Soybean Oil Replacement by Palm Fatty Acid Distillate in Broiler Chicken Diets: Fat Digestibility and Lipid-Class Content along the Intestinal Tract
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Housing and Animals
2.2. Experimental Design and Diets
2.3. Controls and Sampling
2.4. Chemical Analysis
2.5. Calculations
2.6. Statistical Analysis
3. Results
3.1. Characterization of Experimental Oils and Diets
3.2. Growth Performance and Abdominal Fat Deposition
3.3. Lipid-Class Content along the Intestinal Tract
3.4. Apparent Fatty-Acid Digestibility along the Intestinal Tract
3.5. Contribution of Each Intestinal Segment to FA Absorption
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Acknowledgments
Conflicts of Interest
References
- Statista Vegetables. Oils Global Consumption. Available online: https://www.statista.com/statistics/263937/vegetable-oils-global-consumption/ (accessed on 29 January 2021).
- FEDIOL. The European Vegetable Oil and Protein Meal Industry Association—Refining of Edible Oils. Available online: www.fediol.eu/web/refining/1011306087/list1187970096/f1.htm (accessed on 20 March 2021).
- Gibon, V.; De Greyt, W.; Kellens, M. Palm oil refining. Eur. J. Lipid Sci. Technol. 2007, 109, 315–335. [Google Scholar] [CrossRef]
- Varona, E.; Tres, A.; Rafecas, M.; Vichi, S.; Barroeta, A.C.; Guardiola, F. Composition and nutritional value of acid oils and fatty acid distillates used in animal feeding. Animals 2021, 11, 196. [Google Scholar] [CrossRef] [PubMed]
- Vilarrasa, E.; Codony, R.; Esteve-Garcia, E.; Barroeta, A.C. Use of re-esterified oils, differing in their degree of saturation and molecular structure, in broiler chicken diets. Poult. Sci. 2015, 94, 1527–1538. [Google Scholar] [CrossRef]
- Viñado, A.; Castillejos, L.; Rodriguez-Sanchez, R.; Barroeta, A.C. Crude soybean lecithin as alternative energy source for broiler chicken diets. Poult. Sci. 2019, 98, 5601–5612. [Google Scholar] [CrossRef] [PubMed]
- Tancharoenrat, P.; Ravindran, V.; Zaefarian, F.; Ravindran, G. Digestion of fat and fatty acids along the gastrointestinal tract of broiler chickens. Poult. Sci. 2014, 93, 371–379. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Sanchez, R.; Tres, A.; Sala, R.; Guardiola, F.; Barroeta, A. Evolution of lipid classes and fatty acid digestibility along the gastrointestinal tract of broiler chickens fed different fat sources at different ages. Poult. Sci. 2019, 98, 1341–1353. [Google Scholar] [CrossRef] [PubMed]
- Blanch, A.; Barroeta, A.C.; Baucells, M.D.; Puchal, F. The nutritive value of dietary fats in relation to their chemical composition. Apparent fat availability and metabolizable energy in two-week-old chicks. Poult. Sci. 1995, 74, 1335–1340. [Google Scholar] [CrossRef] [PubMed]
