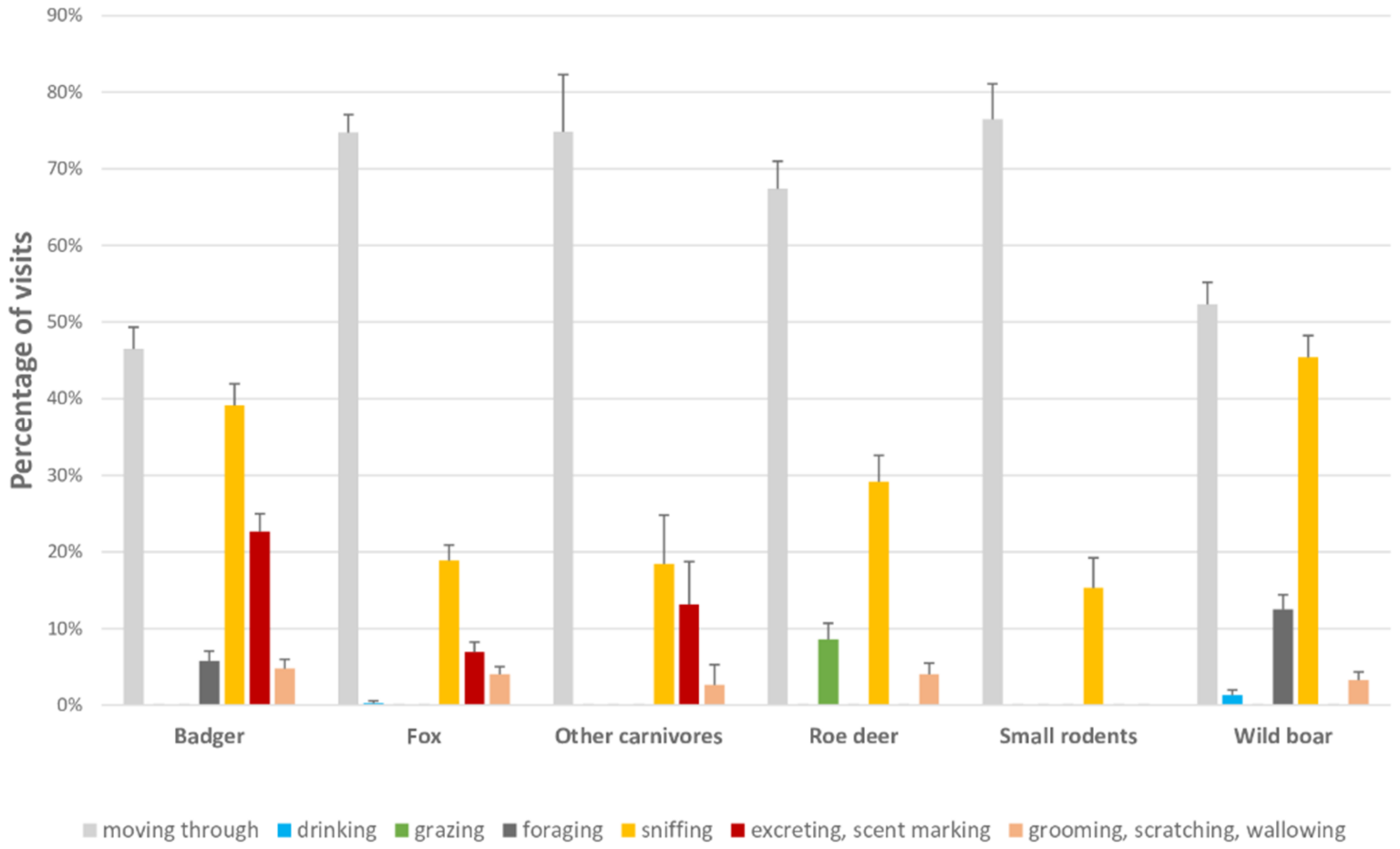
3.2. Frequency and Characterization of Visits Per Species
3.2.1. Badger
3.2.2. Wild Boar
3.2.3. Roe Deer
3.2.4. Fox
3.2.5. Other Carnivores
3.2.6. Small Rodents
4. Discussion
4.1. Methodology
4.2. Spatiotemporal Patterns of Wildlife-Cattle Interactions
4.3. Opportunities of Mycobacteria Transmission
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Roche, B.; Eric Benbow, M.; Merritt, R.; Kimbirauskas, R.; McIntosh, M.; Small, P.L.C.; Williamson, H.; Guégan, J.F. Identifying the Achilles heel of multi-host pathogens: The concept of keystone “host” species illustrated by Mycobacterium ulcerans transmission. Environ. Res. Lett. 2013, 8. [Google Scholar] [CrossRef]
- Kukielka, E.; Barasona, J.A.; Cowie, C.E.; Drewe, J.A.; Gortazar, C.; Cotarelo, I.; Vicente, J. Spatial and temporal interactions between livestock and wildlife in South Central Spain assessed by camera traps. Prev. Vet. Med. 2013, 112, 213–221. [Google Scholar] [CrossRef]
- Carrasco-Garcia, R.; Barasona, J.A.; Gortazar, C.; Montoro, V.; Sanchez-Vizcaino, J.M.; Vicente, J. Wildlife and livestock use of extensive farm resources in South Central Spain: Implications for disease transmission. Eur. J. Wildl. Res. 2015, 62, 65–78. [Google Scholar] [CrossRef]
- Triguero-Ocaña, R.; Barasona, J.A.; Carro, F.; Soriguer, R.C.; Vicente, J.; Acevedo, P. Spatio-temporal trends in the frequency of interspecific interactions between domestic and wild ungulates from Mediterranean Spain. PLoS ONE 2019, 14, e0211216. [Google Scholar] [CrossRef]
- Santos, N.; Richomme, C.; Nunes, T.; Vicente, J.; Alves, P.C.; de la Fuente, J.; Correia-neves, M.; Boschiroli, M.L.; Delahay, R.; Gortázar, C. Quantification of the animal tuberculosis multi- host community offers insights for control. Pathogens 2020, 9, 421. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Hernández, E.; Pizano-Martínez, O.E.; Canto-Alarcón, G.; Flores-Villalva, S.; Quintas-Granados, L.I.; Milián-Suazo, F. Persistence of Mycobacterium bovis under environmental conditions: Is it a real biological risk for cattle? Rev. Med. Microbiol. 2016, 27, 20–24. [Google Scholar] [CrossRef]
- Baldwin, S.L.; Larsenid, S.E.; Ordway, D.; Cassell, G.; Coler, R.N. The complexities and challenges of preventing and treating nontuberculous mycobacterial diseases. PLoS Negl. Trop. Dis. 2019, 13, e0007083. [Google Scholar] [CrossRef] [PubMed]
