Dietary Cinnamon Successfully Enhanced the Growth Performance, Growth Hormone, Antibacterial Capacity, and Immunity of European Sea Bass (Dicentrarchus labrax)
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Fish
2.2. Experimental Diets
2.3. Body Composition Analysis
2.4. Hematological and Blood Biochemical Analysis
2.5. Intestinal Antimicrobial Analysis
2.6. Statistical Analysis
3. Results
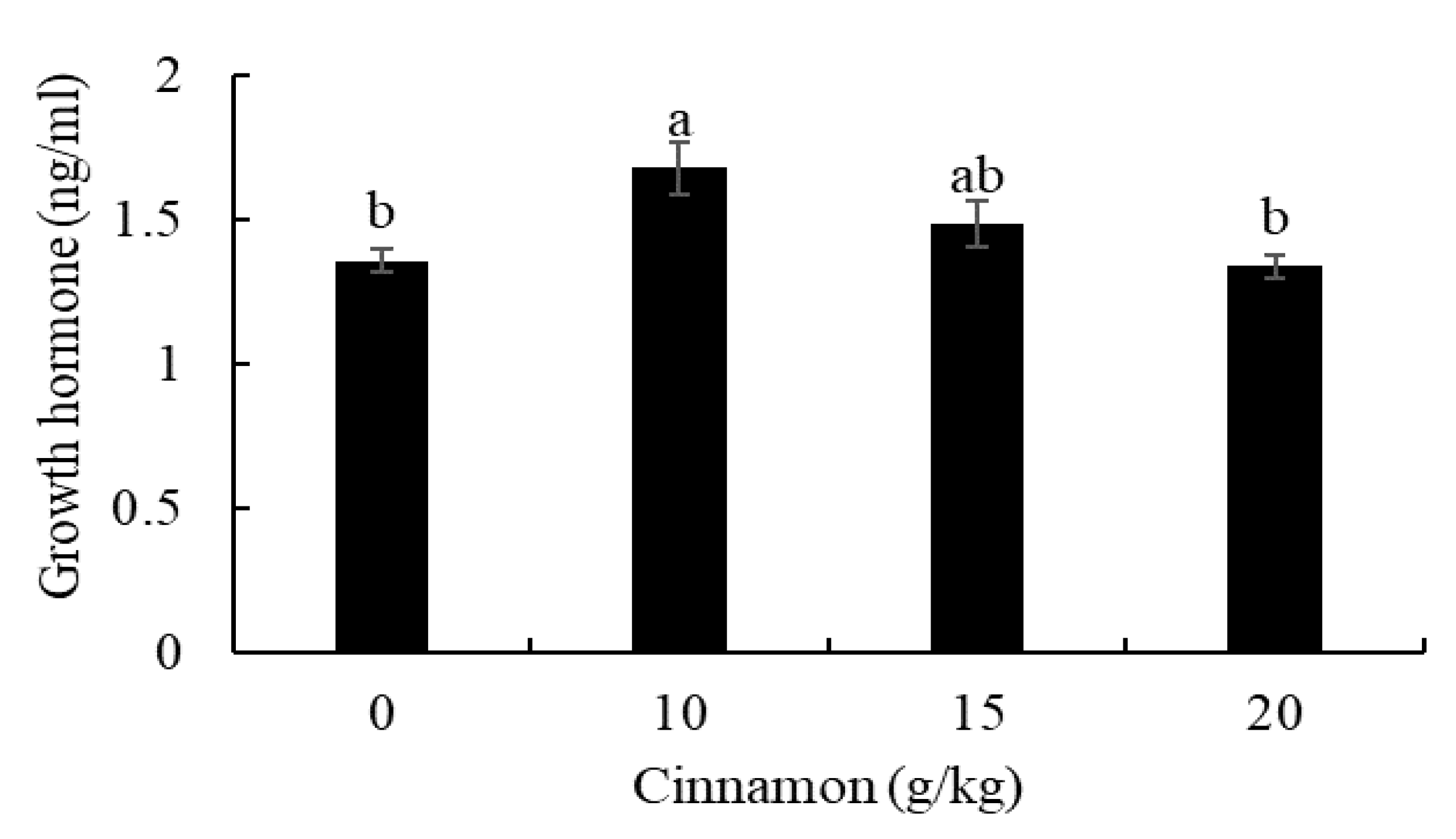
3.1. Growth Performance, Growth Hormone, and Carcass Composition
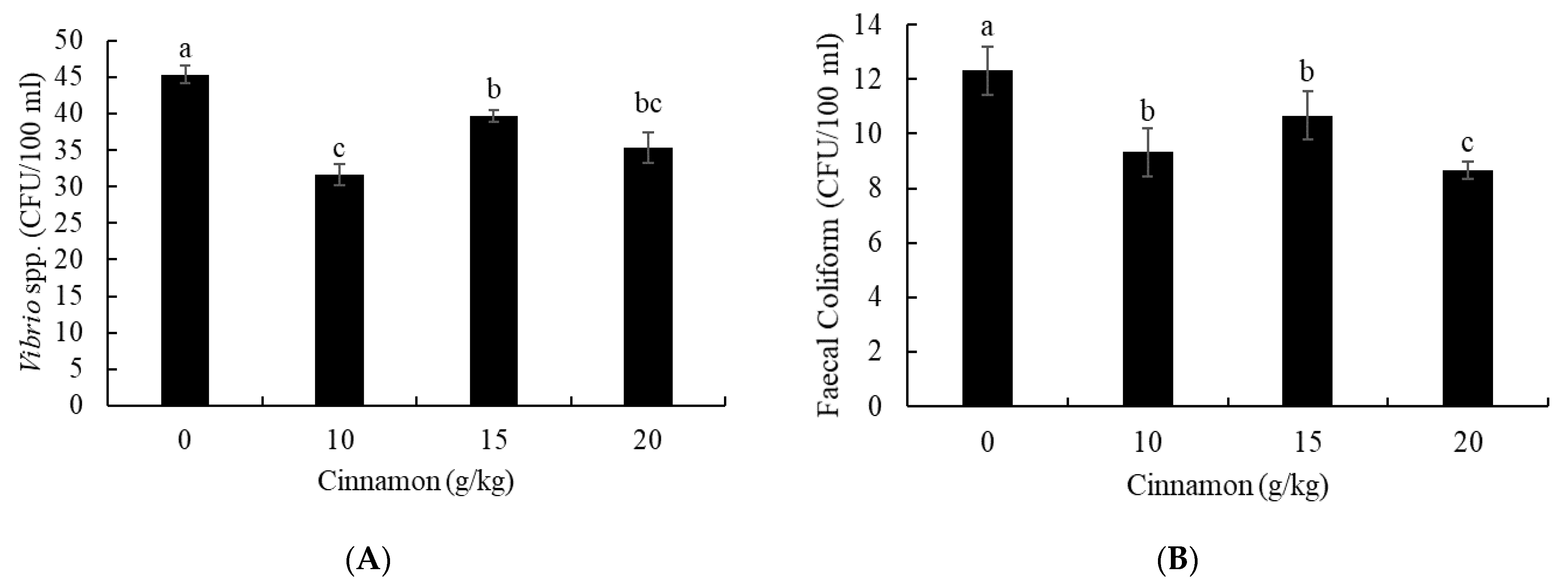
3.2. Intestine Antibacterial Capacity
3.3. Hematology and Blood Biochemical Indices
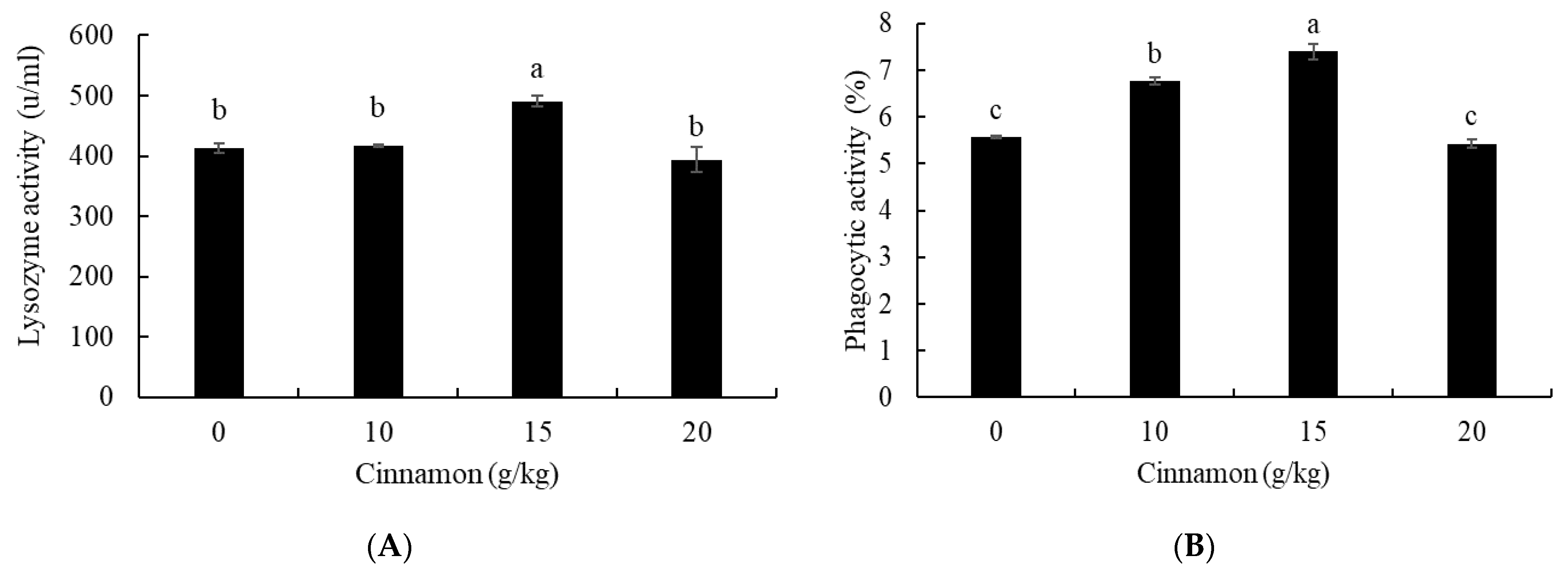
3.4. Lysozyme and Phagocytic Activities
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- See, K.F.; Ibrahim, R.A.; Goh, K.H. Aquaculture efficiency and productivity: A comprehensive review and bibliometric analysis. Aquaculture 2021, 736881. [Google Scholar] [CrossRef]
- Dawood, M.A.O.; Eweedah, N.M.; Khalafalla, M.M.; Khalid, A. Evaluation of fermented date palm seed meal with Aspergillus oryzae on the growth, digestion capacity and immune response of Nile tilapia (Oreochromis niloticus). Aquac. Nutr. 2020, 26, 828–841. [Google Scholar] [CrossRef]
- Tezzo, X.; Bush, S.R.; Oosterveer, P.; Belton, B. Food system perspective on fisheries and aquaculture development in Asia. Agric. Hum. Values 2021, 38, 73–90. [Google Scholar] [CrossRef]
- Dawood, M.; Eweedah, N.M.; Moustafa, E.M.; El-Sharawy, M.E.; Soliman, A.A.; Amer, A.A.; Atia, M.H. Copper Nanoparticles Mitigate the Growth, Immunity, and Oxidation Resistance in Common Carp (Cyprinus carpio). Biol. Trace Elem. Res. 2020, 198, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Ciji, A.; Akhtar, M.S. Stress management in aquaculture: A review of dietary interventions. Rev. Aquac. 2021. [Google Scholar] [CrossRef]
- McArley, T.J.; Sandblom, E.; Herbert, N.A. Fish and hyperoxia—From cardiorespiratory and biochemical adjustments to aquaculture and ecophysiology implications. Fish Fish. 2021, 22, 324–355. [Google Scholar] [CrossRef]
- Erkinharju, T.; Dalmo, R.A.; Hansen, M.; Seternes, T. Cleaner fish in aquaculture: Review on diseases and vaccination. Rev. Aquac. 2021, 13, 189–237. [Google Scholar] [CrossRef]
- Dawood, M. Nutritional immunity of fish intestines: Important insights for sustainable aquaculture. Rev. Aquac. 2021, 13, 642–663. [Google Scholar] [CrossRef]
- Dawood, M.A.; Noreldin, A.E.; Sewilam, H. Long term salinity disrupts the hepatic function, intestinal health, and gills antioxidative status in Nile tilapia stressed with hypoxia. Ecotoxicol. Environ. Saf. 2021, 220, 112412. [Google Scholar] [CrossRef]
