Public Feeding Interactions as Enrichment for Three Zoo-Housed Elephants
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
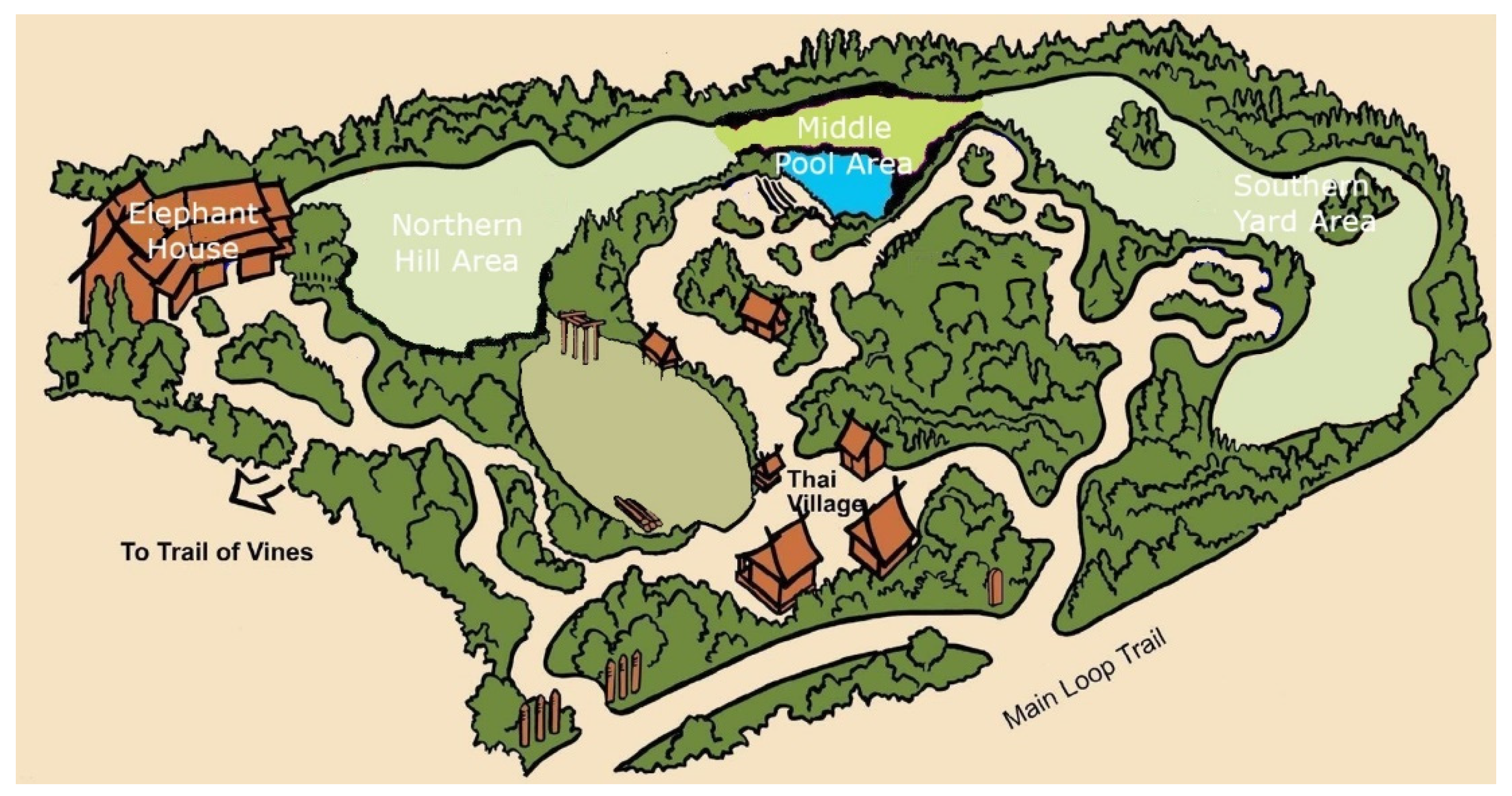
2.1. Subjects and Enclosure
2.2. Materials
2.3. Design and Procedure
2.4. Statistical Analyses
3. Results
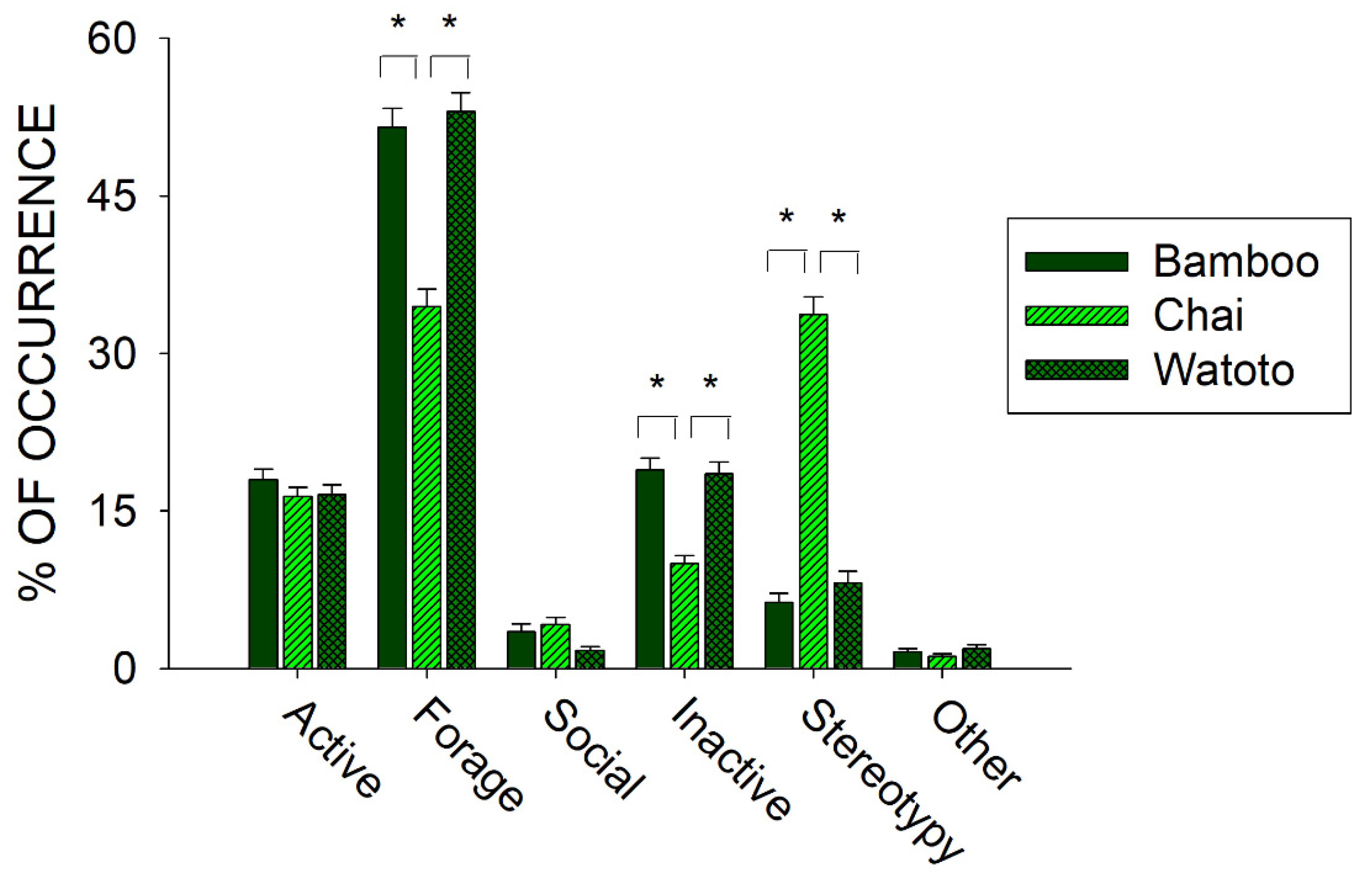
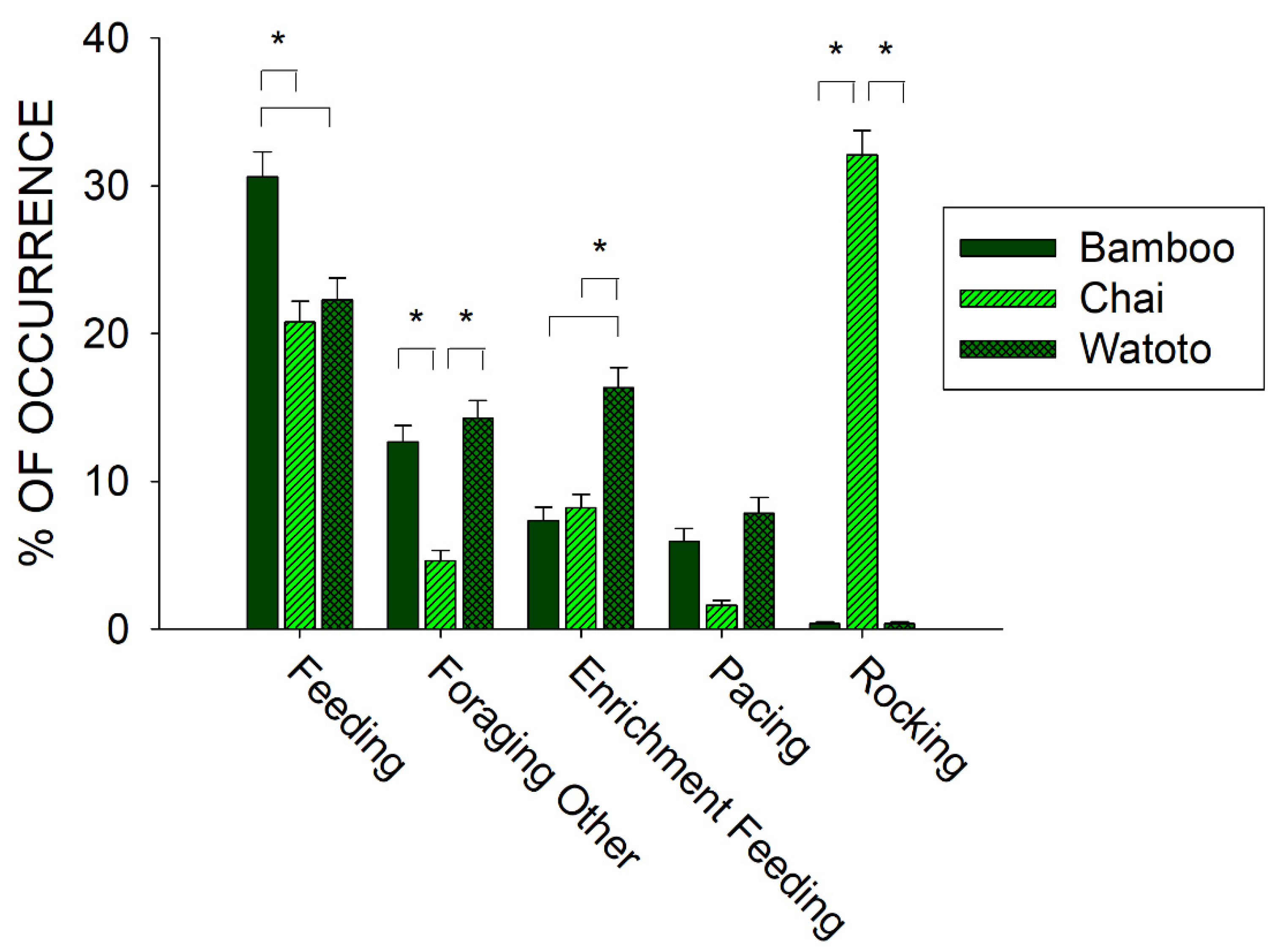
3.1. Prior to Public Feedings: Differences between Elephants
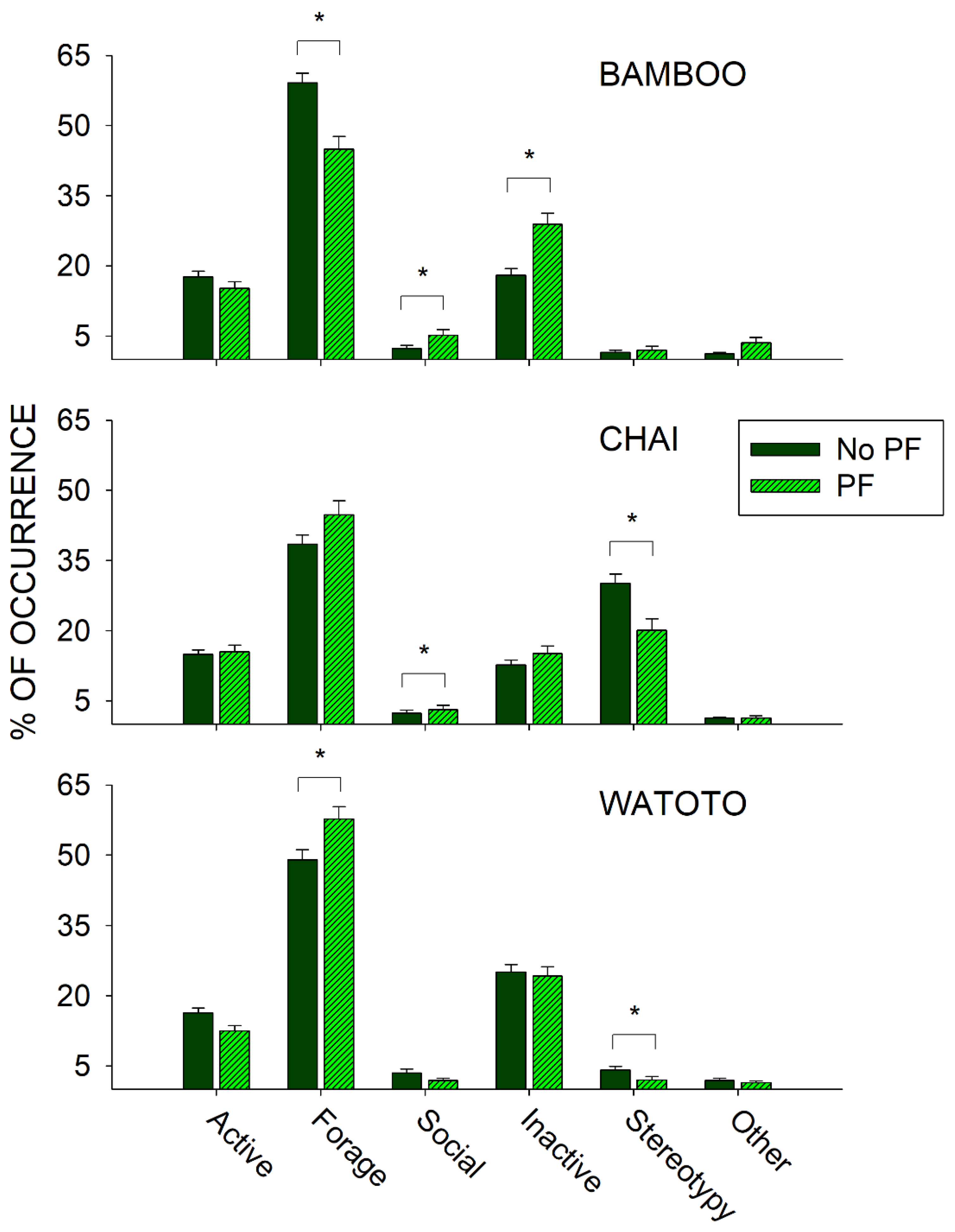
3.2. Public Feed Days Compared with No Public Feed Days
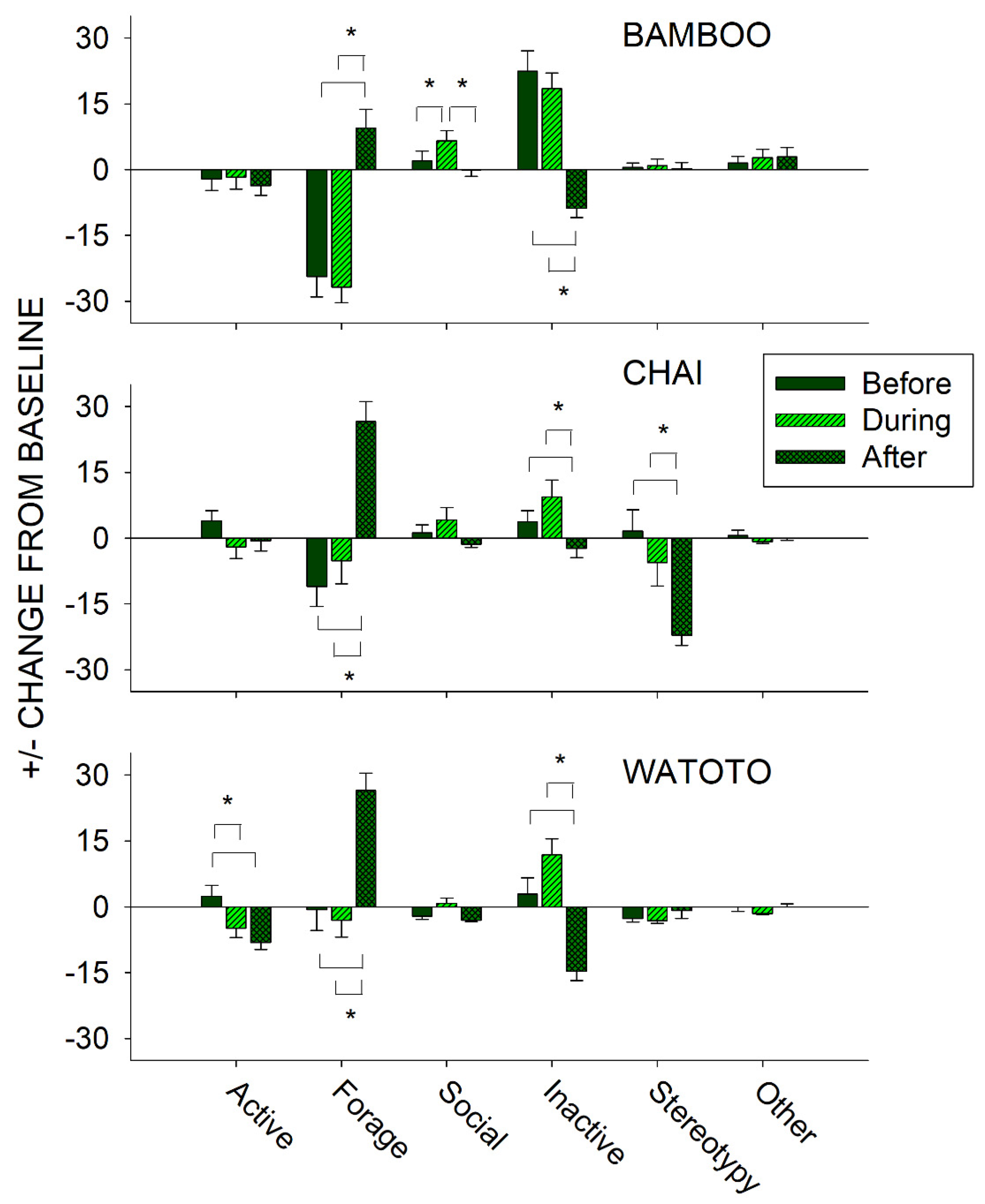
3.3. Before, During, and After a Public Feeding
4. Discussion
4.1. Prior to Public Feedings: Foraging and Stereotypies
4.2. Public Feeding General Effects
4.3. Before and After a Public Feeding
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kleiman, D.G.; Thompson, K.V.; Baer, C.K. (Eds.) Wild Mammals in Captivity: Principles and Techniques for Zoo Management; Uni-versity of Chicago Press: Chicago, IL, USA, 2010. [Google Scholar]
- Maple, T.; Perdue, B.M. Zoo Animal Welfare; Springer: Berlin/Heidelberg, Germany, 2013. [Google Scholar]
- Shepherdson, D.; Mellen, J.; Hutchins, M. (Eds.) Second Nature: Environmental Enrichment for Captive Animals; Smithsonian Institution Press: Washington, DC, USA, 1998. [Google Scholar]
- Clubb, R.; Mason, G. A Review of the Welfare of Zoo Elephants in Europe; RSPCA: Horsham, UK, 2002. [Google Scholar]
- Clubb, R.; Rowcliffe, M.; Lee, P.C.; Mar, K.U.; Moss, C.J.; Mason, G.J. Compromised Survivorship in Zoo Elephants. Science 2008, 322, 1649. [Google Scholar] [CrossRef]
- Clubb, R.; Rowcliffe, M.; Mar, K.U.; Lee, P.; Moss, C.; Mason, G.J. Fecundity and population viability in female zoo elephants: Problems and possible solutions. Anim. Welf. 2009, 18, 237–247. [Google Scholar]
- Wiese, R.J.; Willis, K. Calculation of longevity and life expectancy in captive elephants. Zoo Biol. 2004, 23, 365–373. [Google Scholar] [CrossRef]
