Recommendations for Abnormal Behaviour Ethograms in Monkey Research
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethical Approval
2.2. Subjects and Housing
2.3. Behavioural Observations
2.4. Statistical Analyses
3. Results
3.1. Descriptive Statistics
3.2. Do the Hypothesized Subcategories of Abnormal Behaviours have Convergent Validity?
3.3. Forming New Subcategories
3.4. Are Any of the Newly Created Subcategories Related to Each Other?
4. Discussion
4.1. Self-Sucking
4.2. Hair-Plucking
4.3. Threat-Biting
4.4. Being ‘Withdrawn’
4.5. Self-Biting and Correlates
4.6. Bouncing and Correlates
4.7. Pacing and Head-Twisting Subcategory
4.8. Flipping and Eye-Poking Subcategory
4.9. Strengths and Limitations
4.10. Future Research
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Independent Variables | Unadjusted Odds Ratio and p-Value | Adjusted Odds Ratio * | 95% CI * | p-Value * | Positively and Significantly Predicts Bouncing? * (Result the Same if Age, Sex and Scanning Level not Controlled for?) |
|---|---|---|---|---|---|
| Flipping presence vs. absence | 1.29 (0.219) | 0.23 | 0.03–1.80 | 0.885 | No (Unadjusted relationship is also non-significant, yet positive) |
| Twirling presence vs. absence | 2.37 (0.013) | 0.64 | 0.08–5.20 | 0.662 | No (Unadjusted relationship is positive and significant) |
| Swinging presence vs. absence | 3.08 (<0.001) | 2.60 | 0.99–6.82 | 0.026 | Yes (Unadjusted relationship is also positive and significant) |
| Rocking presence vs. absence | 2.78 (<0.001) | 2.60 | 0.90–7.50 | 0.039 | Yes (Unadjusted relationship is also positive and significant) |
| Independent Variables | Unadjusted Odds Ratio and p-Value | Adjusted Odds Ratio * | 95% CI * | p-Value * | Positively and Significantly Predicts Hair-Plucking? * (Result the Same if Age, Sex and Scanning Level not Controlled for?) |
|---|---|---|---|---|---|
| Self-clasping presence vs. absence | 0.48 (0.835) | 0.33 | 0.04–2.63 | 0.835 | No (Unadjusted relationship is also negative) |
| Eye-poking presence vs. absence 2 | 0.27 (0.912) | 0.19 | 0.03–1.45 | 0.946 | No (Unadjusted relationship is also negative) |
| Independent Variables | Unadjusted Odds Ratio and p-Value | Adjusted Odds Ratio * | 95% CI * | p-Value * | Positively and Significantly Predicts Floating Limb? * (Result the Same if Age, Sex and Scanning Level not Controlled for?) |
|---|---|---|---|---|---|
| Leg-lifting presence vs. absence | 10.51 (<0.001) | 8.45 | 0.88–80.78 | 0.032 | Yes (Unadjusted relationship is also positive and significant) |
| Withdrawn presence vs. absence 2 | 3.83 (0.073) | 2.14 | 0.42–10.95 | 0.182 | No (Unadjusted relationship is also positive and non-significant) |
| Behaviour/Subcategory | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1. Self-bite/self-hit/ self-injurious behaviour | ---- | 10.39 * | 4.47 * | 1.01 | 1.04 | 1.26 | 0.24– 0.28 2a,c | 1.35 | 2.02 3,* | 1.57 3,* | 0.58 | 2.57 * | 0.44 |
| 2. Floating limb/leg-lift | ---- | ---- | 6.24 3,* | 1.15 | 0.95 | 0.66 | 0.09 2a | 1.72 3,* | 2.70 3,* | 1.72 3,* | 0.61 | 16.67 2a,* | 2.00 |
| 3. Self-clasp | ---- | ---- | ---- | 1.26 | 0.84 | 0.50 | 0.12 2a | 1.43 | 1.40 | 2.52 3,* | 0.41 | 0.48 | 1.42 |
| 4. Twirl | ---- | ---- | ---- | ---- | 3.63– 4.14 2b,c,* | 2.68 * | 6.01 * | 3.88 3,* | 0.84 | 1.00 | 0.12 | 0.62 | 1.17 |
| 5. Bounce/rock/swing | ---- | ---- | ---- | ---- | ---- | 3.36 * | 0.30– 0.58 2a,b | 1.18 | 0.48 | 0.15 2a | 0.30 | 0.45 | 1.93 |
| 6. Hang | ---- | ---- | ---- | ---- | ---- | ---- | 0.28 2a | 2.10 3,* | 0.29 | 1.00 | 0.09 | 0.44 | 0.84 |
| 7. Pace/head-twist | ---- | ---- | ---- | ---- | ---- | ---- | ---- | 0.32 2a | 1.30 | 0.02 2a | 0.18 | 0.99 | 0.28 |
| 8. Flip | ---- | ---- | ---- | ---- | ---- | ---- | ---- | ---- | 2.54 * | 0.69 2a | 0.30 | 0.94 | 0.55 |
| 9. Eye-poke | ---- | ---- | ---- | ---- | ---- | ---- | ---- | ---- | ---- | 1.48 | 0.17 | 2.94 3,* | 1.64 |
| 10. Self-suck | ---- | ---- | ---- | ---- | ---- | ---- | ---- | ---- | ---- | ---- | 0.09 3,* | 0.74 | 0.44 |
| 11. Hair-pluck | ---- | ---- | ---- | ---- | ---- | ---- | ---- | ---- | ---- | ---- | ---- | 0.87 | 2.98 |
| 12. Threat-bite | ---- | ---- | ---- | ---- | ---- | ---- | ---- | ---- | ---- | ---- | ---- | ---- | 2.50 |
| 13. Withdrawn | ---- | ---- | ---- | ---- | ---- | ---- | ---- | ---- | ---- | ---- | ---- | ---- | ---- |
References
- Mason, G.; Rushen, J. Stereotypic Animal Behaviour: Fundamentals and Applications to Welfare, 2nd ed.; CABI: London, UK, 2006. [Google Scholar]
- Prescott, M.J.; Nixon, M.E.; Farningham, D.A.H.; Naiken, S. Laboratory macaques: When to wean? Appl. Anim. Behav. Sci. 2012, 137, 194–207. [Google Scholar] [CrossRef]
