Natural Endotoxemia in Dogs—A Hidden Condition That Can Be Treated with a Potential Probiotic Containing Bacillus subtilis, Bacillus licheniformis and Pediococcus acidilactici: A Study Model
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Clinical Investigations
2.3. Determination of Endotoxemia
2.4. Statistical Analysis
3. Results
3.1. Clinical Examination
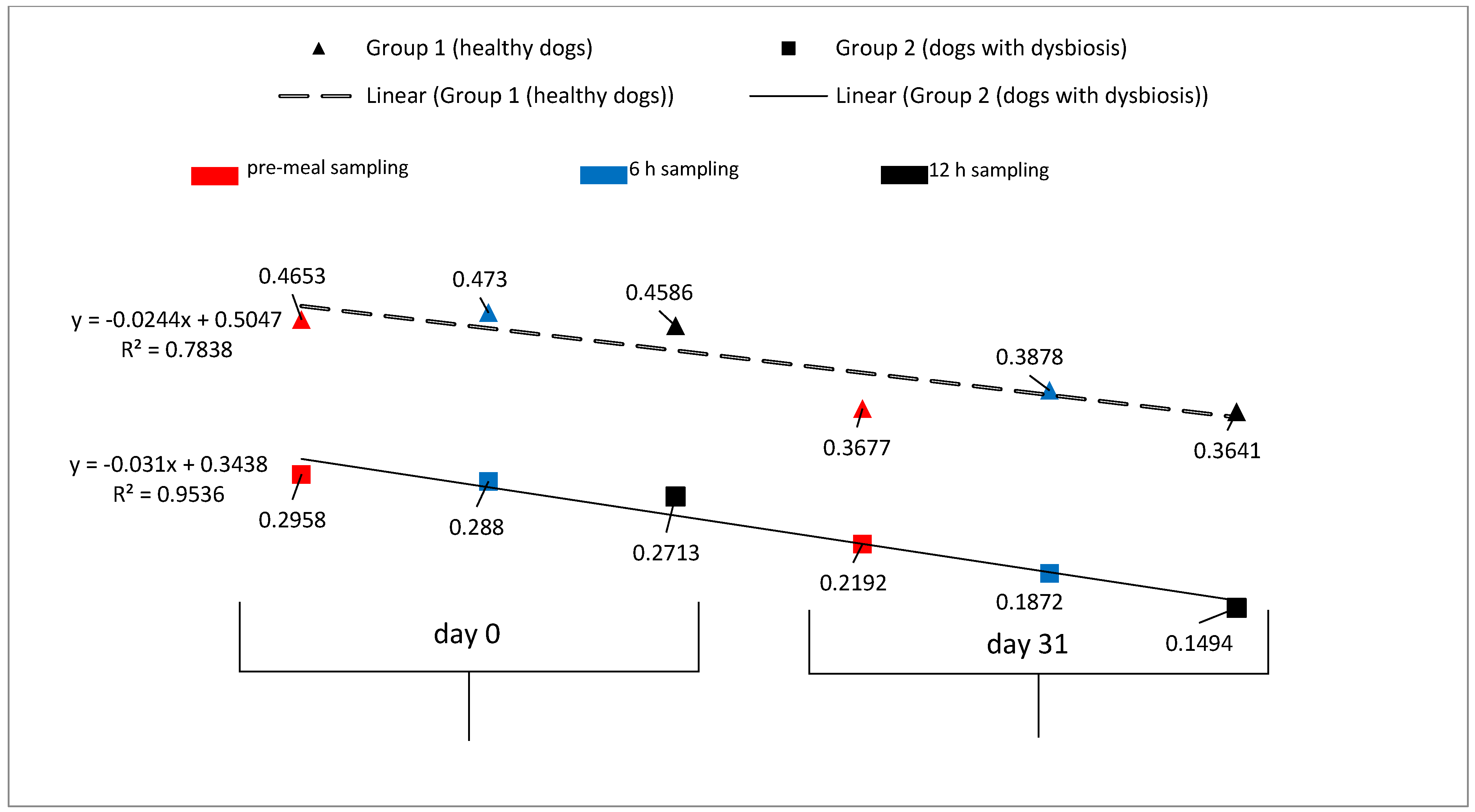
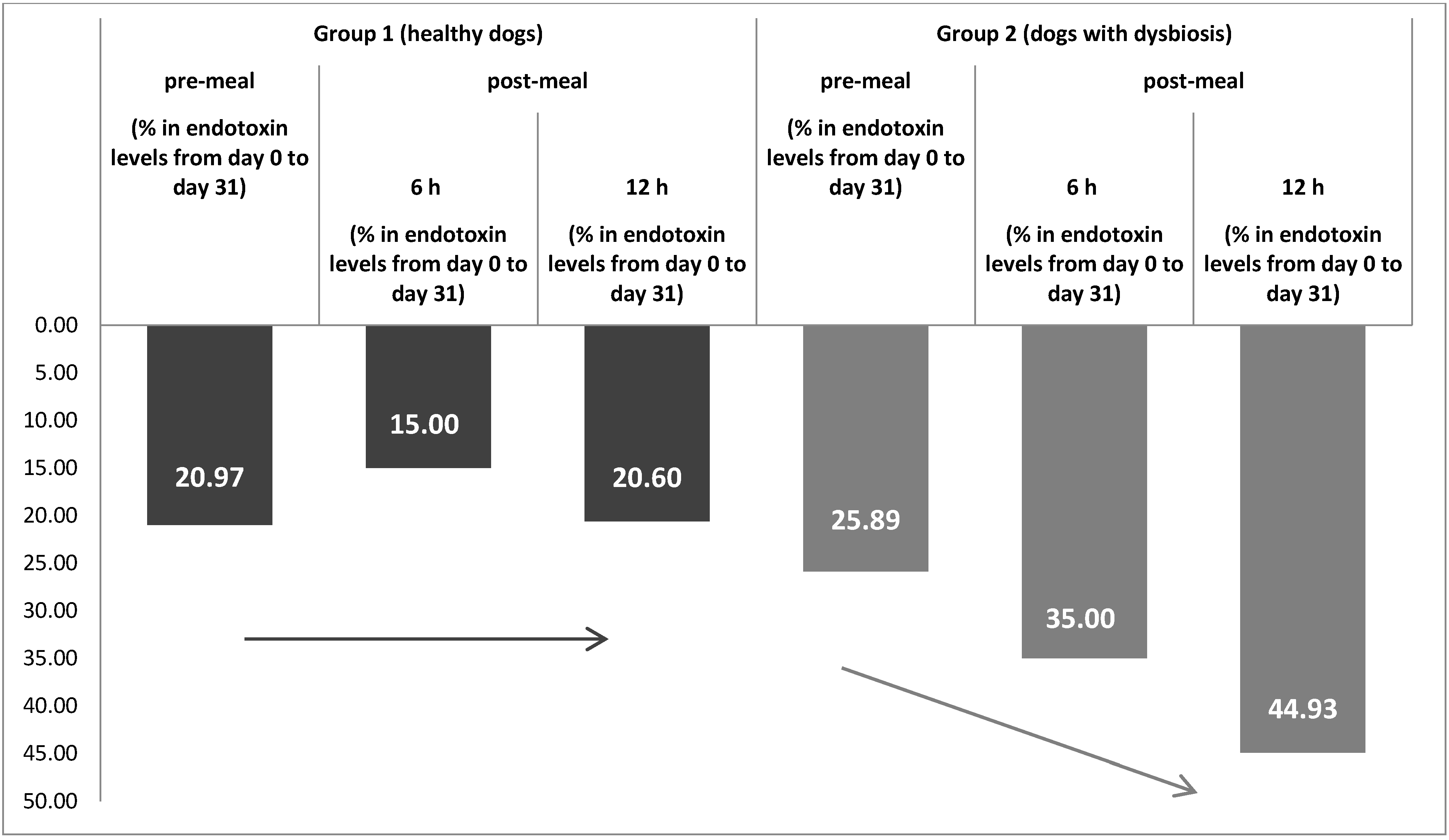
3.2. Endotoxemia
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Gallo, A.; Passaro, G.; Gasbarrini, A.; Landolfi, R.; Montalto, M. Modulation of microbiota as treatment for intestinal inflammatory disorders: An uptodate. World J. Gastroenterol. 2016, 22, 7186–7202. [Google Scholar] [CrossRef] [PubMed]
- Barko, P.C.; Michael, M.A.; Swanson, K.S.; Williams, D.A. The gastrointestinal microbiome: A review. J. Vet. Intern. Med. 2018, 32, 9–25. [Google Scholar] [CrossRef] [PubMed]
- Ceapa, C.; Wopereis, H.; Rezaiki, L.; Kleerebezem, M.; Knol, J.; Oozeer, R. Influence of fermented milk products, prebiotics and probiotics on microbiota composition and health. Best Pract. Res. Clin. Gastroenterol. 2013, 27, 139–155. [Google Scholar] [CrossRef] [PubMed]