- Faust, L.J.; Thompson, S.D.; Earnhardt, J.M. Is reversing the decline of Asian elephants in North American zoos possible? An individual-based modeling approach. Zoo Biol. 2006, 25, 201–218. [Google Scholar] [CrossRef]
- Rees, P.A. Asian elephants in zoos face global extinction: Should zoos accept the inevitable? Oryx 2003, 37, 20–22. [Google Scholar] [CrossRef]
- Wiese, R.J. Asian elephants are not self-sustaining in North America. Zoo Biol. 2000, 19, 299–309. [Google Scholar] [CrossRef]
- Wiese, R.J.; Willis, K. Population management of zoo elephants. Int. Zoo Yearb. 2006, 40, 80–87. [Google Scholar] [CrossRef]
- Carlstead, K.; Mench, J.A.; Meehan, C.; Brown, J.L. An Epidemiological Approach to Welfare Research in Zoos: The Elephant Welfare Project. J. Appl. Anim. Welf. Sci. 2013, 16, 319–337. [Google Scholar] [CrossRef] [PubMed]
- Hutchins, M. Variation in nature: Its implications for zoo elephant management. Zoo Biol. 2006, 25, 161–171. [Google Scholar] [CrossRef]
- Hutchins, M.; Thompson, S.D. Zoo and aquarium research: Priority setting for the coming decades. Zoo Biol. 2008, 27, 488–497. [Google Scholar] [CrossRef]
- Mason, G.J.; Veasey, J.S. How should the psychological well-being of zoo elephants be objectively investigated? Zoo Biol. 2010, 29, 237–255. [Google Scholar] [CrossRef]
- Mason, G.J.; Veasey, J.S. What do population-level welfare indices suggest about the well-being of zoo elephants? Zoo Biol. 2010, 29, 256–273. [Google Scholar] [CrossRef] [PubMed]
- Veasey, J.S. Assessing the Psychological Priorities for Optimising Captive Asian Elephant (Elephas maximus) Welfare. Animals 2019, 10, 39. [Google Scholar] [CrossRef] [PubMed]
- Williams, E.; Chadwick, C.; Yon, L.; Asher, L. A review of current indicators of welfare in captive elephants (Loxodonta africana and Elephas maximus). Anim. Welf. 2018, 27, 235–249. [Google Scholar] [CrossRef]
