Positive Human Contact and Housing Systems Impact the Responses of Piglets to Various Stressors
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals and Treatments
- Housing system
- FC—Farrowing crate
- LP—Loose pen
- Human contact treatment
- +HC—Positive human contact
- C—Routine human contact
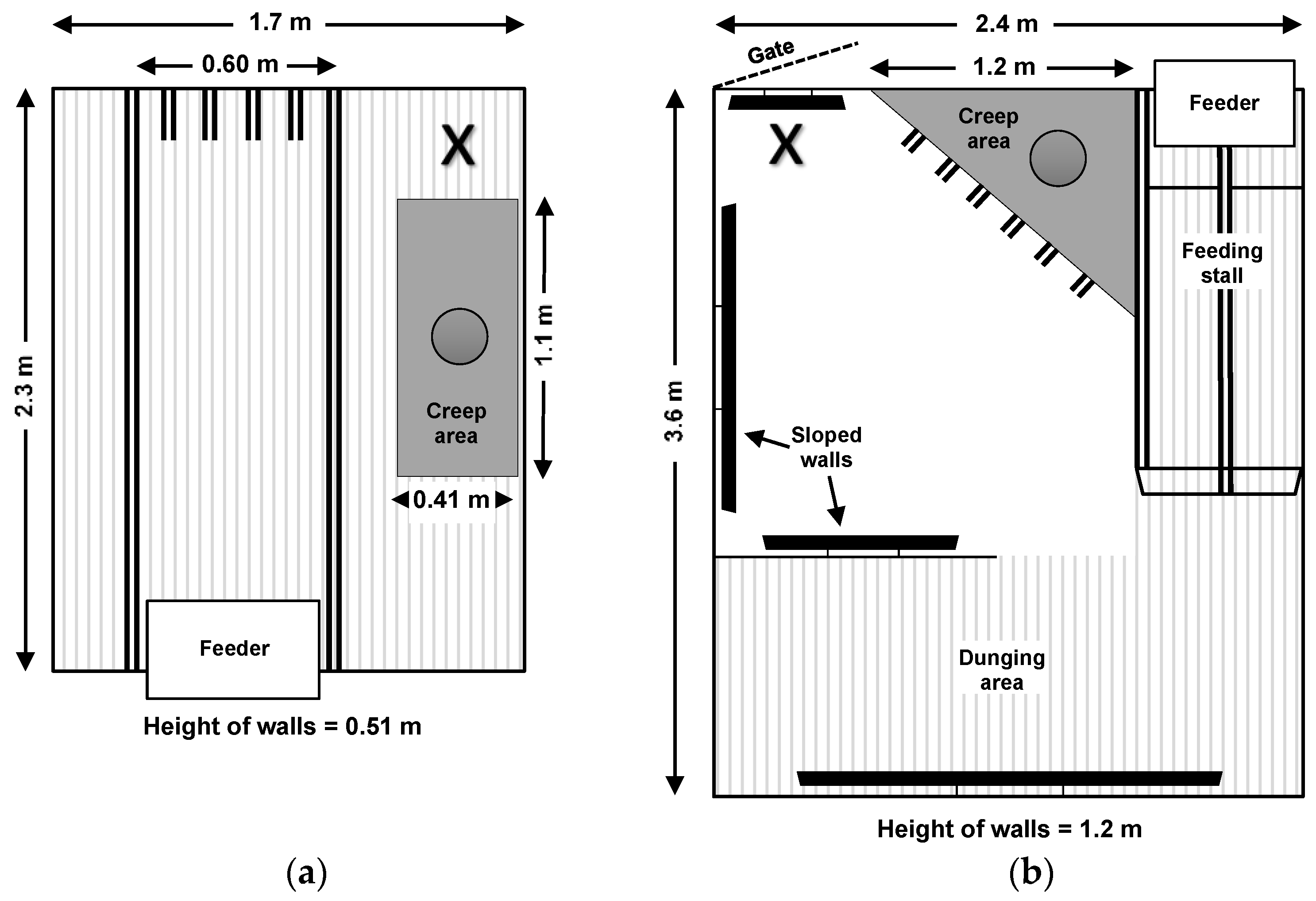
2.1.1. Housing Systems during the Farrowing and Lactation Period
2.1.2. Human Contact Treatment during the Lactation Period
2.2. Human Contact Treatment Allocation
2.3. Management of Animals
2.4. Measurements
2.4.1. Behavioural and Physiological Responses to Routine Husbandry Procedures
2.4.2. Behavioural Responses to Novelty and Humans in a Standard Test
2.4.3. Behavioural and Physiological Responses to Weaning
2.4.4. Sow Reproductive Performance and Piglet Growth, Injuries and Survival
2.4.5. Sample Collection Details and Assay Characteristics
2.5. Statistical Analysis
3. Results
3.1. Responses to Routine Husbandry Procedures at 3 d and 3 wk of Age
3.1.1. Behavioural Responses to Processing and Vaccination
3.1.2. Physiological Responses to Processing
3.2. Responses to Novelty and Humans in a Standard Test at 2 wk of Age
3.2.1. Behavioural Responses to Capture before and after Testing
3.2.2. Behavioural Responses to Novel and Human Stimuli during the Test
3.3. Responses to Weaning
3.3.1. Piglet Behaviour after Weaning
3.3.2. Physiological Responses after Weaning
3.4. Sow Reproductive Performance and Piglet Growth, Injuries and Survival
3.4.1. Injury Scores
3.4.2. Piglet Weights
3.4.3. Piglet Survival during Lactation
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Iacoviella, B.M.; Charney, D.S. Cognitive and behavioral components of resilience to stress. In Stress Resilience: Molecular and Behavioral Aspects; Chen, A., Ed.; Academic Press: London, UK, 2019; p. 23. [Google Scholar]
- Parker, K.J.; Maestripieri, D. Identifying key features of early stressful experiences that produce stress vulnerability and resilience in primates. Neurosci. Biobehav. Rev. 2011, 35, 1466–1483. [Google Scholar] [CrossRef] [PubMed]
- Lyons, D.; Parker, K.; Katz, M.; Schatzberg, A. Developmental cascades linking stress inoculation, arousal regulation, and resilience. Front. Behav. Neurosci. 2009, 3. [Google Scholar] [CrossRef] [PubMed]
- Lyons, D.M.; Parker, K.J.; Schatzberg, A.F. Animal models of early life stress: Implications for understanding resilience. Dev. Psychobiol. 2010, 52, 402–410. [Google Scholar] [CrossRef] [PubMed]
- Baxter, E.M.; Lawrence, A.B.; Edwards, S.A. Alternative farrowing systems: Design criteria for farrowing systems based on the biological needs of sows and piglets. Animal 2011, 5, 580–600. [Google Scholar] [CrossRef] [PubMed]
- Martin, J.E.; Ison, S.H.; Baxter, E.M. The influence of neonatal environment on piglet play behaviour and post-weaning social and cognitive development. Appl. Anim. Behav. Sci. 2015, 163, 69–79. [Google Scholar] [CrossRef]