- Caipang, C.M.A.; Suharman, I.; Avillanosa, A.L.; Gonzales-Plasus, M.M. Influence of Phytogenic Feed Additives on The Health Status in The Gut and Disease Resistance of Cultured Fish. IOP Conf. Ser. Earth Environ. Sci. 2021, 695, 012024. [Google Scholar] [CrossRef]
- Raza, S.H.A.; Naqvi, S.R.Z.; Abdelnour, S.A.; Schreurs, N.; Mohammedsaleh, Z.M.; Khan, I.; Shater, A.F.; El-Hack, M.E.A.; Khafaga, A.F.; Quan, G.; et al. Beneficial effects and health benefits of Astaxanthin molecules on animal production: A review. Res. Vet. Sci. 2021, 138, 69–78. [Google Scholar] [CrossRef] [PubMed]
- Ahmadifar, E.; Yousefi, M.; Karimi, M.; Raieni, R.F.; Dadar, M.; Yilmaz, S.; Dawood, M.A.O.; Abdel-Latif, H.M. Benefits of Dietary Polyphenols and Polyphenol-Rich Additives to Aquatic Animal Health: An Overview. Rev. Fish. Sci. Aquac. 2020, 1–34. [Google Scholar] [CrossRef]
- Zhu, F. A review on the application of herbal medicines in the disease control of aquatic animals. Aquaculture 2020, 526, 735422. [Google Scholar] [CrossRef]
- Hernández-Contreras, Á.; Hernández, M.D. Chapter 14—Application of aromatic plants and their extracts in aquaculture. In Feed Additives; Florou-Paneri, P., Christaki, E., Giannenas, I., Eds.; Academic Press: Cambridge, MA, USA, 2020; pp. 239–259. [Google Scholar]
- Hayatgheib, N.; Moreau, E.; Calvez, S.; Lepelletier, D.; Pouliquen, H. A review of functional feeds and the control of Aeromonas infections in freshwater fish. Aquac. Int. 2020, 28, 1083–1123. [Google Scholar] [CrossRef]
- Anastasiou, T.I.; Mandalakis, M.; Krigas, N.; Vézignol, T.; Lazari, D.; Katharios, P.; Dailianis, T.; Antonopoulou, E. Comparative Evaluation of Essential Oils from Medicinal-Aromatic Plants of Greece: Chemical Composition, Antioxidant Capacity and Antimicrobial Activity against Bacterial Fish Pathogens. Molecules 2019, 25, 148. [Google Scholar] [CrossRef] [PubMed]
- Ganeva, V.O.; Korytář, T.; Pecková, H.; McGurk, C.; Mullins, J.; Yanes-Roca, C.; Gela, D.; Lepič, P.; Policar, T.; Holzer, A.S. Natural feed additives modulate immunity and mitigate infection with Sphaerospora molnari (myxozoa: Cnidaria) in common carp: A pilot study. Pathogens 2020, 9, 1013. [Google Scholar] [CrossRef] [PubMed]
- Xu, A.; Shang-Guan, J.; Li, Z.; Gao, Z.; Huang, Y.C.; Chen, Q. Effects of dietary Chinese herbal medicines mixture on feeding attraction activity, growth performance, nonspecific immunity and digestive enzyme activity of Japanese seabass (Lateolabrax japonicus). Aquac. Rep. 2020, 17, 100304. [Google Scholar] [CrossRef]
- Lee, P.-T.; Chen, H.-Y.; Liao, Z.-H.; Huang, H.-T.; Chang, T.-C.; Huang, C.-T.; Lee, M.-C.; Nan, F.-H. Effects of three medicinal herbs Bidens pilosa, Lonicera japonica, and Cyathula officinalis on growth and non-specific immune responses of cobia (Rachycentron canadum). Fish Shellfish Immunol. 2020, 106, 526–535. [Google Scholar] [CrossRef]