- Yon, L.; Williams, E.; Harvey, N.D.; Asher, L. Development of a behavioural welfare assessment tool for routine use with captive elephants. PLoS ONE 2019, 14, e0210783. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.L.; Carlstead, K.; Bray, J.D.; Dickey, D.; Farin, C.; Ange-van Heugten, K. Individual and environmental risk factors associated with fecal glucocorticoid metabolite concentrations in zoo-housed Asian and African elephants. PLoS ONE 2019, 14, e0217326. [Google Scholar] [CrossRef]
- Carlstead, K.; Paris, S.; Brown, J.L. Good keeper-elephant relationships in North American zoos are mutually beneficial to welfare. Appl. Anim. Behav. Sci. 2019, 211, 103–111. [Google Scholar] [CrossRef]
- Edwards, K.L.; Miller, M.A.; Carlstead, K.; Brown, J.L. Relationships between housing and management factors and clinical health events in elephants in North American zoos. PLoS ONE 2019, 14, e0217774. [Google Scholar] [CrossRef]
- Greco, B.J.; Meehan, C.L.; Heinsius, J.L.; Mench, J.A. Why pace? The influence of social, housing, management, life history, and demographic characteristics on locomotor stereotypy in zoo elephants. Appl. Anim. Behav. Sci. 2017, 194, 104–111. [Google Scholar] [CrossRef]
- Greco, B.J.; Meehan, C.L.; Hogan, J.N.; Leighty, K.A.; Mellen, J.; Mason, G.J.; Mench, J.A. The days and nights of zoo elephants: Using epidemiology to better understand stereotypic behavior of African elephants (Loxodonta africana) and Asian elephants (Elephas maximus) in North American zoos. PLoS ONE 2016, 11, e0144276. [Google Scholar] [CrossRef] [PubMed]
- Greco, B.J.; Meehan, C.L.; Miller, L.J.; Shepherdson, D.J.; Morfeld, K.A.; Andrews, J.; Baker, A.M.; Carlstead, K.; Mench, J.A. Elephant Management in North American Zoos: Environmental Enrichment, Feeding, Exercise, and Training. PLoS ONE 2016, 11, e0152490. [Google Scholar] [CrossRef]
- Meehan, C.L.; Hogan, J.N.; Bonaparte-Saller, M.K.; Mench, J.A. Housing and social environments of African (Loxodonta africana) and Asian (Elephas maximus) elephants in North American zoos. PLoS ONE 2016, 11, e0146703. [Google Scholar] [CrossRef] [PubMed]
