PK/PD Analysis of Marbofloxacin by Monte Carlo Simulation against Mycoplasmaagalactiae in Plasma and Milk of Lactating Goats after IV, SC and SC-Long Acting Formulations Administration
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Experimental Design
2.3. Long-Acting Formulation Preparation
2.4. Analytical Method
2.5. MICs Determination
2.6. Pharmacokinetic Analysis
2.7. PK/PD Analysis
2.8. Statistical Analysis
3. Results
3.1. Pharmacokinetic Analysis
3.2. M. agalactiae MICs Determination
3.3. PK/PD Analysis and Monte Carlo Simulation
4. Discussion
4.1. Pharmacokinetic Analysis
4.2. MICs Determination and PK/PD Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Prats-van der Ham, M.; Tatay-Dualde, J.; Ambroset, C.; De la Fe, C.; Tardy, F. The moderate drift towards less tetracycline-susceptible isolates of contagious agalactia causative agents might result from different molecular mechanisms. Vet. Microbiol. 2018, 220, 39–46. [Google Scholar] [CrossRef]
- Corrales, J.C.; Esnal, A.; De la Fe, C.; Sanchez, A.; Assuncao, P.; Poveda, J.B.; Contreras, A. Contagious agalactia in small ruminants. Small Rumin. Res. 2007, 68, 154–166. [Google Scholar] [CrossRef]
- Ariza-Miguel, J.; Rodríguez-Lázaro, D.; Hernández, M. A survey of Mycoplasma agalactiae in dairy sheep farms in Spain. BMC Vet. Res. 2012, 8, 171. [Google Scholar] [CrossRef]
- Poumarat, F.; Gautier-Bouchardon, A.V.; Bergonier, D.; Gay, E.; Tardy, F. Diversity and variation in antimicrobial susceptibility patterns over time in Mycoplasma agalactiae isolates collected from sheep and goats in France. J. Appl. Microbiol. 2016, 120, 1208–1218. [Google Scholar] [CrossRef]
- Agnone, A.; La Manna, M.P.; Loria, G.R.; Puleio, R.; Villari, S.; Nicholas, R.A.; Guggino, G.; Sireci, G. Timing of activation of CD4+ memory cells as a possible marker to establish the efficacy of vaccines against contagious agalactia in sheep. Vet. Immunol. Immunopathol. 2013, 152, 252–259. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Martín, A.; Amores, J.; Paterna, A.; De la Fe, C. Contagious agalactia due to Mycoplasma spp. in small dairy ruminants: Epidemiology and prospects for diagnosis and control. Vet. J. 2013, 198, 48–56. [Google Scholar] [CrossRef]
- EMA; Committee for Medicinal Products for Veterinary use (CVMP). Categorisation of Antibiotics in the European Union. EMA/CVMP/CHMP/682198/2017. 2020. Available online: https://www.ema.europa.eu/en/news/categorisation-antibiotics-used-animals-promotes-responsible-use-protect-public-animal-health (accessed on 20 February 2021).
- Gautier-Bouchardon, A.V. Antimicrobial Resistance in Mycoplasma spp. In Antimicrobial Resistance in Bacteria from Livestock and Companion Animals; ASM PRESS: Washington, DC, USA, 2018; pp. 425–446. [Google Scholar] [CrossRef]
- Drugeon, H.; Thomas, V.; Gaillandeau, L.; Thomas, E. Antibacterial activity of marbofloxacin against bovine respiratory isolates. In: Proceedings of the 7th EAVPT International Congress, Madrid, Spain. J. Vet. Pharmacol. Ther. 1997, 20 (Suppl. S1), 138–139. [Google Scholar]
- Meunier, D.; Acar, J.F.; Martel, J.L.; Kroemer, S.; Valle, M. Seven years survey of susceptibility to marbofloxacin of bovine pathogenic strains from eight European countries. Int. J. Antimicrob. Agents. 2004, 24, 268–278. [Google Scholar] [CrossRef]
- Martínez, M.; McDemott, P.; Walker, R. Pharmacology of the fluorquinolones: A perpective for the use in domestic animals. Vet. J. 2006, 172, 10–28. [Google Scholar] [CrossRef]
- EMA. Committee for Veterinary Medicinal Products. Marbofloxacin (Extensión to All Food Producing Species). Summary Report. 2009. Available online: http://www.ema.europa.eu/docs/en_GB/document_library/Maximum_Residue_Limits_-_Report/2009/11/WC500014864.pdf (accessed on 29 May 2013).