- Cronin, G.M.; Simpson, G.J.; Hemsworth, P.H. The effects of the gestation and farrowing environments on sow and piglet behaviour and piglet survival and growth in early lactation. Appl. Anim. Behav. Sci. 1996, 46, 175–192. [Google Scholar] [CrossRef]
- Thodberg, K.; Jensen, K.H.; Herskin, M.S. Nursing behaviour, postpartum activity and reactivity in sows: Effects of farrowing environment, previous experience and temperament. Appl. Anim. Behav. Sci. 2002, 77, 53–76. [Google Scholar] [CrossRef]
- Singh, C.; Verdon, M.; Cronin, G.M.; Hemsworth, P.H. The behaviour and welfare of sows and piglets in farrowing crates or lactation pens. Animal 2017, 11, 1210–1221. [Google Scholar] [CrossRef]
- Yonezawa, T.; Takahashi, A.; Imai, S.; Okitsu, A.; Komiyama, S.; Irimajiri, M.; Matsuura, A.; Yamazaki, A.; Hodate, K. Effects of outdoor housing of piglets on behavior, stress reaction and meat characteristics. Asian Australas J. Anim. Sci. 2012, 25, 886–894. [Google Scholar] [CrossRef]
- De Jonge, F.H.; Bokkers, E.A.M.; Schouten, W.G.P.; Helmond, F.A. Rearing piglets in a poor environment: Developmental aspects of social stress in pigs. Physiol. Behav. 1996, 60, 389–396. [Google Scholar] [CrossRef]
- Chaloupkova, H.; Illmann, G.; Neuhauserova, K.; Tomanek, M.; Valis, L. Preweaning housing effects on behavior and physiological measures in pigs during the suckling and fattening periods. J. Anim. Sci. 2007, 1741–1749. [Google Scholar] [CrossRef] [PubMed]
- Backus, B.L.; McGlone, J.J. Evaluating environmental enrichment as a method to alleviate pain after castration and tail docking in pigs. Appl. Anim. Behav. Sci. 2018, 204, 37–42. [Google Scholar] [CrossRef]
- Oostindjer, M.; van den Brand, H.; Kemp, B.; Bolhuis, J.E. Effects of environmental enrichment and loose housing of lactating sows on piglet behaviour before and after weaning. Appl. Anim. Behav. Sci. 2011, 134, 31–41. [Google Scholar] [CrossRef]
- Oostindjer, M.; Bolhuis, J.E.; Mendl, M.; Held, S.; Gerrits, W.; van den Brand, H.; Kemp, B. Effects of environmental enrichment and loose housing of lactating sows on piglet performance before and after weaning. J. Anim. Sci. 2010, 88, 3554–3562. [Google Scholar] [CrossRef]
- Piñeiro, M.; Piñeiro, C.; Carpintero, R.; Morales, J.; Campbell, F.M.; Eckersall, P.D.; Toussaint, M.J.M.; Lampreave, F. Characterisation of the pig acute phase protein response to road transport. Vet. J. 2007, 173, 669–674. [Google Scholar] [CrossRef] [PubMed]
- Salamano, G.; Mellia, E.; Candiani, D.; Ingravalle, F.; Bruno, R.; Ru, G.; Doglione, L. Changes in haptoglobin, C-reactive protein and pig-MAP during a housing period following long distance transport in swine. Vet. J. 2008, 177, 110–115. [Google Scholar] [CrossRef]
- Murata, H. Stress and acute phase protein response: An inconspicuous but essential linkage. Vet. J. 2007, 173, 473–474. [Google Scholar] [CrossRef]
- Sommavilla, R.; Hötzel, M.J.; Dalla Costa, O.A. Piglets’ weaning behavioural response is influenced by quality of human–animal interactions during suckling. Animal 2011, 5, 1426–1431. [Google Scholar] [CrossRef]
