Pointing Out Some Issues Regarding Reproduction Management in Murciano-Granadina Goats
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics
2.2. Goats and General Proczedures
2.3. Experimental Data and Sample Collection
2.4. Statistical Analysis of Results
3. Results
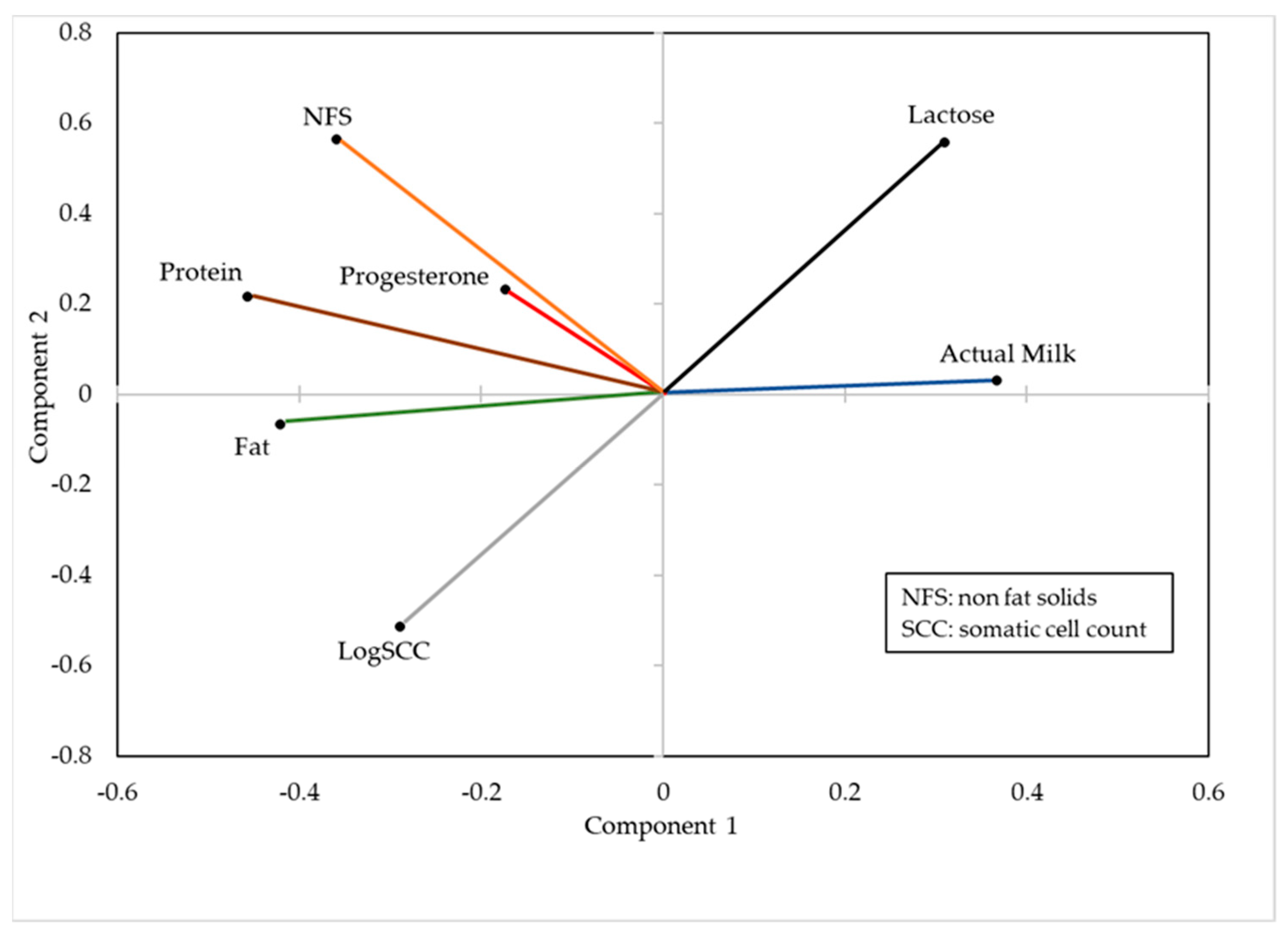
3.1. Principal Components
3.2. Components of Variance for Progesterone Values in Milk
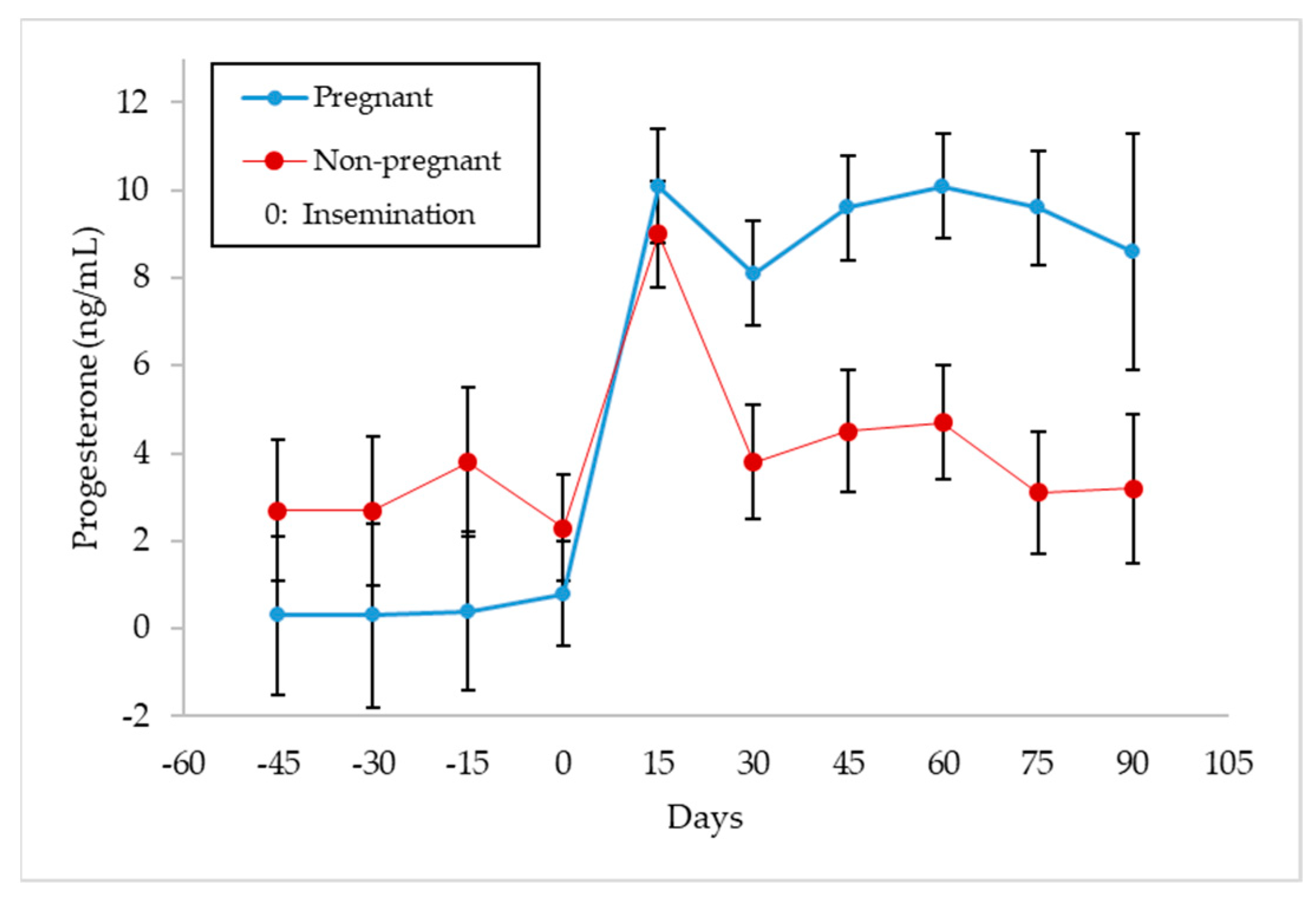
3.3. Progesterone Concentrations in Milk
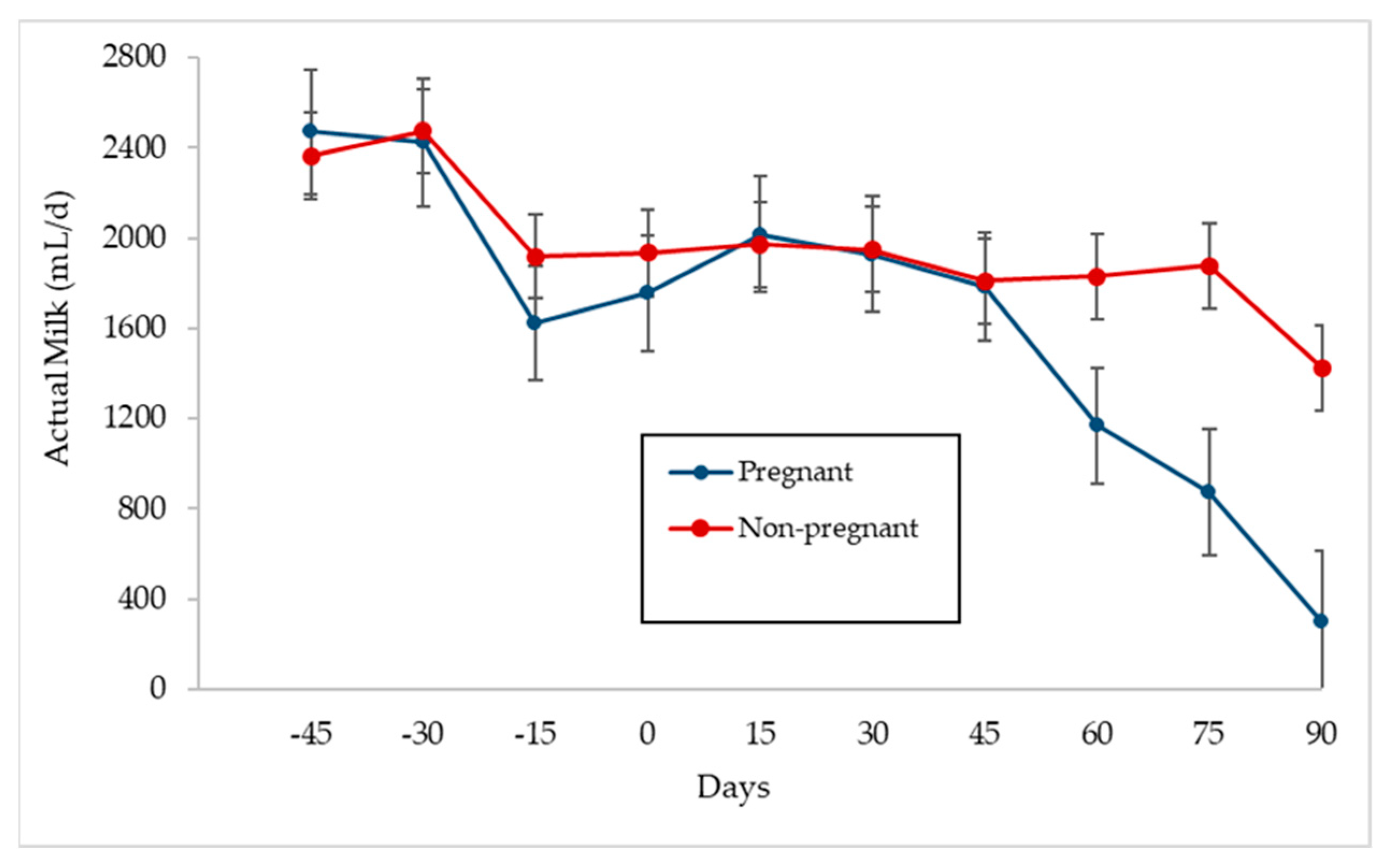
3.4. Relationship between Milk Production and Pregnancy
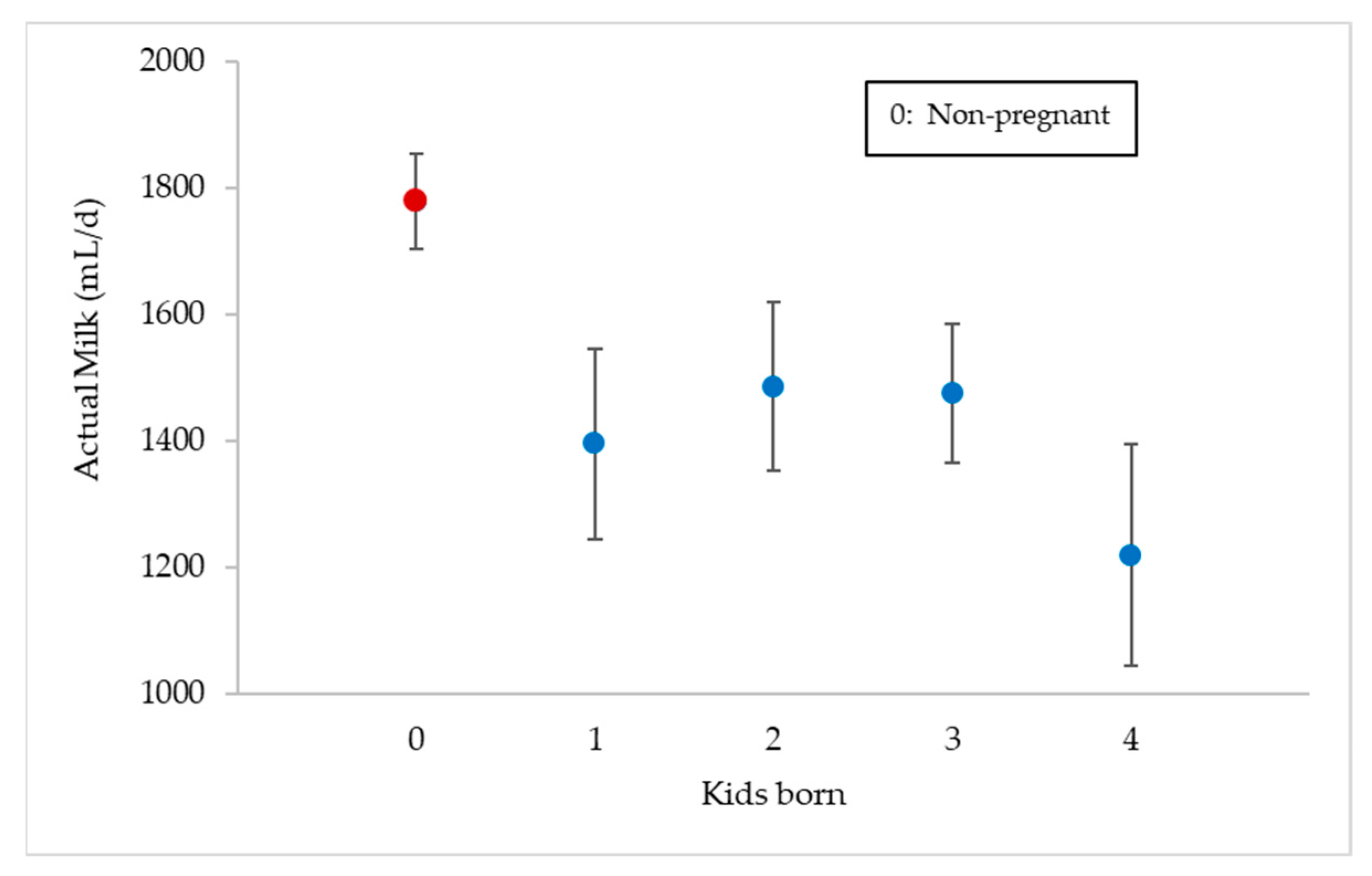
3.5. Effect of the Number of Kids Born
3.6. Effect of Pregnancy on Milk Components