- Ebrahimi, E.; Haghjou, M.; Nematollahi, A.; Goudarzian, F. Effects of rosemary essential oil on growth performance and hematological parameters of young great sturgeon (Huso huso). Aquaculture 2020, 521, 734909. [Google Scholar] [CrossRef]
- Salomón, R.; Firmino, J.P.; Reyes-López, F.E.; Andree, K.B.; González-Silvera, D.; Esteban, M.A.; Tort, L.; Quintela, J.C.; Pinilla-Rosas, J.M.; Vallejos-Vidal, E.; et al. The growth promoting and immunomodulatory effects of a medicinal plant leaf extract obtained from Salvia officinalis and Lippia citriodora in gilthead seabream (Sparus aurata). Aquaculture 2020, 524, 735291. [Google Scholar] [CrossRef]
- El-Hack, M.E.A.; Alagawany, M.; Abdel-Moneim, A.-M.E.; Mohammed, N.G.; Khafaga, A.F.; Bin-Jumah, M.; Othman, S.I.; Allam, A.A.; ElNesr, S.S. Cinnamon (Cinnamomum zeylanicum) Oil as a Potential Alternative to Antibiotics in Poultry. Antibiotics 2020, 9, 210. [Google Scholar] [CrossRef] [PubMed]
- Heshmati, J.; Sepidarkish, M.; Morvaridzadeh, M.; Farsi, F.; Tripathi, N.; Razavi, M.; Rezaeinejad, M. The effect of cinnamon supplementation on glycemic control in women with polycystic ovary syndrome: A systematic review and meta-analysis. J. Food Biochem. 2021, 45, e13543. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.-C.; Wu, S.-J.; Chang, C.-H.; Ng, L.-T.; Huang, L.-T. Antioxidant activity of Cinnamomum cassia. Phytother. Res. 2003, 17, 726–730. [Google Scholar] [CrossRef]
- Gruenwald, J.; Freder, J.; Armbruester, N. Cinnamon and Health. Crit. Rev. Food Sci. Nutr. 2010, 50, 822–834. [Google Scholar] [CrossRef] [PubMed]
- Charles, D.J. Antioxidant Properties of Spices, Herbs and Other Sources; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 2012. [Google Scholar]
- Ahmad, M.H.; El Mesallamy, A.M.D.; Samir, F.; Zahran, F. Effect of Cinnamon (Cinnamomum zeylanicum) on Growth Performance, Feed Utilization, Whole-Body Composition, and Resistance toAeromonas hydrophilain Nile Tilapia. J. Appl. Aquac. 2011, 23, 289–298. [Google Scholar] [CrossRef]
- Zhou, Y.; Jiang, W.-D.; Zhang, J.-X.; Feng, L.; Wu, P.; Liu, Y.; Jiang, J.; Kuang, S.-Y.; Tang, L.; Peng, Y.; et al. Cinnamaldehyde improves the growth performance and digestion and absorption capacity in grass carp (Ctenopharyngodon idella). Fish Physiol. Biochem. 2020, 46, 1589–1601. [Google Scholar] [CrossRef]
- Ravardshiri, M.; Bahram, S.; Javadian, S.R.; Bahrekazemi, M. Cinnamon Promotes Growth Performance, Digestive Enzyme, Blood Parameters, and Antioxidant Activity of Rainbow Trout (Oncorhynchus mykiss) in Low-Carbohydrate Diets. Turk. J. Fish. Aquat. Sci. 2021, 21, 309–322. [Google Scholar] [CrossRef]