- Waxman, S.; Rodríguez, C.; González, F.; De Vicente, M.L.; San Andrés, M.I.; San Andrés, M.D. Pharmacokinetic behavior of marbofloxacin after intravenous and intramuscular administrations in adult goats. J. Vet. Pharmacol. Ther. 2001, 24, 375–378. [Google Scholar] [CrossRef]
- Dova, S.W.; San Andrés, M.D.; González, F.; San Andrés, M.I.; De Lucas, J.J.; Rodríguez, C. Pharmacokinetic behavior and pharmacokinetic/pharmacodynamic integration of marbofloxacin after subcutaneous administration in goats. Vet. J. 2007, 174, 425–427. [Google Scholar] [CrossRef]
- Sidhu, P.K.; Landoni, M.F.; Aliabadi, F.S.; Lees, P. Pharmacokinetic and pharmacodynamic modelling of marbofloxacin administered alone and in combination with tolfenamic acid in goats. Vet. J. 2010, 184, 219–229. [Google Scholar] [CrossRef]
- Bhardwaj, P.; Sidhu, P.K.; Lonare, M.K.; Kaur, R.; Dumka, V.K.; Rampal, S. Pharmacokinetic-pharmacodynamic integration of marbofloxacin after single and repeated intravenous administration in goats. Res. Vet. Sci. 2018, 121, 111–115. [Google Scholar] [CrossRef] [PubMed]
- Bhardwaj, P.; Sidhu, P.K.; Saini, S.P.S.; Rampal, S. Pharmacokinetic-pharmacodynamic relationship of marbofloxacin for Escherichia coli and Pasturella multocida following repeated intramuscular administration in goats. J. Vet. Pharmacol. Ther. 2019, 42, 430–439. [Google Scholar] [CrossRef]
- Waxman, S.; San Andrés, M.D.; González, F.; De Lucas, J.J.; San Andrés, M.I.; Rodríguez, C. Influence of Escherichia coli endotoxin-induced fever on the pharmacokinetic behavior of marbofloxacin after intravenous administration in goats. J. Vet. Pharmacol. Ther. 2003, 26, 65–69. [Google Scholar] [CrossRef]
- Waxman, S.; San Andrés, M.D.; González, F.; San Andrés, M.I.; De Lucas, J.J.; Rodríguez, C. Age-related changes in the pharmacokinetics of marbofloxacin after intravenous administration in goats. J. Vet. Pharmacol. Ther. 2004, 27, 31–35. [Google Scholar] [CrossRef]
- Thomas, V.; Deleforge, J.; Boisrame, B. Pharmacokinectics of marbofloxacin in pre-ruminant and ruminant cattle. In Sixth EAVPT Congress Proceedings; Blackwell Scientific Publications: Edinburgh, Scotland, 1994; pp. 60–61. [Google Scholar]
- Thomas, V.; Deleforge, J.; Boisrame, B.; Espinasse, J. Pharmacokinetics of marbofloxacin in healthy and sick pre-ruminant calves. In Sixth EAVPT Congress Proceedings; Blackwell Scientific Publications: Edinburgh, Scotland, 1994; p. 61. [Google Scholar]
