Seroprevalence of Toxoplasma gondii among Sylvatic Rodents in Poland
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
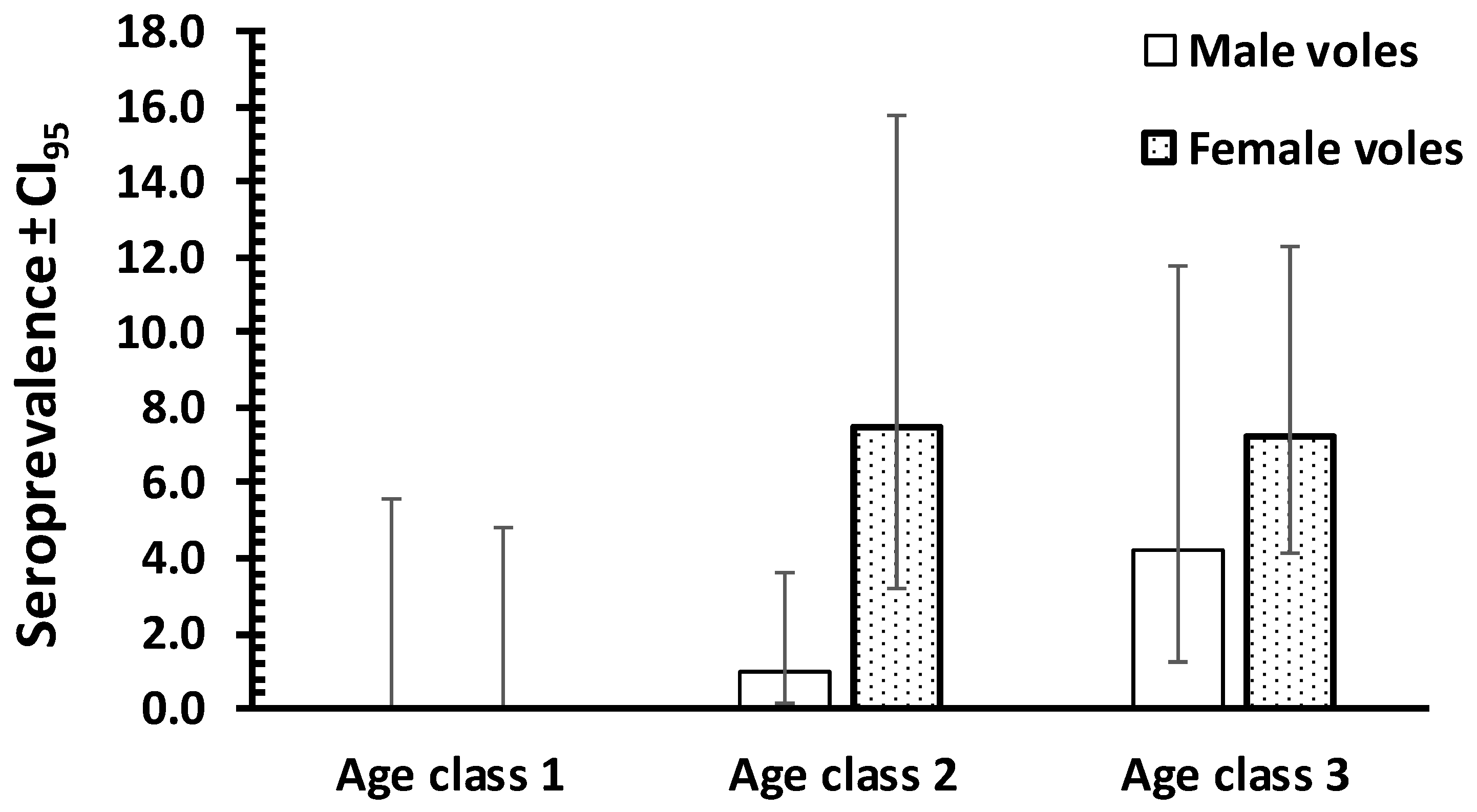
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Grzybek, M.; Bajer, A.; Bednarska, M.M.; Al-Sarraf, M.; Behnke-Borowczyk, J.; Harris, P.D.; Price, S.J.; Brown, G.S.; Osborne, S.-J.; Siński, E.; et al. Long-term spatiotemporal stability and dynamic changes in helminth infracommunities of bank voles (Myodes glareolus) in NE Poland. Parasitology 2015, 142, 1722–1743. [Google Scholar] [CrossRef]
- Behnke, J.M.; Bajer, A.; Harris, P.D.; Newington, L.; Pidgeon, E.; Rowlands, G.; Sheriff, C.; Kuliś-Malkowska, K.; Siński, E.; Gilbert, F.S.; et al. Temporal and between-site variation in helminth communities of bank voles (Myodes glareolus) from N.E. Poland: 2. The infracommunity level. Parasitology 2008, 135, 999–1018. [Google Scholar] [CrossRef] [PubMed]
- Rabalski, L.; Kosinski, M.; Smura, T.; Aaltonen, K.; Kant, R.; Sironen, T.; Szewczyk, B.; Grzybek, M. Detection and molecular characterisation of SARS-CoV-2 in farmed mink (Neovison vison) in Poland. bioRxiv 2020. [Google Scholar] [CrossRef]
- Achazi, K.; Růžek, D.; Donoso-Mantke, O.; Schlegel, M.; Ali, H.S.; Wenk, M.; Schmidt-Chanasit, J.; Ohlmeyer, L.; Rühe, F.; Vor, T.; et al. Rodents as Sentinels for the Prevalence of Tick-Borne Encephalitis Virus. Vector-Borne Zoonotic Dis. 2011, 11, 641–647. [Google Scholar] [CrossRef] [PubMed]
- Grzybek, M.; Cybulska, A.; Tołkacz, K.; Alsarraf, M.; Behnke-Borowczyk, J.; Szczepaniak, K.; Strachecka, A.; Paleolog, J.; Moskwa, B.; Behnke, J.M.; et al. Seroprevalence of Trichinella spp. infection in bank voles (Myodes glareolus)—A long term study. Int. J. Parasitol. Parasites Wildl. 2019, 9. [Google Scholar] [CrossRef]