- Baker, K.C.; Bloomsmith, M.A.; Oettinger, B.; Neu, K.; Griffis, C.; Schoof, V.A.M. Comparing options for pair housing rhesus macaques using behavioral welfare measures. Am. J. Primatol. 2014, 76, 30–42. [Google Scholar] [CrossRef] [PubMed]
- Baker, K.C.; Bloomsmith, M.A.; Oettinger, B.; Neu, K.; Griffis, C.; Schoof, V.; Maloney, M. Benefits of pair housing are consistent across a diverse population of rhesus macaques. Appl. Anim. Behav. Sci. 2012, 137, 148–156. [Google Scholar] [CrossRef] [PubMed]
- Gottlieb, D.H.; Capitanio, J.P.; McCowan, B. Risk factors for stereotypic behavior and self-biting in rhesus macaques (Macaca mulatta): Animal’s history, current environment, and personality. Am. J. Primatol. 2013, 75, 995–1008. [Google Scholar] [CrossRef]
- Gottlieb, D.H.; Maier, A.; Coleman, K. Evaluation of environmental and intrinsic factors that contribute to stereotypic behavior in captive rhesus macaques (Macaca mulatta). Appl. Anim. Behav. Sci. 2015, 171, 184–191. [Google Scholar] [CrossRef]
- Novak, M.A. Self-injurious behavior in rhesus monkeys: New insights into its etiology, physiology, and treatment. Am. J. Primatol. 2003, 59, 3–19. [Google Scholar] [CrossRef]
- Fontenot, M.B.; Wilkes, M.N.; Lynch, C.S. Effects of outdoor housing on self-injurious and stereotypic behavior in adult male rhesus macaques (Macaca mulatta). J. Am. Assoc. Lab. Anim. Sci. 2006, 45, 35–43. [Google Scholar]
- Vandeleest, J.J.; McCowan, B.; Capitanio, J.P. Early rearing interacts with temperament and housing to influence the risk for motor stereotypy in rhesus monkeys (Macaca mulatta). Appl. Anim. Behav. Sci. 2011, 132, 81–89. [Google Scholar] [CrossRef]
- Zhang, Z.; Mao, Y.; Feng, X.; Zheng, N.; Lü, L.; Ma, Y.; Qin, D.; Hu, X. Early adversity contributes to chronic stress induced depression-like behavior in adolescent male rhesus monkeys. Behav. Brain Res. 2016, 306, 154–159. [Google Scholar] [CrossRef]
- Carlsson, H.; Schapiro, S.J.; Farah, I.; Hau, J. Use of primates in research: A global overview. Am. J. Primatol. 2004, 237, 225–237. [Google Scholar] [CrossRef]
- Lankau, E.W.; Turner, P.V.; Mullan, R.J.; Galland, G.G. Use of nonhuman primates in research in North America. J. Am. Assoc. Lab. Anim. Sci. 2014, 53, 278–282. [Google Scholar]
- Rommeck, I.; Anderson, K.; Heagerty, A.; Cameron, A.; McCowan, B. Risk factors and remediation of self-injurious and self-abuse behavior in rhesus macaques. J. Appl. Anim. Welf. Sci. 2009, 12, 61–72. [Google Scholar] [CrossRef]
- Mason, G. Stereotypic behaviour in captive animals: Fundamentals and implications for welfare and beyond. In Stereotypic Animal Behaviour: Fundamentals and Applications to Welfare, 2nd ed.; Mason, G., Rushen, J., Eds.; CABI: London, UK, 2006; pp. 325–356. [Google Scholar]
- Lutz, C.; Well, A.; Novak, M. Stereotypic and self-injurious behavior in rhesus macaques: A survey and retrospective analysis of environment and early experience. Am. J. Primatol. 2003, 60, 1–15. [Google Scholar] [CrossRef]
- Lutz, C.K.; Coleman, K.; Worlein, J.; Novak, M.A. Hair loss and hair-pulling in rhesus macaques (Macaca mulatta). J. Am. Assoc. Lab. Anim. Sci. 2013, 52, 454–457. [Google Scholar]
- Jorgensen, M.J.; Kinsey, J.H.; Novak, M.A. Risk factors for self-injurious behavior in captive rhesus monkeys (Macaca mulatta). Am. J. Primatol. 1998, 45, 187. [Google Scholar]
- Bentson, K.L.; Crockett, C.M.; Wahl, K.L.; Runeson, E.P.; Bellanca, R.U.; Lee, G.H.; Thom, J.P.; Montgomery, H.B.; Yi, M.H.; Mccomas, J.G.S.; et al. Floating limb behaviors and self-biting are associated in laboratory monkeys. Am. J. Primatol. 2010, 72, 725–733. [Google Scholar] [CrossRef]
- Hennessy, M.B.; Chun, K.; Capitanio, J.P. Depressive-like behavior, its sensitization, social buffering, and altered cytokine responses in rhesus macaques moved from outdoor social groups to indoor housing. Soc. Neurosci. 2017, 12, 65–75. [Google Scholar] [CrossRef]
- Hennessy, M.B.; McCowan, B.; Jiang, J.; Capitanio, J.P. Depressive-like behavioral response of adult male rhesus monkeys during routine animal husbandry procedure. Front. Behav. Neurosci. 2014, 8, 1–8. [Google Scholar] [CrossRef]
- Risch, N.; Herrell, R.; Lehner, T.; Liang, K.; Eaves, L.; Hoh, J.; Griem, A.; Kovacs, M.; Ott, J.; Merikangas, K.R. Interaction between the serotonin transporter Gene (5-HTTLPR), stressful life events, and risk of depression: A meta-analysis. JAMA 2009, 301, 2462–2471. [Google Scholar] [CrossRef]
- Gilmer, W.S.; Mckinney, W.T. Early experience and depressive disorders: Human and non-human primate studies. J. Affect. Disord. 2003, 75, 97–113. [Google Scholar] [CrossRef]
