Tandem Mass Tag (TMT) Proteomic Analysis of Saliva in Horses with Acute Abdominal Disease
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
- Diseased horses (n = 7; 4 stallions and 3 mares; mean age = 8 years (range 1–18); Andalusians (n = 5) and crossbred (n = 2)). The presence of AAD was diagnosed based on history, physical examination (abdominal auscultation, rectal examination, and nasogastric intubation), and additional diagnostic tests, including complete blood count (CBC), serum biochemistry profile, abdominal ultrasound, and/or abdominocentesis. Final diagnoses were impaction of ascending colon (n = 3), right dorsal colon displacement (n = 2), and spasmodic colon (n = 2).
- Healthy horses (n = 6, 3 geldings, 2 stallions and 1 mare; mean age = 12 years (range 4–15); Andalusians (n = 4) and crossbred (n = 2)). Horses were found healthy based on history, clinical examination, CBC, and serum biochemistry profile.
- Diseased horses (n = 8; 7 geldings and 2 stallion; mean age = 10 years (range 4–16); Andalusian (n = 2), Warmblood (n = 2), Holsteiner (n = 1) and crossbred (n = 3)). Presence of AAD was diagnosed as described above. Final diagnoses were spasmodic colon (n = 3), right dorsal colon displacement (n = 3), and ascending colon impaction (n = 2).
- Healthy horses (n = 8; 4 geldings and 4 stallions; mean age = 7 years (range 3–11); Andalusians (n = 7) and crossbred (n = 1)).
2.2. Sampling of Saliva Specimens
2.3. Liquid Chromatography—Tandem Mass Spectrometry (LC-MS/MS)
2.4. Bioinformatics
2.5. Lactoferrin Analysis
2.6. Statistical Analysis
3. Results
3.1. Proteomic Changes in the Saliva of Horses with Acute Abdominal Disease
3.2. Bioinformatics
3.3. Changes in Salivary Lactoferrin Concentration in Horses with Acute Abdominal Disease Measured with the ELISA Assay
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Muñoz-Prieto, A.; Pons-Fuster, E.; López-Jornet, P. Salivary Markers in Inflammatory and Autoimmune Diseases BT-Saliva in Health and Disease: The Present and Future of a Unique Sample for Diagnosis; Tvarijonaviciute, A., Martínez-Subiela, S., López-Jornet, P., Lamy, E., Eds.; Springer International Publishing: Cham, Switzerland, 2020; pp. 177–192. [Google Scholar] [CrossRef]
- Nunes, L.A.S.; Brenzikofer, R.; Macedo, D.V. Reference intervals for saliva analytes collected by a standardized method in a physically active population. Clin. Biochem. 2011, 44, 1440–1444. [Google Scholar] [CrossRef]