- Schneider, M.; Thomas, V.; Boisrame, B.; Deleforge, J. Pharmacokinetics of marbofloxacin in dogs after oral and parenteral administration. J. Vet. Pharmacol. Ther. 1996, 19, 56–61. [Google Scholar] [CrossRef]
- Aliabadi, F.S.; Lees, P. Pharmacokinetics and pharmacokinetic/pharmacodynamic integration of marbofloxacin in calf serum, exudate and transudate. J. Vet. Pharmacol. Ther. 2002, 25, 161–174. [Google Scholar] [CrossRef]
- Bousquet-Melou, A.; Bernard, S.; Schneider, M.; Toutain, P.L. Pharmacokinetics of marbofloxacin in horses. Equine Vet. J. 2002, 34, 366–372. [Google Scholar] [CrossRef] [PubMed]
- Sidhu, P.K.; Landoni, M.F.; Aliabadi, F.S.; Lees, P. PK-PD integration and modeling of marbofloxacin in sheep. Res. Vet. Sci. 2010, 88, 134–141. [Google Scholar] [CrossRef] [PubMed]
- Petracca, K.; Riond, J.L.; Graser, T.; Wanner, M. Pharmacokinetics of the gyrase inhibitor marbofloxacin: Influence of pregnancy and lactation in sows. Zentralbl. Veterinarmed. A 1993, 40, 73–79. [Google Scholar] [CrossRef]
- Shem-Tov, M.; Ziv, G.; Glickman, A.; Saran, A. Pharmacokinetics and penetration of marbofloxacin from blood into the milk of cows and ewes. Zentralbl. Veterinarmed. A 1997, 44, 511–519. [Google Scholar] [CrossRef]
- Schneider, M.; Vallé, M.; Woehrlé, F.; Boisramé, B. Pharmacokinetics of marbofloxacin in lactating cows after repeated intramuscular administrations and pharmacodynamics against mastitis isolated strains. J. Dairy Sci. 2004, 87, 202–211. [Google Scholar] [CrossRef]
- Lorenzutti, A.M.; Litterio, N.J.; Himelfarb, M.A.; Zarazaga, M.D.P.; San Andrés, M.I.; De Lucas, J.J. Pharmacokinetics, milk penetration and PK/PD analysis by Monte Carlo simulation of marbofloxacin, after intravenous and intramuscular administration to lactating goats. J. Vet. Pharmacol. Ther. 2017, 40, 629–640. [Google Scholar] [CrossRef]
- Zhang, L.; Parsons, D.L.; Navarre, C.; Kompella, U.B. Development and in-vitro evaluation of sustained release poloxamer 407 (P407) gel formulations of ceftiofur. J. Control Release 2002, 85, 73–81. [Google Scholar] [CrossRef]
- Cárceles, C.M.; Serrano, J.M.; Marín, P.; Escudero, E.; Fernández-Varón, E. Pharmacokinetics of moxifloxacin in rabbits after intravenous, subcutaneous and a long-acting poloxamer 407 gel formulation administration. J. Vet. Med. A Physiol. Pathol. Clin. Med. 2006, 53, 300–304. [Google Scholar] [CrossRef]
- Marín, P.; Escudero, E.; Fernández-Varón, E.; Ramírez, M.J.; Cárceles, C.M. Pharmacokinetics and milk penetration of difloxacin after a long-acting formulation for subcutaneous administration to lactating goats. J. Dairy Sci. 2010, 93, 3056–3064. [Google Scholar] [CrossRef] [PubMed]