- Mineka, S.; Suomi, S.J.; DeLizio, R. Multiple separations in adolescent monkeys: An opponent-process interpretation. J. Exp. Psychol. Gen. 1981, 110, 56–85. [Google Scholar] [CrossRef] [PubMed]
- Willard, S.L.; Shively, C.A. Modeling depression in adult female cynomolgus monkeys (Macaca fascicularis). Am. J. Primatol. 2012, 542, 528–542. [Google Scholar] [CrossRef] [PubMed]
- Qin, D.; Rizak, J.; Chu, X.; Li, Z.; Yang, S.; Lü, L.; Yang, L.; Yang, Q.; Yang, B.; Pan, L.; et al. A spontaneous depressive pattern in adult female rhesus macaques. Sci. Rep. 2015, 5, 11267. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Xu, F.; Xie, L.; Ji, Y.; Cheng, K.; Zhou, Q.; Wang, T.; Shively, C.; Wu, Q.; Gong, W.; et al. Depression-like behavioral phenotypes by social and social plus visual isolation in the adult female Macaca fascicularis. PLoS ONE 2013, 8. [Google Scholar] [CrossRef]
- Laudenslager, M.L.; Boccia, M.L.; Berger, C.L.; Gennaro-Ruggles, M.M.; McFerran, B.; Reite, M.L. Total cortisol, free cortisol, and growth hormone associated with brief social separation experiences in young macaques. Dev. Psychobiol. 1995, 28, 199–211. [Google Scholar] [CrossRef]
- Hinde, R.A. Mother-infant separation in rhesus monkeys. J. Psychosom. Res. 1972, 16, 227–228. [Google Scholar] [CrossRef]
- Spencer-Booth, Y.; Hinde, R.A. The effects of separating rhesus monkey infants from their mothers for six days. J. Child Psychol. Psychiatry 1967, 7, 179–197. [Google Scholar] [CrossRef]
- Lilienfeld, S.O.; Waldman, I.D.; Israel, A.C. A critical examination of the use of the term and concept of comorbidity in psychopathology research. Clin. Psychol. Sci. Pract. 1994, 1, 71–83. [Google Scholar] [CrossRef]
- Martin, P.; Bateson, P. Measuring Behaviour—An Introductory Guide, 3rd ed.; Cambridge University Press: Cambridge, UK, 2007; pp. 116–117. [Google Scholar]
- Hopper, L.M.; Freeman, H.D.; Ross, S.R. Reconsidering coprophagy as an indicator of negative welfare for captive chimpanzees. Appl. Anim. Behav. Sci. 2016, 176, 112–119. [Google Scholar] [CrossRef]
- Ferreira, R.G.; Mendl, M.; Wagner, P.G.C.; Araujo, T.; Nunes, D.; Mafra, A.L. Coping strategies in captive capuchin monkeys (Sapajus spp.). Appl. Anim. Behav. Sci. 2016, 176, 120–127. [Google Scholar] [CrossRef]
- Rommeck, I.; Gottlieb, D.H.; Strand, S.C.; McCowan, B. The effects of four nursery rearing strategies on infant behavioral development in rhesus macaques (Macaca mulatta). J. Am. Assoc. Lab. Anim. Sci. 2009, 48, 395–401. [Google Scholar]
- Poirier, C.; Bateson, M. Pacing stereotypies in laboratory rhesus macaques: Implications for animal welfare and the validity of neuroscientific findings. Neurosci. Biobehav. Rev. 2017. [Google Scholar] [CrossRef]
- Pomerantz, O.; Meiri, S.; Terkel, J. Socio-ecological factors correlate with levels of stereotypic behavior in zoo-housed primates. Behav. Processes 2013, 98, 85–91. [Google Scholar] [CrossRef]
- Kroshko, J.; Clubb, R.; Harper, L.; Mellor, E.; Moehrenschlager, A.; Mason, G. Stereotypic route tracing in captive Carnivora is predicted by species-typical home range sizes and hunting styles. Anim. Behav. 2016, 117, 197–209. [Google Scholar] [CrossRef]
- Lewis, M.; Gluck, J.; Beauchamp, A.; Keresztury, M.; Mailman, R. Long-term effects of early social isolation in Macaca mulatta: Changes in dopamine receptor function following apomorphine challenge. Brain Res. 1990, 513, 67–73. [Google Scholar] [CrossRef]
- Fontenot, M.B.; Padgett, E.E.; Dupuy, A.M.; Lynch, C.R.; De Petrillo, P.B.; Higley, J.D. The effects of fluoxetine and buspirone on self-injurious and stereotypic behavior in adult male rhesus macaques. Comp. Med. 2005, 55, 67–74. [Google Scholar]
- Bauman, M.D.; Toscano, J.E.; Babineau, B.A.; Mason, W.A.; Amaral, D.G. Emergence of stereotypies in juvenile monkeys (Macaca mulatta) with neonatal amygdala or hippocampus lesions. Behav. Neurosci. 2008, 122, 1005–1015. [Google Scholar] [CrossRef]
- Moadab, G.; Bliss-moreau, E.; Amaral, D.G. Adult social behavior with familiar partners following neonatal amygdala or hippocampus damage. Behav. Neurosci. 2015, 129, 339–350. [Google Scholar] [CrossRef]
- Novak, M.A.; Kinsey, J.H.; Jorgensen, M.J.; Hazen, T.J. Effects of puzzle feeders on pathological behavior in individually housed rhesus monkeys. Am. J. Primatol. 1998, 46, 213–227. [Google Scholar] [CrossRef]
- Bayne, K.; Dexter, S.; Mainzer, H.; McCully, C.; Campbell, G.; Yamada, F. The use of artificial turf as a foraging substrate for individually housed rhesus monkeys (Macaca Mulatta). Anim. Welf. 1992, 1, 39–53. [Google Scholar]