- Prado-Oviedo, N.A.; Bonaparte-Saller, M.K.; Malloy, E.J.; Meehan, C.L.; Mench, J.A.; Carlstead, K.; Brown, J.L. Evaluation of Demographics and Social Life Events of Asian (Elephas maximus) and African Elephants (Loxodonta africana) in North American Zoos. PLoS ONE 2016, 11, e0154750. [Google Scholar] [CrossRef] [PubMed]
- Davey, G. Visitors’ Effects on the Welfare of Animals in the Zoo: A Review. J. Appl. Anim. Welf. Sci. 2007, 10, 169–183. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, E.J.; Tamborski, M.A.; Pickens, S.R.; Timberlake, W. Animal–visitor interactions in the modern zoo: Conflicts and interventions. Appl. Anim. Behav. Sci. 2009, 120, 1–8. [Google Scholar] [CrossRef]
- Godinez, A.M.; Fernandez, E.J. What is the Zoo Experience? How Zoos Impact a Visitor’s Behaviors, Perceptions, and Conservation Efforts. Front. Psychol. 2019, 10, 1746. [Google Scholar] [CrossRef]
- Hosey, G.R. Zoo animals and their human audiences: What is the visitor effect? Anim. Welf. 2000, 9, 343–357. [Google Scholar]
- Sherwen, S.L.; Hemsworth, P.H. The Visitor Effect on Zoo Animals: Implications and Opportunities for Zoo Animal Welfare. Animals 2019, 9, 366. [Google Scholar] [CrossRef]
- Hediger, H. Man and Animal in the Zoo; Delacort Press: New York, NY, USA, 1969. [Google Scholar]
- Kreger, M.D.; Mench, J.A. Visitor—Animal interactions at the zoo. Anthrozoös 1995, 8, 143–158. [Google Scholar] [CrossRef]
- Law, G.; Reid, A. Enriching the lives of bears in zoos. Int. Zoo Yearb. 2010, 44, 65–74. [Google Scholar] [CrossRef]
- D’Cruze, N.; Khan, S.; Carder, G.; Megson, D.; Coulthard, E.; Norrey, J.; Groves, G. A global review of animal—visitor interactions in modern zoos and aquariums and their implications for wild animal welfare. Animals 2019, 9, 332. [Google Scholar] [CrossRef] [PubMed]
- Linge, J.H.V. How to out-zoo the zoo. Tourism Manag. 1992, 13, 115–117. [Google Scholar] [CrossRef]