- Tallet, C.; Sy, K.; Prunier, A.; Nowak, R.; Boissy, A.; Boivin, X. Behavioural and physiological reactions of piglets to gentle tactile interactions vary according to their previous experience with humans. Livest. Sci. 2014, 167, 331–341. [Google Scholar] [CrossRef]
- Tanida, H.; Miura, A.; Tanaka, T.; Yoshimoto, T. Behavioural response to humans in individually handled weanling pigs. Appl. Anim. Behav. Sci. 1995, 42, 249–259. [Google Scholar] [CrossRef]
- Hemsworth, P.H.; Coleman, G.J. Human-Livestock Interactions: The Stockperson and the Productivity and Welfare of Intensively Farmed Animals, 2nd ed.; CABI Publishing: Wallingford, UK, 2011. [Google Scholar]
- Muns, R.; Rault, J.L.; Hemsworth, P. Positive human contact on the first day of life alters the piglet’s behavioural response to humans and husbandry practices. Physiol. Behav. 2015, 151, 162–167. [Google Scholar] [CrossRef] [PubMed]
- Hayes, M.E.; Hemsworth, L.M.; Morrison, R.S.; Butler, K.L.; Rice, M.; Rault, J.-L.; Hemsworth, P.H. Effects of positive human contact during gestation on the behaviour, physiology and reproductive performance of sows. Animals 2021, 11, 214. [Google Scholar] [CrossRef] [PubMed]
- Brajon, S.; Laforest, J.-P.; Bergeron, R.; Tallet, C.; Hötzel, M.-J.; Devillers, N. Persistency of the piglet’s reactivity to the handler following a previous positive or negative experience. Appl. Anim. Behav. Sci. 2015, 162, 9–19. [Google Scholar] [CrossRef]
- Hemsworth, P.H.; Barnett, J.L. The effects of early contact with humans on the subsequent level of fear of humans in pigs. Appl. Anim. Behav. Sci. 1992, 35, 83–90. [Google Scholar] [CrossRef]
- De Oliveira, D.; Paranhos da Costa, M.J.R.; Zupan, M.; Rehn, T.; Keeling, L.J. Early human handling in non-weaned piglets: Effects on behaviour and body weight. Appl. Anim. Behav. Sci. 2015, 164, 56–63. [Google Scholar] [CrossRef]
- Zupan, M.; Rehn, T.; de Oliveira, D.; Keeling, L.J. Promoting positive states: The effect of early human handling on play and exploratory behaviour in pigs. Animal 2016, 10, 135–141. [Google Scholar] [CrossRef]
- Büttner, K.; Czycholl, I.; Basler, H.; Krieter, J. Effects of an intensified human–animal interaction on tail biting in pigs during the rearing period. J. Agric. Sci. 2018, 156, 1039–1046. [Google Scholar] [CrossRef]
- De Oliveira, D.; Keeling, L.J.; Paranhos da Costa, M.J.R. Individual variation over time in piglet’s reactions to early handling and its association to weight gain. Appl. Anim. Behav. Sci. 2019, 215, 7–12. [Google Scholar] [CrossRef]
- Villain, A.S.; Lanthony, M.; Guérin, C.; Noûs, C.; Tallet, C. Manipulable object and human contact: Preferences and modulation of emotional states in weaned piglets. Front. Vet. Sci. 2020, 7. [Google Scholar] [CrossRef]
- Luna, D.; González, C.; Byrd, C.J.; Palomo, R.; Huenul, E.; Figueroa, J. Do domestic pigs acquire a positive perception of humans through observational social learning? Animals 2021, 11, 127. [Google Scholar] [CrossRef] [PubMed]