- Behnke, J.M. Structure in parasite component communities in wild rodents: Predictability, stability, associations and interactions or pure randomness? Parasitology 2008, 135, 751–766. [Google Scholar] [CrossRef]
- Wilson, D.E.; Reeder, D.M. Mammal Species of the World, 3rd ed.; Johns Hopkins University Press: Baltimore, MD, USA, 2005; Volume 1, ISBN 9780801882210. [Google Scholar]
- Waindok, P.; Özbakış-Beceriklisoy, G.; Janecek-Erfurth, E.; Springer, A.; Pfeffer, M.; Leschnik, M.; Strube, C. Parasites in brains of wild rodents (Arvicolinae and Murinae) in the city of Leipzig, Germany. Int. J. Parasitol. Parasites Wildl. 2019, 10, 211–217. [Google Scholar] [CrossRef]
- Grzybek, M.; Sironen, T.; Mäki, S.; Tołkacz, K.; Alsarraf, M.; Strachecka, A.; Paleolog, J.; Biernat, B.; Szczepaniak, K.; Behnke-Borowczyk, J.; et al. Zoonotic Virus Seroprevalence among Bank Voles, Poland, 2002–2010. Emerg. Infect. Dis. 2019, 25, 1607–1609. [Google Scholar] [CrossRef]
- Johnson, H.J.; Koshy, A.A. Latent Toxoplasmosis Effects on Rodents and Humans: How Much is Real and How Much is Media Hype? MBio 2020, 11. [Google Scholar] [CrossRef]
- Hill, D.; Dubey, J.P. Toxoplasma gondii: Transmission, diagnosis and prevention. Clin. Microbiol. Infect. 2002, 8, 634–640. [Google Scholar] [CrossRef]
- Tenter, A.M.; Heckeroth, A.R.; Weiss, L.M. Toxoplasma gondii: From animals to humans. Int. J. Parasitol. 2000, 30, 1217–1258. [Google Scholar] [CrossRef]
- ECDC. Annual Epidemiological Report for 2017; ECDC: Stockholm, Sweden, 2019. [Google Scholar]
- NIPH—NIH National Institute of Public Health—National Institute of Hygiene in Poland. Available online: http://wwwold.pzh.gov.pl/oldpage/epimeld/index_p.html (accessed on 20 December 2020).