- Zumbado, M.E.; Scheele, C.W.; Kwakernaak, C. Chemical composition, digestibility, and metabolizable energy content of different fat and oil by-products. J. Appl. Poult. Res. 1999, 8, 263–271. [Google Scholar] [CrossRef]
- Tancharoenrat, P.; Ravindran, V.; Zaefarian, F.; Ravindran, G. Influence of age on the apparent metabolisable energy and total tract apparent fat digestibility of different fat sources for broiler chickens. Anim. Feed Sci. Technol. 2013, 186, 186–192. [Google Scholar] [CrossRef]
- Rodriguez-Sanchez, R.; Tres, A.; Sala, R.; Garcés-Narro, C.; Guardiola, F.; Gasa, J.; Barroeta, A.C. Effects of dietary free fatty-acid content and saturation degree on lipid-class composition and fatty-acid digestibility along the gastrointestinal tract in broiler starter chickens. Poult. Sci. 2019, 98, 4929–4941. [Google Scholar] [CrossRef]
- Roll, A.P.; Vilarrasa, E.; Tres, A.; Barroeta, A.C. The different molecular structure and glycerol-to-fatty acid ratio of palm oils affect their nutritive value in broiler chicken diets. Animal 2018, 12, 240–248. [Google Scholar] [CrossRef]
- Aviagen. Ross Broiler Management Handbook; Aviagen, Ed.; Newbridge: Scotland, UK, 2014. [Google Scholar]
- Fundación Española para el Desarrollo de la Nutrición Animal. Necesidades Nutricionales para Avicultura: Pollos de Carne y Aves de Puesta; FEDNA: Madrid, Spain, 2008; ISBN 9788409065295. [Google Scholar]
- American Oil Chemists’ Society (AOCS). Official Method Ca 2d-25. Moisture and volatile matter, vacuum oven method. In Official Methods and Recommended Practices of the American Oil Chemists’ Society; AOCS Press: Champaign, IL, USA, 2017. [Google Scholar]
- International Standard. ISO 663:2017. Animal and Vegetable Fats and Oils, Determination of Insoluble Impurities Content; ISO: Geneva, Switzerland, 2017. [Google Scholar]
- American Oil Chemists’ Society (AOCS). Official Method Ca 6b-53. Unsaponifiable matter. In Official Methods and Recommended Practices of the American Oil Chemists’ Society; AOCS Press: Champaign, IL, USA, 2017. [Google Scholar]
- IUPAC (International Union of Pure and Applied Chemistry). Determination of polymerized triglycerides in oils and fats by high performance liquid chromatography (method 2508). In Standard Methods for the Analysis of Oils, Fats and Derivatives; IUPAC, Ed.; Blackwell Scientific Publications: Oxford, UK, 1991. [Google Scholar]