- Leidig, M.S.; Hertrampf, B.; Failing, K.; Schumann, A.; Reiner, G. Pain and discomfort in male piglets during surgical castration with and without local anaesthesia as determined by vocalisation and defence behaviour. Appl. Anim. Behav. Sci. 2009, 116, 174–178. [Google Scholar] [CrossRef]
- Center for Conservation Bioacoustics. Raven Pro: Interactive Sound Analysis Software; Version 1.5; The Cornell Lab of Ornithology: Ithaca, NY, USA, 2014. [Google Scholar]
- Widowski, T.M.; Cottrell, T.; Dewey, C.E.; Friendship, R.M. Observations of piglet-directed behavior patterns and skin lesions in eleven commercial swine herds. J. Swine Health Prod. 2003, 11, 181–185. [Google Scholar]
- VSN International. Genstat for Windows, 18th ed.; VSN International: Hemel Hempstead, UK, 2015. [Google Scholar]
- Hinde, R.A. Animal Behaviour: A Synthesis of Ethology and Comparative Psychology, 2nd ed.; McGraw-Hill: New York, NY, USA, 1970. [Google Scholar]
- Moberg, G.P. Biological response to stress: Implications for animal welfare. In The Biology of Animal Stress; Moberg, G.P., Mench, J.A., Eds.; CABI Publishing: Wallingford, UK, 2000; pp. 1–21. [Google Scholar]
- Hemsworth, P.H.; Barnett, J.L.; Hansen, C. The influence of handling by humans on the behaviour, reproduction and corticosteroids of male and female pigs. Appl. Anim. Behav. Sci. 1986, 15, 303–314. [Google Scholar] [CrossRef]
- Hemsworth, P.H.; Barnett, J.L.; Hansen, C.; Gonyou, H.W. The influence of early contact with humans on subsequent behavioural response of pigs to humans. Appl. Anim. Behav. Sci. 1986, 15, 55–63. [Google Scholar] [CrossRef]
- Jones, R.B.; Waddington, D. Modification of fear in domestic chicks, Gallus gallus domesticus, via regular handling and early environmental enrichment. Anim. Behav. 1992, 43, 1021–1033. [Google Scholar] [CrossRef]
- Turpin, D.L.; Langendijk, P.; Sharp, C.; Pluske, J.R. Improving welfare and production in the peri-weaning period: Effects of co-mingling and intermittent suckling on the stress response, performance, behaviour, and gastrointestinal tract carbohydrate absorption in young pigs. Livest. Sci. 2017, 203, 82–91. [Google Scholar] [CrossRef]
- Van der Meulen, J.; Koopmans, S.J.; Dekker, R.A.; Hoogendoorn, A. Increasing weaning age of piglets from 4 to 7 weeks reduces stress, increases post-weaning feed intake but does not improve intestinal functionality. Animal 2010, 4, 1653–1661. [Google Scholar] [CrossRef]
- Moeser, A.J.; Vander Klok, C.; Ryan, K.A.; Wooten, J.G.; Little, D.; Cook, V.L.; Blikslager, A.T. Stress signaling pathways activated by weaning mediate intestinal dysfunction in the pig. Am. J. Physiol. Gastrointest. Liver Physiol. 2007, 292, G173–G181. [Google Scholar] [CrossRef]
- Puppe, B.; Tuchscherer, M.; Tuchscherer, A. The effect of housing conditions and social environment immediately after weaning on the agonistic behaviour, neutrophil/lymphocyte ratio, and plasma glucose level in pigs. Livest. Prod. Sci. 1997, 48, 157–164. [Google Scholar] [CrossRef]