- Bourgeois, S.R.; Brent, L. Modifying the behaviour of singly caged baboons: Evaluating the effectiveness of four enrichment techniques. Anim. Welf. 2005, 14, 71–81. [Google Scholar]
- Polanco, A.; Díez-Leon, M.; Mason, G. Stereotypic behaviours are heterogeneous in their triggers and treatments in the American mink, Neovison vison, a model carnivore. Anim. Behav. 2018, 141, 105–114. [Google Scholar] [CrossRef]
- Díez-León, M.; Bursian, S.; Galicia, D.; Napolitano, A.; Palme, R.; Mason, G. Environmentally enriching American mink (Neovison vison) increases lymphoid organ weight and skeletal symmetry, and reveals differences between two sub-types of stereotypic behaviour. Appl. Anim. Behav. Sci. 2016, 177, 59–69. [Google Scholar] [CrossRef]
- Pomerantz, O.; Paukner, A.; Terkel, J. Some stereotypic behaviors in rhesus macaques (Macaca mulatta) are correlated with both perseveration and the ability to cope with acute stressors. Behav. Brain Res. 2012, 230, 274–280. [Google Scholar] [CrossRef]
- Lutz, C.K.; Williams, P.C.; Sharp, R.M. Abnormal behavior and associated risk factors in captive baboons (Papio hamadryas spp.). Am. J. Primatol. 2014, 76, 355–361. [Google Scholar] [CrossRef]
- Bashaw, M.J.; Tarou, L.R.; Maki, T.S.; Maple, T.L. A survey assessment of variables related to stereotypy in captive giraffe and okapi. Appl. Anim. Behav. Sci. 2001, 73, 235–247. [Google Scholar] [CrossRef]
- Normando, S.; Meers, L.; Samuels, W.E.; Faustini, M.; Ödberg, F.O. Variables affecting the prevalence of behavioural problems in horses. Can riding style and other management factors be significant? Appl. Anim. Behav. Sci. 2011, 133, 186–198. [Google Scholar] [CrossRef]
- Waters, A.J.; Nicol, C.J.; French, N.P. Factors influencing the development of stereotypic and redirected behaviours in young horses: Findings of a four year prospective epidemiological study. Equine Vet. J. 2002, 34, 572–579. [Google Scholar] [CrossRef]
- Greco, B.J.; Meehan, C.L.; Heinsius, J.L.; Mench, J.A. Why pace? The influence of social, housing, management, life history, and demographic characteristics on locomotor stereotypy in zoo elephants. Appl. Anim. Behav. Sci. 2017, 194, 104–111. [Google Scholar] [CrossRef]
- Nierenberg, A.A.; Miyahara, S.; Spencer, T.; Wisniewski, S.R.; Otto, M.W.; Simon, N.; Pollack, M.H.; Ostacher, M.J.; Yan, L.; Siegel, R.; et al. Clinical and diagnostic implications of lifetime attention-deficit/ hyperactivity disorder comorbidity in adults with bipolar disorder: Data from the first 1000 STEP-BD participants. Biol. Psychiatry 2005, 57, 1467–1473. [Google Scholar] [CrossRef]
- Krug, I.; Treasure, J.; Anderluh, M.; Bellodi, L.; Cellini, E.; di Bernardo, M.; Granero, R.; Karwautz, A.; Nacmias, B.; Penelo, E.; et al. Present and lifetime comorbidity of tobacco, alcohol and drug use in eating disorders: A European multicenter study. Drug Alcohol Depend. 2008, 97, 169–179. [Google Scholar] [CrossRef]
- Bellanca, R.U.; Crockett, C.M. Factors predicting increased incidence of abnormal behavior in male pigtailed macaques. Am. J. Primatol. 2002, 58, 57–69. [Google Scholar] [CrossRef]
- United States Department of Agriculture (USDA). Animal Welfare Act, Food Secuirity Act of 1985, Subtitle F-Animal Welfare; 1985. Available online: https://www.nal.usda.gov/awic/public-law-99-198-food-security-act-1985-subtitle-f-animal-welfare (accessed on 1 November 2019).
- Budaev, S.V. Using principal components and factor analysis in animal behaviour research: Caveats and guidelines. Ethology 2010, 116, 472–480. [Google Scholar] [CrossRef]
- Ruxton, G.D.; Neuhäuser, M. When should we use one-tailed hypothesis testing? Methods Ecol. Evol. 2010, 1, 114–117. [Google Scholar] [CrossRef]
- Cross, H.A.; Harlow, H.F. Prolonged and progressive effects of partial isolation on the behavior of macaque monkeys. J. Exp. Res. Personal. 1965, 1, 39–49. [Google Scholar]
- Duke, D.C.; Keeley, M.L.; Geffken, G.R.; Storch, E.A. Trichotillomania: A current review. Clin. Psychol. Rev. 2010, 30, 181–193. [Google Scholar] [CrossRef]
- McCowan, B. (University of California, Oakland, CA, USA). Personal communication, 2020.
- Gaither, A.M.; Baker, K.C.; Gilbert, M.H.; Blanchard, J.L.; Liu, D.X.; Luchins, K.R.; Bohm, R.P. Videotaped behavior as a predictor of clinical outcome in rhesus macaques (Macaca mulatta). Comp. Med. 2014, 64, 193–199. [Google Scholar]
- Canales, J.Z.; Cordás, T.A.; Fiquer, J.T.; Cavalcante, A.F.; Moreno, R.A. Posture and body image in individuals with major depressive disorder: A controlled study. Rev. Bras. Psiquiatr. 2010, 32, 375–380. [Google Scholar] [CrossRef]
