A Case of Concurrent Molybdenosis, Secondary Copper, Cobalt and Selenium Deficiency in a Small Sheep Herd in Northern Germany
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Case Presentation
2.2. Clinical Examination
2.3. Farm Visit
2.4. Further Diagnostics
2.4.1. Blood and Liver Samples
2.4.2. Faecal Samples
2.4.3. Post Mortem Investigation
2.4.4. Nutritional Assessment
3. Results
3.1. Clinical Examination
3.2. Farm Visit
3.3. Further Diagnostics
3.3.1. Blood and Liver Samples
3.3.2. Faecal Samples
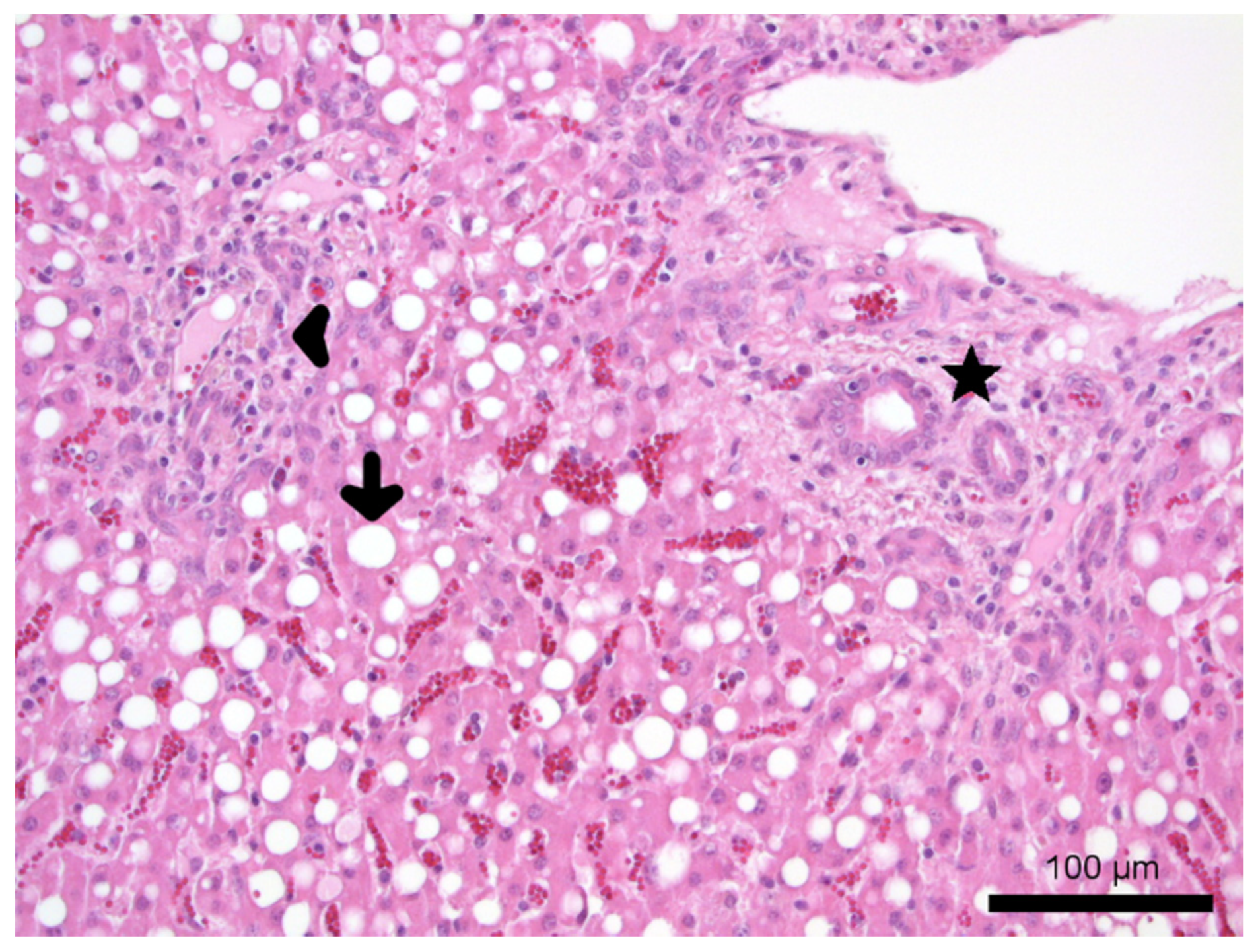
3.3.3. Post Mortem Investigation
3.3.4. Nutritional Assessment
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sardesai, V.M. Molybdenum: An essential trace element. Nutr. Clin. Pract. 1993, 8, 277–281. [Google Scholar] [CrossRef]
- Gooneratne, S.R.; Buckley, W.T.; Christensen, D.A. Review of Copper deficiency and metabolism in ruminants. Can. J. Anim. Sci. 1989, 69, 819–845. [Google Scholar] [CrossRef]
- Majak, W.; Steinke, D.; Lysyk, T.; Ogilvie, K.; McGillivray, J. Efficacy of copper supplementation in the prevention of molybdenosis in cattle. Rangel. Ecol. Manag. 2006, 59, 285–292. [Google Scholar] [CrossRef]
- Allen, J.D.; Gawthrone, J.M. Involvement of the solid phase rumen digesta in the interaction between copper, molybdenum and sulfur in sheep. Br. J. Nutr. 1987, 58, 265–276. [Google Scholar] [CrossRef] [PubMed]
- Gardner, W.C.; Broersma, K.; Popp, J.D.; Mir, Z.; Mir, P.S.; Buckley, W.T. Copper and health status of grazing high-molybdenum forage from reclaimed mining tailing site. Can. J. Anim. Sci. 2003, 83, 479–485. [Google Scholar] [CrossRef]
- National Research Council. Mineral Tolerance of Domestic Animals; National Academy Press: Washington, DC, USA, 1980. [Google Scholar]
- Laiblin, C.; Stöber, M. Fütterungs-, stoffwechsel-, mangel- und vergiftungs-bedingte Krankheiten mit Beteiligung mehrerer Organsysteme. In Innere Medizin und Chirurgie des Rindes, 5th ed.; Dirksen, G., Gründer, H.D., Stöber, M., Eds.; Parey in MVS Medizinverlage: Stuttgart, Germany, 2006; pp. 1266–1272. [Google Scholar]
- O’Connor, G.A.; Brobst, R.B.; Chaney, R.L.; Kincaid, R.L.; McDowell, L.R.; Pierzynski, G.M.; Rubin, A.; Van Riper, G.G. A modified risk assessment to establish molybdenum standards for land application of biosolids. J. Environ. Qual. 2001, 30, 1490–1507. [Google Scholar] [CrossRef] [PubMed]
- Miltimore, J.E.; Mason, J.L. Copper to molybdenum ratio and molybdenum and copper concentrations in ruminant feeds. Can. J. Anim. Sci. 1971, 51, 193–200. [Google Scholar] [CrossRef]