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Whitlock, B.K.; Coffman, E.A. Review of pregnancy diagnosis techniques in cattle and small ruminants. Clin. Theriogenol. 2010, 2, 275–288. [Google Scholar]
- Zobel, G.; Weary, D.M.; Leslie, K.E.; von Keyserlingk, M.A.G. Cessation of lactation: Effects on animal welfare. J. Dairy Sci. 2015, 98, 8263–8277. [Google Scholar] [CrossRef]
- Karen, A.; Kovács, P.; Beckers, J.F.; Szenci, O. Pregnancy diagnosis in sheep: Review of the most practical methods. Acta Vet. Brno 2001, 70, 115–126. [Google Scholar] [CrossRef]
- Available online: http://www.webmedcentral.com/article_view/1305 (accessed on 26 January 2021).
- Rioux, P.; Rajotte, D. Progesterone in milk: A simple experiment illustrating the estrous cycle and enzyme immunoassay. Adv. Physiol. Educ. 2004, 28, 64–67. [Google Scholar] [CrossRef][Green Version]
- Holtz, W. Recent developments in assisted reproduction in goats. Small Rumin. Res. 2005, 60, 95–110. [Google Scholar] [CrossRef]
- De Montigny, G.; Millerioux, P.; Jeanguyot, N.; Humblot, P.; Thibier, M. Milk fat progesterone concentrations in goats and early pregnancy diagnosis. Theriogenology 1982, 17, 423–431. [Google Scholar] [CrossRef]
- Murray, R.D.; Newstead, R. Determination of steroid hormones in goats’ milk and plasma as an aid to pregnancy diagnosis using an ELISA. Vet. Rec. 1988, 122, 158–161. [Google Scholar] [CrossRef] [PubMed]
- Patton, S. Milk. The fluid made by the mammary gland is a remarkable blend of complex biological molecules. How the gland does its work is the subject of active investigation. Small Rumin. Res. 1974, 221, 59–68. [Google Scholar]