- Fiquer, J.T.; Moreno, R.A.; Brunoni, A.R.; Barros, V.B.; Fernandes, F.; Gorenstein, C. What is the nonverbal communication of depression? Assessing expressive differences between depressive patients and healthy volunteers during clinical interviews. J. Affect. Disord. 2018, 238, 636–644. [Google Scholar] [CrossRef]
- Michalak, J.; Troje, N.; Fischer, J.; Vollmar, P.; Heidenreich, T.; Schulte, D. Embodiment of Sadness and Depression—Gait Patterns Associated with Dysphoric Mood. Psychosom. Med. 2009, 587, 580–587. [Google Scholar] [CrossRef]
- Mallapur, A.; Choudhury, B.C. Behavioral abnormalities in captive nonhuman primates. J. Appl. Anim. Welf. Sci. 2003, 6, 275–284. [Google Scholar] [CrossRef] [PubMed]
- Harlow, H.F.; Mc Kinney, W.T. Nonhuman primates and psychoses. J. Autism Child. Schizophr. 1971, 1, 368–375. [Google Scholar] [CrossRef] [PubMed]
- Novak, M.A.; Suomi, S.J. Abnormal behavior in nonhuman primates and models of development. In Primate Models of Children’s Health and Developmental Disabilities; Academic Press: Cambridge, MA, USA, 2006; pp. 141–160. [Google Scholar]
- Polanco, A. A Tinbergian review of self-injurious behaviors in laboratory rhesus macaques. Appl. Anim. Behav. Sci. 2016, 179, 1–10. [Google Scholar] [CrossRef]
- Harlow, H.F.; Suomi, S.J. Social recovery by isolation-reared monkeys. Proc. Natl. Acad. Sci. USA 1971, 68, 1534–1538. [Google Scholar] [CrossRef] [PubMed]
- Kaufman, M.E. The effects of institutionalization on development of stereotyped and social behaviors in mental defectives. Am. J. Ment. Defic. 1967, 71, 581–585. [Google Scholar] [PubMed]
- Mason, W.A.; Berkson, G. Effects of maternal mobility on the development of rocking and other behaviors in rhesus monkeys: A study with artificial mothers. Dev. Psychobiol. 1975, 8, 197–211. [Google Scholar] [CrossRef] [PubMed]
- Bonadonna, P. Effects of a vestibular stimulation program on stereotypic rocking behavior. Am. J. Occup. Ther. 1981, 35, 775–781. [Google Scholar] [CrossRef][Green Version]
- Thelen, E. Determinants of amounts of stereotyped behavior in normal human infants. Ethol. Sociobiol. 1980, 1, 141–150. [Google Scholar] [CrossRef]
- Shafer, R.L.; Newell, K.M.; Lewis, M.H.; Bodfish, J.W. A cohesive framework for motor stereotypy in typical and atypical development: The role of sensorimotor integration. Front. Integr. Neurosci. 2017, 11, 1–8. [Google Scholar] [CrossRef]
- Goldman, S.; Wang, C.; Salgado, M.W.; Greene, P.E.; Kim, M.; Rapin, I. Motor stereotypies in children with autism and other developmental disorders. Dev. Med. Child Neurol. 2009, 51, 30–38. [Google Scholar] [CrossRef]
- Bildsøe, M.; Heller, K.E.; Jeppesen, L.L. Effects of immobility stress and food restriction on stereotypies in low and high stereotyping female ranch mink. Behav. Processes 1991, 25, 179–189. [Google Scholar] [CrossRef]
- Polanco, A.; Campbell, D.L.M.; Díez-León, M.; Mason, G. Towards a taxonomy of stereotypic behaviours in the American mink (Neovison vison), a model Carnivore: Homogeneous or heterogeneous? Appl. Anim. Behav. Sci. 2017, 194, 95–103. [Google Scholar] [CrossRef]
- Favell, J.E.; McGimsey, J.F.; Schell, R.M. Treatment of self-injury by providing alternate sensory activities. Anal. Interv. Dev. Disablities 1982, 2, 83–104. [Google Scholar] [CrossRef]
- Powell, S.B.; Newman, H.A.; Pendergast, J.F.; Lewis, M.H. A rodent model of spontaneous stereotype. Initial characterization of developmental, environmental, and neurobiological factors. Physiol. Behav. 1999, 66, 355–363. [Google Scholar] [CrossRef]
- Tanimura, Y.; Yang, M.C.K.; Ottens, A.K.; Lewis, M.H. Development, and temporal organization of repetitive behavior in an animal model. Dev. Psychobiol. 2010, 52, 813–824. [Google Scholar] [CrossRef]
- Lutz, C.K. A cross-species comparison of abnormal behavior in three species of singly-housed old world monkeys. Appl. Anim. Behav. Sci. 2018, 199, 52–58. [Google Scholar] [CrossRef]
- Poirier, C.; Oliver, C.J.; Bueno, J.C.; Flecknell, P.; Bateson, M. Pacing behaviour in laboratory macaques is an unreliable indicator of acute stress. Sci. Rep. 2019, 9, 7476. [Google Scholar] [CrossRef] [PubMed]
- Sandson, J.; Albert, M.L. Varieties of Perseveration. Neuropsychologia 1984, 22, 715–732. [Google Scholar] [CrossRef]
- MacLellan, A.; Fureix, C.; Polanco, A.; Mason, G. Can animals develop depression? A multidisciplinary assessment of depression-like responses. Behaviour 2021. under revision. [Google Scholar]
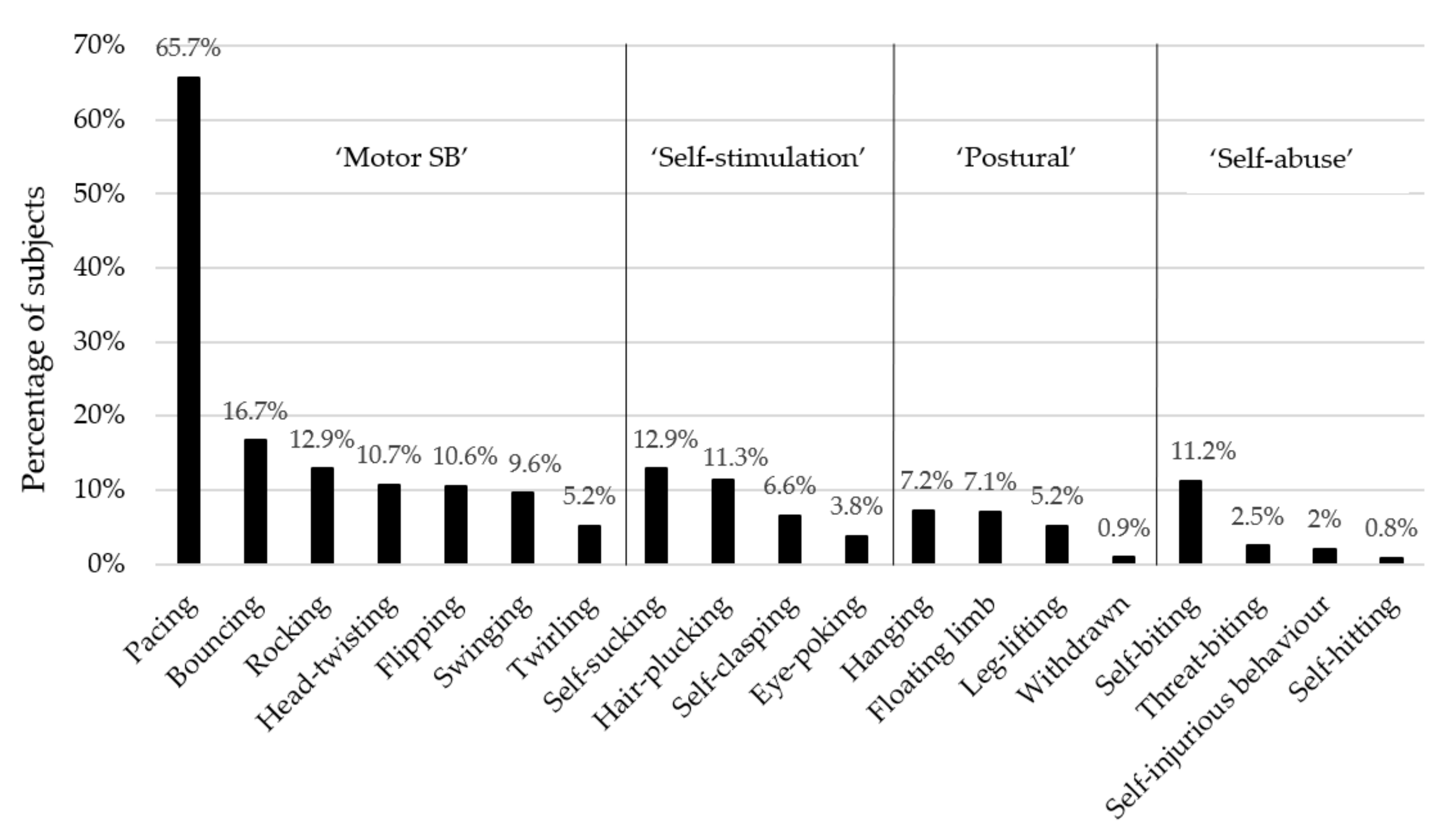
| Behavioural Subcategory | Individual Behaviour | Description | Comorbid with Other Behaviours? * | Does Prior Work Support or Contradict the Subcategory’s Construct Validity? |