- Hoefsloot, W.; Van Ingen, J.; Andrejak, C.; Ängeby, K.; Bauriaud, R.; Bemer, P.; Beylis, N.; Boeree, M.J.; Cacho, J.; Chihota, V.; et al. The geographic diversity of nontuberculous mycobacteria isolated from pulmonary samples: An NTM-NET collaborative study. Eur. Respir. J. 2013, 42, 1604–1613. [Google Scholar] [CrossRef]
- Biet, F.; Boschiroli, M.L. Non-tuberculous mycobacterial infections of veterinary relevance. Res. Vet. Sci. 2014, 97, S69–S77. [Google Scholar] [CrossRef] [PubMed]
- Hruska, K.; Kaevska, M. Mycobacteria in water, soil, plants and air: A review. Vet. Med. 2012, 57, 623–679. [Google Scholar] [CrossRef]
- Eppleston, J.; Begg, D.J.; Dhand, N.K.; Watt, B.; Whittington, R.J. Environmental survival of Mycobacterium avium subsp. paratuberculosis in different climatic zones of eastern Australia. Appl. Environ. Microbiol. 2014, 80, 2337–2342. [Google Scholar] [CrossRef]
- Gortazar, C.; Boadella, M. Animal tuberculosis in Spain: A multihost system. In Zoonotic Tuberculosis: Mycobacterium Bovis and Other Pathogenic Mycobacteria, 3rd ed.; John Wiley & Sons, Inc.: New York, NY, USA, 2014; pp. 349–356. ISBN 9781118474310. [Google Scholar]
- Napp, S.; Allepuz, A.; Mercader, I.; Nofrarias, M.; Lopez-Soria, S.; Domingo, M.; Romero, B.; Bezos, J.; De Val Perez, B. Evidence of goats acting as domestic reservoirs of bovine tuberculosis. Vet. Rec. 2013, 172, 663. [Google Scholar] [CrossRef] [PubMed]
- Muñoz-Mendoza, M.; Romero, B.; del Cerro, A.; Gortázar, C.; García-Marín, J.F.; Menéndez, S.; Mourelo, J.; de Juan, L.; Sáez, J.L.; Delahay, R.J.; et al. Sheep as a Potential Source of Bovine TB: Epidemiology, Pathology and Evaluation of Diagnostic Techniques. Transbound. Emerg. Dis. 2015, 63, 635–646. [Google Scholar] [CrossRef] [PubMed]
- Parra, A.; Fernández-Llario, P.; Tato, A.; Larrasa, J.; García, A.; Alonso, J.M.; Hermoso De Mendoza, M.; Hermoso De Mendoza, J. Epidemiology of Mycobacterium bovis infections of pigs and wild boars using a molecular approach. Vet. Microbiol. 2003, 97, 123–133. [Google Scholar] [CrossRef]
- Ministerio de Agricultura Pesca y Alimentación Programa Nacional de Erradicación de Tuberculosis Bovina Presentado Por España Para el Año 2020. Available online: https://www.mapa.gob.es/es/ganaderia/temas/sanidad-animal-higiene-ganadera/pnetb_2020final_tcm30-523317.PDF (accessed on 7 October 2020).