- Abdel-Tawwab, M.; Samir, F.; El-Naby, A.S.A.; Monier, M.N. Antioxidative and immunostimulatory effect of dietary cinnamon nanoparticles on the performance of Nile tilapia, Oreochromis niloticus (L.) and its susceptibility to hypoxia stress and Aeromonas hydrophila infection. Fish Shellfish Immunol. 2018, 74, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Basto, A.; Matos, E.; Valente, L.M. Nutritional value of different insect larvae meals as protein sources for European sea bass (Dicentrarchus labrax) juveniles. Aquaculture 2020, 521, 735085. [Google Scholar] [CrossRef]
- Ashry, A.; Hassan, A.; Habiba, M.; El-Zayat, A.; El-Sharnouby, M.; Sewilam, H.; Dawood, M. The Impact of Dietary Curcumin on the Growth Performance, Intestinal Antibacterial Capacity, and Haemato-Biochemical Parameters of Gilthead Seabream (Sparus aurata). Animals 2021, 11, 1779. [Google Scholar] [CrossRef] [PubMed]
- Association of Official Analytical Chemists (AOAC). Official Methods of Analysis of Official Analytical Chemists International, 16th ed.; AOAC: Washington, DC, USA, 1998. [Google Scholar]
- Houston, A. Blood and Circulation/Methods for Fish Biology; American Fisheries Society: New York, NY, USA, 1990. [Google Scholar]
- Blaxhall, P.C.; Daisley, K.W. Routine haematological methods for use with fish blood. J. Fish Biol. 1973, 5, 771–781. [Google Scholar] [CrossRef]
- Dacie, J.V.; Lewis, S.M. Practical Hematology, 6th ed.; Churchill Livingstone: London, UK, 1999. [Google Scholar]
- Reitman, S.; Frankel, S. A colorimeter method for determination of serum glutamic oxaloacetic acid and glutamic pyruvic acid transferases. Am. J. Clin. Pathol. 1957, 28, 56–63. [Google Scholar] [CrossRef]
- Kid, P.; King, E. Colorimetric determination of alkaline phosphatase activity. J. Clin. Pathol. 1954, 6, 322. [Google Scholar]
- Lugo, J.M.; Rodriguez, A.; Helguera, Y.; Morales, R.; Gonzalez, O.; Acosta, J.; Besada, V.; Sanchez, A.; Estrada, M.P. Recombinant novel pituitary adenylate cyclase-activating polypeptide from African catfish (Clarias gariepinus) authenticates its biological function as a growth-promoting factor in low vertebrates. J. Endocrinol. 2008, 197, 583–597. [Google Scholar] [CrossRef]
- Cai, W.-Q.; Li, S.-F.; Ma, J.-Y. Diseases resistance of Nile tilapia (Oreochromis niloticus), blue tilapia (Oreochromis aureus) and their hybrid (female Nile tilapia × male blue tilapia) to Aeromonas sobria. Aquaculture 2004, 229, 79–87. [Google Scholar] [CrossRef]
- Kawahara, E.; Ueda, T.; Nomura, S. In Vitro Phagocytic Activity of White-Spotted Char Blood Cells after Injection with Aeromonas salmonicida Extracellular Products. Fish Pathol. 1991, 26, 213–214. [Google Scholar] [CrossRef]
- Demers, N.E.; Bayne, C.J. The immediate effects of stress on hormones and plasma lysozyme in rainbow trout. Dev. Comp. Immunol. 1997, 21, 363–373. [Google Scholar] [CrossRef]