|---|---|---|---|---|
| ‘Motor’ stereotypic behaviours [5,9,13] | Pace | Walking back and forth or in a circular pattern, for at least three repetitions or 5 or more seconds. | ‘Self-stimulation’ SBs [34]; cf. [13]; floating limb [13]; cf. [34]; self-injurious behaviours [13]; cf. [34], including self-biting [5]. | Individual ‘motor’ behaviours showed distinct risk factors [15], and rocking formed its own component in a PCA [34], thereby suggesting this subcategory does not have construct validity. |
| Flip | Turning a somersault, usually in a backwards fashion, for at least three repetitions or 5 or more seconds. | |||
| Twirl | Swinging in a circle or spinning, for at least three repetitions or 5 or more seconds. | |||
| Swing | Moving back and forth suspended from above, for at least three repetitions or 5 or more seconds. | |||
| Bounce | Moving jerkily, usually up and down, for at least three repetitions or 5 or more seconds. | |||
| Head-twist | Moving or lifting the head with a sudden motion, for at least two repetitions or 5 or more seconds. | |||
| Rock | Moving back and forth or from side to side, especially gently or rhythmically, for at least three repetitions or 5 or more seconds. | |||
| ‘Self-stimulation’ [13,15,34] | Self-clasp | Embracing or hugging oneself, for at least 15 s. | ‘Motor’ SBs [34]; cf. [13]. | Individual ‘self-stimulation’ behaviours showed distinct risk factors [15]; thereby suggesting this subcategory does not have construct validity. |
| Hair-pluck | Excessive pulling of one’s hair; often leads to over-groomed appearance. | |||
| Self-suck | Sucking various parts of one’s body including digits, tail, and male genitalia. | |||
| Eye-poke | ‘Saluting’ gesture of hand over eye; often involves a digit (frequently the thumb) being pressed into the eye-socket. | |||
| ‘Postural’ [13] | Hang | Hanging from the cage ceiling with 1–4 limbs, for at least 15 s. | No prior data. | No prior data comparing individual forms. |
| Floating limb | Arm or leg being lifted seemingly without the animal’s knowledge; sometimes used to self-groom as though the animal is being groomed by another; often results in self-biting when animal sees limb. | Self-biting [13,34]; ‘motor’ SBs [13]; cf. [34]. | ||
| Leg-lift | Arm or leg reaches around the back of the body or is propped on the back, for at least ten seconds. | No prior data. | ||
| Withdrawn | Facing a corner for an extended period of time; usually acting socially withdrawn, for at least 30 s. | No prior data. | ||
| ‘Self-Abuse’ [13,55] | Self-bite | Biting oneself; usually involves biting arms, legs, shoulders or genitals. | Floating limb [13,34]; ‘motor’ SBs [5,13]; cf. [34]. | Self-biting and self-injurious behaviour share the same risk factors [15], therefore supporting this subcategory’s validity (although other forms were not assessed). |
| Threat-bite | Biting hand, wrist, or forearm while staring at an observer, mirror, or conspecific in a threatening manner. | |||
| Self-hit | Striking oneself forcefully. | |||
| Self-injurious behaviour | Self-biting, scratching, or some other form of self-mutilation which results in injury. If the self-biting incident did not result in injury, then it was categorized as ‘self-biting’. |
| Independent Variable/Dependent Variable | Age | Male Sex (Referent = Female) | Moderate Scanning Level (Referent = Rare) | Frequent Scanning Level (Referent = Rare) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Odds Ratio | 95% CI | p-Value | Odds Ratio | 95% CI | p-Value | Odds Ratio | 95% CI | p-Value | Odds Ratio | 95% CI | p-Value | |
| Pacing | 0.97 | 0.95–0.99 | 0.004 | 0.80 | 0.63–1.02 | 0.076 | 3.00 | 2.25–4.00 | <0.001 | 9.17 | 6.39–13.14 | <0.001 |
| Bouncing | 0.95 | 0.92–0.97 | <0.001 | 1.81 | 1.32–2.47 | <0.001 | 1.72 | 1.16–2.55 | 0.007 | 1.92 | 1.30–2.84 | 0.001 |
| Rocking | 1.03 | 1–1.06 | 0.031 | 1.33 | 0.94–1.86 | 0.106 | 2.78 | 1.69–4.58 | <0.001 | 3.70 | 2.27–6.02 | <0.001 |
| Head-twisting | 0.93 | 0.89–0.96 | <0.001 | 1.20 | 0.83–1.73 | 0.338 | 2.74 | 1.48–5.09 | 0.001 | 6.59 | 3.71–11.71 | <0.001 |
| Flipping | 0.80 | 0.75–0.85 | <0.001 | 1.04 | 0.72–1.50 | 0.844 | 1.72 | 1.01–2.93 | 0.045 | 3.25 | 1.99–5.33 | <0.001 |
| Swinging | 0.84 | 0.80–0.89 | <0.001 | 1.03 | 0.70–1.52 | 0.890 | 1.54 | 0.89–2.68 | 0.124 | 3.02 | 1.82–5.01 | <0.001 |
| Twirling | 0.92 | 0.87–0.97 | 0.004 | 1.38 | 0.83–2.32 | 0.216 | 1.64 | 0.76–3.55 | 0.209 | 3.62 | 1.81–7.26 | <0.001 |
| Self-sucking | 0.64 | 0.59–0.70 | <0.001 | 1.83 | 1.29–2.58 | 0.001 | 0.97 | 0.63–1.51 | 0.904 | 1.53 | 1.02–2.31 | 0.04 |