- Sauer, F.J.; Gerber, V.; Frei, S.; Bruckmaier, R.M.; Groessl, M. Salivary cortisol measurement in horses: Immunoassay or LC-MS/MS? Domest. Anim. Endocrinol. 2020, 72, 106445. [Google Scholar] [CrossRef]
- Contreras-Aguilar, M.D.; Lamy, E.; Escribano, D.; Cerón, J.J.; Tecles, F.; Quiles, A.J.; Hevia, M.L. Changes in Salivary Analytes of Horses Due to Circadian Rhythm and Season: A Pilot Study. Animals 2020, 10, 1486. [Google Scholar] [CrossRef]
- Proudman, C.J. A two year, prospective survey of equine colic in general practice. Equine Vet. J. 1992, 24, 90–93. [Google Scholar] [CrossRef] [PubMed]
- Tinker, M.K.; White, N.A.; Lessard, P.; Thatcher, C.D.; Pelzer, K.D.; Davis, B.; Carmel, D.K. Prospective study of equine colic incidence and mortality. Equine Vet. J. 1997, 29, 448–453. [Google Scholar] [CrossRef]
- Curtis, L.; Burford, J.H.; England, G.C.W.; Freeman, S.L. Risk factors for acute abdominal pain (colic) in the adult horse: A scoping review of risk factors, and a systematic review of the effect of management-related changes. PLoS ONE 2019, 14, e0219307. [Google Scholar] [CrossRef] [PubMed]
- Cohen, N.D.; Matejka, P.L.; Honnas, C.M.; Hooper, R.N. Case-control study of the association between various management factors and development of colic in horses. Texas Equine Colic Study Group. J. Am. Vet. Med. Assoc. 1995, 206, 667–673. [Google Scholar] [PubMed]
- Morris, D.D.; Moore, J.N.; Ward, S. Comparison of age, sex, breed, history and management in 229 horses with colic. Equine Vet. J. 1989, 21, 129–132. [Google Scholar] [CrossRef]
- Reeves, M.J.; Salman, M.D.; Smith, G. Risk factors for equine acute abdominal disease (colic): Results from a multi-center case-control study. Prev. Vet. Med. 1996, 26, 285–301. [Google Scholar] [CrossRef]
- McClure, J.T.; Kobluk, C.; Voller, K.; Geor, R.J.; Ames, T.R.; Sivula, N. Fecalith impaction in four miniature foals. J. Am. Vet. Med. Assoc. 1992, 200, 205–207. [Google Scholar]
- Vatistas, N.J.; Snyder, J.R.; Wilson, W.D.; Drake, C.; Hildebrand, S. Surgical treatment for colic in the foal (67 cases): 1980–1992. Equine Vet. J. 1996, 28, 139–145. [Google Scholar] [CrossRef]
- Contreras-Aguilar, M.D.; Escribano, D.; Martínez-Subiela, S.; Martín-Cuervo, M.; Lamy, E.; Tecles, F.; Cerón, J.J. Changes in saliva analytes in equine acute abdominal disease: A sialochemistry approach. BMC Vet. Res. 2019, 15, 187. [Google Scholar] [CrossRef]