- Kousoulaki, K.; Saether, B.-S.; Albrektsen, S.; Noble, C.; Sæther, B. Review on European sea bass (Dicentrarchus labrax, Linnaeus, 1758) nutrition and feed management: A practical guide for optimizing feed formulation and farming protocols. Aquac. Nutr. 2015, 21, 129–151. [Google Scholar] [CrossRef]
- Amiza, M.; Zakiah, J.; Ng, L.; Lai, K. Fermentation of tempoyak using isolated tempoyak culture. Res. J. Microbiol. 2010, 5, 903–914. [Google Scholar]
- Mehdipour, Z.; Afsharmanesh, M. Evaluation of synbiotic and cinnamon (Cinnamomum verum) as antibiotic growth promoter substitutions on growth performance, intestinal microbial populations and blood parameters in Japanese quail. J. Livest. Sci. Technol. 2018, 6, 1–8. [Google Scholar]
- Kot, B.; Kwiatek, K.; Janiuk, J.; Witeska, M.; Pękala-Safińska, A. Antibacterial Activity of Commercial Phytochemicals against Aeromonas Species Isolated from Fish. Pathogens 2019, 8, 142. [Google Scholar] [CrossRef]
- Yang, Y.-F.; Zhao, L.-L.; Shao, Y.-X.; Liao, X.-D.; Zhang, L.-Y.; Lu, L.; Luo, X.-G. Effects of dietary graded levels of cinnamon essential oil and its combination with bamboo leaf flavonoid on immune function, antioxidative ability and intestinal microbiota of broilers. J. Integr. Agric. 2019, 18, 2123–2132. [Google Scholar] [CrossRef]
- Bennick, A. Interaction of Plant Polyphenols with Salivary Proteins. Crit. Rev. Oral Biol. Med. 2002, 13, 184–196. [Google Scholar] [CrossRef]
- Takasao, N.; Tsuji-Naito, K.; Ishikura, S.; Tamura, A.; Akagawa, M. Cinnamon extract promotes type i collagen biosynthesis via activation of igf-i signaling in human dermal fibroblasts. J. Agric. Food Chem. 2012, 60, 1193–1200. [Google Scholar] [CrossRef] [PubMed]
- Hlebowicz, J.; Hlebowicz, A.; Lindstedt, S.; Björgell, O.; Höglund, P.; Holst, J.J.; Darwiche, G.; Almér, L.-O. Effects of 1 and 3 g cinnamon on gastric emptying, satiety, and postprandial blood glucose, insulin, glucose-dependent insulinotropic polypeptide, glucagon-like peptide 1, and ghrelin concentrations in healthy subjects. Am. J. Clin. Nutr. 2009, 89, 815–821. [Google Scholar] [CrossRef] [PubMed]
- Dawood, M.; El Basuini, M.; Zaineldin, A.; Yilmaz, S.; Hasan, T.; Ahmadifar, E.; El Asely, A.; Abdel-Latif, H.; Alagawany, M.; Abu-Elala, N.; et al. Antiparasitic and Antibacterial Functionality of Essential Oils: An Alternative Approach for Sustainable Aquaculture. Pathogens 2021, 10, 185. [Google Scholar] [CrossRef]
- Dawood, M.A.; Noreldin, A.E.; Ali, M.A.; Sewilam, H. Menthol essential oil is a practical choice for intensifying the production of Nile tilapia (Oreochromis niloticus): Effects on the growth and health performances. Aquaculture 2021, 543, 737027. [Google Scholar] [CrossRef]