- National Research Council. Nutritient Requirements of Beef Cattle, 7th ed.; National Academy Press: Washington, DC, USA, 2000. [Google Scholar]
- Mills, C.F.; Davis, G.K. Molybdenum. In Trace Elements in Human and Animal Nutrition; Academic Press Inc.: London, UK, 1987; Volume 1, pp. 429–463. [Google Scholar]
- Raisbeck, M.F.; Siemion, R.S.; Smith, M.A. Modest copper supplementation blocks molybdenosis in cattle. J. Vet. Diagn. Invest. 2006, 18, 566–572. [Google Scholar] [CrossRef] [PubMed]
- Humphries, W.R. Control of hypocupremia in cattle by addition of copper to water supplies. Vet. Rec. 1980, 106, 359–362. [Google Scholar] [CrossRef]
- Frank, A. ‘Mysterious’ moose disease in Sweden. Similarities to copper deficiency and/or molybdenosis in cattle and sheep. Biochemical background of clinical signs and organ lesions. Sci. Total Environ. 1998, 209, 17–26. [Google Scholar] [CrossRef]
- Howell, J.M. Nutrition and the nervous system in farm animals. World Rev. Nutr. Diet. 1970, 12, 377–412. [Google Scholar] [PubMed]
- Buck, B.C.; Ulrich, R.; Taube, V.; Jacobsen, B.; Ganter, M. Osteopenie in Folge eines Kupfermangels bei einer zwergwüchsigen Thüringerwald Ziege. Tierärztl Prax 2012, 40, 45–52. [Google Scholar]
- Underwood, E.J. Mineral imbalances in farm animals and their study and diagnosis with isotopic tracers. Energy Rev. 1976, 14, 591–619. [Google Scholar]
- Humann, E. Investigations of Pathogenesis and Diagnostic of Chronic Copper Poisoning in Sheep. Ph.D. Thesis, University of Vet Med Hannover, Hanover, Germany, 1997. [Google Scholar]
- López-Alonso, M.; Miranda, M. Copper Supplementation, a Challenge in Cattle. Animals 2020, 10, 1890. [Google Scholar] [CrossRef] [PubMed]
- Humann-Ziehank, E.; Coenen, M.; Ganter, M.; Bickhardt, K. Long-Term Observation of Subclinical Chronic Copper Poisoning in Two Sheep Breeds. J. Vet. Med. A 2001, 48, 429–439. [Google Scholar] [CrossRef]
- Martinovich, D. Sheep diseases in Northland associated with suspected toxic forage. In Proceedings of the New Zealand Veterinary Association Sheep Society; Annual Seminar 1974, Volume, Jan 1974; The Society of Sheep and Beef Cattle Veterinarians of the New Zealand Veterinary Association: Wellington, New Zealand, 1974; p. 99. [Google Scholar]
- Smith, R.A.; Marston, H.R. Production, absorption and excretion of vitamin B12 in sheep. Br. J. Nutr. 1970, 24, 857–877. [Google Scholar] [CrossRef]