- Tołkacz, K.; Alsarraf, M.; Kowalec, M.; Dwużnik, D.; Grzybek, M.; Behnke, J.M.; Bajer, A. Bartonella infections in three species of Microtus: Prevalence and genetic diversity, vertical transmission and the effect of concurrent Babesia microti infection on its success. Parasites Vectors 2018, 11, 491. [Google Scholar] [CrossRef] [PubMed]
- Behnke, J.M.; Barnard, C.J.; Bajer, A.; Bray, D.; Dinmore, J.; Frake, K.; Osmond, J.; Race, T.; Sinski, E. Variation in the helminth community structure in bank voles (Clethrionomys glareolus) from three comparable localities in the Mazury Lake District region of Poland. Parasitology 2001, 123, 401–414. [Google Scholar] [CrossRef] [PubMed]
- Grzybek, M.; Bajer, A.; Behnke-Borowczyk, J.; Al-Sarraf, M.; Behnke, J.M. Female host sex-biased parasitism with the rodent stomach nematode Mastophorus muris in wild bank voles (Myodes glareolus). Parasitol. Res. 2014, 114, 523–533. [Google Scholar] [CrossRef]
- Tołkacz, K.; Bednarska, M.; Alsarraf, M.; Dwużnik, D.; Grzybek, M.; Welc-Falęciak, R.; Behnke, J.M.; Bajer, A. Prevalence, genetic identity and vertical transmission of Babesia microti in three naturally infected species of vole, Microtus spp. (Cricetidae). Parasites Vectors 2017, 10, 1–12. [Google Scholar] [CrossRef]
- Antolová, D.; Reiterová, K.; Stanko, M.; Zalesny, G.; Fričová, J.; Dvorožňáková, E. Small mammals: Paratenic hosts for species of Toxocara in eastern Slovakia. J. Helminthol. 2013, 87, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Essbauer, S.; Schmidt, J.; Conraths, F.J.; Friedrich, R.; Koch, J.; Hautmann, W.; Pfeffer, M.; Wölfel, R.; Finke, J.; Dobler, G.; et al. A new Puumala hantavirus subtype in rodents associated with an outbreak of Nephropathia epidemica in South-East Germany in 2004. Epidemiol. Infect. 2006, 134. [Google Scholar] [CrossRef]
- Reiterová, K.; Antolová, D.; Zaleśny, G.; Stanko, M.; Špilovská, S.; Mošanský, L. Small rodents—Permanent reservoirs of toxocarosis in different habitats of Slovakia. Helminthologia 2013, 50. [Google Scholar] [CrossRef]
- Reperant, L.A.; Hegglin, D.; Tanner, I.; Fischer, C.; Deplazes, P. Rodents as shared indicators for zoonotic parasites of carnivores in urban environments. Parasitology 2009, 136, 329–337. [Google Scholar] [CrossRef]
- Hughes, H.P.; Van Knapen, F.; Atkinson, H.J.; Balfour, A.H.; Lee, D.L. A new soluble antigen preparation of Toxoplasma gondii and its use in serological diagnosis. Clin. Exp. Immunol. 1982, 49, 239–246. [Google Scholar]
- Opsteegh, M.; Teunis, P.; Mensink, M.; Züchner, L.; Titilincu, A.; Langelaar, M.; van der Giessen, J. Evaluation of ELISA test characteristics and estimation of Toxoplasma gondii seroprevalence in Dutch sheep using mixture models. Prev. Vet. Med. 2010, 96, 232–240. [Google Scholar] [CrossRef]