- Goel, B.; Mishra, S. Medicinal and Nutritional Perspective of Cinnamon: A Mini-Review. Eur. J. Med. Plants 2020, 10–16. [Google Scholar] [CrossRef]
- Dalsgaard, A. The occurrence of human pathogenic Vibrio spp. and Salmonella in aquaculture*. Int. J. Food Sci. Technol. 1998, 33, 127–138. [Google Scholar] [CrossRef]
- Hammer, K.A.; Carson, C.; Riley, T.V. Antimicrobial activity of essential oils and other plant extracts. J. Appl. Microbiol. 1999, 86, 985–990. [Google Scholar] [CrossRef]
- Chang, S.-T.; Chen, P.-F.; Chang, S.-C. Antibacterial activity of leaf essential oils and their constituents from cinnamomum osmophloeum. J. Ethnopharmacol. 2001, 77, 123–127. [Google Scholar] [CrossRef]
- Tiihonen, K.; Kettunen, H.; Bento, M.; Saarinen, M.; Lahtinen, S.; Ouwehand, A.; Schulze, H.; Rautonen, N. The effect of feeding essential oils on broiler performance and gut microbiota. Br. Poult. Sci. 2010, 51, 381–392. [Google Scholar] [CrossRef]
- Jamroz, D.; Wertelecki, T.; Houszka, M.; Kamel, C. Influence of diet type on the inclusion of plant origin active substances on morphological and histochemical characteristics of the stomach and jejunum walls in chicken. J. Anim. Physiol. Anim. Nutr. 2006, 90, 255–268. [Google Scholar] [CrossRef] [PubMed]
- Zemheri-Navruz, F.; Acar, Ü.; Yılmaz, S. Dietary supplementation of olive leaf extract increases haematological, serum biochemical parameters and immune related genes expression level in common carp (Cyprinus carpio) juveniles. Fish Shellfish Immunol. 2019, 89, 672–676. [Google Scholar] [CrossRef]
- Dawood, M.A.O.; Ali, M.F.; Amer, A.A.; Gewaily, M.S.; Mahmoud, M.M.; Alkafafy, M.; Assar, D.H.; Soliman, A.A.; Van Doan, H. The influence of coconut oil on the growth, immune, and antioxidative responses and the intestinal digestive enzymes and histomorphometry features of Nile tilapia (Oreochromis niloticus). Fish Physiol. Biochem. 2021, 1–12. [Google Scholar] [CrossRef]
- Fazio, F. Fish hematology analysis as an important tool of aquaculture: A review. Aquaculture 2019, 500, 237–242. [Google Scholar] [CrossRef]
- Rodgers, M.S.; Chang, C.-C.; Kass, L. Elliptocytes and tailed poikilocytes correlate with severity of iron-deficiency anemia. Am. J. Clin. Pathol. 1999, 111, 672–675. [Google Scholar] [CrossRef]
- Ahmadifar, E.; Moghadam, M.S.; Dawood, M.; Hoseinifar, S.H. Lactobacillus fermentum and/or ferulic acid improved the immune responses, antioxidative defence and resistance against Aeromonas hydrophila in common carp (Cyprinus carpio) fingerlings. Fish Shellfish Immunol. 2019, 94, 916–923. [Google Scholar] [CrossRef] [PubMed]
- Shourbela, R.M.; El-Hawarry, W.N.; Elfadadny, M.R.; Dawood, M.A. Oregano essential oil enhanced the growth performance, immunity, and antioxidative status of Nile tilapia (Oreochromis niloticus) reared under intensive systems. Aquaculture 2021, 542, 736868. [Google Scholar] [CrossRef]