- Muñoz-Mendoza, M.; Marreros, N.; Boadella, M.; Gortázar, C.; Menéndez, S.; de Juan, L.; Bezos, J.; Romero, B.; Copano, M.F.; Amado, J.; et al. Wild boar tuberculosis in Iberian Atlantic Spain: A different picture from Mediterranean habitats. BMC Vet. Res. 2013, 9, 176. [Google Scholar] [CrossRef] [PubMed]
- Varela-Castro, L.; Torrontegi, O.; Sevilla, I.A.; Barral, M. Detection of wood mice (Apodemus sylvaticus) carrying non-tuberculous mycobacteria able to infect cattle and interfere with the diagnosis of bovine tuberculosis. Microorganisms 2020, 8, 374. [Google Scholar] [CrossRef]
- Acevedo, P.; Prieto, M.; Quirós, P.; Merediz, I.; de Juan, L.; Infantes-Lorenzo, J.A.; Triguero-Ocaña, R.; Balseiro, A. Tuberculosis Epidemiology and Badger (Meles meles) Spatial Ecology in a Hot-Spot Area in Atlantic Spain. Pathogens 2019, 8, 292. [Google Scholar] [CrossRef]
- Blanco Vázquez, C.B.; Barral, T.D.; Romero, B.; Queipo, M.; Merediz, I.; Quirós, P.; Armenteros, J.Á.; Juste, R.; Domínguez, L.; Domínguez, M.; et al. Spatial and Temporal Distribution of Mycobacterium tuberculosis Complex Infection in Eurasian Badger (Meles meles) and Cattle in Asturias, Spain. Animals 2021, 11, 1294. [Google Scholar] [CrossRef]
- Varela-Castro, L.; Alvarez, V.; Sevilla, I.A.; Barral, M. Risk factors associated to a high Mycobacterium tuberculosis complex seroprevalence in wild boar (Sus scrofa) from a low bovine tuberculosis prevalence area. PLoS ONE 2020, 15, e0231559. [Google Scholar] [CrossRef] [PubMed]
- Varela-Castro, L.; Gerrikagoitia, X.; Alvarez, V.; Geijo, M.V.; Barral, M.; Sevilla, I.A. A long-term survey on Mycobacterium tuberculosis complex in wild mammals from a bovine tuberculosis low prevalence area. Eur. J. Wildl. Res. 2021, 67, 1–8. [Google Scholar] [CrossRef]
- Balseiro, A.; Merediz, I.; Sevilla, I.A.; García-Castro, C.; Gortázar, C.; Prieto, J.M.; Delahay, R.J. Infection of Eurasian badgers (Meles meles) with Mycobacterium avium complex (MAC) bacteria. Vet. J. 2011, 188, 231–233. [Google Scholar] [CrossRef]
- Rowcliffe, J.M.; Field, J.; Turvey, S.T.; Carbone, C. Estimating animal density using camera traps without the need for individual recognition. J. Appl. Ecol. 2008, 45, 1228–1236. [Google Scholar] [CrossRef]
- Niedballa, J.; Wilting, A.; Sollmann, R.; Hofer, H.; Courtiol, A. Assessing analytical methods for detecting spatiotemporal interactions between species from camera trapping data. Remote Sens. Ecol. Conserv. 2019. [Google Scholar] [CrossRef]
- Niedballa, J.; Sollmann, R.; Courtiol, A.; Wilting, A. camtrapR: An R package for efficient camera trap data management. Methods Ecol. Evol. 2016, 7, 1457–1462. [Google Scholar] [CrossRef]
- Triguero-Ocaña, R.; Vicente, J.; Palencia, P.; Laguna, E.; Acevedo, P. Quantifying wildlife-livestock interactions and their spatio-temporal patterns: Is regular grid camera trapping a suitable approach? Ecol. Indic. 2020, 117, 106565. [Google Scholar] [CrossRef]
- Gobierno Vasco euskadi.eus. Available online: https://www.euskadi.eus/inicio/ (accessed on 5 May 2020).
- Payne, A.; Philipon, S.; Hars, J.; Dufour, B.; Gilot-Fromont, E. Wildlife Interactions on Baited Places and Waterholes in a French Area Infected by Bovine Tuberculosis. Front. Vet. Sci. 2017, 3, 122. [Google Scholar] [CrossRef]
- Payne, A.; Chappa, S.; Hars, J.; Dufour, B.; Gilot-Fromont, E. Wildlife visits to farm facilities assessed by camera traps in a bovine tuberculosis-infected area in France. Eur. J. Wildl. Res. 2016, 62, 33–42. [Google Scholar] [CrossRef]
- Burnham, K.P.; Anderson, D.R. Model Selection and Multimodel Inference: A Practical Information-Theoretic Approach, 2nd ed.; Springer: Berlin/Heidelberg, Germany, 2002. [Google Scholar]
- Zuur, A.F.; Ieno, E.N.; Walker, N.; Saveliev, A.A.; Smith, G.M. Mixed Effects Models and Extensions in Ecology with R; Springer: New York, NY, USA, 2009. [Google Scholar]
- R Core Team 2020: R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2021; Available online: https://www.R-project.org/ (accessed on 31 July 2021).