- Ulvund, M.J.; Pestalozzi, M. Ovine white-liver disease (OWLD) in Norway: Clinical symptoms and preventive measures. Acta Vet. Scan. 1990, 31, 53–62. [Google Scholar] [CrossRef]
- Vellema, P.; Moll, L.; Barkema, H.W.; Schukken, Y.H. Effect of cobalt supplementation on serum vitamin B12 levels, weight gain and survival rate in lambs grazing cobalt-deficient pastures. Vet. Quart. 1997, 19, 1–5. [Google Scholar] [CrossRef]
- Sargison, N.D.; Scott, P.R.; Wilson, D.J.; Bell, G.J.C.; Mauchline, S.; Rhind, S.M. Hepatic encephalopathy associated with cobalt deficiency and white liver disease in lambs. Vet. Rec. 2001, 149, 770–772. [Google Scholar] [CrossRef]
- Rice, D.A.; McLoughlin, M.; Blachflower, W.J.; McMurray, C.H.; Goodall, E.A. Sequential changes in plasma methylmalonic acid and vitamin B12 in sheep eating cobalt-deficient grass. Biol. Trace Elem. Res. 1989, 22, 153–164. [Google Scholar] [CrossRef]
- Mohammed, R.; Lamand, M. Cardivascular lesions in cobalt–vitamin B12 deficient sheep. Ann. Vet. Res. 1986, 17, 447–450. [Google Scholar]
- MacPherson, A.; Moon, F.E.; Voss, R.C. Biochemical aspects of cobalt deficiency in sheep with special reference to vitamin status and a possible involvement in the aetiology of cerebrocortical necrosis. Br. Vet. J. 1976, 132, 294–308. [Google Scholar] [CrossRef]
- Lee, H.J.; Marston, H.R. Requirement for cobalt of sheep grazed on cobalt-deficient pastures. Aust. J. Agric. Res. 1969, 20, 905–918. [Google Scholar] [CrossRef]
- Martson, H.R.; Allen, S.H.; Smith, R.H. Primary metablic defect supervening on vitamin B12 deficiency in sheep. Nature 1961, 190, 1085–1092. [Google Scholar]
- Gawthrone, J.M. The excretion of methylmalonic and formiminoglutamic acids during the induction and remission of vitamin B12 deficiency in sheep. Aust. J. Biol. Sci. 1968, 21, 789–794. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ulvund, M.J. Ovine white-liver disease (OWLD). Pathology. Acta Vet. Scand. 1990, 31, 309–324. [Google Scholar] [CrossRef]
- Muth, O.H.; Schubert, J.R.; Oldfield, J.E. White muscle disease (myopathy) in lambs and calves. VII. Etiology and prophylaxis. Am. J. Vet. Res. 1961, 22, 466–469. [Google Scholar]
- Andrews, E.D.; Hartley, W.J.; Grant, A.B. Se-responsive diseases if animals in New Zealand. N. Z. Vet. J. 1968, 16, 3–17. [Google Scholar] [CrossRef]
- Bostedt, H. Serumenzymatische Untersuchungen bei Lämmern im Alter von 10–30 Tagen; gleichzeitig ein Beitrag zur Prophylaxe der enzootischen Muskeldystrophie. Berl. Münch. Tierärztl. Wschr. 1976, 89, 169–174. [Google Scholar]