- Ambrose, P.G. Monte Carlo simulation in the evaluation of susceptibility breakpoints: Predicting the future: Insights from the society of infectious diseases pharmacists. Pharmacotherapy 2006, 26, 129–134. [Google Scholar] [CrossRef] [PubMed]
- Craig, W.A. Pharmacokinetic/pharmacodynamic parameters: Rationale for antibacterial dosing of mice and men. Clin. Infect. Dis. 1998, 26, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Martinez, M.N.; Papich, M.G.; Drusano, G.L. Dosing regimen matters: The importance of early intervention and rapid attainment of the pharmacokinetic/pharmacodynamic target. Antimicrob. Agents Chemother. 2012, 56, 2795–2805. [Google Scholar] [CrossRef] [PubMed]
- Martinez, M.N.; Toutain, P.-L.; Turnidge, J. The pharmacodynamics of antimicrobial agents. In Antimicrobial Therapy in Veterinary Medicine; Wiley-Blackwell: Hoboken, NJ, USA, 2013; pp. 79–103. [Google Scholar]
- Walker, R.D. Fluoroquinolones. In Antimicrobial Therapy in Veterinary Medicine; Iowa State University Press: Ames, IA, USA, 2000; pp. 315–338. [Google Scholar]
- McKellar, Q.A.; Sanchez Bruni, S.F.; Jones, D.G. Pharmacokinetic/pharmacodynamic relationships of antimicrobial drugs used in veterinary medicine. J. Vet. Pharmacol. Ther. 2004, 27, 503–514. [Google Scholar] [CrossRef]
- Papich, M.G. Pharmacokinetic-pharmacodynamic (PK-PD) modeling and the rational selection of dosage regimes for the prudent use of antimicrobial drugs. Vet. Microbiol. 2014, 171, 480–486. [Google Scholar] [CrossRef] [PubMed]
- Toutain, P.L.; Sidhu, P.K.; Lees, P.; Rassouli, A.; Pelligand, L. VetCAST Method for Determination of the Pharmacokinetic-Pharmacodynamic Cut-Off Values of a Long-Acting Formulation of Florfenicol to Support Clinical Breakpoints for Florfenicol Antimicrobial Susceptibility Testing in Cattle. Front. Microbiol. 2019, 10, 1310. [Google Scholar] [CrossRef]
- Toutain, P.L.; Bousquet-Mélou, A.; Damborg, P.; Ferran, A.A.; Mevius, D.; Pelligand, L.; Veldman, K.T.; Lees, P. En Route towards European Clinical Breakpoints for Veterinary Antimicrobial Susceptibility Testing: A Position Paper Explaining the VetCAST Approach. Front. Microbiol. 2017, 15, 2344. [Google Scholar] [CrossRef]
- Clinical and Laboratory Standards Institute. Understanding Susceptibility Test Data as a Component of Antimicrobial Stewardship in Veterinary Settings; CLSI: Wayne, PA, USA, 2019. [Google Scholar]