- Drewe, J.A.; O’Connor, H.M.; Weber, N.; McDonald, R.A.; Delahay, R.J. Patterns of direct and indirect contact between cattle and badgers naturally infected with tuberculosis. Epidemiol. Infect. 2013, 141, 1467–1475. [Google Scholar] [CrossRef] [PubMed]
- Zabala, J.; Garin, I.; Zuberogoitia, I.; Aihartza, J. Habitat selection and diet of badgers (Meles meles) in Biscay (northern Iberian Peninsula). Ital. J. Zool. 2002, 69, 233–238. [Google Scholar] [CrossRef]
- Mullen, E.M.; MacWhite, T.; Maher, P.K.; Kelly, D.J.; Marples, N.M.; Good, M. The avoidance of farmyards by European badgers Meles meles in a medium density population. Appl. Anim. Behav. Sci. 2015, 171, 170–176. [Google Scholar] [CrossRef]
- Tolhurst, B.A.; Delahay, R.J.; Walker, N.J.; Ward, A.I.; Roper, T.J. Behaviour of badgers (Meles meles) in farm buildings: Opportunities for the transmission of Mycobacterium bovis to cattle ? Appl. Anim. Behav. Sci. 2009, 117, 103–113. [Google Scholar] [CrossRef]
- Judge, J.; Mcdonald, R.A.; Walker, N.; Delahay, R.J. Effectiveness of Biosecurity Measures in Preventing Badger Visits to Farm Buildings. PLoS ONE 2011, 6, e28941. [Google Scholar] [CrossRef] [PubMed]
- Woodroffe, R.; Donnelly, C.A.; Ham, C.; Jackson, S.Y.B.; Moyes, K.; Chapman, K.; Stratton, N.G.; Cartwright, S.J. Badgers prefer cattle pasture but avoid cattle: Implications for bovine tuberculosis control. Ecol. Lett. 2016, 19, 1201–1208. [Google Scholar] [CrossRef] [PubMed]
- Hoem, S.A.; Melis, C.; Linnell, J.D.C.; Andersen, R. Fighting behaviour in territorial male roe deer Capreolus capreolus: The effects of antler size and residence. Eur. J. Wildl. Res. 2007, 53, 1–8. [Google Scholar] [CrossRef]
- Duncan, P.; Tixier, H.; Hofmann, R.; Lechner-Doll, M. Feeding strategies and the physiology of digestion in roe deer. In The European Roe Deer: The Biology of Success; Scandinavian University Press: Oslo, Norway, 1998; pp. 91–116. [Google Scholar]
- Palomares, F.; Delibes, M. Spatio-temporal Ecology and Behaviour of European Genets in Southwestern Spain. J. Mammal. 1994, 75, 714–724. [Google Scholar] [CrossRef]
- Torre, I.; Arrizabalaga, A.; Díaz, M. Ratón de campo Apodemus sylvaticus (Linnaeus, 1758). Galemys 2002, 14, 1–26. [Google Scholar]
- Montgomery, W.I.; Dowie, M. The Distribution and Population Regulation of the Wood Mouse Apodemus sylvaticus on Field Boundaries of Pastoral Farmland. J. Appl. Ecol. 1993, 30, 783. [Google Scholar] [CrossRef]
- Barasona, J.A.; Latham, M.C.; Acevedo, P.; Armenteros, J.A.; Latham, A.D.M.; Gortazar, C.; Carro, F.; Soriguer, R.C.; Vicente, J. Spatiotemporal interactions between wild boar and cattle: Implications for cross-species disease transmission. Vet. Res. 2014, 45, 122. [Google Scholar] [CrossRef]
- Caron, A.; Cappelle, J.; Cumming, G.S.; De Garine-Wichatitsky, M.; Gaidet, N. Bridge hosts, a missing link for disease ecology in multi-host systems. Vet. Res. 2015, 46, 1–11. [Google Scholar] [CrossRef]
- Millán, J.; Jiménez, M.Á.; Viota, M.; Candela, M.G.; Peña, L.; León-Vizcaíno, L. Disseminated Bovine Tuberculosis in a Wild Red Fox (Vulpes vulpes) in Southern Spain. J. Wildl. Dis. 2008, 44, 701–706. [Google Scholar] [CrossRef]
- Michelet, L.; De Cruz, K.; Hénault, S.; Tambosco, J.; Richomme, C.; Réveillaud, É.; Gares, H.; Moyen, J.L.; Boschiroli, M.L. Mycobacterium bovis infection of red fox, France. Emerg. Infect. Dis. 2018, 24, 1151–1153. [Google Scholar] [CrossRef]