| Hair-plucking | 1.17 | 1.13–1.20 | <0.001 | 0.96 | 0.69–1.38 | 0.825 | 1.21 | 0.79–1.87 | 0.382 | 1.00 | 0.63–1.57 | 0.985 |
| Self-clasping | 0.99 | 0.95–1.03 | 0.77 | 0.86 | 0.54–1.36 | 0.513 | 1.10 | 0.59–2.03 | 0.765 | 1.91 | 1.09–3.35 | 0.023 |
| Eye-poking | 0.96 | 0.91–1.02 | 0.198 | 1.29 | 0.71–2.35 | 0.399 | 2.13 | 0.90–5.04 | 0.085 | 2.99 | 1.31–6.83 | 0.009 |
| Hanging | 0.93 | 0.88–0.97 | 0.002 | 1.35 | 0.87–2.10 | 0.184 | 1.13 | 0.65–1.97 | 0.659 | 1.33 | 0.78–2.29 | 0.297 |
| Floating limb | 1.02 | 0.98–1.05 | 0.393 | 1.46 | 0.94–2.28 | 0.096 | 2.14 | 0.95–4.82 | 0.067 | 8.00 | 3.90–16.41 | <0.001 |
| Leg-lifting | 0.95 | 0.90–1.00 | 0.036 | 1.21 | 0.72–2.01 | 0.472 | 2.28 | 0.92–5.66 | 0.075 | 6.89 | 3.05–15.58 | <0.001 |
| Withdrawn | 1.15 | 1.06–1.25 | 0.001 | 1.35 | 0.41–4.44 | 0.624 | 1.04 | 0.21–5.20 | 0.959 | 1.84 | 0.44–7.74 | 0.407 |
| Self-biting | 0.98 | 0.95–1.01 | 0.173 | 1.81 | 1.25–2.61 | 0.002 | 2.61 | 1.35–5.07 | 0.005 | 9.52 | 5.22–17.37 | <0.001 |
| Threat-biting | 0.99 | 0.93–1.06 | 0.770 | 2.29 | 1.06–4.93 | 0.035 | 1.31 | 0.35–4.91 | 0.691 | 6.03 | 2.05–17.74 | 0.001 |
| Self-injurious behaviour | 0.99 | 0.92–1.06 | 0.793 | 1.58 | 0.70–3.59 | 0.272 | 0.69 | 0.19–2.47 | 0.571 | 2.61 | 1.00–6.88 | 0.051 |
| Self-hitting | 0.88 | 0.75–1.04 | 0.131 | 3.96 | 0.82–19.13 | 0.087 | 3.15 | 0.33–30.37 | 0.322 | 5.54 | 0.64–47.63 | 0.119 |
| Independent Variables | Unadjusted Odds Ratio and p-Value | Adjusted Odds Ratio * | 95% CI * | p-Value * | Positively and Significantly Predicts Pacing? * (Result the Same if Age, Sex and Scanning Level not Controlled for?) |
|---|---|---|---|---|---|
| Flipping presence vs. absence (rarely scanned group) | 0.33 (0.985) | 0.31 | 0.11–0.87 | 0.987 | No (Unadjusted relationship is also negative) |
| Flipping presence vs. absence (moderately scanned group) | 1.28 (0.261) | 1.07 | 0.49–2.34 | 0.436 | No (Unadjusted relationship is also positive and non-significant) |
| Flipping presence vs. absence (frequently scanned group) | 1.47 (0.201) | 1.29 | 0.52–3.20 | 0.289 | No (Unadjusted relationship is also positive and non-significant) |
| Twirling presence vs. absence | 3.23 (<0.001) | 1.66 | 0.48–5.70 | 0.210 | No (Unadjusted relationship is positive and significant) |
| Swinging presence vs. absence | 1.84 (0.005) | 0.77 | 0.32–1.83 | 0.723 | No (Unadjusted relationship is positive and significant) |
| Bouncing presence vs. absence | 0.69 (0.989) | 0.27 | 0.13–0.58 | 0.999 | No (Unadjusted relationship is also negative) |
| Head-twisting presence vs. absence | 4.77 (<0.001) | 2.89 | 0.96–8.73 | 0.029 | Yes (Unadjusted relationship is also positive and significant) |
| Rocking presence vs. absence (rarely scanned group) | 0.11 (0.999) | 0.12 | 0.03–0.49 | 0.998 | No (Unadjusted relationship is also negative) |
| Rocking presence vs. absence (moderately scanned group) | 0.41 (0.999) | 0.45 | 0.25–0.79 | 0.997 | No (Unadjusted relationship is also negative) |
| Rocking presence vs. absence (frequently scanned group) | 0.64 (0.883) | 0.66 | 0.32–1.36 | 0.871 | No (Unadjusted relationship is also negative) |
| Independent Variables | Unadjusted Odds Ratio and p-Value | Adjusted Odds Ratio * | 95% CI * | p-Value * | Positively and Significantly Predicts Self-Sucking? * (Result the Same if Age, Sex and Scanning Level not Controlled for?) |
|---|---|---|---|---|---|
| Self-clasping presence vs. absence | 2.52 (<0.001) | 2.55 | 0.45–14.33 | 0.144 | No (Unadjusted relationship is positive and significant) |
| Eye-poking presence vs. absence | 1.48 (0.166) | 2.24 | 0.08–64.81 | 0.319 | No (Unadjusted relationship is also positive and non-significant) |
| Hair-plucking presence vs. absence 2 | 0.09 (0.999) | 0.59 | 0.12–2.80 | 0747 | No (Unadjusted relationship is also negative) |
| Independent Variables | Unadjusted Odds Ratio and p-Value | Adjusted Odds Ratio * | 95% CI * | p-Value * | Positively and Significantly Predicts Hanging? * (Result the Same if Age, Sex and Scanning Level not Controlled for?) |
|---|---|---|---|---|---|
| Floating limb presence vs. absence 2 | 0.81 (0.675) | 0.74 | 0.29–1.93 | 0.723 | No (Unadjusted relationship is also negative) |
| Leg-lifting presence vs. absence 2 | 0.64 (0.764) | 0.53 | 0.16–1.75 | 0.851 | No (Unadjusted relationship is also negative) |
| Withdrawn presence vs. absence 3 | 0.84 (0.561) | 0.84 | 0–5.19 | 0.561 | No (Unadjusted relationship is also negative) |
| Independent Variables | Unadjusted Odds Ratio and p-Value | Adjusted Odds Ratio * | 95% CI * | p-Value * | Positively and Significantly Predicts Self-Biting? * (Result the Same if Age, Sex and Scanning Level not Controlled for?) |