- Guardiola, F.; Codony, R.; Rafecas, M.; Boatella, J.; López, A. Fatty acid composition and nutritional value of fresh eggs, from large- and small-scale farms. J. Food Compos. Anal. 1994, 7, 171–188. [Google Scholar] [CrossRef]
- American Oil Chemists’ Society. Official Methods of Analysis of AOAC International, 18th ed.; AOAC International: Gaithersburg, MD, USA, 2005; ISBN 5935584-77-3 A. [Google Scholar]
- Short, F.J.; Gorton, P.; Wiseman, J.; Boorman, K.N. Determination of titanium dioxide added as an inert marker in chicken digestibility studies. Anim. Feed Sci. Technol. 1996, 5, 215–221. [Google Scholar] [CrossRef]
- Sukhija, P.S.; Palmquist, D.L. Rapid method for determination of total fatty acid content and composition of feedstuffs and feces. J. Agric. Food Chem. 1988, 36, 1202–1206. [Google Scholar] [CrossRef]
- Cortinas, L.; Villaverde, C.; Galobart, J.; Baucells, M.D.; Codony, R.; Barroeta, A.C. Fatty acid content in chicken thigh and breast as affected by dietary polyunsaturation level. Poult. Sci. 2004, 83, 1155–1164. [Google Scholar] [CrossRef]
- Renner, R. Site of fat absorption in the chick. Poult. Sci. 1965, 44, 861–864. [Google Scholar] [CrossRef]
- Jimenez-Moya, B.; Martin, D.; Soler-Rivas, D.; Barroeta, A.C.; Tres, A.; Sala, R. Acid versus crude oils for broiler chicken diets: In vitro lipid digestion and bioaccessibility. Anim. Feed Sci. Technol. 2021. under review. [Google Scholar]
- Krogdahl, A. Digestion and absorption of lipids in poultry. J. Nutr. 1985, 115, 675–685. [Google Scholar] [CrossRef]
- Noy, Y.; Sklan, D. Digestion and absorption in the young chick. Poult. Sci. 1995, 74, 366–373. [Google Scholar] [CrossRef]
- Freeman, C.P. Properties of fatty acids in dispersions of emulsified lipid and bile salt and the significance of these properties in fat absorption in the pig and the sheep. Br. J. Nutr. 1969, 23, 249–263. [Google Scholar] [CrossRef]
- Small, D.M. The effects of glyceride structure on absorption and metabolism. Annu. Rev. Nutr. 1991, 11, 413–434. [Google Scholar] [CrossRef] [PubMed]
- Leeson, S.; Summers, J.D. Commercial Poultry Nutrition, 3rd ed.; Nottingham University Press: Nottingham, UK, 2005; ISBN 9781904761785. [Google Scholar]
- Ravindran, V.; Tancharoenrat, P.; Zaefarian, F.; Ravindran, G. Fats in poultry nutrition: Digestive physiology and factors influencing their utilisation. Anim. Feed Sci. Technol. 2016, 213, 1–21. [Google Scholar] [CrossRef]
- Wiseman, J.; Salvador, F. The influence of free fatty acid content and degree of saturation on the apparent metabolizable energy value of fats fed to broilers. Poult. Sci. 1991, 70, 573–582. [Google Scholar] [CrossRef] [PubMed]