- Lisci, G.A.; Ramírez, R.C.; Gómez, P.L. Ginecología Endocrina; Limusa S.A.: Ciudad de México, México, 1992. [Google Scholar]
- Nepal, S.; Subedi, D.; Kaphle, K. Pregnancy Diagnosis with Progesterone ELISA Kit in Farm Animals, Its Accuracy and Application. Nepal. Vet. J. 2019, 36, 111–117. [Google Scholar] [CrossRef]
- Bretzlaff, K.N.; Elmore, R.G.; Nuti, L.C. Use of an enzyme immunoassay to determine concentrations of progesterone in caprine plasma and milk. J. Am. Vet. Med. Assoc. 1898, 194, 664–668. [Google Scholar]
- Mann, G.E.; Lamming, G.E.; Fray, M.D. Plasma oestradiol and progesterone during early pregnancy in the cow and the effects of treatment with buserelin. Anim. Reprod. Sci. 1995, 37, 121–131. [Google Scholar] [CrossRef]
- Blasco, E.; Gómez, E.A.; Vicente, C.; Vidal, G.; Peris, C. Factors affecting milking speed in Murciano-Granadina breed goats. J. Dairy Sci. 2016, 99, 10102–10108. [Google Scholar] [CrossRef] [PubMed]
- El Hadi, A. The Impact of Shering and Hormonal Treatments (Melatonine or Cabergoline) in Lactating Dairy Ewes. Ph.D. Thesis, Universitat Autònoma de Barcelona, Barcelona, Spain, 2020. [Google Scholar]
- Caja, G.; El Hadi, A.; Such, X.; Salama, A.A.K. Suppression of prolactin and reduction of milk secretion by effect of cabergoline in lactating dairy ewes. J. Dairy Sci. 2020, 103, 12033–12044. [Google Scholar] [CrossRef] [PubMed]
- Available online: https://cima.aemps.es/cima/pdfs/es/p/69669/P_69669.pdf (accessed on 3 February 2021).