|---|---|---|---|---|---|
| Threat-biting presence vs. absence (rarely scanned group) | 11.00 (0.022) | 9.02 | 0.86–94.56 | 0.034 | Yes (Unadjusted relationship is also positive and significant) |
| Threat-biting presence vs. absence (moderately scanned group) | 3.00 (0.167) | 2.76 | 0.29–25.84 | 0.188 | No (Unadjusted relationship is also positive and non-significant) |
| Threat-biting presence vs. absence (frequently scanned group) | 1.00 (0.495) | 0.89 | 0.31–2.53 | 0.587 | No (Unadjusted relationship is also non-significant) |
| Self-hitting presence vs. absence 2 | 6.48 (0.003) | 4.04 | 0.96–16.98 | 0.029 | Yes (Unadjusted relationship is also positive and significant) |
| Self-injurious behaviour presence vs. absence 2 | 7.19 (<0.001) | 5.43 | 2.18–13.49 | <0.001 | Yes (Unadjusted relationship is also positive and significant) |
| Behaviour/Subcategory | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1. Self-bite/self-hit/ self-injurious behaviour | ---- | 6.36– 6.37 * | 4.32–4.50 1* | 0.62– 0.66 1 | 0.42– 0.46 | 0.77– 0.85 | 0.23– 0.49 3a,3c | 0.94– 0.95 1 | 1.40– 1.41 1 | 1.47– 1.59 1 | 0.61– 0.63 1 | 6.11– 7.11 4* | 0.44 2 |
| 2. Floating limb/leg-lift | ---- | ---- | 3.16–3.23 | 0.77– 0.78 1 | 0.25– 0.26 | 0.58– 0.59 1 | 0.09 3a | 1.25– 1.29 1 | 4.26–4.72 | 0.51– 1.21 | 0.59– 0.611 | 8.31– 9.52 3a* | 2.00 2 |
| 3. Self-clasp | ---- | ---- | ---- | 1.04– 1.08 1 | 0.19 | 0.47– 0.48 1 | 0.11– 0.12 3a | 1.02– 1.24 1 | 2.66 | 1.58– 2.55 | 0.33– 0.40 | 0.39– 0.40 1 | 1.39– 1.411 |
| 4. Twirl | ---- | ---- | ---- | ---- | 3.43– 3.74 3b,3c* | 2.25 1* | 4.50– 4.80 1* | 0.97– 1.29 | 0.62– 0.711 | 0.44– 0.52 | 0.15– 0.18 1 | 0.41– 0.43 1 | 1.17 2 |
| 5. Bounce/rock/swing | ---- | ---- | ---- | ---- | ---- | 2.07– 2.10* | 0.29– 0.58 3a,3b | 0.47– 0.54 | 0.39 | 0.11– 0.15 3a | 0.08– 0.09 | 0.33– 0.35 1 | 2.06– 2.20 2 |
| 6. Hang | ---- | ---- | ---- | ---- | ---- | ---- | 0.27– 0.30 3a | 1.08– 1.19 | 0.25– 0.28 1 | 0.47– 0.62 1 | 0.10– 0.12 1 | 0.38– 0.42 1 | 0.84 2 |
| 7. Pace/head-twist | ---- | ---- | ---- | ---- | ---- | ---- | ---- | 0.30– 0.37 3a | 0.75– 0.80 1 | 0.11– 0.12 3a | 0.02 | 0.46– 0.47 1 | 0.19– 0.26 1 |
| 8. Flip | ---- | ---- | ---- | ---- | ---- | ---- | ---- | ---- | 2.26– 2.53 1* | 0.21– 0.24 3a | 1.11– 1.13 1 | 0.76– 0.77 | 0.55 2 |
| 9. Eye-poke | ---- | ---- | ---- | ---- | ---- | ---- | ---- | ---- | ---- | 1.19– 2.24 | 0.19 1 | 2.01– 2.07 1 | 1.64 2 |
| 10. Self-suck | ---- | ---- | ---- | ---- | ---- | ---- | ---- | ---- | ---- | ---- | 0.42– 0.59 1 | 0.72– 0.85 1 | 0.44 2 |
| 11. Hair-pluck | ---- | ---- | ---- | ---- | ---- | ---- | ---- | ---- | ---- | ---- | ---- | 0.85– 0.90 1 | 1.12– 1.22 1 |
| 12. Threat-bite | ---- | ---- | ---- | ---- | ---- | ---- | ---- | ---- | ---- | ---- | ---- | ---- | 2.50 2 |
| 13. Withdrawn | ---- | ---- | ---- | ---- | ---- | ---- | ---- | ---- | ---- | ---- | ---- | ---- | ---- |
| Step 1—Creating Initial Subcategories (Based on Table 3, Table 4, Table 5 and Table 6 and Table A1, Table A2 and Table A3) | Step 2—Expanding Subcategories (Based on Table 7) |
|---|---|
| Self-biting, self-hitting, and self-injurious behaviours; | 1. ‘Self-biting, self-hitting, self-injurious behaviour, floating limb, leg-lift, self-clasping’ subcategory |
| Floating limb and leg-lift; | |
| Self-clasping; | |
| Bouncing, rocking, and swinging; | 2. ‘Bouncing, rocking, swinging, twirling and hanging’ subcategory |
| Twirling; | |
| Hanging; | |
| Pacing and head-twisting; | 3. ‘Pacing and head-twisting’ subcategory |
| Flipping; | 4. ‘Flipping and eye-poking’ subcategory |
| Eye-poking; | |
| Self-sucking; | 5. ‘Self-sucking’ |
| Hair-plucking; | 6. ‘Hair-plucking’ |
| Threat-biting; | 7. ‘Threat-biting’ |
| Withdrawn; | 8. ‘Withdrawn’ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Polanco, A.; McCowan, B.; Niel, L.; Pearl, D.L.; Mason, G. Recommendations for Abnormal Behaviour Ethograms in Monkey Research. Animals 2021, 11, 1461. https://doi.org/10.3390/ani11051461
Polanco A, McCowan B, Niel L, Pearl DL, Mason G. Recommendations for Abnormal Behaviour Ethograms in Monkey Research. Animals. 2021; 11(5):1461. https://doi.org/10.3390/ani11051461
Chicago/Turabian StylePolanco, Andrea, Brenda McCowan, Lee Niel, David L. Pearl, and Georgia Mason. 2021. "Recommendations for Abnormal Behaviour Ethograms in Monkey Research" Animals 11, no. 5: 1461. https://doi.org/10.3390/ani11051461
APA StylePolanco, A., McCowan, B., Niel, L., Pearl, D. L., & Mason, G. (2021). Recommendations for Abnormal Behaviour Ethograms in Monkey Research. Animals, 11(5), 1461. https://doi.org/10.3390/ani11051461