- Young, R.J.; Garrett, R.L. Effect of oleic and linoleic acids on the absorption of saturated fatty acids in the chick. J. Nutr. 1963, 81, 321–329. [Google Scholar] [CrossRef]
- Batal, A.B.; Parsons, C.M. Effects of age on nutrient digestibility in chicks fed different diets. Poult. Sci. 2002, 81, 400–407. [Google Scholar] [CrossRef]
- Angel, R.; Kim, S.W.; Li, W.; Jimenez-Moreno, E. Velocidad de paso y ph intestinal en aves: Implicaciones para la digestión y el uso de enzimas. In Proceedings of the XXIX Curso de Especialización FEDNA, Madrid, Spain, 6–7 November 2013. [Google Scholar]

| Ingredients, % | Starter Diet (from 0 d to 22 d) | Grower-Finisher Diet (from 23 d to 35 d) |
|---|---|---|
| Wheat | 54.49 | 44.02 |
| Soybean meal 47% | 35.40 | 27.25 |
| Barley | - | 18.58 |
| Experimental fats 1 | 6.00 | 6.00 |
| Calcium carbonate | 1.44 | 1.39 |
| Monocalcium phosphate | 0.99 | 1.20 |
| Titanium dioxide | 0.50 | 0.50 |
| Vitamin and mineral premix 2 | 0.40 | 0.40 |
| Sodium chloride | 0.40 | 0.35 |
| DL-Methionine | 0.23 | 0.17 |
| L-Lysine | 0.15 | 0.12 |
| L-Threonine | - | 0.02 |
| Item | S | PFAD | P |
|---|---|---|---|
| Moisture (g/100 g) | ND | 0.01 | ND |
| Insoluble impurities (g/100 g) | 1.27 | 3.76 | 0.59 |
| Unsaponifiable matter (g/100 g) | 0.99 | 1.34 | 0.21 |
| Fatty acid composition (%) 2 | |||
| C16:0 | 10.98 | 46.59 | 43.94 |
| C18:0 | 3.47 | 6.62 | 4.64 |
| C18:1 n-9 | 25.11 | 34.96 | 38.43 |
| C18:2 n-6 | 51.70 | 8.49 | 9.70 |
| C18:3 n-3 | 5.34 | 0.29 | 0.13 |
| Minor fatty acids | 3.40 | 3.05 | 3.15 |
| SFA | 15.86 | 55.13 | 50.64 |
| cis-MUFA | 27.06 | 35.87 | 39.44 |
| trans-C18:1 | 0.04 | 0.22 | 0.08 |
| PUFA | 57.04 | 8.78 | 9.83 |
| UFA:SFA | 5.29 | 0.82 | 0.98 |
| Lipid class composition (%) 3 | |||
| TAG | 96.27 | 4.01 | 92.46 |
| DAG | 3.23 | 3.04 | 7.54 |
| MAG | ND | ND | ND |
| FFA | 0.50 | 92.94 | ND |
| T (mg/kg) | 1007.31 | 42.79 | 199.40 |
| T3 (mg/kg) | ND | 52.59 | 431.87 |
| Item | Starter Diets (from 0 to 22 d) | Grower-Finisher Diets (from 23 to 35 d) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| S6 | S4-PA2 | S2-PA4 | PA6 | P6 | S6 | S4-PA2 | S2-PA4 | PA6 | P6 | |
| Macronutrient content | ||||||||||
| Dry matter (g/100 g) | 91.00 | 91.03 | 91.14 | 90.89 | 90.93 | 90.14 | 90.27 | 90.37 | 90.43 | 90.02 |
| Crude protein (g/100 g) | 23.61 | 23.87 | 23.47 | 23.60 | 23.15 | 21.04 | 22.03 | 21.45 | 20.59 | 20.84 |
| Crude fat (g/100 g) | 7.51 | 7.39 | 8.16 | 7.78 | 7.70 | 8.18 | 8.08 | 8.36 | 8.10 | 7.49 |
| Crude fiber (g/100 g) | 3.29 | 3.14 | 3.14 | 2.86 | 3.20 | 3.08 | 3.10 | 3.32 | 3.13 | 3.41 |
| Ash (g/100 g) | 5.54 | 5.58 | 6.92 | 7.13 | 7.09 | 6.21 | 6.69 | 6.51 | 6.46 | 5.75 |