- Tatay-Dualde, J.; Prats-van der Ham, M.; de la Fe, C.; Paterna, A.; Sánchez, A.; Corrales, J.C.; Contreras, A.; Gómez-Martín, A. Mutations in the quinolone resistance determining region conferring resistance to fluoroquinolones in Mycoplasma agalactiae. Vet. Microbiol. 2017, 207, 63–68. [Google Scholar] [CrossRef]
- Schmolka, I.R. Artificial skin. I. Preparation and properties of pluronic F-127 gels for treatment of burns. J. Biomed. Mater. Res. 1972, 6, 571–582. [Google Scholar] [CrossRef]
- Siefert, H.M.; Kohlsdorferc, C.; Steinkec, W.; Witt, A. Pharmacokinetics of the 8-methoxyquinolone, moxifloxacin: Tissue distribution in male rats. J. Antimicrob. Chemother. 1999, 43, 61–67. [Google Scholar] [CrossRef]
- Marenda, M.S.; Sagné, E.; Poumarat, F.; Citti, C. Suppression subtractive hybridization as a basis to assess Mycoplasma agalactiae and Mycoplasma bovis genomic diversity and species-specific sequences. Microbiol. Read. 2005, 151, 475–489. [Google Scholar] [CrossRef]
- Hannan, P.C. Guidelines and recommendations for antimicrobial minimum inhibitory concentration (MIC) testing against veterinary mycoplasma species. International Research Programme on Comparative Mycoplasmology. Vet. Res. 2000, 31, 373–395. [Google Scholar] [CrossRef]
- Albers, A.C.; Fletcher, R.D. Simple method for quantitation of viable mycoplasmas. Appl. Environ. Microbiol. 1982, 43, 958–960. [Google Scholar] [CrossRef]
- Asín-Prieto, E.; Rodríguez-Gascón, A.; Isla, A. Applications of the pharmacokinetic/pharmacodynamic (PK/PD) analysis of antimicrobial agents. J. Infect. Chemother. 2015, 21, 319–329. [Google Scholar] [CrossRef]
- Lees, P.; Pelligand, L.; Illambas, J.; Potter, T.; Lacroix, M.; Rycroft, A.; Toutain, P.L. Pharmacokinetic/pharmacodynamic integration and modelling of amoxicillin for the calf pathogens Mannheimia haemolytica and Pasteurella multocida. J. Vet. Pharmacol. Ther. 2015, 38, 457–470. [Google Scholar] [CrossRef]
- Toutain, P.L.; Bousquet-Mélou, A. Plasma terminal half-life. J. Vet. Pharmacol. Ther. 2004, 27, 427–439. [Google Scholar] [CrossRef]
- Toutain, P.L.; Bousquet-Mélou, A. Bioavailability and its assessment. J. Vet. Pharmacol. Ther. 2004, 27, 455–466. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Varón, E.; Villamayor, L.; Escudero, E.; Espuny, A.; Cárceles, C.M. Pharmacokinetics and milk penetration of moxifloxacin after intravenous and subcutaneous administration to lactating goats. Vet. J. 2006, 172, 302–307. [Google Scholar] [CrossRef]