- Knight, C.H.; Wilde, C.J. Milk production in concurrently pregnant and lactating goats mated out of season. J. Dairy Res. 1988, 55, 487–493. [Google Scholar] [CrossRef]
- Salama, A.A.K.; Caja, G.; Such, X.; Casals, R.; Albanell, E. Effect of pregnancy and extended lactation on milk production in dairy goats milked once daily. J. Dairy Sci. 2005, 88, 3894–3904. [Google Scholar] [CrossRef]
- Ali, A.; Shook, G.E. An optimum transformation for somatic cell concentration in milk. J. Dairy Sci. 1980, 63, 487–490. [Google Scholar] [CrossRef]
- Vacca, G.; Stocco, G.; Dettori, M.L.; Pira, E.; Bittante, G.; Pazzola, M. Milk yield, quality, and coagulation properties of 6 breeds of goats: Environmental and individual variability. J. Dairy Sci. 2018, 101, 7236–7247. [Google Scholar] [CrossRef]
- Currò, S.; Manuelian, C.L.; De Marchi, M.; Claps, S.; Rufrano, D.; Neglia, G. Effects of breed and stage of lactation on milk fatty acid composition of Italian goat breeds. Animals 2019, 9, 764. [Google Scholar] [CrossRef] [PubMed]
- Paschino, P.; Stocco, G.; Dettori, M.L.; Pazzola, M.; Marongiu, M.L.; Pilo, C.E.; Cipolat-Gotet, C.; Vacca, G. Characterization of milk composition, coagulation properties, and cheese-making ability of goats reared in extensive farms. J. Dairy Sci. 2020, 103, 5830–5843. [Google Scholar] [CrossRef]
- Leitner, G.; Merin, U.; Silanikove, N. Changes in milk composition as affected by subclinical mastitis in goats. J. Dairy Sci. 2004, 87, 1719–1726. [Google Scholar] [CrossRef]
- Stocco, G.; Pazzola, M.; Dettori, M.L.; Cipolat-Gotet, A.S.; Vacca, G.M. Variation in caprine milk composition and coagulation as affected by udder health indicators. Int. Dairy J. 2019, 98, 9–16. [Google Scholar] [CrossRef]
- Alabart, J.L.; Lahoz, B.; Muñoz, F.; Mozo, R.; Legua, A.; Sánchez, P.; Folch, J.; Andueza, D. Diagnóstico precoz de gestación en ovejas a partir de muestras de plasma sanguíneo analizadas mediante espectroscopía en el infrarrojo cercano (NIRS). In Proceedings of the XXXIX Jornadas Científicas de la Sociedad Española de Ovinotecnia y Caprinotecnia, Ourense, Spain, 17–19 September 2014. [Google Scholar]
- Pérez-Baena, I.; Jarque-Duran, M.; Gómez, E.A.; Díaz, J.R.; Peris, C. Terminal Crossbreeding of Murciano-Granadina Goats to Boer Bucks: Effects on Reproductive Performance of Goats and Growth of Kids in Artificial Rearing. Animals 2021, 11, 986. [Google Scholar] [CrossRef] [PubMed]
- Nebel, R.L.; McGuilliard, M.L. Interactions of High Milk Yield and Reproductive Performance in Dairy Cows. J. Dairy Sci. 1993, 76, 3257–3268. [Google Scholar] [CrossRef]
- Van Binsbergen, R.; Bouwman, A.C.; Veerkamp, R.F. Genetic parameters for atypical reproductive patterns in dairy cows estimated from in-line milk progesterone profiles. J. Dairy Sci. 2019, 102, 11104–11115. [Google Scholar] [CrossRef] [PubMed]
- Blaszczyk, B.; Udala, J.; Gaczarzewicz, D. Changes in estradiol, progesterone, melatonine, prolactine and thyroxine concentratios in blood plasma of goats following induced estrus in and outside the natural breeding season. Small Rumin. Res. 2004, 51, 209–219. [Google Scholar] [CrossRef]
- Available online: https://www.researchgate.net/publication/308308304 (accessed on 29 January 2021).