| Gross energy, kcal/kg | 4367 | 4402 | 4368 | 4332 | 4332 | 4339 | 4355 | 4320 | 4308 | 4324 |
| Fatty acid composition (%) | ||||||||||
| C14:0 | - | 0.40 | 0.66 | 0.91 | 0.87 | 0.06 | 0.39 | 0.65 | 0.90 | 0.85 |
| C16:0 | 14.43 | 21.99 | 30.79 | 39.07 | 37.38 | 13.24 | 21.93 | 30.24 | 39.18 | 36.85 |
| C18:0 | 3.48 | 4.19 | 5.07 | 5.79 | 4.29 | 3.35 | 4.19 | 4.97 | 5.69 | 4.17 |
| C18:1 n-9 | 22.83 | 25.07 | 27.57 | 30.31 | 32.34 | 22.61 | 25.31 | 27.85 | 29.96 | 32.49 |
| C18:1 n-7 | 1.46 | 1.28 | 1.05 | 0.78 | 0.83 | 1.50 | 1.25 | 1.00 | 0.74 | 0.80 |
| C18:2 n-6 | 50.78 | 41.48 | 30.59 | 20.30 | 21.37 | 52.04 | 41.29 | 30.99 | 20.72 | 22.02 |
| C18:3 n-3 | 5.27 | 4.13 | 2.83 | 1.55 | 1.46 | 5.50 | 4.20 | 2.93 | 1.61 | 1.60 |
| Minor fatty acids | 1.75 | 1.46 | 1.45 | 1.29 | 1.46 | 1.69 | 1.44 | 1.38 | 1.19 | 1.22 |
| SFA | 18.72 | 27.26 | 37.14 | 46.14 | 43.13 | 17.70 | 27.20 | 36.38 | 46.07 | 42.47 |
| MUFA | 25.24 | 27.13 | 29.44 | 32.01 | 34.04 | 24.76 | 27.31 | 29.70 | 31.60 | 33.92 |
| PUFA | 56.04 | 45.61 | 33.42 | 21.85 | 22.83 | 57.54 | 45.49 | 33.92 | 22.33 | 23.62 |
| UFA:SFA | 4.16 | 2.60 | 1.66 | 1.14 | 1.30 | 4.54 | 2.61 | 1.70 | 1.15 | 1.34 |
| Lipid class composition (%) | ||||||||||
| TAG | 71.88 | 54.76 | 37.54 | 14.98 | 78.67 | 76.67 | 58.78 | 37.77 | 14.20 | 78.53 |
| DAG | 11.73 | 10.05 | 7.47 | 4.67 | 10.54 | 10.23 | 9.03 | 7.28 | 5.58 | 10.44 |
| MAG | 2.19 | 2.10 | 1.66 | 1.19 | 1.84 | 2.49 | 2.20 | 1.93 | 1.77 | 2.31 |
| FFA | 14.20 | 33.08 | 53.33 | 79.17 | 8.96 | 10.61 | 29.99 | 53.01 | 78.44 | 8.72 |
| Item | Dietary Treatments 2 | SEM 3 | p-Value | ||||
|---|---|---|---|---|---|---|---|
| S6 | S4-PA2 | S2-PA4 | PA6 | P6 | |||
| From 0 to 22 d | |||||||
| ADFI, g/d/bird | 48.7 | 53.2 | 50.7 | 54.0 | 54.5 | 2.25 | 0.335 |
| ADG, g/d/bird | 37.2 | 39.1 | 38.6 | 39.7 | 40.6 | 1.24 | 0.373 |
| FCR, g/g | 1.31 | 1.36 | 1.31 | 1.36 | 1.34 | 0.036 | 0.733 |
| BW at 22 d, g | 856 | 899 | 888 | 913 | 933 | 27.1 | 0.361 |
| From 23 to 35 d | |||||||
| ADFI, g/d/bird | 134 | 141 | 141 | 144 | 143 | 2.92 | 0.148 |
| ADG, g/d/bird | 87.8 | 90.2 | 89.8 | 90.3 | 89.9 | 2.18 | 0.929 |
| FCR, g/g | 1.53 | 1.57 | 1.57 | 1.59 | 1.60 | 0.022 | 0.175 |
| BW at 35 d, g | 1997 | 2072 | 2055 | 2086 | 2101 | 44.4 | 0.526 |
| From 0 to 35 d | |||||||
| ADFI, g/d/bird | 80.3 | 85.8 | 84.3 | 87.3 | 87.5 | 2.01 | 0.130 |
| ADG, g/d/bird | 56.0 | 58.1 | 57.6 | 58.5 | 58.9 | 1.27 | 0.535 |
| FCR, g/g | 1.43 | 1.48 | 1.46 | 1.49 | 1.49 | 0.017 | 0.154 |
| Abdominal fat, g | 29.62 | 30.35 | 29.52 | 25.66 | 32.36 | 1.938 | 0.136 |