- Ryan, C.; Saward, J. The Zoo as Ecotourism Attraction—Visitor Reactions, Perceptions and Management Implications: The Case of Hamilton Zoo, New Zealand. J. Sustain. Tour. 2004, 12, 245–266. [Google Scholar] [CrossRef]
- Woods, B. Good zoo/bad zoo: Visitor experiences in captive settings. Anthrozoös 2002, 15, 343–360. [Google Scholar] [CrossRef]
- Collins, C.; Corkery, I.; Haigh, A.; McKeown, S.; Quirke, T.; O’Riordan, R. The effects of environmental and visitor variables on the behavior of free-ranging ring-tailed lemurs (Lemur catta) in captivity. Zoo Biol. 2017, 36, 250–260. [Google Scholar] [CrossRef]
- Farrand, A.; Hosey, G.; Buchanan-Smith, H.M. The visitor effect in petting zoo-housed animals: Aversive or enriching? Appl. Anim. Behav. Sci. 2014, 151, 117–127. [Google Scholar] [CrossRef]
- Manna, D.; Rodeano, M.; Ferrero, E.A. A lemur mixed exhibit at Parco Zoo Punta Verde, Italy. Int. Zoo News 2007, 361, 452–457. [Google Scholar]
- Orban, D.A.; Siegford, J.; Snider, R.J. Effects of guest feeding programs on captive giraffe behavior. Zoo Biol. 2016, 35, 157–166. [Google Scholar] [CrossRef] [PubMed]
- Smith, L.; Broad, S. Do Zoo Visitors Attend to Conservation Messages? A Case Study of an Elephant Exhibit. Tour. Rev. Int. 2007, 11, 225–235. [Google Scholar] [CrossRef]
- Swanagan, J.S. Factors Influencing Zoo Visitors’ Conservation Attitudes and Behavior. J. Environ. Educ. 2000, 31, 26–31. [Google Scholar] [CrossRef]
- Jones, H.; McGregor, P.K.; Farmer, H.L.A.; Baker, K.R. The influence of visitor interaction on the behavior of captive crowned lemurs (Eulemur coronatus) and implications for welfare. Zoo Biol. 2016, 35, 222–227. [Google Scholar] [CrossRef]
- Ramont, M.; Leahy, M.; Cronin, K.A. Domestic animal welfare at the zoo: The impact of an animal visitor interaction program on chickens. Anim. Behav. Cog. 2021, 8, 1–14. [Google Scholar] [CrossRef]
- Altmann, J. Observational Study of Behavior: Sampling Methods. Behaviour 1974, 49, 227–266. [Google Scholar] [CrossRef] [PubMed]
- Carlstead, K.; Seidensticker, J.; Baldwin, R. Environmental enrichment for zoo bears. Zoo Biol. 1991, 10, 3–16. [Google Scholar] [CrossRef]