- Turpin, D.L.; Langendijk, P.; Tai-Yuan, C.; Lines, D.; Pluske, J.R. Intermittent suckling causes a transient increase in cortisol that does not appear to compromise selected measures of piglet welfare and stress. Animals 2016, 6, 24. [Google Scholar] [CrossRef]
- Murata, H.; Shimada, N.; Yoshioka, M. Current research on acute phase proteins in veterinary diagnosis: An overview. Vet. J. 2004, 168, 28–40. [Google Scholar] [CrossRef]
- De, U.K.; Nandi, S.; Mukherjee, R.; Gaur, G.K.; Verma, M.R. Identification of some plasma biomarkers associated with early weaning stress in crossbred piglets. Comp. Clin. Path. 2017, 26, 343–349. [Google Scholar] [CrossRef]
- Pomorska-Mol, M.; Kwit, K.; Markowska-Daniel, I. Major acute phase proteins in pigs from birth until slaughter. Bull. Vet. Inst. Pulawy 2012, 56, 553–557. [Google Scholar] [CrossRef]
- Sauerwein, H.; Schmitz, S.; Hiss, S. The acute phase protein haptoglobin and its relation to oxidative status in piglets undergoing weaning-induced stress. Redox Rep. 2005, 10, 295–302. [Google Scholar] [CrossRef] [PubMed]
- Ott, S.; Soler, L.; Moons, C.P.; Kashiha, M.A.; Bahr, C.; Vandermeulen, J.; Janssens, S.; Gutiérrez, A.M.; Escribano, D.; Cerón, J.J.; et al. Different stressors elicit different responses in the salivary biomarkers cortisol, haptoglobin, and chromogranin A in pigs. Res. Vet. Sci. 2014, 97, 124–128. [Google Scholar] [CrossRef]
- Kutzer, T.; Bünger, B.; Kjaer, J.B.; Schrader, L. Effects of early contact between non-littermate piglets and of the complexity of farrowing conditions on social behaviour and weight gain. Appl. Anim. Behav. Sci. 2009, 121, 16–24. [Google Scholar] [CrossRef]
- Chaloupková, H.; Illmann, G.; Bartoš, L.; Špinka, M. The effect of pre-weaning housing on the play and agonistic behaviour of domestic pigs. Appl. Anim. Behav. Sci. 2007, 103, 25–34. [Google Scholar] [CrossRef]
- Turner, S.P.; Roehe, R.; D’Eath, R.B.; Ison, S.H.; Farish, M.; Jack, M.C.; Lundeheim, N.; Rydhmer, L.; Lawrence, A.B. Genetic validation of postmixing skin injuries in pigs as an indicator of aggressiveness and the relationship with injuries under more stable social conditions. J. Anim. Sci. 2009, 87, 3076–3082. [Google Scholar] [CrossRef]
- Morrison, R.; Baxter, E. Developing Commercially Viable, Confinement Free Farrowing and Lactation Systems—Part 1: PigSAFE System. Final Report for the Co-Operative Research Centre for High Integrity Australian Pork, 1A-105. 2014. Available online: http://porkcrc.com.au/wp-content/uploads/2013/12/1A-105-Part-1-Report.pdf (accessed on 29 May 2020).
- Morrison, R.; Athorn, R.; McDonald, E. Developing Commercially Viable, Confinement Free Farrowing and Lactation Systems—Part 2: Utilising confinement free systems to maximise economic performance. Final Report Prepared for the Co-Operative Research Centre for High Integrity Australian Pork, 1A-105. 2015. Available online: http://porkcrc.com.au/wp-content/uploads/2015/05/1A-105-Part-2-Final-Report.pdf (accessed on 29 May 2020).