| Abdominal fat, % | 1.46 | 1.47 | 1.42 | 1.23 | 1.53 | 0.082 | 0.080 |
| Item | Dietary Treatments | SEM 3 | p-Value | ||||
|---|---|---|---|---|---|---|---|
| S6 | S4-PA2 | S2-PA4 | PA6 | P6 | |||
| Upper Jejunum | |||||||
| TAG | 0.53 ab | 0.57 a | 0.44 ab | 0.19 b | 0.49 ab | 0.082 | 0.027 |
| DAG | 1.30 | 1.60 | 1.91 | 2.37 | 1.99 | 0.413 | 0.440 |
| MAG | 0.18 ab | 0.28 a | 0.16 ab | 0.12 b | 0.15 ab | 0.033 | 0.018 |
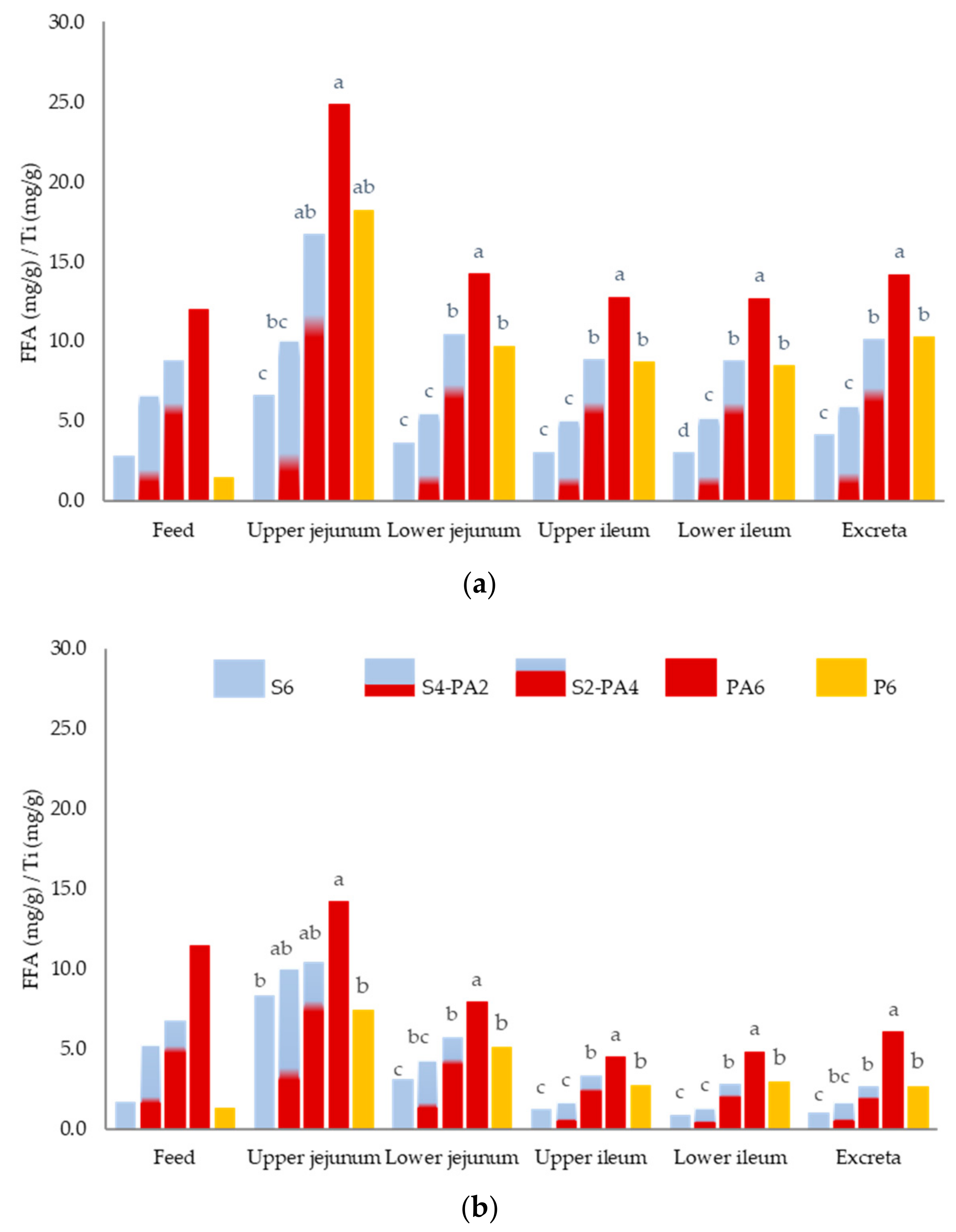
| FFA | 6.58 c | 10.04 bc | 16.69 ab | 24.82 a | 18.20 ab | 2.215 | <0.001 |
| Lower Jejunum | |||||||
| TAG | 0.34 ab | 0.34 ab | 0.47 a | 0.32 b | 0.30 b | 0.031 | 0.008 |
| DAG | 0.82 ab | 0.69 b | 1.16 a | 0.98 ab | 0.76 b | 0.091 | 0.008 |
| MAG | 0.18 | 0.18 | 0.21 | 0.17 | 0.17 | 0.025 | 0.729 |
| FFA | 3.65 c | 5.50 c | 10.45 b | 14.21 a | 9.70 b | 0.471 | <0.001 |
| Upper Ileum | |||||||
| TAG | 0.25 | 0.24 | 0.25 | 0.32 | 0.24 | 0.053 | 0.775 |
| DAG | 0.64 | 0.55 | 0.72 | 0.87 | 0.62 | 0.121 | 0.426 |
| MAG | 0.16 | 0.15 | 0.15 | 0.16 | 0.13 | 0.030 | 0.929 |
| FFA | 3.05 c | 5.02 c | 8.82 b | 12.76 a | 8.69 b | 0.530 | <0.001 |
| Lower Ileum | |||||||
| TAG | 0.32 | 0.29 | 0.27 | 0.33 | 0.19 | 0.045 | 0.239 |
| DAG | 0.64 | 0.60 | 0.76 | 0.71 | 0.45 | 0.084 | 0.114 |
| MAG | 0.23 | 0.25 | 0.25 | 0.23 | 0.15 | 0.029 | 0.137 |
| FFA | 3.02 d | 5.23 c | 8.78 b | 12.70 a | 8.47 b | 0.471 | <0.001 |
| Excreta | |||||||
| TAG | 0.38 | 0.39 | 0.28 | 0.27 | 0.29 | 0.044 | 0.219 |
| DAG | 0.98 | 0.95 | 1.33 | 1.20 | 0.80 | 0.167 | 0.205 |
| MAG | 0.19 | 0.13 | 0.15 | 0.13 | 0.21 | 0.031 | 0.282 |
| FFA | 4.16 c | 5.98 c | 10.11 b | 14.18 a | 10.24 b | 0.794 | <0.001 |
| Item | Dietary Treatments | SEM 3 | p-Value | ||||
|---|---|---|---|---|---|---|---|
| S6 | S4-PA2 | S2-PA4 | PA6 | P6 | |||
| Upper Jejunum | |||||||
| TAG | 0.21 | 0.20 | 0.20 | 0.20 | 0.26 | 0.048 | 0.871 |
| DAG | 1.32 | 1.69 | 1.47 | 2.22 | 1.20 | 0.256 | 0.073 |
| MAG | 0.24 | 0.27 | 0.30 | 0.27 | 0.17 | 0.048 | 0.436 |
| FFA | 8.28 b | 10.05 ab | 10.40 ab | 14.18 a | 7.40 b | 1.296 | 0.011 |
| Lower Jejunum | |||||||
| TAG | 0.10 | 0.26 | 0.27 | 0.23 | 0.22 | 0.045 | 0.108 |
| DAG | 0.43 | 0.61 | 0.61 | 0.72 | 0.58 | 0.115 | 0.496 |
| MAG | 0.18 | 0.26 | 0.19 | 0.23 | 0.14 | 0.041 | 0.342 |
| FFA | 3.10 c | 4.29 bc | 5.72 b | 7.89 a | 5.08 b | 0.412 | <0.001 |
| Upper Ileum | |||||||
| TAG | 0.14 | 0.16 | 0.13 | 0.13 | 0.09 | 0.021 | 0.254 |
| DAG | 0.16 | 0.21 | 0.18 | 0.17 | 0.17 | 0.021 | 0.482 |
| MAG | 0.11 | 0.12 | 0.16 | 0.14 | 0.11 | 0.015 | 0.090 |
| FFA | 1.20 c | 1.65 c | 3.32 b | 4.47 a | 2.67 b | 0.189 | <0.001 |
| Lower Ileum | |||||||