- Fernandez, E.J. Stereotypies and Foraging: Appetitive Search Behaviors and Stereotypies in Captive Animals; VDM Publishing: Saarbrücken, Germany, 2010. [Google Scholar]
- Fernandez, E.J. Appetitive search behaviors and stereotypies in polar bears (Ursus maritimus). Behav. Process. 2021, 182, 104299. [Google Scholar] [CrossRef]
- Fernandez, E.J.; Timberlake, W. Foraging devices as enrichment in captive walruses (Odobenus rosmarus). Behav. Process. 2019, 168, 103943. [Google Scholar] [CrossRef] [PubMed]
- Forthman, D.L.; Elder, S.D.; Bakeman, R.; Kurkowski, T.W.; Noble, C.C.; Winslow, S.W. Effects of feeding enrichment on behavior of three species of captive bears. Zoo Biol. 1992, 11, 187–195. [Google Scholar] [CrossRef]
- Kurt, F.; Garai, M. Stereotypies in Captive Asian Elephants—A Symptom of Social Isolation; International Elephant and Rhino Research Symposium: Vienna, Austria, 2001. [Google Scholar]
- Lawrence, A.; Terlouw, E.M.C. A review of behavioral factors involved in the development and continued performance of stereotypic behaviors in pigs. J. Anim. Sci. 1993, 71, 2815–2825. [Google Scholar] [CrossRef]
- Shepherdson, D.J.; Carlstead, K.; Mellen, J.D.; Seidensticker, J. The influence of food presentation on the behavior of small cats in confined environments. Zoo Biol. 1993, 12, 203–216. [Google Scholar] [CrossRef]
- Rees, P.A. Activity budgets and the relationship between feeding and stereotypic behaviors in Asian elephants (Elephas maximus) in a Zoo. Zoo Biol. 2009, 28, 79–97. [Google Scholar] [CrossRef] [PubMed]
- Posta, B.; Huber, R.; Moore, D.E. The effects of housing on zoo elephant behavior: A quantitative case study of diurnal and seasonal variation. Int. J. Comp. Psychol. 2013, 26, 37–52. [Google Scholar]
- Rees, P.A. Low environmental temperature causes an increase in stereotypic behaviour in captive Asian elephants (Elephas maximus). J. Therm. Biol. 2004, 29, 37–43. [Google Scholar] [CrossRef]