- Bickardt, K.; König, G. Blutmesswerte von gesunden Mutterschafen der Merino- und Schwarzkopfrasse zur Zeit der Geburt (Referenzwerte). Dtsch Tierärztl. Wschr. 1999, 106, 445–451. [Google Scholar]
- Humann, E.; Risse, R.; Bruegmann, M.; Henze, P.; Ganter, M. Liver biopsy in sheep: Experiences with two different techniques. Tierärztl. Umsch. 1999, 54, 151–157. [Google Scholar]
- Humann-Ziehank, E.; Tegtmeyer, P.; Seelig, B.; Roehrig, P.; Ganter, M. Variation of serum selenium concentrations in German sheep flocks and implications for herd health management consultancy. Acta Vet. Scan. 2013, 55, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Benedek, L. Allatorv. Lapok 1943, 66, 139. [Google Scholar]
- Naumann, C.; Bassler, R. Methoden der landwirtschaftlichen Forschungs- und Untersuchungsanstalt, Biochemische Untersuchung von Futtermitteln, Methodenbuch 3 (Einschließlich der Achten Ergänzungen); VDLUFA: Darmstadt, Germany, 2012. [Google Scholar]
- Kamphues, J.; Coenen, M.; Wolf, P.; Liesegang, A.; Eder, K.; Männer, K.; Iben, C.; Zebeli, Q.; Kienzle, E.; Zentek, J. Supplemente zur Tierernährung für Studium und Praxis; M. & H. Schaper GmbH: Hannover, Germany, 2014; pp. 232, 294–295. [Google Scholar]
- El-Wahab, A.A.; Visscher, C.; Teitge, F.; Steinhagen, D. Choice preference of diets with different protein levels depending on water temperature in Nile tilapia. J. World Aquacult. Soc. 2020, 51, 512–526. [Google Scholar] [CrossRef]
- Lineva, A.; Kirchner, R.; Kienzle, E.; Kamphues, J.; Dobenecker, B. A pilot study on in vitro solubility of phosphorus from mineral sources, feed ingredients and compound feed for pigs, poultry, dogs and cats. J. Anim. Physiol. Anim. Nutr. 2019, 103, 317–323. [Google Scholar] [CrossRef]
- Visscher, C.; Middendorf, L.; Günther, R.; Engels, A.; Leibfacher, C.; Möhle, H.; Düngelhoef, K.; Weier, S.; Haider, W.; Radko, D. Fat content, fatty acid pattern and iron content in livers of turkeys with hepatic lipidosis. Lipids Health Dis. 2017, 16, 98. [Google Scholar] [CrossRef]
- Neustädter, L.-T.; Kamphues, J.; Ratert, C. Influences of different dietary contents of macrominerals on the availability of trace elements in horse. J. Anim. Physiol. Anim. Nutr. 2018, 102, e633–e640. [Google Scholar] [CrossRef] [PubMed]