- Available online: https://www.researchgate.net/publication/333776052 (accessed on 29 January 2021).
- Khan, J.R.; Ludri, R.S. Hormonal profiles during periparturient period in single and twin fetus bearing goats. Asian-Aust. J. Anim. Sci. 2002, 15, 346–351. [Google Scholar] [CrossRef]
- Jain, G.C.; Arora, R.C.; Pandey, R.S. Milk progesterone content and pregnancy diagnosis in goats. Transb. Emerg. Dis. 1980, 27, 103–108. [Google Scholar]
- Engeland, I.V.; Ropstad, E.; Andresen, Ø.; Eik, L.O. Pregnancy diagnosis in dairy goats using progesterone assay kits and oestrous observation. Anim. Repr. Sci. 1997, 47, 237–243. [Google Scholar] [CrossRef]
- González-Stagnaro, C.; Corteel, J.M.; Baril, G. Cinética de la progesterona plasmática durante el celo natural e inducido pr tratamientos hormonales en cabras lecheras. Rev. Científica FCV-LUZ 1992, II, 12–21. [Google Scholar]
- De Castro, T.; Rubianes, E.; Manchaca, A.; Rivero, A. Ovarian dynamics, serum estradiol and progesterone concentrations during the interovulatory interval in goats. Theriogenology 1999, 52, 399–411. [Google Scholar] [CrossRef]
- Górecki, M.T.; Wójtowski, J.; Kaczmartek, P.; Danków, R.; Casi-Sokolinska, D.; Nowak, K.W. Concentrations of progesterone and 17β-estradiol in blood and milk and those of natural inhibitors in milk goats in various physiological stages. Arch. Tierz. 2004, 47, 90–96. [Google Scholar]
- Dawson, L.J. Pregnancy diagnosis in goats. In Proceedings of the 14th Annual Goat Field Day; Langston University: Langston, OK, USA, 1999; pp. 97–103. [Google Scholar]
- Available online: https://www.msdvetmanual.com/management-and-nutrition/management-of-reproduction-goats/pregnancy-determination-in-goats (accessed on 29 January 2021).
- Fleming, S.A.; Van Camp, S.D.; Chapin, H.M. Serum progesterone determination as an aid for pregnancy diagnosis in goats bred out of season. Can. Vet. J. 1990, 31, 104–107. [Google Scholar] [PubMed]
- Pennington, J.A.; Hoffman, W.F.; Schultz, L.H.; Spahr, S.L.; Lodge, J.R. Milk progesterone for pregnancy diagnosis in dairy goats. J. Dairy Sci. 1982, 65, 2011–2014. [Google Scholar] [CrossRef]
- Delpino, A.; Conzález-Stagnaro, C. Evaluación delo comportamiento reproductivo en pequeños rumiantes tropicales utilizando los perfiles de progesterona. Rev. Científica FCV-LUZ 1993, III, 231–243. [Google Scholar]
- Kalkan, C.; Cetin, H.; Kaygusuzoglu, E.; Yilmaz, B.; Ciftci, M.; Yildiz, H.; Yildiz, A.; Deveci, H.; Apaydin, A.M.; Ocal, H. An investigation on plasma progesterone levels during pregnancy and parturition in the Ivesi sheep. Acta Vet. Hung. 1996, 44, 335–340. [Google Scholar]
- Chauhan, F.S.; Waziri, M.A. Evaluation of rectal-abdominal palpation technique and hormonal diagnosis of pregnancy in small ruminants. Indian J. Anim. Reprod. 1991, 12, 63–67. [Google Scholar]