- Brown, S.A. Fluoroquinolones in animal health. J. Vet Pharmacol. Ther. 1996, 19, 1–14. [Google Scholar] [CrossRef]
- Atkinson, H.C.; Begg, E.J. Prediction of drug distribution into human milk from physicochemical characteristics. Clin. Pharmacokinet. 1990, 18, 151–167. [Google Scholar] [CrossRef]
- McManaman, J.L.; Neville, M.C. Mammary physiology and milk secretion. Adv. Drug. Deliv. Rev. 2003, 55, 629–641. [Google Scholar] [CrossRef]
- Pulido, M.M.; Molina, A.J.; Merino, G.; Mendoza, G.; Prieto, J.G.; Alvarez, A.I. Interaction of enrofloxacin with breast cancer resistance protein (BCRP/ABCG2): Influence of flavonoids and role in milk secretion in sheep. J. Vet. Pharmacol. Ther. 2006, 29, 279–287. [Google Scholar] [CrossRef]
- Schrickx, J.A.; Fink-Gremmels, J. Implications of ABC transporters on the disposition of typical veterinary medicinal products. Eur. J. Pharmacol. 2008, 585, 510–519. [Google Scholar] [CrossRef]
- Wu, H.J.; Luo, J.; Wu, N.; Matand, K.; Zhang, L.J.; Han, X.F.; Yang, B.J. Cloning, sequence and functional analysis of goat ATP-binding cassette transporter G2 (ABCG2). Mol. Biotechnol. 2008, 39, 21–27. [Google Scholar] [CrossRef]
- AliAbadi, F.S.; Lees, P. Antibiotic treatment for animals: Effect on bacterial population and dosage regimen optimisation. Int. J. Antimicrob. Agents 2000, 14, 307–313. [Google Scholar] [CrossRef]
- Mitchell, J.D.; McKellar, Q.A.; McKeever, D.J. Pharmacodynamics of antimicrobials against Mycoplasma mycoides mycoides small colony, the causative agent of contagious bovine pleuropneumonia. PLoS ONE 2012, 7, 44158. [Google Scholar] [CrossRef]
- Zhang, N.; Gu, X.; Ye, X.; Wu, X.; Zhang, B.; Zhang, L.; Shen, X.; Jiang, H.; Ding, H. The PK/PD Interactions of Doxycycline against Mycoplasma gallisepticum. Front. Microbiol. 2016, 4, 653. [Google Scholar] [CrossRef] [PubMed]
- Xiao, X.; Lan, W.; Zhao, Y.; Li, R.; Liu, Y.; Liu, J.; Wang, Z. In vivo Pharmacokinetic and Pharmacodynamic (PK/PD) Modeling and Establishment of the PK/PD Cutoff of Florfenicol against Pasteurella multocida in Ducks. Front. Microbiol. 2021, 11, 616685. [Google Scholar] [CrossRef]
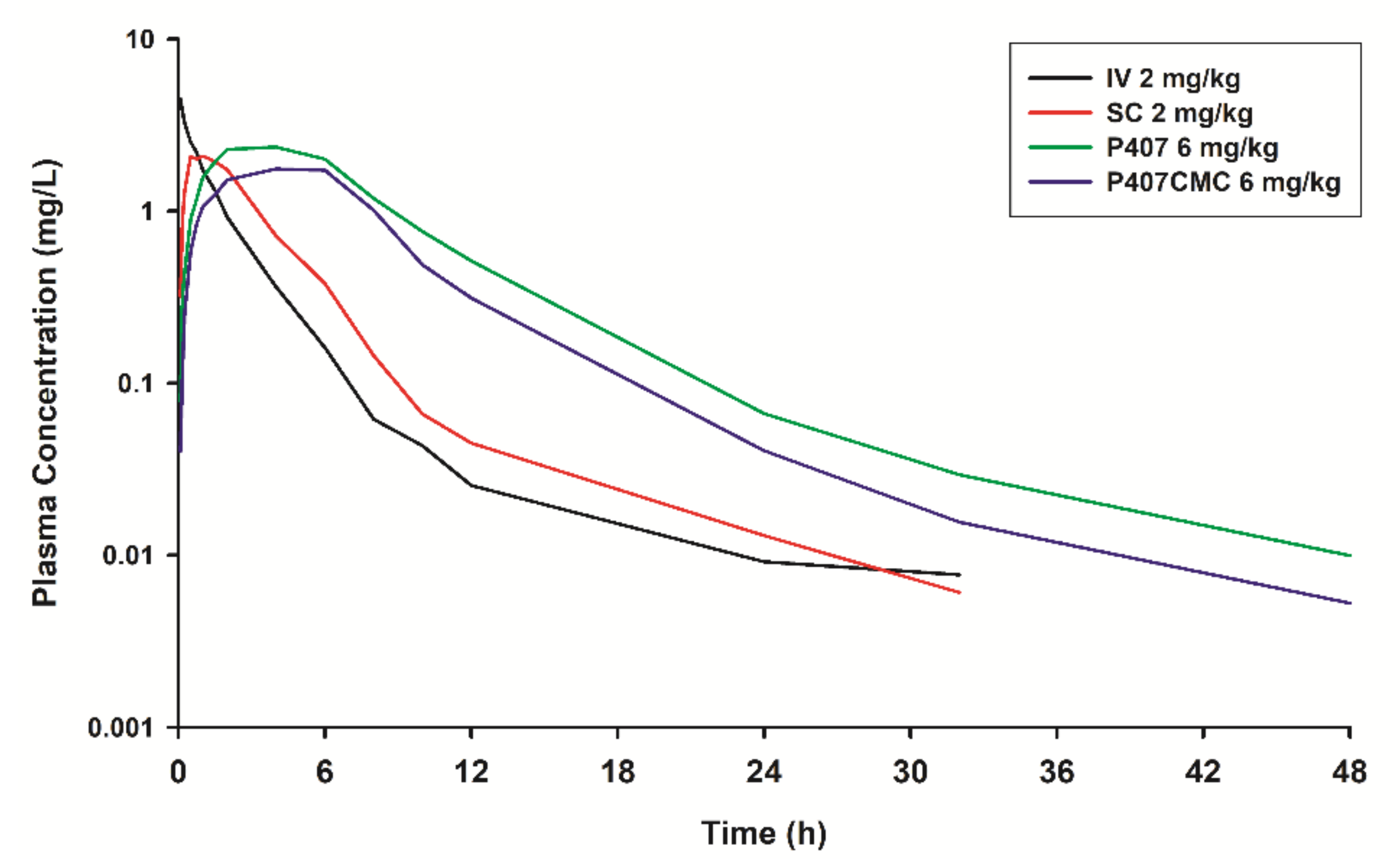

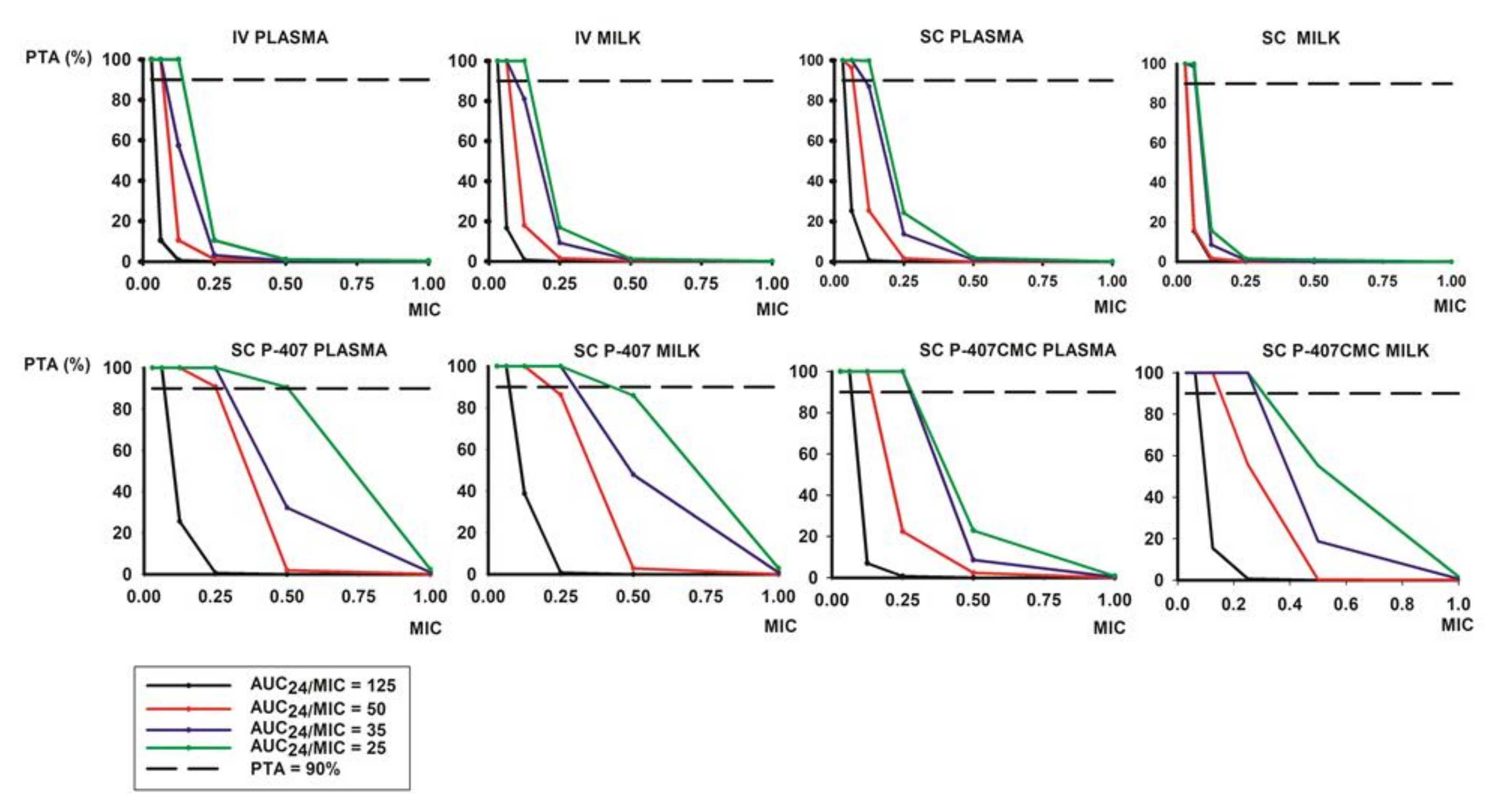
| Parameters | IV | SC | SC-P407 | SC-PCMC |
|---|---|---|---|---|
| λz (1/h) | 0.10 (0.08–0.15) | 0.11 (0.09–0.15) | 0.05 (0.03–0.08) a,b | 0.06 (0.014–0.08) a,b |
| t1/2λz (h) | 7.12 (4.72–9.13) | 6.57 (4.68–7.79) | 13.92 (8.99–24.42) a,b | 12.19 (8.86–17.14) a,b |