- Lange, M.; Höltershinken, M.; Scholz, H.; Vogt, C. Critical evaluation of internal standards and measuring conditions for the simultaneous determination of iodine and cobalt in bovine serum and urine (WedPo57). In Proceedings of the European Winter Conference on Plasma Spectrochemistry, Taormina, Italy, 18–23 February 2007. [Google Scholar]
- Russel, A. Body condition scoring of sheep. In Pract. 1984, 6, 91–93. [Google Scholar] [CrossRef] [PubMed]
- Dag Scoring, LambPlus, Profit through Science. Available online: http://www.sheep.ie/services/lambplus/files/dag_scoring.pdf (accessed on 3 February 2016).
- Weiss, D.J.; Wardrop, K.J. Schalm’s Veterinary Hematology; Wiley-Blackwell: Ames, IA, USA, 2010. [Google Scholar]
- Bickhardt, K.; Dudziak, D.; Ganter, M.; Henze, P. Untersuchungen zur Altersabhängigkeit hämatologischer und blutchemischer Meßgrößen bei gesunden Schaflämmern—Ein Beitrag zur Definition von Referenzwerten beim Schaf. Dtsch. Tierärztl. Wschr. 1999, 106, 445–451. [Google Scholar]
- Puls, R. Mineral Levels in Animal Health, 2nd ed.; Sherpa International: Clearbrook, BC, Canada, 1994. [Google Scholar]
- Dohm, A. Der Schwefel- und Sulfat-Gehalt in Grund- und Kraftfuttermitteln–Einflüsse und Mögliche Bedeutung (Sulfur and Sulfate Contents in Roughages and Concentrates–Influence Sand Possible Significance). Ph.D. Thesis, University of Veterinary Medicine Hannover, Hanover, Germany, 2015. [Google Scholar]
- Mac Pherson, A.; Brown, N.A.; Hemingway, R.C. The relation between the concentration of copper in the blood and livers of sheep. Vet. Rec. 1964, 76, 643–645. [Google Scholar]
- West, D.M.; Bruère, A.N.; Ridler, A.L. Clinical aspects of trace element requirements of grazing ruminants with particularly reference to sheep and cattle. In The Sheep: Health, Disease and Production, 3rd ed.; VetLearn®: Wellington, New Zealand, 2009; pp. 129–180. [Google Scholar]
- Nookabkaew, S.; Rangkadilok, N.; Prachoom, N.; Satayavivad, J. Concentrations of Trace Elements in Organic Fertilizers and Animal Manures and Feeds and Cadmium Contamination in Herbal Tea (Gynostemma pentaphyllum Makino). J. Agric. Food Chem. 2016, 64, 3119–3126. [Google Scholar] [CrossRef] [PubMed]
- Barlow, R.M. Recent Advances in Swayback. Proc. R. Soc. Med. 1958, 51, 748–752. [Google Scholar] [CrossRef] [PubMed]
- Ulvund, M.J. Ovine white-liver disease (OWLD). Changes in blood chemistry. Acta Vet. Scand. 1990, 31, 277–286. [Google Scholar] [CrossRef]
- Ulvund, M.J. Ovine White-Liver Disease (OWLD). Manifestation of Cobalt/Vitamin B12 Deficiency in Lambs. Ph.D. Thesis, Norwegian College of Veterinary Medicine, Oslo, Norway, 1990. [Google Scholar]