- Naguleswaran, A.; Hemphill, A.; Rajapakse, R.P.V.J.; Sager, H. Elaboration of a crude antigen ELISA for serodiagnosis of caprine neosporosis: Validation of the test by detection of Neospora caninum-specific antibodies in goats from Sri Lanka. Vet. Parasitol. 2004, 126, 257–262. [Google Scholar] [CrossRef]
- Grzybek, M.; Alsarraf, M.; Tołkacz, K.; Behnke-Borowczyk, J.; Biernat, B.; Stańczak, J.; Strachecka, A.; Guz, L.; Szczepaniak, K.; Paleolog, J.; et al. Seroprevalence of TBEV in bank voles from Poland-a long-term approach. Emerg. Microbes Infect. 2018, 7, 145. [Google Scholar] [CrossRef]
- Jittapalapong, S.; Sarataphan, N.; Maruyama, S.; Hugot, J.-P.; Morand, S.; Herbreteau, V. Toxoplasmosis in Rodents: Ecological Survey and First Evidences in Thailand. Vector-Borne Zoonotic Dis. 2011, 11, 231–237. [Google Scholar] [CrossRef]
- Galeh, T.M.; Sarvi, S.; Montazeri, M.; Moosazadeh, M.; Nakhaei, M.; Shariatzadeh, S.A.; Daryani, A. Global Status of Toxoplasma gondii Seroprevalence in Rodents: A Systematic Review and Meta-Analysis. Front. Vet. Sci. 2020, 7. [Google Scholar] [CrossRef]
- Weber, J.-M.; Dailly, L. Food habits and ranging behaviour of a group of farm cats (Felis catus) in a Swiss mountainous area. J. Zool. 1998, 245, S0952836998286092. [Google Scholar] [CrossRef]
- Afonso, E.; Poulle, M.L.; Lemoine, M.; Villena, I.; Aubert, D.; Gilot-Fromont, E. Prevalence of Toxoplasma gondii in small mammals from the Ardennes region, France. Folia Parasitol. 2007, 54, 313–314. [Google Scholar] [CrossRef] [PubMed]
- Afonso, E.; Thulliez, P.; Pontier, D.; Gilot-Fromonte, E. Toxoplasmosis in prey species and consequences for prevalence in feral cats: Not all prey species are equal. Parasitology 2007, 134, 1963–1971. [Google Scholar] [CrossRef]
- Pavlova, E.V.; Kirilyuk, E.V.; Naidenko, S.V. Occurrence Pattern of Influenza A Virus, Coxiella burnetii, Toxoplasma gondii, and Trichinella sp. in the Pallas Cat and Domestic Cat and Their Potential Prey Under Arid Climate Conditions. Arid Ecosyst. 2016, 6, 277–283. [Google Scholar] [CrossRef]
- Lehrer, E.W.; Fredebaugh, S.L.; Schooley, R.L.; Mateus-Pinilla, N.E. Prevalence of Antibodies to Toxoplasma gondii in Woodchucks across an Urban–Rural Gradient. J. Wildl. Dis. 2010, 46, 977–980. [Google Scholar] [CrossRef] [PubMed]
- Mercier, A.; Garba, M.; Bonnabau, H.; Kane, M.; Rossi, J.-P.; Darde, M.-L.; Dobigny, G. Toxoplasmosis seroprevalence in urban rodents: A survey in Niamey, Niger. Mem. Inst. Oswaldo Cruz 2013, 108, 399–407. [Google Scholar] [CrossRef]
- Murphy, R.G.; Williams, R.H.; Hughes, J.M.; Hide, G.; Ford, N.J.; Oldbury, D.J. The urban house mouse (Mus domesticus) as a reservoir of infection for the human parasite Toxoplasma gondii: An unrecognised public health issue? Int. J. Environ. Health Res. 2008, 18, 177–185. [Google Scholar] [CrossRef]