- Contreras-Aguilar, M.D.; Escribano, D.; Martín-Cuervo, M.; Tecles, F.; Cerón, J.J. Salivary alpha-amylase activity and cortisol in horses with acute abdominal disease: A pilot study. BMC Vet. Res. 2018, 14, 1–7. [Google Scholar] [CrossRef]
- Contreras-Aguilar, M.D.; Martínez-Subiela, S.; Cerón, J.J.; Martín-Cuervo, M.; Tecles, F.; Escribano, D. Salivary alpha-amylase activity and concentration in horses with acute abdominal disease: Association with outcome. Equine Vet. J. 2019, 51, 569–574. [Google Scholar] [CrossRef]
- Bilić, P.; Kuleš, J.; Galan, A.; De Pontes, L.G.; Guillemin, N.; Horvatić, A.; Sabes, A.F.; Mrljak, V.; Eckersall, P.D. Proteomics in Veterinary Medicine and Animal Science: Neglected Scientific Opportunities with Immediate Impact. Proteomics 2018, 18, e1800047. [Google Scholar] [CrossRef]
- Bhawal, R.; Oberg, A.L.; Zhang, S.; Kohli, M. Challenges and Opportunities in Clinical Applications of Blood-Based Proteomics in Cancer. Cancers 2020, 12, 2428. [Google Scholar] [CrossRef]
- Baeumlisberger, D.; Arrey, T.N.; Rietschel, B.; Rohmer, M.; Papasotiriou, D.G.; Mueller, B.; Beckhaus, T.; Karas, M. Labeling elastase digests with TMT: Informational gain by identification of poorly detectable peptides with MALDI-TOF/TOF mass spectrometry. Proteomics 2010, 10, 3905–3909. [Google Scholar] [CrossRef]
- Baldini, C.; Giusti, L.; Ciregia, F.; Da Valle, Y.; Giacomelli, C.; Donadio, E.; Ferro, F.; Galimberti, S.; Donati, V.; Bazzichi, L.; et al. Correspondence between salivary proteomic pattern and clinical course in primary Sjögren syndrome and non-Hodgkin’s lymphoma: A case report. J. Transl. Med. 2011, 9, 188. [Google Scholar] [CrossRef] [PubMed]
- Jacobsen, S.; Top Adler, D.M.; Bundgaard, L.; Sørensen, M.A.; Andersen, P.H.; Bendixen, E. The use of liquid chromatography tandem mass spectrometry to detect proteins in saliva from horses with and without systemic inflammation. Vet. J. 2014, 202, 483–488. [Google Scholar] [CrossRef]
- Martinez-Subiela, S.; Horvatic, A.; Escribano, D.; Pardo-Marin, L.; Kocaturk, M.; Mrljak, V.; Burchmore, R.; Ceron, J.J.; Yilmaz, Z. Identification of novel biomarkers for treatment monitoring in canine leishmaniosis by high-resolution quantitative proteomic analysis. Vet. Immunol. Immunopathol. 2017, 191, 60–67. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Galán, A.; Horvatić, A.; Kuleš, J.; Bilić, P.; Gotić, J.; Mrljak, V. LC-MS/MS analysis of the dog serum phosphoproteome reveals novel and conserved phosphorylation sites: Phosphoprotein patterns in babesiosis caused by Babesia canis, a case study. PLoS ONE 2018, 13, e0207245. [Google Scholar] [CrossRef]