- Dawood, M.A.; Gewaily, M.S.; Monier, M.N.; Younis, E.M.; Van Doan, H.; Sewilam, H. The regulatory roles of yucca extract on the growth rate, hepato-renal function, histopathological alterations, and immune-related genes in common carp exposed with acute ammonia stress. Aquaculture 2021, 534, 736287. [Google Scholar] [CrossRef]
- Magnadóttir, B. Innate immunity of fish (overview). Fish Shellfish Immunol. 2006, 20, 137–151. [Google Scholar] [CrossRef] [PubMed]
| Ingredients (g/kg) | Cinnamon (g/kg) | |||
|---|---|---|---|---|
| 0 | 10 | 15 | 20 | |
| Fish meal (67%) | 400 | 400 | 400 | 400 |
| Shrimp meal | 110 | 110 | 110 | 110 |
| Soybean (48%) | 150 | 150 | 150 | 150 |
| Yellow corn (7.5%) | 50 | 50 | 50 | 50 |
| Corn gluten (60%) | 50 | 50 | 50 | 50 |
| Wheat flour | 100 | 90 | 85 | 80 |
| Fish oil | 100 | 100 | 100 | 100 |
| Di-calcium phosphate | 10 | 10 | 10 | 10 |
| Vitamin and mineral premix 1 | 30 | 30 | 30 | 30 |
| Cinnamon | 0 | 10 | 15 | 20 |
| Total | 1000 | 1000 | 1000 | 1000 |
| Chemical analysis | ||||
| Dry matter (%) | 91.9 | 91.12 | 91.42 | 91.35 |
| Crude protein (%) | 46 | 45.99 | 46 | 45.99 |
| Crude lipids (%) | 15.28 | 15.3 | 15.33 | 15.37 |
| Ash (%) | 10.43 | 10.39 | 10.54 | 10.6 |
| Crude fiber (%) | 2.19 | 2.16 | 2.19 | 2.17 |
| Nitrogen free extract | 26.09 | 26.15 | 25.94 | 25.87 |
| Gross energy (Kcal/100 g) 2 | 511.83 | 512.21 | 512.76 | 511.67 |
| Item | Cinnamon (g/kg) | |||
|---|---|---|---|---|
| 0 | 10 | 15 | 20 | |
| Initial weight (g) | 7.22 ± 0.18 | 6.96 ± 0.09 | 7.05 ± 0.12 | 7.09 ± 0.38 |
| Final weight (g) | 38.84 ± 0.65 c | 51.29 ± 1.45 a | 42.25 ± 0.56 b | 38.01 ± 1.14 c |
| Weight gain (g) | 31.62 ± 0.72 c | 44.33 ± 1.47 a | 35.20 ± 0.64 b | 30.92 ± 1.05 c |
| Specific growth rate (%/day) | 1.87 ± 0.04 b | 2.22 ± 0.04 a | 1.99 ± 0.03 b | 1.87 ± 0.06 b |
| Feed intake (g/fish) | 63.92 ± 1.48 | 60.63 ± 1.20 | 58.15 ± 1.79 | 62.45 ± 1.66 |
| Feed conversion ratio | 2.02 ± 0.00 a | 1.37 ± 0.02 c | 1.65 ± 0.05 b | 2.02 ± 0.03 a |
| Protein efficiency ratio | 1.08 ± 0.00 c | 1.59 ± 0.03 a | 1.32 ± 0.04 b | 1.08 ± 0.01 c |
| Survival (%) | 91.67 ± 4.41 | 91.67 ± 4.41 | 93.33 ± 1.67 | 91.67 ± 4.41 |
| Item | Initial | Cinnamon (g/kg) | |||
|---|---|---|---|---|---|
| 0 | 10 | 15 | 20 | ||
| Dry matter (%) | 33.01 ± 1.12 | 33.72 ± 0.13 | 34.22 ± 0.21 | 33.63 ± 0.16 | 33.52 ± 0.13 |
| Crude protein (%) | 52.77 ± 0.23 | 53.58 ± 0.32 | 54.08 ± 0.18 | 53.78 ± 0.27 | 53.42 ± 0.15 |
| Lipids (%) | 25.77 ± 0.64 | 27.78 ± 0.19 * | 27.55 ± 0.16 * | 27.97 ± 0.33 * | 28.08 ± 0.13 * |
| Ash (%) | 16.76 ± 0.13 | 16.73 ± 0.25 | 16.83 ± 0.26 | 16.84 ± 0.34 | 17.18 ± 0.23 |