- Sivertsen, T.; Plassen, C. Hepatic Cobalt and Copper Levels in Lambs in Norway. Acta Vet. Scand. 2004, 45, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Ulvund, M.J.; Pestalozzi, M. Ovine white-liver disease (OWLD). Botanical and chemical composition of pasture grass. Acta Vet. Scan. 1990, 31, 257–265. [Google Scholar] [CrossRef]
| Id | Sample Id | Age | Sex | Health Status | Tissue Examined | Date |
|---|---|---|---|---|---|---|
| lamb 1– presented in the clinic | 1 | 6 months | m | emaciation, photodermatitis, conjunctivitis, apathy | blood samples, faecal sample, post mortem investigation, liver samples | 5/10/15 |
| lamb 2– farm visit | 2 | 6 months | m | unremarkable | blood samples, pooled faecal sample | 29/10/15 |
| lamb 3– farm visit | 3 | 6 months | m | emaciation, photodermatitis in healing | blood samples, pooled faecal sample | 29/10/15 |
| ewe 1– farm visit | 4 | 7 years | f | emaciation, shaggy, pale wool | blood samples, pooled faecal sample | 29/10/15 |
| ewe 2– farm visit | 5 | 3 years | f | unremarkable | blood samples, pooled faecal sample | 29/10/15 |
| liver 1– farm visit, frozen | 6 | adult, concrete age u | f | u | liver samples | slaughtered in autumn 2014 |
| liver 2– farm visit, frozen | 7 | adult, concrete age u | f | u | liver samples | slaughtered in autumn 2014 |
| Sample ID | 1 | 2 | 3 | 4 | 5 | 6 | 7 |
|---|---|---|---|---|---|---|---|
| Selenium *** (s: 80–500 µg/L l: 0.25–1.5 mg/kg FM) | s: 83.5 | s: 125.2 | s: 112.3 | s: 60.9 ↓ | s: 125.3 | l: 0.109 ↓ | l: 0.099 ↓ |
| Copper *** (s: 7–24 µmol/L; l: 10–120 mg/kg FM) | s: 10.9 l: 1.45 ↓ | s: 5.8 ↓ | s: 5.7 ↓ | s: 13.7 | s: 12.3 | l: 24.2 | l: 9.1 ↓ |
| Cobalt *** (l: 0.025–0.085 mg/kg FM) | l: 0.008 ↓ | - | - | - | - | l: 0.015 ↓ | l: 0.020 ↓ |
| Molybdenum *** (l: 1.5–6 mg/kg DM) | - | - | - | - | - | l: 7 ↑ | l: 8.5 ↑ |
| Haemoglobin * (90–140 g/L) | 84 ↓ | 90 | 109 | - | - | - | - |
| Packed Cell volume * (0.27–0.41 L/L) | 0.24 ↓ | 0.3 | 0.36 | ||||
| MCH * (13–14 pg) | 10.4 ↓ | 10.5 ↓ | 10.7 ↓ | - | - | - | - |
| MCV * (34–46 fL) | 32 ↓ | 35.1 | 35.3 | ||||
| MCHC * (290–340 g/L) | 323 | 300 | 303 | ||||
| Bilirubin ** (0–10 µmol/L) | 13.13 ↑ | - | - | - | - | - | - |
| Protein ** (52–70 g/L) | 71.9 ↑ | 62.5 | 66.2 | ||||
| Albumin ** (27–39 g/L) | 24.2 ↓ | 36 | 31.8 | - | - | - | - |
| ASAT ** (30–80 U/L) | 589 ↑ | 63 | 47 | - | - | - | - |
| CK ** (10–230 U/L) | 87 | 78 | 100 | - | - | - | - |