| TAG | 0.09 | 0.11 | 0.09 | 0.07 | 0.07 | 0.022 | 0.676 |
| DAG | 0.14 | 0.17 | 0.11 | 0.18 | 0.17 | 0.020 | 0.141 |
| MAG | 0.13 | 0.14 | 0.21 | 0.21 | 0.15 | 0.022 | 0.051 |
| FFA | 0.87 c | 1.32 c | 2.77 b | 4.82 a | 2.92 b | 0.257 | <0.001 |
| Excreta | |||||||
| TAG | 0.12 | 0.21 | 0.18 | 0.15 | 0.15 | 0.021 | 0.080 |
| DAG | 0.13 | 0.20 | 0.14 | 0.24 | 0.15 | 0.027 | 0.055 |
| MAG | 0.09 b | 0.12 ab | 0.12 ab | 0.16 a | 0.13 ab | 0.011 | 0.005 |
| FFA | 0.96 c | 1.63 bc | 2.62 b | 6.04 a | 2.66 b | 0.265 | <0.001 |
| Item | Dietary Treatments 1 | SEM 4 | p-Value | ||||
|---|---|---|---|---|---|---|---|
| S6 | S4-PA2 | S2-PA4 | PA6 | P6 | |||
| AME, kcal/kg 2 | 3348 a | 3340 a | 3074 b | 2760 c | 3014 b | 26.08 | <0.001 |
| Upper Jejunum 3 | |||||||
| TFA | 0.61 a | 0.61 a | 0.29 b | 0.05b | 0.20 b | 0.071 | <0.001 |
| SFA | 0.20 ab | 0.36 a | 0.19 ab | -0.01b | 0.19 ab | 0.076 | 0.044 |
| MUFA | 0.51 a | 0.51 a | 0.35 ab | 0.16 b | 0.31 ab | 0.059 | <0.001 |
| PUFA | 0.78a | 0.71 a | 0.34 b | 0.02 b | 0.04 b | 0.078 | <0.001 |
| Lower Jejunum 3 | |||||||
| TFA | 0.72 a | 0.67 a | 0.51 b | 0.30 c | 0.48 b | 0.020 | <0.001 |
| SFA | 0.60 a | 0.47 b | 0.32 c | 0.12 d | 0.37 c | 0.026 | <0.001 |
| MUFA | 0.69 a | 0.67 ab | 0.58 b | 0.44 c | 0.58 b | 0.025 | <0.001 |
| PUFA | 0.77 ab | 0.78 a | 0.67 b | 0.48 c | 0.56 c | 0.026 | <0.001 |
| Upper Ileum 3 | |||||||
| TFA | 0.74 a | 0.68 a | 0.53 b | 0.35 c | 0.51 b | 0.027 | <0.001 |
| SFA | 0.65 a | 0.49 b | 0.32 c | 0.12 d | 0.36 bc | 0.033 | <0.001 |
| MUFA | 0.73 a | 0.69 a | 0.61 ab | 0.52 b | 0.62 ab | 0.031 | <0.001 |
| PUFA | 0.78 a | 0.78 a | 0.69 ab | 0.59 b | 0.63 b | 0.028 | <0.001 |
| Lower Ileum 3 | |||||||
| TFA | 0.79 a | 0.73 a | 0.65 b | 0.41 c | 0.62 b | 0.020 | <0.001 |
| SFA | 0.69 a | 0.55 b | 0.47 b | 0.18 c | 0.49 b | 0.022 | <0.001 |
| MUFA | 0.76 a | 0.74 a | 0.71 a | 0.56 b | 0.74 a | 0.025 | <0.001 |
| PUFA | 0.83 a | 0.83 a | 0.79 a | 0.68 b | 0.75 ab | 0.028 | 0.002 |
| Excreta 3 | |||||||
| TFA | 0.80 a | 0.73 ab | 0.63 bc | 0.47 d | 0.60 c | 0.023 | <0.001 |
| SFA | 0.64 a | 0.53 b | 0.43 bc | 0.23 d | 0.39 c | 0.025 | <0.001 |
| MUFA | 0.79 a | 0.77 ab | 0.74 ab | 0.68 b | 0.73 ab | 0.021 | 0.018 |
| PUFA | 0.85 a | 0.81 a | 0.75 ab | 0.67 b | 0.80 a | 0.031 | 0.004 |
| Item | Dietary Treatments 1 | SEM 4 | p-Value | ||||
|---|---|---|---|---|---|---|---|
| S6 | S4-PA2 | S2-PA4 | PA6 | P6 | |||
| AME, kcal/kg 2 | 3364 a | 3379 a | 3212 bc | 3121 c | 3279 ab | 32.48 | <0.001 |
| Upper Jejunum 3 | |||||||
| TFA | 0.48 ab | 0.53 a | 0.32 ab | 0.29 b | 0.51 a | 0.052 | 0.005 |
| SFA | 0.21 b | 0.46 ab | 0.26 ab | 0.25 ab | 0.48 a | 0.062 | 0.009 |