- Biourge, V.; Vallet, C.; Levesque, A.; Sergheraert, R.; Chevalier, S.; Roberton, J.L. The use of probiotics in the diet of dogs. J. Nutr. 1998, 128 (Suppl. 12), 2730S–2732S. [Google Scholar] [CrossRef] [PubMed]
- Schmitz, S.; Suchodolski, J. Understanding the canine intestinal microbiota and its modification by pro-, pre- and synbiotics- what is the evidence? Vet. Med. Sci. 2016, 2, 71–94. [Google Scholar] [CrossRef] [PubMed]
- Logan, N.A. Bacillus species of medical and veterinary importance. J. Med. Microbiol. 1988, 25, 157–165. [Google Scholar] [CrossRef] [PubMed]
- Hill, C.; Guarner, F.; Reid, G.; Gibson, G.R.; Merenstein, D.J.; Pot, B.; Morelli, L.; Canani, R.B.; Flint, H.J.; Salminen, S.; et al. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 506–514. [Google Scholar] [CrossRef] [PubMed]
- Joint FAO/WHO Working Group Report on Drafting Guidelines for the Evaluation of Probiotics in Food London, Ontario, Canada. 30 April and 1 May 2002. Available online: https://www.who.int/foodsafety/fs_management/en/probiotic_guidelines.pdf (accessed on 4 May 2021).