- Keulen-Kromhout, G.V. Zoo enclosures for bears. Int. Zoo Yearb. 1978, 18, 177–186. [Google Scholar] [CrossRef]
- Bassett, L.; Buchanan-Smith, H.M. Effects of predictability on the welfare of captive animals. Appl. Anim. Behav. Sci. 2007, 102, 223–245. [Google Scholar] [CrossRef]
- Bloomsmith, M.A.; Lambeth, S.P. Effects of predictable versus unpredictable feeding schedules on chimpanzee behavior. Appl. Anim. Behav. Sci. 1995, 44, 65–74. [Google Scholar] [CrossRef]
- Schneider, M.; Nogge, G.; Kolter, L. Implementing unpredictability in feeding enrichment for Malayan sun bears (Helarctos malayanus). Zoo Biol. 2014, 33, 54–62. [Google Scholar] [CrossRef]
- Stoinski, T.S.; Daniel, E.; Maple, T.L. A preliminary study of the behavioral effects of feeding enrichment on African elephants. Zoo Biol. 2000, 19, 485–493. [Google Scholar] [CrossRef]
- Wagman, J.D.; Lukas, K.E.; Dennis, P.M.; Willis, M.A.; Carroscia, J.; Gindlesperger, C.; Schook, M.W. A work-for-food enrichment program increases exploration and decreases stereotypies in four species of bears. Zoo Biol. 2018, 37, 3–15. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, E.J.; Kinley, R.C.; Timberlake, W. Training penguins to interact with enrichment devices for lasting effects. Zoo Biol. 2019, 38, 481–489. [Google Scholar] [CrossRef]


| Behavioral Class and Behaviors (Abbreviations) | Definitions |
|---|---|
| Active | |
| Enrichment Contact (EC) | Manipulating any nonfood enrichment item. |
| Locomotion (Lo) | Directed nonrepetitive movement. |
| Digging/Dusting (DD) | Manipulating substrate or throwing substrate on body. |
| Play/Bathe (PB) | Splashing, kicking, bouncing, or other animated behavior (must be alone). |
| Forage | |
| Feeding (FE) | Eating any food provided by the keepers. |
| Foraging Other (FO) | Reaching over the fence, browsing on exhibit shrubs, or grazing on exhibit grasses. |
| Enrichment Feeding (EF) | Eating any item inside an enrichment device. |