| Cmax (mg/L) | 2.37 (1.04–2.75) | 2.62 (1.74–4.06) | 1.99 (1.54–2.95) | |
| Tmax (h) | 1.00 (1.00–1.50) | 3.00 (2.00–4.00) b | 4.00 (2.00–6.00) b | |
| AUC0-inf (mg·h/L) | 6.82 (5.53–8.93) | 8.86 (4.06–12.00) | 21.99 (17.73–27.78) | 17.24 (13.47–20.00) |
| AUC24 (mg·h/L) | 6.82 (5.53–8.93) | 8.74 (3.92–11.71) | 20.25 (16.33–26.53) | 16.48 (12.95–19.02) |
| MRT (h) | 3.05 (2.20–3.56) | 4.04 (3.35–4.87) | 9.63 (6.15–10.21) a,b | 7.69 (7.07–9. 11) a,b |
| MAT (h) | 1.54 (0.19–1.80) | 6.49 (2.59–7.18) b | 5.35 (4.25–5.56) b | |
| VSS (L/kg) | 0.91 (0.75–1.14) | |||
| Cl (L/h/kg) | 0.29 (0.22–0.35) | |||
| F% | 93.92 (64.91–113.40) | 103.27 (69.19–117.10) | 81.74 (68.31–106.47) |
| Parameters | IV | SC | SC-P407 | SC-P407-CMC |
|---|---|---|---|---|
| λz (1/h) | 0.10 (0.06–0.18) | 0.10 (0.07–0.14) | 0.07 (0.04–0.12) a,b | 0.09 (0.06–0.12) a,b |
| t1/2λz (h) | 7.22 (3.87–11.37) | 7.16 (4.83–9.68) | 9.30 (86.00–17.44) a,b | 7.74 (6.02–11.16) a,b |
| Cmax (µg/mL) | 1.78 (0.71–2.67) | 1.41 (0.85–3.14) | 3.26 (1.46–4.63) | 2.38 (1.78–2.66) |
| Tmax (h) | 2.00 (1.00–4.00) | 2.00 (2.00–4.00) | 4.00 (2.00–6.00) a,b | 4.00 (2.00–6.00) a,b |
| AUC (µg·h/mL) | 7.98 (4.45–11.61) | 6.96 (4.15–14.08) | 23.36 (14.84–35.54) | 19.34 (16.09–28.08) |
| AUC24 (µg·h/mL) | 7.83 (4.31–11.52) | 6.84 (4.05–13.87) | 22.89 (14.28–34.92) | 18.93 (15.79–26.41) |
| MRT (h) | 4.79 (4.28–5.80) | 5.05 (4.62–5.62) | 7.73 (6.62–8.68) a,b | 7.85 (6.27–10.43) a,b |
| Cmax-milk/Cmax-plasma | 0.72 (0.47–1.51) | 1.20 (0.47–1.89) | 1.11 (0.90–1.50) | |
| AUCmilk/AUCplasma | 1.04 (0.79–1.37) | 0.93 (0.72–1.59) | 0.95 (0.76–1.68) | 1.23 (0.81–1.59) |
| Recovery (%) | 0.76 (0.11–2.69) | 0.59 (0.24–1.34) | 0.60 (0.51–1.52) | 0.52 (0.40–0.76) |
| Plasma | IV | SC | SC-P407 n | SC-P407-CMC n |
|---|---|---|---|---|
| Cmax (mg/L) | 2.37 (1.04–2.75) | 0.87 (0.58–1.35) b | 0.66 (0.51–0.98) b | |
| AUC (mg·h/L) | 6.82 (5.53–8.93) | 8.86 (4.06–12.00) | 7.33 (5.91–9.26) | 5.75 (4.49–6.67) a,b,c |
| AUC24 (mg·h/L) | 6.82 (5.53–8.93) | 8.74 (3.92–11.71) | 6.75 (5.44–8.84) | 5.49 (4.32–6.34) a,b,c |
| Milk | IV | SC | SC-P407 n | SC-P407-CMC n |
| Cmax (mg/L) | 1.78 (0.71–2.67) | 1.41 (0.85–3.14) | 1.09 (0.49–1.54) | 0.79 (0.59–0.89) b |
| AUC (mg·h/L) | 7.98 (4.45–11.61) | 6.96 (4.15–14.08) | 7.79 (4.95–11.85) | 6.45 (5.36–9.36) |
| AUC24 (mg·h/L) | 7.83 (4.31–11.52) | 6.84 (4.05–13.87) | 7.63 (4.76–11.64) | 6.31 (5.26–8.80) |
| Parameters | IV | SC | SC-P407 | SC-P407-CMC |
|---|---|---|---|---|