- Taheri, H.R.; Moravej, H.; Malakzadegan, A.; Tabandeh, F.; Zaghari, M.; Shivazad, M.; Adibmoradi, M. Efficacy of Pediococcus acidlactici-based probiotic on intestinal coliforms and villus height, serum cholesterol level and performance of broiler chickens. Afr. J. Biotechnol. 2010, 9, 7564–7567. [Google Scholar]
- Herstad, H.K.; Nesheim, B.B.; L’Abée-Lund, T.; Larsen, S.; Skancke, E. Effects of a probiotic intervention in acute canine gastroenteritis—A controlled clinical trial. J. Small Anim. Pract. 2010, 51, 34–38. [Google Scholar] [CrossRef] [PubMed]
- Mcfarlin, K.B.; Henning, A.L.; Bownam, E.M.; Gary, M.A.; Carbajal, K.M. Oral spore-based probiotic supplementantion was associated with reduced incidence of post-prandial dietary endotoxin, triglycerides, and disease risk biomarkers. World J. Gastrointest. Pathophysiol. 2017, 8, 117–126. [Google Scholar] [CrossRef] [PubMed]
- Kirshnan, K. Metabolic endotoxemia, A driving force behind chronic illness. In Aapi’s Nutrition Guide to Optimal Health Using Principles of Functional Medicine & Nutritional Genomics, 3rd ed.; Laidlaw, S.H., Ed.; American Association of Physicians of Indian Origin: Oakbrook, IL, USA, 2017; pp. 269–286. [Google Scholar]
- de Vries, F.; Leuschner, J.; Jilma, B.; Derhaschnig, U. Establishment of a Low Dose Canine Endotoxemia Model to Test Anti-Inflammatory Drugs: Effects of Prednisolone. Int. J. Immunopathol. Pharmacol. 2013, 26, 861–869. [Google Scholar] [CrossRef] [PubMed]
- Deventer, S.V.; Buller, H.R.; Cate, J.W.T.; Aarden, L.A.; Hack, C.E.; Sturk, A. Experimental endotoxemia in humans: Analysis of cytokine release and coagulation, fibrinolytic, and complement pathways. Blood 1990, 76, 2520–2526. [Google Scholar] [CrossRef] [PubMed]
- Zailinawati, A.H.; Schattner, P.; Mazza, D. Doing a Pilot Study: Why Is It Essential? Malays. Fam. Physician 2006, 1, 70–73. [Google Scholar]
- Stewart, P.W. Small or Pilot Study, GCRC Protocols Which Propose “Pilot Studies”; Cincinnati Children’s Hospital Medical Center: Cincinnati, OH, USA, 1999. [Google Scholar]
- Lancaster, G.A.; Dodd, S.; Williamson, P.R. Design and analysis of pilot studies: Recommendations for good practice. J. Eval. Clin. Pract. 2004, 10, 307–312. [Google Scholar] [CrossRef] [PubMed]
- Kraemer, H.C.; Mintz, J.; Noda, A.; Tinklenberg, J.; Yesavage, J.A. Caution regarding the use of pilot studies to guide power calculations for study proposals. Arch. Gen. Psychiatry 2006, 63, 484–489. [Google Scholar] [CrossRef] [PubMed]
- Fenwick, N.; Griffin, G.; Gauthier, C. The welfare of animals used in science: How the “Three Rs” ethic guides improvements. Can. Vet. J. 2009, 50, 523–530. [Google Scholar] [PubMed]
- Three Rs Microsite. Canadian Council on Animal Care Ottawa: Canadian Council on Animal Care Launched 2008. Available online: https://ccac.ca/en/three-rs-and-ethics/ (accessed on 18 March 2021).