- King, R.L.; Baxter, E.M.; Matheson, S.M.; Edwards, S.A. Consistency is key: Interactions of current and previous farrowing system on litter size and piglet mortality. Animal 2019, 13, 180–188. [Google Scholar] [CrossRef]
- Thodberg, K.; Jensen, K.H.; Herskin, M.S. Nest building and farrowing in sows: Relation to the reaction pattern during stress, farrowing environment and experience. Appl. Anim. Behav. Sci. 2002, 77, 21–42. [Google Scholar] [CrossRef]
- Jarvis, S.; Van der Vegt, B.J.; Lawrence, A.B.; McLean, K.A.; Deans, L.A.; Chirnside, J.; Calvert, S.K. The effect of parity and environmental restriction on behavioural and physiological responses of pre-parturient pigs. Appl. Anim. Behav. Sci. 2001, 71, 203–216. [Google Scholar] [CrossRef]
- Hemsworth, P.H. Key determinants of pig welfare: Implications of animal management and housing design on livestock welfare. Anim. Prod. Sci. 2018, 58, 1375–1386. [Google Scholar] [CrossRef]
- Baxter, E.M.; Andersen, I.L.; Edwards, S.A. Sow welfare in the farrowing crate and alternatives. In Advances in Pig Welfare; Špinka, M., Ed.; Woodhead Publishing: Duxford, UK, 2018; pp. 27–72. [Google Scholar] [CrossRef]

| Measurement | Human Contact | Housing System | p-Value | ||||
|---|---|---|---|---|---|---|---|
| +HC | C | FC | LP | Human Contact | Housing System | Interaction | |
| Processing at 3 d of age | |||||||
| Escape behaviour score at capture and iron injection | 1.32 (1.06, 1.57) | 1.70 (1.44, 1.95) | 1.32 (1.06, 1.57) | 1.70 (1.44, 1.95) | 0.042 | 0.043 | 0.691 |
| Escape behaviour score at oral treatment administration | 1.35 (1.16, 1.55) | 1.68 (1.49, 1.88) | 1.45 (1.25, 1.64) | 1.59 (1.39, 1.79) | 0.026 | 0.337 | 0.251 |
| Escape behaviour score at tail clipping | 1.61 (1.38, 1.84) | 1.61 (1.37, 1.84) | 1.69 (1.46, 1.91) | 1.53 (1.29, 1.76) | 0.970 | 0.340 | 0.126 |
| Duration of vocalisations (s) | 4.47 (3.41, 5.52) | 6.30 (5.31, 7.29) | 5.68 (4.62, 6.74) | 5.08 (4.09, 6.07) | 0.018 | 0.407 | 0.693 |
| Number of vocalisations | 7.20 (5.87, 8.84) | 9.53 (7.88, 11.5) | 7.94 (6.47, 9.75) | 8.70 (7.19, 10.5) | 0.067 | 0.577 | 0.618 |
| Cortisol (ng/mL) | 48.8 (40.1, 59.2) | 51.2 (42.3, 61.8) | 45.6 (37.7, 55.1) | 54.6 (44.9, 66.3) | 0.711 | 0.214 | 0.368 |
| Vaccination at 3 wk of age | |||||||
| Escape behaviour score at capture and vaccination | 1.49 (1.32, 1.65) | 1.86 (1.69, 2.03) | 1.77 (1.60, 1.93) | 1.57 (1.39, 1.74) | 0.005 | 0.114 | 0.107 |
| Measurement | Human Contact | Housing System | Human Contact | ||||
|---|---|---|---|---|---|---|---|
| +HC | C | FC | LP | Human Contact | Housing System | Interaction | |
| Response to capture | |||||||
| Escape behaviour score at capture from home pen pre-test | 1.21 (0.881, 1.54) | 2.14 (1.81, 2.47) | 1.50 (1.17, 1.83) | 1.85 (1.52, 2.18) | <0.001 | 0.148 | 0.911 |
| Escape behaviour score at capture from arena post-test | 1.48 (1.28, 1.68) | 2.13 (1.93, 2.32) | 1.91 (1.71, 2.11) | 1.69 (1.49, 1.89) | <0.001 | 0.290 | 0.172 |