- DeFeo, M.L.; Dubey, J.P.; Mather, T.N.; Rhodes, R.C., III. Epidemiologic investigation of seroprevalence of antibodies to Toxoplasma gondii in cats and rodents. Am. J. Vet. Res. 2002, 63, 1714–1717. [Google Scholar] [CrossRef] [PubMed]
- Ding, H.; Gao, Y.-M.; Deng, Y.; Lamberton, P.H.L.; Lu, D.-B. A systematic review and meta-analysis of the seroprevalence of Toxoplasma gondii in cats in mainland China. Parasites Vectors 2017, 10, 27. [Google Scholar] [CrossRef]
- Grzybek, M.; Tołkacz, K.; Sironen, T.; Mäki, S.; Alsarraf, M.; Behnke-Borowczyk, J.; Biernat, B.; Nowicka, J.; Vaheri, A.; Henttonen, H.; et al. Zoonotic Viruses in Three Species of Voles from Poland. Animals 2020, 10, 1820. [Google Scholar] [CrossRef]
- Meerburg, B.G.; Singleton, G.R.; Kijlstra, A. Rodent-borne diseases and their risks for public health. Crit. Rev. Microbiol. 2009, 35, 221–270. [Google Scholar] [CrossRef]
- Thompson, R.C.A.; Lymbery, A.J.; Smith, A. Parasites, emerging disease and wildlife conservation. Int. J. Parasitol. 2010, 40, 1163–1170. [Google Scholar] [CrossRef]
| Species | Negative | Positive | Total | Seroprevalence (%) ± CL95 |
|---|---|---|---|---|
| M. agrestis | 7 | 3 | 10 | 30.0 [8.7–61.9] |
| M. arvalis | 37 | 9 | 46 | 19.6 [9.1–36.8] |
| A. oeconomus | 12 | 2 | 14 | 14.3 [2.6–42.6] |
| Myodes graelous | 489 | 18 | 507 | 3.6 [2.6–4.9] |
| Overall | 545 | 32 | 577 | 5.5 [4.2–7.3] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Grzybek, M.; Antolová, D.; Tołkacz, K.; Alsarraf, M.; Behnke-Borowczyk, J.; Nowicka, J.; Paleolog, J.; Biernat, B.; Behnke, J.M.; Bajer, A. Seroprevalence of Toxoplasma gondii among Sylvatic Rodents in Poland. Animals 2021, 11, 1048. https://doi.org/10.3390/ani11041048
Grzybek M, Antolová D, Tołkacz K, Alsarraf M, Behnke-Borowczyk J, Nowicka J, Paleolog J, Biernat B, Behnke JM, Bajer A. Seroprevalence of Toxoplasma gondii among Sylvatic Rodents in Poland. Animals. 2021; 11(4):1048. https://doi.org/10.3390/ani11041048
Chicago/Turabian StyleGrzybek, Maciej, Daniela Antolová, Katarzyna Tołkacz, Mohammed Alsarraf, Jolanta Behnke-Borowczyk, Joanna Nowicka, Jerzy Paleolog, Beata Biernat, Jerzy M. Behnke, and Anna Bajer. 2021. "Seroprevalence of Toxoplasma gondii among Sylvatic Rodents in Poland" Animals 11, no. 4: 1048. https://doi.org/10.3390/ani11041048
APA StyleGrzybek, M., Antolová, D., Tołkacz, K., Alsarraf, M., Behnke-Borowczyk, J., Nowicka, J., Paleolog, J., Biernat, B., Behnke, J. M., & Bajer, A. (2021). Seroprevalence of Toxoplasma gondii among Sylvatic Rodents in Poland. Animals, 11(4), 1048. https://doi.org/10.3390/ani11041048