| GLDH ** (2–12 U/L) | 525 ↑ | 7 | 9 | - | - | - | - |
| GGT ** (5–32 U/L) | 135 ↑ | 34 ↑ | 35 ↑ | - | - | - | - |
| Faecal Sample | Coccidial Oocysts/g Faeces | Gastrointestinal Nematodes Eggs/g Faeces | Nematodirus Eggs/g Faeces | Trichuris | Capillaria | Strongyloides Eggs/g Faeces | Moniezia spp. | Dicrocoelium dentriticum | Fasciola hepatica |
|---|---|---|---|---|---|---|---|---|---|
| lamb 1 | 5 | 45 | 0 | − | − | 3 | − | − | − |
| ram lambs | 17 | 7 | 33 | + | − | 10 | − | − | − |
| female lambs | 12 | 202 | 0 | + | − | 17 | − | − | − |
| yearling ewes | 5 | 117 | 2 | − | − | 4 | − | − | − |
| ewes and rams | 0 | 4 | 0 | − | − | 3 | − | − | − |
| Reference Values | Pasture 1 | Pasture 2 | |
|---|---|---|---|
| DM (g/kg original substance) | - | 205 | 214 |
| Ca (g/kg DM) | 7.1 * | 4.66 ↓ | 5.46 ↓ |
| P (g/kg DM) | 3.4 * | 4.02 | 3.55 |
| S (g/kg DM) | 2.58 ** | 3.79 ↑ | 2.60 |
| Fe (mg/kg DM) | 50–280 *** | 404 ↑ | 531 ↑ |
| Cu (mg/kg DM) | 5–10 *** | 9.26 | 8.61 |
| Co (mg/kg DM) | 0.1–0.2 * | 0.124 | 0.16 |
| Se (mg/kg DM) | 0.4–1 *** | 0.112 ↓ | 0.018 ↓ |
| Mo (mg/kg DM) | 0.5–3.5 *** | 11.3 ↑ | 6.34 ↑ |
| Cu: Mo-ratio | Set point value: 6:1, 2–3:1 limits of acceptability, <2:1 toxic **** | 0.8:1 ↓ | 1.4:1 ↓ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Helmer, C.; Hannemann, R.; Humann-Ziehank, E.; Kleinschmidt, S.; Koelln, M.; Kamphues, J.; Ganter, M. A Case of Concurrent Molybdenosis, Secondary Copper, Cobalt and Selenium Deficiency in a Small Sheep Herd in Northern Germany. Animals 2021, 11, 1864. https://doi.org/10.3390/ani11071864
Helmer C, Hannemann R, Humann-Ziehank E, Kleinschmidt S, Koelln M, Kamphues J, Ganter M. A Case of Concurrent Molybdenosis, Secondary Copper, Cobalt and Selenium Deficiency in a Small Sheep Herd in Northern Germany. Animals. 2021; 11(7):1864. https://doi.org/10.3390/ani11071864
Chicago/Turabian StyleHelmer, Carina, Regina Hannemann, Esther Humann-Ziehank, Sven Kleinschmidt, Mareike Koelln, Josef Kamphues, and Martin Ganter. 2021. "A Case of Concurrent Molybdenosis, Secondary Copper, Cobalt and Selenium Deficiency in a Small Sheep Herd in Northern Germany" Animals 11, no. 7: 1864. https://doi.org/10.3390/ani11071864
APA StyleHelmer, C., Hannemann, R., Humann-Ziehank, E., Kleinschmidt, S., Koelln, M., Kamphues, J., & Ganter, M. (2021). A Case of Concurrent Molybdenosis, Secondary Copper, Cobalt and Selenium Deficiency in a Small Sheep Herd in Northern Germany. Animals, 11(7), 1864. https://doi.org/10.3390/ani11071864