| Response to empty arena | |||||||
| Number of sections entered | 4.53 (3.66, 5.39) | 4.94 (4.08, 5.80) | 5.30 (4.44, 6.16) | 4.16 (3.30, 5.03) | 0.517 | 0.072 | 0.763 |
| Response to traffic cone | |||||||
| Latency to approach 0.6 m (s) | 20.5 (13.2, 27.8) | 25.8 (18.7, 32.9) | 15.5 (8.17, 22.8) | 30.8 (23.8, 38.0) | 0.291 | 0.005 | 0.107 |
| Latency to interact (s) | 27.3 (20.1, 34.6) | 30.0 (23.0. 37.1) | 22.8 (15.6, 30.1) | 34.5 (27.5, 41.6) | 0.559 | 0.027 | 0.105 |
| Number of interactions | 2.76 (2.02, 3.78) | 2.67 (1.95, 3.66) | 3.14 (2.29, 4.30) | 2.35 (1.71, 3.22) | 0.970 | 0.202 | 0.206 |
| Time spent within 0.6 m (s) | 20.6 (14.5, 26.7) | 20.7 (14.7, 26.7) | 22.2 (16.1, 28.3) | 19.1 (13.1, 25.1) | 0.982 | 0.448 | 0.218 |
| Response to human hand | |||||||
| Latency to approach 0.6 m (s) | 26.8 (19.7, 33.9) | 29.0 (22.1, 35.9) | 20.9 (13.9, 28.0) | 34.9 (28.0, 41.8) | 0.643 | 0.009 | 0.534 |
| Latency to interact (s) | 34.5 (27.6, 41.2) | 39.5 (32.7, 46.2) | 31.0 (24.2, 37.8) | 42.9 (36.0, 49.8) | 0.231 | 0.008 | 0.799 |
| Number of interactions | 1.34 (0.813, 2.21) | 1.23 (0.747, 2.03) | 1.59 (0.964, 2.64) | 1.04 (0.627, 1.71) | 0.769 | 0.132 | 0.169 |
| Time spent within 0.6 m (s) | 14.2 (9.10, 19.4) | 13.5 (8.55, 18.5) | 15.8 (10.6, 20.9) | 12.0 (7.02, 17.0) | 0.863 | 0.326 | 0.445 |
| Response to standing human | |||||||
| Latency to approach 0.6 m (s) | 24.0 (16.6, 31.4) | 34.9 (27.7. 42.0) | 24.1 (16.7, 31.4) | 34.8 (27.6, 42.0) | 0.048 | 0.058 | 0.420 |
| Latency to interact (s) | 30.4 (22.1, 38.7) | 41.2 (32.9, 49.4) | 31.0 (22.7, 39.3) | 40.6 (32.2, 49.0) | 0.042 | 0.078 | 0.546 |
| Number of interactions | 2.50 (1.39, 4.49) | 1.79 (0.995, 3.21) | 2.38 (1.32, 4.30) | 1.87 (1.04, 3.38) | 0.322 | 0.444 | 0.501 |
| Time spent within 0.6 m (s) | 20.7 (13.2, 28.1) | 16.1 (8.68, 23.6) | 20.4 (12.9, 27.8) | 16.4 (8.85, 24.0) | 0.355 | 0.423 | 0.393 |
| Measurement | Human Contact | Housing System | p-Value | ||||||
|---|---|---|---|---|---|---|---|---|---|
| +HC | C | Odds Ratio | FC | LP | Odds Ratio | Human Contact | Housing System | Interaction | |
| Upright, stationary | 71.9 | 67.1 | 1.25 (0.74, 2.12) | 59.1 | 78.3 | 2.49 (1.47, 4.22) | 0.639 | 0.002 | 0.365 |
| Upright, walking | 34.8 | 37.3 | 0.90 (0.67, 1.21) | 32.0 | 40.3 | 1.43 (1.06, 1.93) | 0.300 | 0.024 | 0.290 |
| Vocalising | 2.45 | 2.31 | 1.06 (0.81, 1.39) | 1.93 | 2.93 | 1.53 (1.17, 2.00) | 0.947 | 0.004 | 0.389 |
| Nosing pen mate | 19.4 | 20.4 | 1.06 (0.83, 1.36) | 17.8 | 22.2 | 1.32 (1.03, 1.69) | 0.461 | 0.035 | 0.420 |
| Nosing pen floor 1 | 8.55 | 11.4 | 0.83 (0.43, 1.61) | 6.75 | 13.2 | 2.03 (1.04, 3.96) | 0.580 | 0.038 | 0.145 |
| Play behaviour | 0.621 | 1.04 | 0.59 (0.34, 1.04) | 0.885 | 0.729 | 0.82 (0.47, 1.44) | 0.124 | 0.649 | 0.299 |
| Aggressive behaviour | 1.52 | 1.24 | 1.24 (0.54, 2.82) | 1.28 | 1.47 | 1.15 (0.50, 2.63) | 0.684 | 0.666 | 0.115 |