- Dowle, A.A.; Wilson, J.; Thomas, J.R. Comparing the Diagnostic Classification Accuracy of iTRAQ, Peak-Area, Spectral-Counting, and emPAI Methods for Relative Quantification in Expression Proteomics. J. Proteome Res. 2016, 15, 3550–3562. [Google Scholar] [CrossRef]
- Cerón, J.J. Acute phase proteins, saliva and education in laboratory science: An update and some reflections. BMC Vet. Res. 2019, 15, 197. [Google Scholar] [CrossRef]
- Evennett, N.; Cerigioni, E.; Hall, N.J.; Pierro, A.; Eaton, S. Smooth muscle actin as a novel serologic marker of severe intestinal damage in rat intestinal ischemia-reperfusion and human necrotising enterocolitis. J. Surg. Res. 2014, 191, 323–330. [Google Scholar] [CrossRef]
- Lechuga, S.; Baranwal, S.; Li, C.; Naydenov, N.G.; Kuemmerle, J.F.; Dugina, V.; Chaponnier, C.; Ivanov, A.I. Loss of γ-cytoplasmic actin triggers myofibroblast transition of human epithelial cells. Mol. Biol. Cell 2014, 25, 3133–3146. [Google Scholar] [CrossRef] [PubMed]
- Vance, S.J.; McDonald, R.E.; Cooper, A.; Kennedy, M.W.; Smith, B.O. Resonance assignments for latherin, a natural surfactant protein from horse sweat. Biomol. NMR Assign. 2014, 8, 213–216. [Google Scholar] [CrossRef][Green Version]
- Vance, S.J.; McDonald, R.E.; Cooper, A.; Smith, B.O.; Kennedy, M.W. The structure of latherin, a surfactant allergen protein from horse sweat and saliva. J. R. Soc. Interface 2013, 10, 20130453. [Google Scholar] [CrossRef]
- Zimecki, M.; Actor, J.K.; Kruzel, M.L. The potential for Lactoferrin to reduce SARS-CoV-2 induced cytokine storm. Int. Immunopharmacol. 2021, 95, 107571. [Google Scholar] [CrossRef]
- Berlutti, F.; Pantanella, F.; Natalizi, T.; Frioni, A.; Paesano, R.; Polimeni, A.; Valenti, P. Antiviral properties of lactoferrin—A natural immunity molecule. Molecules 2011, 16, 6992–7018. [Google Scholar] [CrossRef]
- Nibbering, P.H.; Ravensbergen, E.; Welling, M.M.; Van Berkel, L.A.; Van Berkel, P.H.C.; Pauwels, E.K.J.; Nuijens, J.H. Human lactoferrin and peptides derived from its N terminus are highly effective against infections with antibiotic-resistant bacteria. Infect. Immun. 2001, 69, 1469–1476. [Google Scholar] [CrossRef]
- Valenti, P.; Antonini, G. Lactoferrin. Cell. Mol. Life Sci. 2005, 62, 2576–2587. [Google Scholar] [CrossRef]
- Vorland, L.H. Lactoferrin: A multifunctional glycoprotein. APMIS Acta Pathol. Microbiol. Immunol. Scand. 1999, 107, 971–981. [Google Scholar] [CrossRef]