- Richomme, C.; Réveillaud, E.; Moyen, J.L.; Sabatier, P.; de Cruz, K.; Michelet, L.; Boschiroli, M.L. Mycobacterium bovis infection in red foxes in four animal tuberculosis endemic areas in France. Microorganisms 2020, 8, 1070. [Google Scholar] [CrossRef]
- Matos, A.C.; Figueira, L.; Martins, M.H.; Matos, M.; Morais, M.; Dias, A.P.; Pinto, M.L.; Coelho, A.C. Disseminated Mycobacterium bovis Infection in Red Foxes ( Vulpes vulpes ) with Cerebral Involvement Found in Portugal. Vector-Borne Zoonotic Dis. 2014, 14, 531–533. [Google Scholar] [CrossRef]
- Matos, A.C.; Figueira, L.; Martins, M.H.; Pinto, M.L.; Matos, M.; Coelho, A.C. New insights into Mycobacterium bovis prevalence in wild mammals in Portugal. Transbound. Emerg. Dis. 2016, 63, e313–e322. [Google Scholar] [CrossRef]
- Mathews, F.; Macdonald, D.W.; Taylor, G.M.; Gelling, M.; Norman, R.A.; Honess, P.E.; Foster, R.; Gower, C.M.; Varley, S.; Harris, A.; et al. Bovine tuberculosis (Mycobacterium bovis) in British farmland wildlife: The importance to agriculture. Proc. R. Soc. B Biol. Sci. 2006, 273, 357–365. [Google Scholar] [CrossRef] [PubMed]
- Delahay, R.J.; De Leeuw, A.N.S.; Barlow, A.M.; Clifton-Hadley, R.S.; Cheeseman, C.L. The status of Mycobacterium bovis infection in UK wild mammals: A review. Vet. J. 2002, 164, 90–105. [Google Scholar] [CrossRef]
- Delahay, R.J.; Smith, G.C.; Barlow, A.M.; Walker, N.; Harris, A.; Clifton-Hadley, R.S.; Cheeseman, C.L. Bovine tuberculosis infection in wild mammals in the South-West region of England: A survey of prevalence and a semi-quantitative assessment of the relative risks to cattle. Vet. J. 2007, 173, 287–301. [Google Scholar] [CrossRef]
- Smith, N.H.; Crawshaw, T.; Parry, J.; Birtles, R.J. Mycobacterium microti: More diverse than previously thought. J. Clin. Microbiol. 2009, 47, 2551–2559. [Google Scholar] [CrossRef]
- Kipar, A.; Burthe, S.J.; Hetzel, U.; Abo Rokia, M.; Telfer, S.; Lambin, X.; Birtles, R.J.; Begon, M.; Bennett, M. Mycobacterium microti Tuberculosis in Its Maintenance Host, the Field Vole (Microtus agrestis): Characterization of the Disease and Possible Routes of Transmission. Vet. Pathol. 2014, 51, 903–914. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lambert, S.; Hars, J.; Réveillaud, E.; Moyen, J.-L.; Gares, H.; Rambaud, T.; Gueneau, E.; Faure, E.; Boschiroli, M.-L.; Richomme, C. Host status of wild roe deer in bovine tuberculosis endemic areas. Eur. J. Wildl. Res. 2017, 63, 15. [Google Scholar] [CrossRef]
- Balseiro, A.; Orusa, A.O.R.; Robetto, S.; Zoppi, Z.; Dondo, A.; Goria, M.; Gortázar, C.; Marín, J.F.G.; Domenis, L. Tuberculosis in roe deer from Spain and Italy. Vet. Rec. 2009, 164, 468–470. [Google Scholar] [CrossRef] [PubMed]
- Gortázar, C.; Diez-Delgado, I.; Barasona, J.A.; Vicente, J.; De La Fuente, J.; Boadella, M. The Wild Side of Disease Control at the Wildlife-Livestock-Human Interface: A Review. Front. Vet. Sci. 2015, 1, 27. [Google Scholar] [CrossRef] [PubMed]
- Turenne, C.Y.; Collins, D.M.; Alexander, D.C.; Behr, M.A. Mycobacterium avium subsp. paratuberculosis and M. avium subsp. avium are independently evolved pathogenic clones of a much broader group of M. avium organisms. J. Bacteriol. 2008, 190, 2479–2487. [Google Scholar] [CrossRef]