| AUC24/MIC50 (h) plasma | 27.27 (22.13–35.70) | 34.94 (15.69–46.82) | 81.02 (65.33–106.12) a | 95.92 (51.79–76.08) a |
| AUC24/MIC50 (h) milk | 31.32 (17.25–46.08) | 27.35 (16.22–55.47) | 91.55 (57.12–139.70) a | 75.70 (63.15–105.66) a |
| AUC24/MIC90 (h) plasma | 6.82 (5.53–8.93) | 8.74 (3.92–11.71) | 20.25 (16.33–26.53) a | 16.48 (12.95–19.02) a |
| AUC24/MIC90n (h) milk | 7.83 (4.31–11.52) | 6.84 (4.05–13.87) | 22.89 (14.28–34.92) a | 18.93 (15.79–26.41) a |
| MIC50 (μg/mL) | 0.25 | Range (μg/mL) 0.0625–4.00 | ||
| MIC90 (μg/mL) | 1.00 | |||
| Dosage Calculated to Achieve a 90% PTA of Endpoint | ||||
|---|---|---|---|---|
| AUC/MIC Ratio | IV | SC | P407 | P407CMC |
| 125 | 9.65 | 8.47 | 8.83 | 11.57 |
| 50 | 3.85 | 3.48 | 3.45 | 4.61 |
| 30 | 2.81 | 2.44 | 2.35 | 3.2 |
| 25 | 1.96 | 2.27 | 1.73 | 2.3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fernández-Varón, E.; García-Romero, E.; Serrano-Rodríguez, J.M.; Cárceles, C.M.; García-Galán, A.; Cárceles-García, C.; Fernández, R.; Muñoz, C.; de la Fe, C. PK/PD Analysis of Marbofloxacin by Monte Carlo Simulation against Mycoplasmaagalactiae in Plasma and Milk of Lactating Goats after IV, SC and SC-Long Acting Formulations Administration. Animals 2021, 11, 1104. https://doi.org/10.3390/ani11041104
Fernández-Varón E, García-Romero E, Serrano-Rodríguez JM, Cárceles CM, García-Galán A, Cárceles-García C, Fernández R, Muñoz C, de la Fe C. PK/PD Analysis of Marbofloxacin by Monte Carlo Simulation against Mycoplasmaagalactiae in Plasma and Milk of Lactating Goats after IV, SC and SC-Long Acting Formulations Administration. Animals. 2021; 11(4):1104. https://doi.org/10.3390/ani11041104
Chicago/Turabian StyleFernández-Varón, Emilio, Edgar García-Romero, Juan M. Serrano-Rodríguez, Carlos M. Cárceles, Ana García-Galán, Carlos Cárceles-García, Rocío Fernández, Cristina Muñoz, and Christian de la Fe. 2021. "PK/PD Analysis of Marbofloxacin by Monte Carlo Simulation against Mycoplasmaagalactiae in Plasma and Milk of Lactating Goats after IV, SC and SC-Long Acting Formulations Administration" Animals 11, no. 4: 1104. https://doi.org/10.3390/ani11041104
APA StyleFernández-Varón, E., García-Romero, E., Serrano-Rodríguez, J. M., Cárceles, C. M., García-Galán, A., Cárceles-García, C., Fernández, R., Muñoz, C., & de la Fe, C. (2021). PK/PD Analysis of Marbofloxacin by Monte Carlo Simulation against Mycoplasmaagalactiae in Plasma and Milk of Lactating Goats after IV, SC and SC-Long Acting Formulations Administration. Animals, 11(4), 1104. https://doi.org/10.3390/ani11041104