| MUFA | 0.60 ab | 0.66 ab | 0.52 ab | 0.50 b | 0.67 a | 0.042 | 0.015 |
| PUFA | 0.51a | 0.49a | 0.20bc | 0.06c | 0.34ab | 0.063 | <0.001 |
| Lower Jejunum 3 | |||||||
| TFA | 0.81 a | 0.78 a | 0.65 b | 0.62 b | 0.69 b | 0.021 | <0.001 |
| SFA | 0.73 ab | 0.76 a | 0.59 c | 0.55 c | 0.64 bc | 0.031 | <0.001 |
| MUFA | 0.85 a | 0.84 a | 0.76 b | 0.77 b | 0.83 a | 0.016 | <0.001 |
| PUFA | 0.81 a | 0.76 a | 0.62 b | 0.54 b | 0.56 b | 0.031 | <0.001 |
| Upper Ileum 3 | |||||||
| TFA | 0.89 a | 0.89 a | 0.77 bc | 0.72 c | 0.82 b | 0.015 | <0.001 |
| SFA | 0.85 a | 0.86 a | 0.67 bc | 0.61 c | 0.77 ab | 0.030 | <0.001 |
| MUFA | 0.91 a | 0.91 a | 0.85 b | 0.84 b | 0.91 a | 0.008 | <0.001 |
| PUFA | 0.90 a | 0.90 a | 0.82 b | 0.77 c | 0.78 c | 0.008 | <0.001 |
| Lower Ileum 3 | |||||||
| TFA | 0.92 a | 0.92 a | 0.82 b | 0.76 c | 0.84 b | 0.010 | <0.001 |
| SFA | 0.90 a | 0.90 a | 0.71 c | 0.64 d | 0.78 b | 0.017 | <0.001 |
| MUFA | 0.93 a | 0.93 a | 0.88 b | 0.88 b | 0.93 a | 0.006 | <0.001 |
| PUFA | 0.93 a | 0.93 a | 0.87 b | 0.85 b | 0.83 b | 0.013 | <0.001 |
| Excreta 3 | |||||||
| TFA | 0.93 a | 0.92 a | 0.84 b | 0.72 c | 0.84 b | 0.009 | <0.001 |
| SFA | 0.87 a | 0.87 a | 0.76 b | 0.59 c | 0.77 b | 0.016 | <0.001 |
| MUFA | 0.93 a | 0.93 a | 0.89 b | 0.85 c | 0.92 a | 0.005 | <0.001 |
| PUFA | 0.94 a | 0.94 a | 0.90 b | 0.82 c | 0.84 c | 0.009 | <0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jimenez-Moya, B.; Barroeta, A.C.; Tres, A.; Soler, M.D.; Sala, R. Soybean Oil Replacement by Palm Fatty Acid Distillate in Broiler Chicken Diets: Fat Digestibility and Lipid-Class Content along the Intestinal Tract. Animals 2021, 11, 1035. https://doi.org/10.3390/ani11041035
Jimenez-Moya B, Barroeta AC, Tres A, Soler MD, Sala R. Soybean Oil Replacement by Palm Fatty Acid Distillate in Broiler Chicken Diets: Fat Digestibility and Lipid-Class Content along the Intestinal Tract. Animals. 2021; 11(4):1035. https://doi.org/10.3390/ani11041035
Chicago/Turabian StyleJimenez-Moya, Beatriz, Ana C. Barroeta, Alba Tres, María Dolores Soler, and Roser Sala. 2021. "Soybean Oil Replacement by Palm Fatty Acid Distillate in Broiler Chicken Diets: Fat Digestibility and Lipid-Class Content along the Intestinal Tract" Animals 11, no. 4: 1035. https://doi.org/10.3390/ani11041035
APA StyleJimenez-Moya, B., Barroeta, A. C., Tres, A., Soler, M. D., & Sala, R. (2021). Soybean Oil Replacement by Palm Fatty Acid Distillate in Broiler Chicken Diets: Fat Digestibility and Lipid-Class Content along the Intestinal Tract. Animals, 11(4), 1035. https://doi.org/10.3390/ani11041035