- Turenne, C.Y.; Wallace, R.; Behr, M.A. Mycobacterium avium in the Postgenomic Era. Clin. Microbiol. Rev. 2007, 20, 205–229. [Google Scholar] [CrossRef]
- Kasperbauer, S.; Huitt, G. Management of Extrapulmonary Nontuberculous Mycobacterial Infections. Semin. Respir. Crit. Care Med. 2013, 34, 143–150. [Google Scholar] [CrossRef]
- Claeys, T.A.; Robinson, R.T. The Many Lives of Nontuberculous Mycobacteria. J. Bacteriol. 2018, 200, e00739-17. [Google Scholar] [CrossRef] [PubMed]
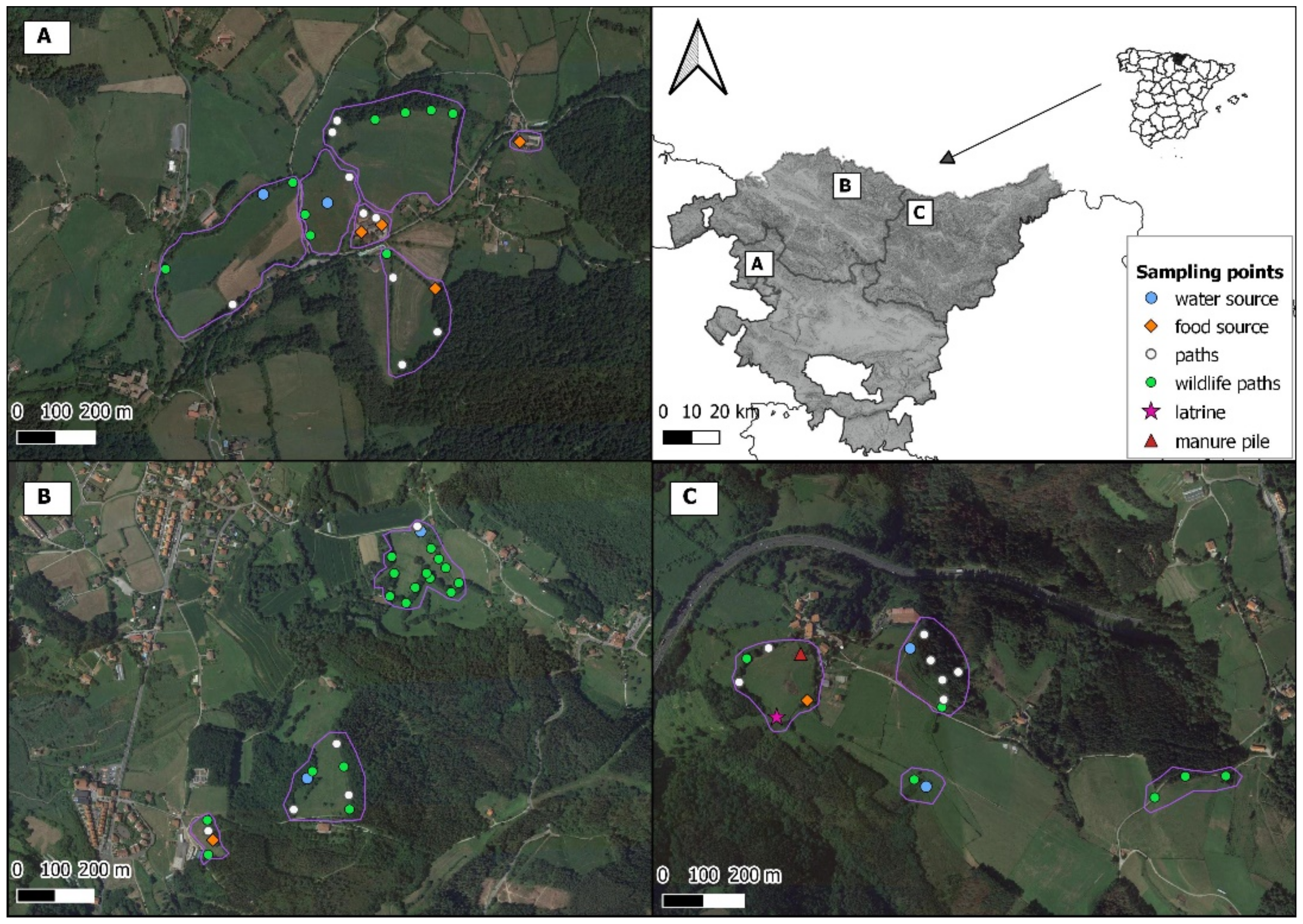
| Type of Place | Number Surveyed | Type of Point | Number Surveyed | Number of Surveillance Hours | Number of Sessions |
|---|---|---|---|---|---|
| Pasture (1070) | 11 | Water source (114) | 6 | 4514.83 | 17 |
| Food source (10) | 3 | 939.60 | 4 | ||
| Manure (12) | 1 | 1343.82 | 4 | ||
| Latrine (54) | 1 | 511.27 | 2 | ||
| Wildlife path (703) | 30 | 20,115.23 | 77 | ||
| Path (177) | 13 | 8590.40 | 30 | ||
| Farm building (22) | 2 | Food source (8) | 3 | 2564.35 | 11 |
| Path (16) | 2 | 1144.52 | 7 | ||
| Forest (61) | 1 | Wildlife path (12) | 1 | 360.23 | 1 |
| Path (49) | 2 | 2756.07 | 8 | ||
| Edge (140) | 3 | Wildlife path (17) | 1 | 1455.08 | 4 |
| Path (109) | 3 | 3689.23 | 12 | ||
| Water source (14) | 1 | 1027.97 | 3 | ||
| Total | 17 | 67 | 49,012.60 | 180 |
| Behavior | Description |
|---|---|
| Grazing (for roe deer) | Feeding from grass, plants or fruits from a surface |
| Foraging (for badger and wild boar) | Searching for food by digging the ground with the snout |
| Sniffing (for all species) | Smelling the ground to search for food or to explore a surface/object |
| Excreting (for all species)/Scent marking (for all carnivores) | Urinating or defecating. For carnivores, lifting the tail and approaching the pelvis to the ground |
| Grooming/Scratching/Wallowing (for all species) | Applying tongue or paws to parts of the body in repeated motions, shaking the body, scraping against a surface, rolling in a water point |
| Drinking (for all species) | Drinking from water sources |
| Moving through (for all species) | Passing through a sampling point without performing any of the aforementioned behaviours |
| Badger (n = 315) | Wild Boar (n = 304) | Roe Deer (n = 175) | Fox (n = 376) | Other Carnivores (n = 38) | Small Rodents (n = 85) | |
|---|---|---|---|---|---|---|
| Frequency of visits (all visits, number per month) | 4.73 ± 0.61 0–46.67 | 4.41 ± 0.58 0–46.33 | 2.76 ± 0.64 0–83.72 | 5.69 ± 0.66 0–59.20 | 0.54 ± 0.16 0–19.66 | 1.85 ± 0.74 0–117.2 |
| Frequency of visits in buildings only (number per month) | 0 | 1.07 ± 0.76 0–12 | 0 | 1.30 ± 0.76 0–12.12 | 0 | 2.04 ± 2.04 0–36.76 |
| Visit duration (min) | 0.89 ± 0.19 0.17–31 | 1.64 ± 0.26 0.17–38 | 2.33 ± 0.60 0.17–56 | 1.31 ± 0.31 0.17–86 | 0.47 ± 0.13 0.17–4 | 5.12 ± 1.68 0.17–95 |
| Number of individuals per visit | 1.05 ± 0.01 1–4 | 2.57 ± 0.10 1–10 | 1.16 ± 0.03 1–3 | 1.04 ± 0.01 1–3 | 1 ± 0 1–1 | 1.08 ± 0.04 1–3 |
| Badger | Wild Boar | Roe Deer | Fox | Small Rodents | |
|---|---|---|---|---|---|
| Number of direct interactions | 2 | 6 | 1 | 8 | 6 |
| Most frequent season | Autumn (2) | Autumn (5) | Summer | Summer (5) | Autumn (4) |
| Most frequent place | Pasture (2) | Pasture (5) | Pasture | Pasture (7) | Farm building (4) |
| Most frequent point | Latrine (2) | Manure (2)/Wildlife path (2) | Wildlife path | Wildlife path (6) | Path (4) |