- Quirke, J.F.; Hanrahan, J.P.; Gosling, J.P. Plasma progesterone levels throughout the oestrous cycle and release of LH at oestrous in sheep with different ovulation rates. J. Reprod. Fertil. 1979, 55, 37–44. [Google Scholar] [CrossRef]
- Vilar, M.J.; Rajala-Schultz, P.J. Dry-off and dairy cow udder health and welfare: Effects of different milk cessation methods. Vet. J. 2020, 262, 105503. [Google Scholar] [CrossRef]
- Caldeira, R.M.; Belo, A.T.; Santos, C.C.; Vazques, M.I.; Portugal, A.V. The effect of long-term feed restriction and over-nutrition on body condition secore, blood metabolites and hormonal profiles in ewes. Small Rumin. Res. 2007, 68, 242–255. [Google Scholar] [CrossRef]
- Roche, J.R. Effect of pregnancy on milk production and bodyweight from identical twin study. J. Dairy Sci. 2003, 86, 777–783. [Google Scholar] [CrossRef]
- Oltenacu, P.A.; Rounsville, T.R.; Milligan, R.A.; Lintz, R.L. Relationship between days open and cumulative milk yield at various intervals from parturition for high- and low-producing cows. J. Dairy Sci. 1980, 63, 1317–1327. [Google Scholar] [CrossRef]
- National Research Council. Nutrient Requirements of Dairy Cattle, 7th ed.; National Academies Press: Washington, DC, USA, 2001. [Google Scholar]




| Progesterone | Variance Component | ||
| Goat | Lactation Period | Others | |
| 65.08 | 30.56 | 4.35 | |
| Progesterone | Season of the Year | p-Value | |||
|---|---|---|---|---|---|
| Spring | Summer | Autumn | Winter | ||
| Minimum (n = 199) | 1.18 ± 0.09 | 1.15 ± 0.15 | 1.27 ± 0.17 | 1.40 ± 0.18 | p = 0.128 |
| Maximum (n = 115) | 11.6 a ± 1.10 | 8.85 b ± 0.91 | 8.28 b ± 1.12 | 11.56 a ± 1.15 | p = 0.0006 |
| Pregnancy Duration | Number of Kids Born | p-Value | |||
| 1 | 2 | 3 | 4 | ||
| 147.2 ± 3.2 | 144.0 ± 2.4 | 146.3 ± 3.5 | 144.3 ± 3.4 | p = 0.421 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fernández, N.; Beltrán, M.C.; Romero, G.; Roca, M.A.; Rodríguez, M.; Balasch, S. Pointing Out Some Issues Regarding Reproduction Management in Murciano-Granadina Goats. Animals 2021, 11, 1781. https://doi.org/10.3390/ani11061781
Fernández N, Beltrán MC, Romero G, Roca MA, Rodríguez M, Balasch S. Pointing Out Some Issues Regarding Reproduction Management in Murciano-Granadina Goats. Animals. 2021; 11(6):1781. https://doi.org/10.3390/ani11061781
Chicago/Turabian StyleFernández, Nemesio, M. Carmen Beltrán, Gema Romero, M. Amparo Roca, Martín Rodríguez, and Sebastián Balasch. 2021. "Pointing Out Some Issues Regarding Reproduction Management in Murciano-Granadina Goats" Animals 11, no. 6: 1781. https://doi.org/10.3390/ani11061781
APA StyleFernández, N., Beltrán, M. C., Romero, G., Roca, M. A., Rodríguez, M., & Balasch, S. (2021). Pointing Out Some Issues Regarding Reproduction Management in Murciano-Granadina Goats. Animals, 11(6), 1781. https://doi.org/10.3390/ani11061781