- Legrand, D. Overview of Lactoferrin as a Natural Immune Modulator. J. Pediatrics 2016, 173, S10–S15. [Google Scholar] [CrossRef]
- de la Rosa, G.; Yang, D.; Tewary, P.; Varadhachary, A.; Oppenheim, J.J. Lactoferrin acts as an alarmin to promote the recruitment and activation of APCs and antigen-specific immune responses. J. Immunol. 2008, 180, 6868–6876. [Google Scholar] [CrossRef]
- Fedorka, C.; Scoggin, K.; Boakari, Y.; Hoppe, N.; Squires, E.; Ball, B.; Troedsson, M. The anti-inflammatory effect of exogenous lactoferrin on breeding-induced endometritis when administered post-breeding in susceptible mares. Theriogenology 2018, 114, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Drago-Serrano, M.; Campos-Rodríguez, R.; Carrero, J.; de la Garza, M. Lactoferrin: Balancing Ups and Downs of Inflammation Due to Microbial Infections. Int. J. Mol. Sci. 2017, 18, 501. [Google Scholar] [CrossRef] [PubMed]
- Escribano, D.; Horvatić, A.; Contreras-Aguilar, M.D.; Guillemin, N.; Cerón, J.J.; Lopez-Arjona, M.; Hevia, M.L.; Eckersall, P.D.; Manteca, X.; Mrljak, V. Identification of possible new salivary biomarkers of stress in sheep using a high-resolution quantitative proteomic technique. Res. Vet. Sci. 2019, 124, 338–345. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Li, Y.; Li, W.; Guo, X.; Zhou, S.; Zheng, H. Genetic polymorphisms in serine protease inhibitor Kazal-type 5 and risk of atopic dermatitis. Medicine 2020, 99, e21256. [Google Scholar] [CrossRef]
- Baldus, S.E.; Engelmann, K.; Hanisch, F.G. MUC1 and the MUCs: A family of human mucins with impact in cancer biology. Crit. Rev. Clin. Lab. Sci. 2004, 41, 189–231. [Google Scholar] [CrossRef]
- Mall, A.S. Analysis of mucins: Role in laboratory diagnosis. J. Clin. Pathol. 2008, 61, 1018–1024. [Google Scholar] [CrossRef]
- Brandtzaeg, P. Secretory immunity with special reference to the oral cavity. J. Oral Microbiol. 2013, 5, 1–24. [Google Scholar] [CrossRef]
- Culp, D.J.; Robinson, B.; Cash, M.N.; Bhattacharyya, I.; Stewart, C.; Cuadra-Saenz, G. Salivary Mucin 19 Glycoproteins. J. Biol. Chem. 2015, 290, 2993–3008. [Google Scholar] [CrossRef]
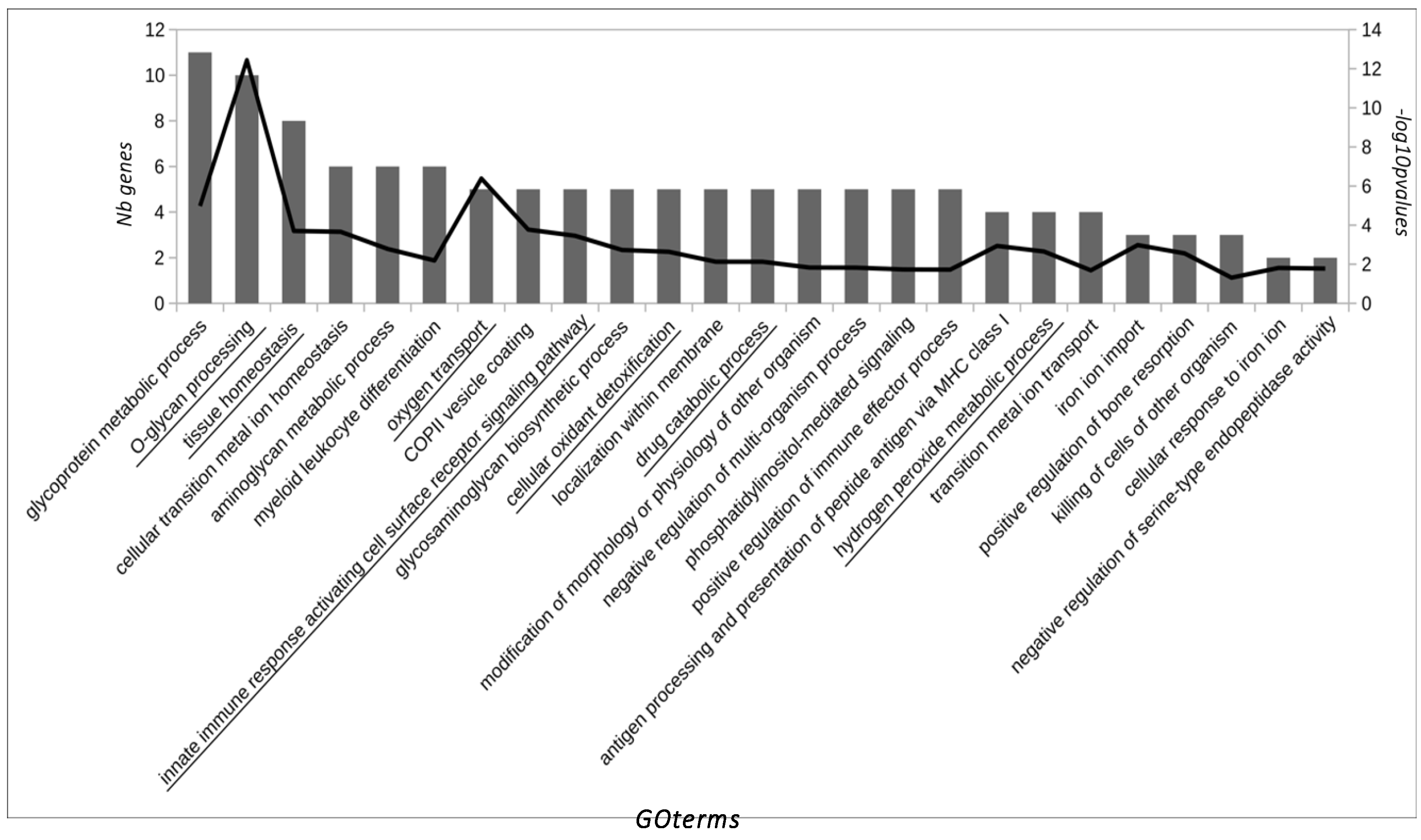
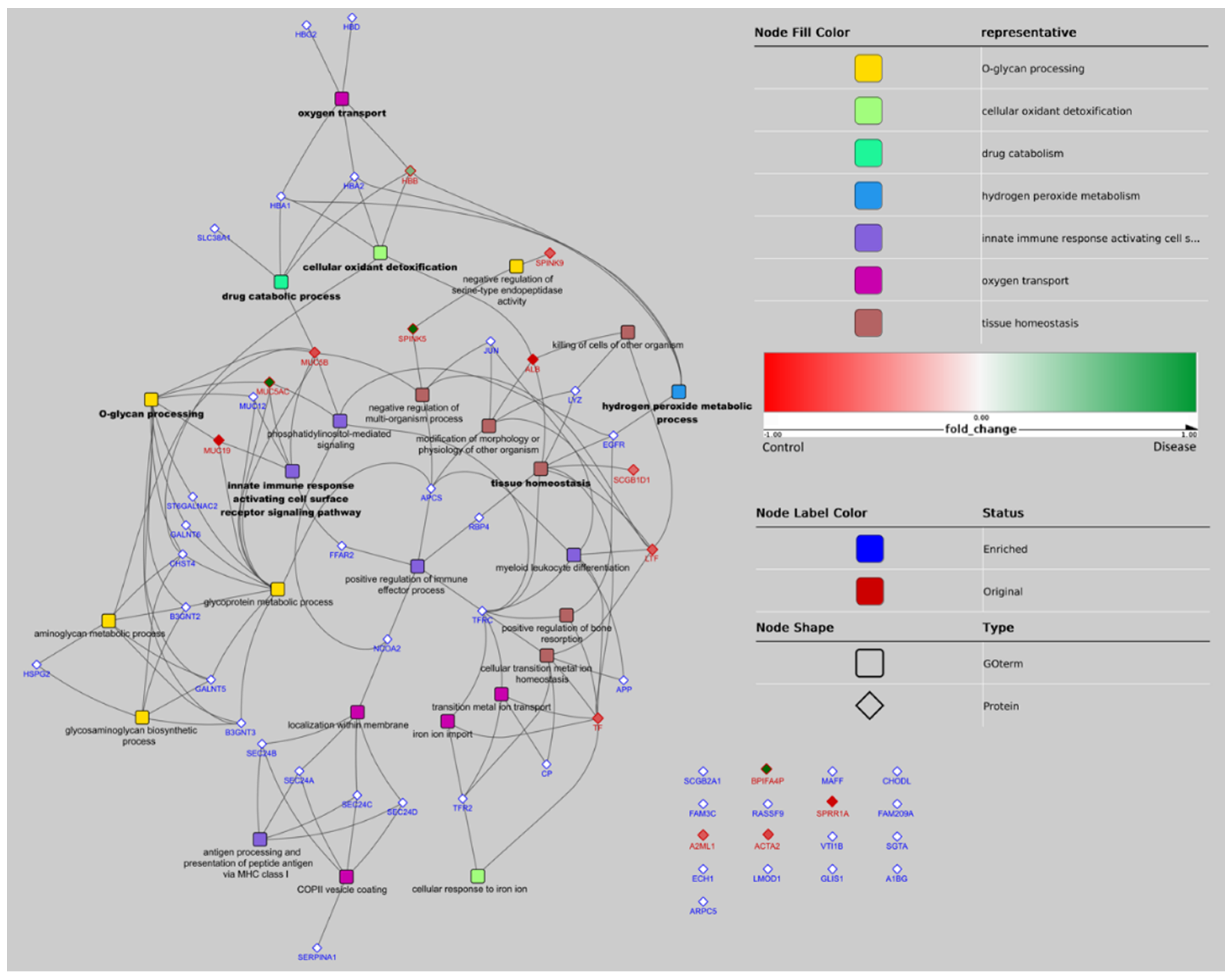

| Protein Accession ID | Protein Name | p-Value | Mean Control | Mean Disease | Fold Change | Regulation in AAD |
|---|---|---|---|---|---|---|
| 335773136 | Actin, gamma-enteric smooth muscle | 0.009 | 1.83 | 0.93 | 0.98 | Down |