| Item | Cinnamon (g/kg) | |||
|---|---|---|---|---|
| 0 | 10 | 15 | 20 | |
| Hemoglobin (g/dL) | 8.99 ± 0.26 b | 11.24 ± 0.36 a | 9.91 ± 0.71 ab | 10.86 ± 0.31 ab |
| Hematocrit (%) | 41.23 ± 0.44 c | 43.98 ± 0.41 a | 45.85 ± 1.14 b | 46.81 ± 0.93 ab |
| Red blood cells (×106/mm3) | 3.31 ± 0.04 b | 4.30 ± 0.08 a | 3.87 ± 0.26 a | 4.13 ± 0.11 a |
| MCV (µm3/cell) | 124.56 ± 1.67 | 102.28 ± 0.31 | 118.48 ± 0.47 | 113.52 ± 0.34 |
| MCH (pg/cell) | 27.16 ± 0.19 | 26.14 ± 0.68 | 25.61 ± 0.40 | 26.29 ± 0.55 |
| MCHC (mg/dL) | 21.81 ± 0.11 | 25.56 ± 0.01 | 21.61 ± 0.25 | 23.21 ± 0.35 |
| White blood cells (×103/mm3) | 26.319 ± 134.14 c | 28.243 ± 146.07 a | 27.46 ± 118.31 b | 27.60 ± 6.61 b |
| Item | Cinnamon (g/kg) | |||
|---|---|---|---|---|
| 0 | 10 | 15 | 20 | |
| ALT (U/I) | 82.22 ± 0.10 | 82.57 ± 0.07 | 83.24 ± 0.16 | 81.49 ± 0.17 |
| AST (U/I) | 80.00 ± 0.58 | 80.33 ± 0.67 | 80.67 ± 0.88 | 79.33 ± 0.67 |
| ALP (U/I) | 68.99 ± 0.84 | 74.53 ± 0.71 | 71.79 ± 0.67 | 69.30 ± 0.49 |
| Total lipids (g/dL) | 933.67 ± 4.37 c | 952.67 ± 3.76 b | 971.67 ± 2.03 a | 960.00 ± 2.65 b |
| Urea (mg/dL) | 4.54 ± 0.04 | 4.56 ± 0.15 | 4.66 ± 0.08 | 4.33 ± 0.13 |
| Creatinine | 0.61 ± 0.05 | 0.62 ± 0.05 | 0.63 ± 0.02 | 0.50 ± 0.04 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Habiba, M.M.; Hussein, E.E.; Ashry, A.M.; El-Zayat, A.M.; Hassan, A.M.; El-Shehawi, A.M.; Sewilam, H.; Van Doan, H.; Dawood, M.A.O. Dietary Cinnamon Successfully Enhanced the Growth Performance, Growth Hormone, Antibacterial Capacity, and Immunity of European Sea Bass (Dicentrarchus labrax). Animals 2021, 11, 2128. https://doi.org/10.3390/ani11072128
Habiba MM, Hussein EE, Ashry AM, El-Zayat AM, Hassan AM, El-Shehawi AM, Sewilam H, Van Doan H, Dawood MAO. Dietary Cinnamon Successfully Enhanced the Growth Performance, Growth Hormone, Antibacterial Capacity, and Immunity of European Sea Bass (Dicentrarchus labrax). Animals. 2021; 11(7):2128. https://doi.org/10.3390/ani11072128
Chicago/Turabian StyleHabiba, Mahmoud M., Ebtehal E. Hussein, Ahmed M. Ashry, Ahmed M. El-Zayat, Aziza M. Hassan, Ahmed M. El-Shehawi, Hani Sewilam, Hien Van Doan, and Mahmoud A.O. Dawood. 2021. "Dietary Cinnamon Successfully Enhanced the Growth Performance, Growth Hormone, Antibacterial Capacity, and Immunity of European Sea Bass (Dicentrarchus labrax)" Animals 11, no. 7: 2128. https://doi.org/10.3390/ani11072128
APA StyleHabiba, M. M., Hussein, E. E., Ashry, A. M., El-Zayat, A. M., Hassan, A. M., El-Shehawi, A. M., Sewilam, H., Van Doan, H., & Dawood, M. A. O. (2021). Dietary Cinnamon Successfully Enhanced the Growth Performance, Growth Hormone, Antibacterial Capacity, and Immunity of European Sea Bass (Dicentrarchus labrax). Animals, 11(7), 2128. https://doi.org/10.3390/ani11072128