| Drinking (Dr) | Mouth contact with pool or other water source. |
| Social | |
| Affiliation (Af) | Conspecific, prosocial behaviors (e.g., play, trunk tangle, or caressing with elephant). |
| Interacting with Keeper (IK) | Any keeper-related interaction. |
| Inactive | |
| Lying Down (LD) | At least two limbs no longer upright (e.g., on side). |
| Standing (St) | Completely upright with no movement. |
| Gate Hugging (GH) | Standing, but in front of a gate and without movement. |
| Stereotypy | |
| Pacing (Pa) | Moving in a repetitive pattern, with completion from point A to B and back to point A (must include at least one full A–B–A movement) or circling. |
| Rocking (Ro) | Moving back and forth without locomotion. Must include at least one full back-and-forth motion. |
| Other | |
| Vocalization (Vo) | Making nondirected vocal sound. |
| Social Aggression (AG) | Aggressive behavior that includes physical contact. |
| Threat/Display (TD) | Aggressive behavior, but without physical contact. |
| Urinate/Defecate (UD) | Urination or defecation. |
| Out of Sight (OS) | Not visible to the observer. |
| Other (Ot) | Engaged in a behavior not listed above. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fernandez, E.J.; Upchurch, B.; Hawkes, N.C. Public Feeding Interactions as Enrichment for Three Zoo-Housed Elephants. Animals 2021, 11, 1689. https://doi.org/10.3390/ani11061689
Fernandez EJ, Upchurch B, Hawkes NC. Public Feeding Interactions as Enrichment for Three Zoo-Housed Elephants. Animals. 2021; 11(6):1689. https://doi.org/10.3390/ani11061689
Chicago/Turabian StyleFernandez, Eduardo J., Bruce Upchurch, and Nancy C. Hawkes. 2021. "Public Feeding Interactions as Enrichment for Three Zoo-Housed Elephants" Animals 11, no. 6: 1689. https://doi.org/10.3390/ani11061689
APA StyleFernandez, E. J., Upchurch, B., & Hawkes, N. C. (2021). Public Feeding Interactions as Enrichment for Three Zoo-Housed Elephants. Animals, 11(6), 1689. https://doi.org/10.3390/ani11061689