| Behaviors observed | Sniffing/ scent marking | Sniffing/foraging/moving through | Sniffing | Moving through/sniffing | Moving through |
| Species | Response Variable | V.E by Fixed Part | V.E by Random Part | Fixed Effect | Level | OR (95% CI) | Estimate | p-Value |
|---|---|---|---|---|---|---|---|---|
| Badger | Frequency of visits | 28.35% | 40.94% | Season | Autumn | 0.87 (0.59–1.26) | −0.14 | 0.454 |
| (ref: summer) | Winter | 2.49 (1.75–3.54) | 0.91 | <0.001 *** | ||||
| Spring | 1.16 (0.86–1.58) | 0.15 | 0.325 | |||||
| Place | Edge | 0.56 (0.34–0.91) | −0.59 | 0.020 * | ||||
| (ref: pasture) | Forest | 1.06 (0.53–2.11) | 0.06 | 0.875 | ||||
| Point | Latrine | 3.81 (1.97–7.35) | 1.34 | <0.001 *** | ||||
| (ref: wildlife path) | Manure | 0.04 (0.01–0.31) | −3.17 | 0.002 ** | ||||
| Path | 0.46 (0.31–0.68) | −0.78 | <0.001 *** | |||||
| Water source | 0.81 (0.54–1.20) | −0.21 | 0.287 | |||||
| Duration of visits | 1.03% | 9.09% | Place | Edge | 0.43 (0.20–0.89) | −0.85 | 0.024 * | |
| (ref: pasture) | Forest | 0.35 (0.15–0.80) | −1.06 | 0.013 * | ||||
| Point | Latrine | 0.43 (0.18–1.03) | −0.84 | 0.059 | ||||
| (ref: wildlife path) | Manure | 0.27 (0.02–3.04) | −1.29 | 0.292 | ||||
| Path | 0.41 (0.25–0.70) | −0.88 | <0.001 *** | |||||
| Water source | 1.60 (0.95–2.70) | 0.47 | 0.079 | |||||
| Wild boar | Frequency of visits | 29.08% | 19.64% | Season | Autumn | 1.57 (1.13–2.18) | 0.45 | 0.007 ** |
| (ref: summer) | Winter | 0.39 (0.23–0.67) | −0.94 | <0.001 *** | ||||
| Spring | 1.38 (1.02–1.88) | 0.32 | 0.038 * | |||||
| Point | Food source | 0.34 (0.13–0.94) | −1.07 | 0.037 * | ||||
| (ref: wildlife path) | Latrine | 9.07 (4.47–18.42) | 2.20 | <0.001 *** | ||||
| Manure | 1.30 (0.49–3.41) | 0.26 | 0.599 | |||||
| Path | 0.77 (0.55–1.08) | −0.26 | 0.128 | |||||
| Water source | 1.23 (0.86–1.76) | 0.21 | 0.260 | |||||
| Number of animals | 10.85% | 0.60% | Season | Autumn | 1.52 (1.20–1.94) | 0.42 | <0.001 *** | |
| (ref: summer) | Winter | 0.70 (0.46–1.07) | −0.35 | 0.103 | ||||
| Spring | 1.04 (0.83–1.30) | 0.04 | 0.746 | |||||
| Period of the day | Dawn | 0.70 (0.34–1.43) | −0.35 | 0.332 | ||||
| (ref: night) | Sunset | 0.59 (0.38–0.92) | −0.53 | 0.020 * | ||||
| Duration of visits | 0.32% | 10.88% | Season | Autumn | 1.12 (0.60–2.07) | 0.11 | 0.730 | |
| (ref: summer) | Winter | 3.41 (1.54–7.57) | 1.23 | 0.003 ** | ||||
| Spring | 1.21 (0.71–2.06) | 0.19 | 0.480 | |||||
| Roe deer | Frequency of visits | 4.87% | 91.50% | Season | Autumn | 1.57 (0.83–2.97) | 0.45 | 0.162 |
| (ref: summer) | Winter | 0.54 (0.25–1.18) | −0.61 | 0.123 | ||||
| Spring | 0.30 (0.21–0.44) | −1.19 | <0.001 *** | |||||
| Duration of visits | 6.21% | 20.20% | Season | Autumn | 0.28 (0.10–0.81) | −1.26 | 0.020 * | |
| (ref: summer) | Winter | 0.95 (0.33–2.72) | −0.05 | 0.925 | ||||
| Spring | 0.10 (0.06–0.16) | −2.32 | <0.001 *** | |||||
| Point | Food source | 22.15 (4.46–110.05) | 3.10 | <0.001 *** | ||||
| (ref: wildlife path) | Path | 1.43 (0.59–3.46) | 0.35 | 0.434 | ||||
| Water source | 1.85 (0.42–8.11) | 0.62 | 0.413 | |||||
| Fox | Frequency of visits | 28.42% | 6.43% | Point | Food source | 0.15 (0.05–0.42) | −1.89 | <0.001 *** |
| (ref: wildlife path) | Latrine | 2.79 (1.42–5.49) | 1.03 | 0.003 ** | ||||
| Manure | 0.48 (0.19–1.23) | −0.73 | 0.127 | |||||
| Path | 1.02 (0.79–1.32) | 0.02 | 0.863 | |||||
| Water source | 0.62 (0.42–0.93) | −0.47 | 0.020 * | |||||