| Tail biting | 0 | 0 | - | 0 | 0 | - | - | - | - |
| Measurement | Human Contact | Housing System | p-Value | ||||
|---|---|---|---|---|---|---|---|
| +HC | C | FC | LP | Human Contact | Housing System | Interaction | |
| 1.5 h post-weaning | |||||||
| Cortisol (ng/mL) | 32.1 (27.0, 38.1) | 31.3 (26.8, 36.7) | 30.1 (25.7, 35.2) | 33.3 (28.1, 39.6) | 0.946 | 0.393 | 0.988 |
| Neutrophil to lymphocyte ratio | 1.15 (0.966, 1.37) | 1.12 (0.937, 1.348) | 1.11 (0.934, 1.32) | 1.16 (0.968, 1.40) | 0.803 | 0.563 | 0.388 |
| 25 h post-weaning | |||||||
| Cortisol (ng/mL) | 33.7 (27.3, 41.4) | 36.2 (29.4, 44.6) | 34.0 (27.7, 41.6) | 35.9 (28.9, 44.6) | 0.652 | 0.597 | 0.012 |
| Haptoglobin (µg/mL) | 1130 (937, 1370) | 697 (574, 846) | 817 (679, 983) | 966 (769, 1173) | 0.002 | 0.212 | 0.139 |
| Measurement | Human Contact | Housing System | p-Value | ||||
|---|---|---|---|---|---|---|---|
| +HC | C | FC | LP | Human Contact | Housing System | Interaction | |
| Injuries | |||||||
| 2 wk of age | 1.03 (0.819, 1.23) | 1.13 (0.919, 1.33) | 0.850 (0.644, 1.06) | 1.30 (1.09, 1.51) | 0.503 | 0.004 | 0.185 |
| 2 d post-weaning | 0.944 (0.675, 1.21) | 1.59 (1.32, 1.85) | 1.31 (1.05, 1.57) | 1.22 (0.939, 1.50) | 0.003 | 0.641 | 0.706 |
| Weights | |||||||
| 3 d of age (kg) | 1.55 (1.41, 1.69) | 1.69 (1.55, 1.83) | 1.60 (1.46, 1.74) | 1.64 (1.50, 1.78) | 0.162 | 0.677 | 0.895 |
| 18 d of age (kg) | 4.43 (4.07, 4.78) | 4.44 (4.08, 4.80) | 4.45 (4.09, 4.81) | 4.42 (4.06, 4.77) | 0.953 | 0.896 | 0.827 |
| Piglet survival | |||||||
| Number born alive 1 | - | - | 11.8 (10.8, 12.8) | 10.9 (9.88, 11.9) | - | 0.226 | - |
| Number stillborn 1 | - | - | 0.750 (0.260, 1.24) | 1.05 (0.376, 1.72) | - | 0.536 | - |
| Number weaned 2 | 8.85 (8.06, 9.64) | 8.90 (8.11, 9.69) | 9.50 (8.71, 10.3) | 8.25 (7.46, 9.04) | 0.931 | 0.035 | 0.146 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hayes, M.E.; Hemsworth, L.M.; Morrison, R.S.; Tilbrook, A.J.; Hemsworth, P.H. Positive Human Contact and Housing Systems Impact the Responses of Piglets to Various Stressors. Animals 2021, 11, 1619. https://doi.org/10.3390/ani11061619
Hayes ME, Hemsworth LM, Morrison RS, Tilbrook AJ, Hemsworth PH. Positive Human Contact and Housing Systems Impact the Responses of Piglets to Various Stressors. Animals. 2021; 11(6):1619. https://doi.org/10.3390/ani11061619
Chicago/Turabian StyleHayes, Megan E., Lauren M. Hemsworth, Rebecca S. Morrison, Alan J. Tilbrook, and Paul H. Hemsworth. 2021. "Positive Human Contact and Housing Systems Impact the Responses of Piglets to Various Stressors" Animals 11, no. 6: 1619. https://doi.org/10.3390/ani11061619
APA StyleHayes, M. E., Hemsworth, L. M., Morrison, R. S., Tilbrook, A. J., & Hemsworth, P. H. (2021). Positive Human Contact and Housing Systems Impact the Responses of Piglets to Various Stressors. Animals, 11(6), 1619. https://doi.org/10.3390/ani11061619