| 953883212 | Latherin isoform X1 | 0.01 | 1.4 | 0.75 | 0.9 | Down |
| 255653068 | Lactotransferrin precursor | 0.01 | 1.25 | 0.97 | 0.37 | Down |
| 255683515 | Hemoglobin subunit beta | 0.01 | 1.17 | 1.09 | 0.1 | Down |
| 545181443 | Secretoglobin family 1D member | 0.015 | 2.31 | 1.02 | 1.18 | Down |
| 349602714 | Alpha-2-macroglobulin | 0.024 | 2.02 | 1.08 | 0.90 | Down |
| 338713575 | Serine protease inhibitor Kazal-type 9 | 0.024 | 1.67 | 1.08 | 0.63 | Down |
| 3581959 | Lactoferrin, partial | 0.024 | 1.25 | 0.97 | 0.37 | Down |
| 953869413 | Mucin-5AC | 0.030 | 1.26 | 1.1 | 0.20 | Down |
| 953851776 | Polymeric immunoglobulin receptor isoform X1 | 0.032 | 1.73 | 1.54 | 0.17 | Down |
| 953857134 | Mucin-19 | 0.009 | 1.15 | 1.76 | −0.61 | Up |
| 953872813 | Serine protease inhibitor Kazal-type 5 isoform X2 | 0.010 | 1.06 | 1.89 | −0.83 | Up |
| 14456405 | Albumin, partial | 0.015 | 0.94 | 1.51 | −0.68 | Up |
| 953869415 | Mucin-5B | 0.026 | 1.15 | 1.31 | −0.19 | Up |
| 953891633 | Odorant-binding protein | 0.030 | 1.07 | 1.35 | −0.34 | Up |
| 953853589 | Cornifin | 0.035 | 0.99 | 1.17 | −0.24 | Up |
| 3892523 | Transferrin, partial | 0.038 | 0.67 | 0.7 | −0.06 | Up |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Muñoz-Prieto, A.; Escribano, D.; Contreras-Aguilar, M.D.; Horvatić, A.; Guillemin, N.; Jacobsen, S.; Cerón, J.J.; Mrljak, V. Tandem Mass Tag (TMT) Proteomic Analysis of Saliva in Horses with Acute Abdominal Disease. Animals 2021, 11, 1304. https://doi.org/10.3390/ani11051304
Muñoz-Prieto A, Escribano D, Contreras-Aguilar MD, Horvatić A, Guillemin N, Jacobsen S, Cerón JJ, Mrljak V. Tandem Mass Tag (TMT) Proteomic Analysis of Saliva in Horses with Acute Abdominal Disease. Animals. 2021; 11(5):1304. https://doi.org/10.3390/ani11051304
Chicago/Turabian StyleMuñoz-Prieto, Alberto, Damián Escribano, María Dolores Contreras-Aguilar, Anita Horvatić, Nicolas Guillemin, Stine Jacobsen, José Joaquín Cerón, and Vladimir Mrljak. 2021. "Tandem Mass Tag (TMT) Proteomic Analysis of Saliva in Horses with Acute Abdominal Disease" Animals 11, no. 5: 1304. https://doi.org/10.3390/ani11051304
APA StyleMuñoz-Prieto, A., Escribano, D., Contreras-Aguilar, M. D., Horvatić, A., Guillemin, N., Jacobsen, S., Cerón, J. J., & Mrljak, V. (2021). Tandem Mass Tag (TMT) Proteomic Analysis of Saliva in Horses with Acute Abdominal Disease. Animals, 11(5), 1304. https://doi.org/10.3390/ani11051304