| Duration of visits | 3.88% | 3.89% | Season | Autumn | 0.44 (0.20–0.97) | −0.83 | 0.042 * | |
| (ref: summer) | Winter | 0.86 (0.43–1.73) | −0.15 | 0.670 | ||||
| Spring | 2.02 (1.10–3.68) | 0.70 | 0.022 * | |||||
| Period of the day | Dawn | 0.89 (0.25–3.19) | −0.12 | 0.859 | ||||
| (ref: night) | Day | 3.76 (1.61–8.78) | 1.33 | 0.002 ** | ||||
| Sunset | 0.60 (0.25–1.44) | −0.51 | 0.254 | |||||
| Place | Edge | 0.38 (0.15–0.97) | −0.97 | 0.042 * | ||||
| (ref: pasture) | Farm building | 1.48 (0.13–16.54) | 0.39 | 0.750 | ||||
| Forest | 0.45 (0.16–1.25) | −0.80 | 0.125 | |||||
| Point | Food source | 2.22 (0.15–33.41) | 0.80 | 0.563 | ||||
| (ref: wildlife path) | Latrine | 0.23 (0.04–1.34) | −1.47 | 0.103 | ||||
| Manure | 0.12 (0.01–1.03) | −2.13 | 0.053. | |||||
| Path | 0.68 (0.33–1.41) | −0.39 | 0.301 | |||||
| Water source | 0.32 (0.13–0.83) | −1.13 | 0.019 * | |||||
| Other carnivores | Frequency of visits | 2.68% | 3.58% | Place (ref: edge) | Pasture | 0.19 (0.06–0.60) | −1.68 | 0.005 ** |
| Point | Water source | 0.20 (0.06–0.67) | −1.61 | 0.009 ** | ||||
| (ref: path) | Wildlife path | 0.34 (0.11–1.02) | −1.08 | 0.054. | ||||
| Duration of visits | 6.9% | 0.00% | Place (ref: edge) | Pasture | 0.36 (0.20–0.66) | −1.02 | <0.001 *** | |
| Small rodents | Frequency of visits | 23.92% | 18.23% | Season | Summer | 0.03 (0.01–0.09) | −3.64 | <0.001 *** |
| (ref: autumn) | Winter | 0.11 (0.05–0.23) | −2.19 | <0.001 *** | ||||
| Spring | 0.15 (0.08–0.29) | −1.91 | <0.001 *** | |||||
| Place (ref: pasture) | Edge | 0.33 (0.15–0.72) | −1.11 | 0.005 ** | ||||
| Farm building | 1.86 (0.16–21.88) | 0.62 | 0.623 | |||||
| Point | Food source | 0.20 (0.03–1.13) | −1.63 | 0.068 | ||||
| (ref: wildlife path) | Path | 0.53 (0.31–0.92) | −0.63 | 0.023 * | ||||
| Water source | 0.42 (0.17–1.04) | −0.86 | 0.060 | |||||
| Duration of visits | 11.08% | 0.00% | Season | Summer | 0.02 (0.00–0.14) | −3.74 | <0.001 *** | |
| (ref: autumn) | Winter | 0.04 (0.01–0.11) | −3.31 | <0.001 *** | ||||
| Spring | 0.02 (0.01–0.06) | −3.74 | <0.001 *** |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Varela-Castro, L.; Sevilla, I.A.; Payne, A.; Gilot-Fromont, E.; Barral, M. Interaction Patterns between Wildlife and Cattle Reveal Opportunities for Mycobacteria Transmission in Farms from North-Eastern Atlantic Iberian Peninsula. Animals 2021, 11, 2364. https://doi.org/10.3390/ani11082364
Varela-Castro L, Sevilla IA, Payne A, Gilot-Fromont E, Barral M. Interaction Patterns between Wildlife and Cattle Reveal Opportunities for Mycobacteria Transmission in Farms from North-Eastern Atlantic Iberian Peninsula. Animals. 2021; 11(8):2364. https://doi.org/10.3390/ani11082364
Chicago/Turabian StyleVarela-Castro, Lucía, Iker A. Sevilla, Ariane Payne, Emmanuelle Gilot-Fromont, and Marta Barral. 2021. "Interaction Patterns between Wildlife and Cattle Reveal Opportunities for Mycobacteria Transmission in Farms from North-Eastern Atlantic Iberian Peninsula" Animals 11, no. 8: 2364. https://doi.org/10.3390/ani11082364
APA StyleVarela-Castro, L., Sevilla, I. A., Payne, A., Gilot-Fromont, E., & Barral, M. (2021). Interaction Patterns between Wildlife and Cattle Reveal Opportunities for Mycobacteria Transmission in Farms from North-Eastern Atlantic Iberian Peninsula. Animals, 11(8), 2364. https://doi.org/10.3390/ani11082364