- Bartko, J.; Derhaschnig, U.; Neels, T.; Nabozny, G.H.; Harcken, C.; Leuschner, J.; de Vries, F.; Jilma, B. Selective glucocorticoid receptor modulation inhibits cytokine responses in a canine model of mild endotoxemia. Pharmacol. Res. 2017, 125, 215–223. [Google Scholar] [CrossRef] [PubMed]
- The Canine Digestion Process, Whole Dog Journal. 2019. Available online: https://www.whole-dog-journal.com/health/digestion/the-canine-digestion-process/ (accessed on 1 July 2019).

| Group 1 | Group 2 | ||||
|---|---|---|---|---|---|
| Patient Code 1 | Age | Sex | Patient Code 2 | Age | Sex |
| 1.1 | 48 months | Male | 2.1 | 7 months | Male |
| 1.2 | 20 months | Female | 2.2 | 69 months | Female |
| 1.3 | 50 months | Female | 2.3 | 4 months | Male |
| 1.4 | 24 months | Female | 2.4 | 24 months | Male |
| 1.5 | 17 months | Female | 2.5 | 53 months | Male |
| 2.6 | 36 months | Male | |||
| DAY/EXPERIMENTAL TIME | ACTIVITIES | |
|---|---|---|
| Day 0 | Blood collection 1: pre-meal | Clinical examination, collection of biological samples |
| Blood collection 2: 6 h post-meal | ||
| Blood collection 3: 12 h post-meal | ||
| DAY 1–DAY 30 → PROBIOTIC ADMINISTRATION | ||
| Day 31 | Blood collection 1: pre-meal | Clinical examination, collection of biological samples |
| Blood collection 2: 6 h post-meal | ||
| Blood collection 3: 12 h post-meal | ||
| Endotoxemia Evaluation Moment | Group 1 1 (n = 5) | Group 2 2 (n = 6) | |||
|---|---|---|---|---|---|
| Pre-A 3 (Day 0) | Post-A 4 (Day 31) | Pre-A (Day 0) | Post-A (Day 31) | ||
| Pre-meal 5 mean ± SD | 0.4653 ± 0.113 | 0.3677 ± 0.226 | 0.2958 ± 0.168 | 0.2192 ± 0.241 | |
| Post-meal 6 mean ± SD | 6 h | 0.4730 ± 0.172 | 0.3878 ± 0.232 | 0.2880 ± 0.200 | 0.1872 ± 0.145 |
| 12 h | 0.4586 ± 0.149 | 0.3641 ± 0.242 | 0.2713 ± 0.170 | 0.1494 ± 0.070 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Matei, M.-C.; Andrei, S.M.; Buza, V.; Cernea, M.S.; Dumitras, D.A.; Neagu, D.; Rafa, H.; Popovici, C.P.; Szakacs, A.R.; Catinean, A.; et al. Natural Endotoxemia in Dogs—A Hidden Condition That Can Be Treated with a Potential Probiotic Containing Bacillus subtilis, Bacillus licheniformis and Pediococcus acidilactici: A Study Model. Animals 2021, 11, 1367. https://doi.org/10.3390/ani11051367
Matei M-C, Andrei SM, Buza V, Cernea MS, Dumitras DA, Neagu D, Rafa H, Popovici CP, Szakacs AR, Catinean A, et al. Natural Endotoxemia in Dogs—A Hidden Condition That Can Be Treated with a Potential Probiotic Containing Bacillus subtilis, Bacillus licheniformis and Pediococcus acidilactici: A Study Model. Animals. 2021; 11(5):1367. https://doi.org/10.3390/ani11051367
Chicago/Turabian StyleMatei, Maria-Catalina, Sanda Maria Andrei, Victoria Buza, Mihai Sorin Cernea, Daria Antonia Dumitras, Daniela Neagu, Horatiu Rafa, Cristian Paul Popovici, Andrei Radu Szakacs, Adrian Catinean, and et al. 2021. "Natural Endotoxemia in Dogs—A Hidden Condition That Can Be Treated with a Potential Probiotic Containing Bacillus subtilis, Bacillus licheniformis and Pediococcus acidilactici: A Study Model" Animals 11, no. 5: 1367. https://doi.org/10.3390/ani11051367
APA StyleMatei, M.-C., Andrei, S. M., Buza, V., Cernea, M. S., Dumitras, D. A., Neagu, D., Rafa, H., Popovici, C. P., Szakacs, A. R., Catinean, A., Stefanut, E., & Stefanut, L. C. (2021). Natural Endotoxemia in Dogs—A Hidden Condition That Can Be Treated with a Potential Probiotic Containing Bacillus subtilis, Bacillus licheniformis and Pediococcus acidilactici: A Study Model. Animals, 11(5), 1367. https://doi.org/10.3390/ani11051367








