Long-Term Immunocastration Protocols Successfully Reduce Testicles’ Size in Bísaro Pigs
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
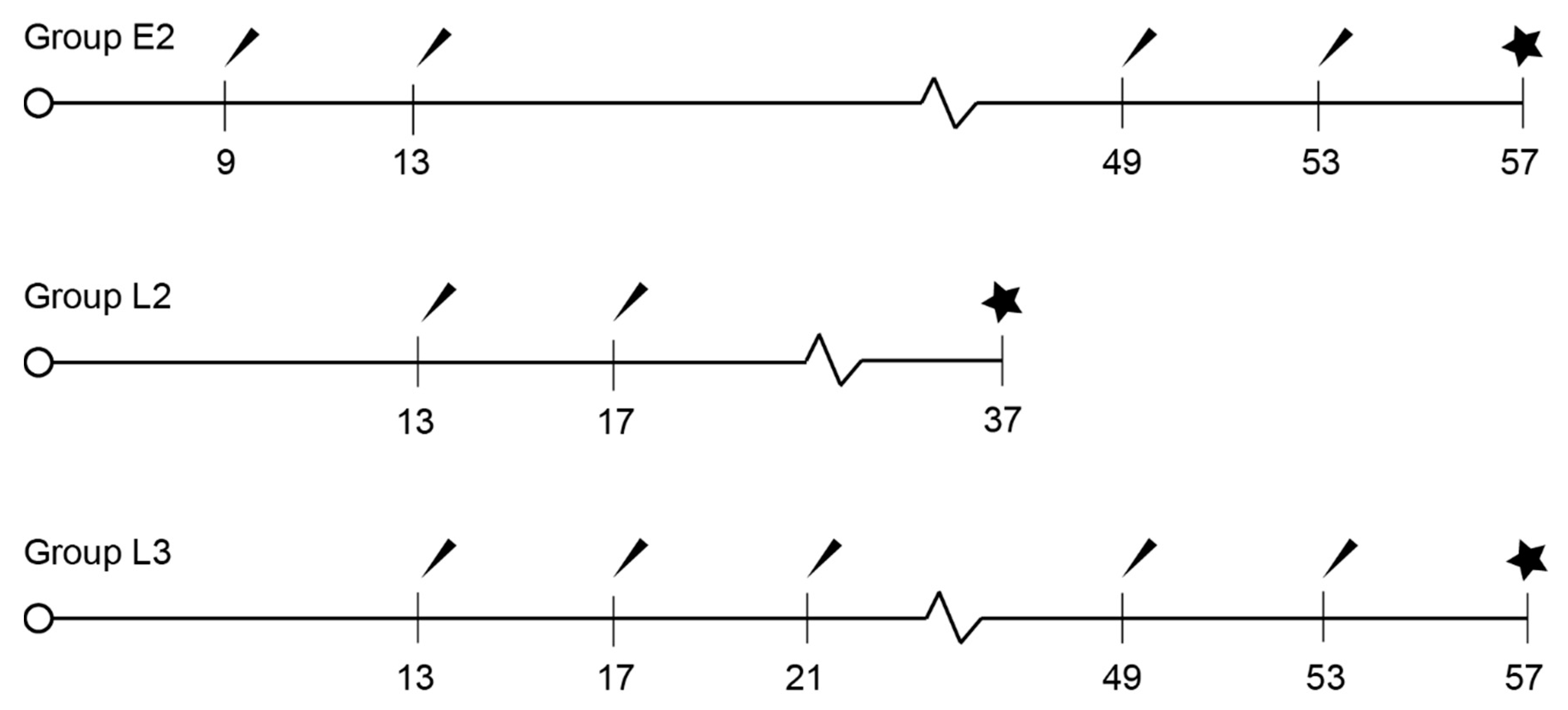
2.1. Vaccination Protocols
2.2. Measurements
2.3. Statistical Analysis
3. Results
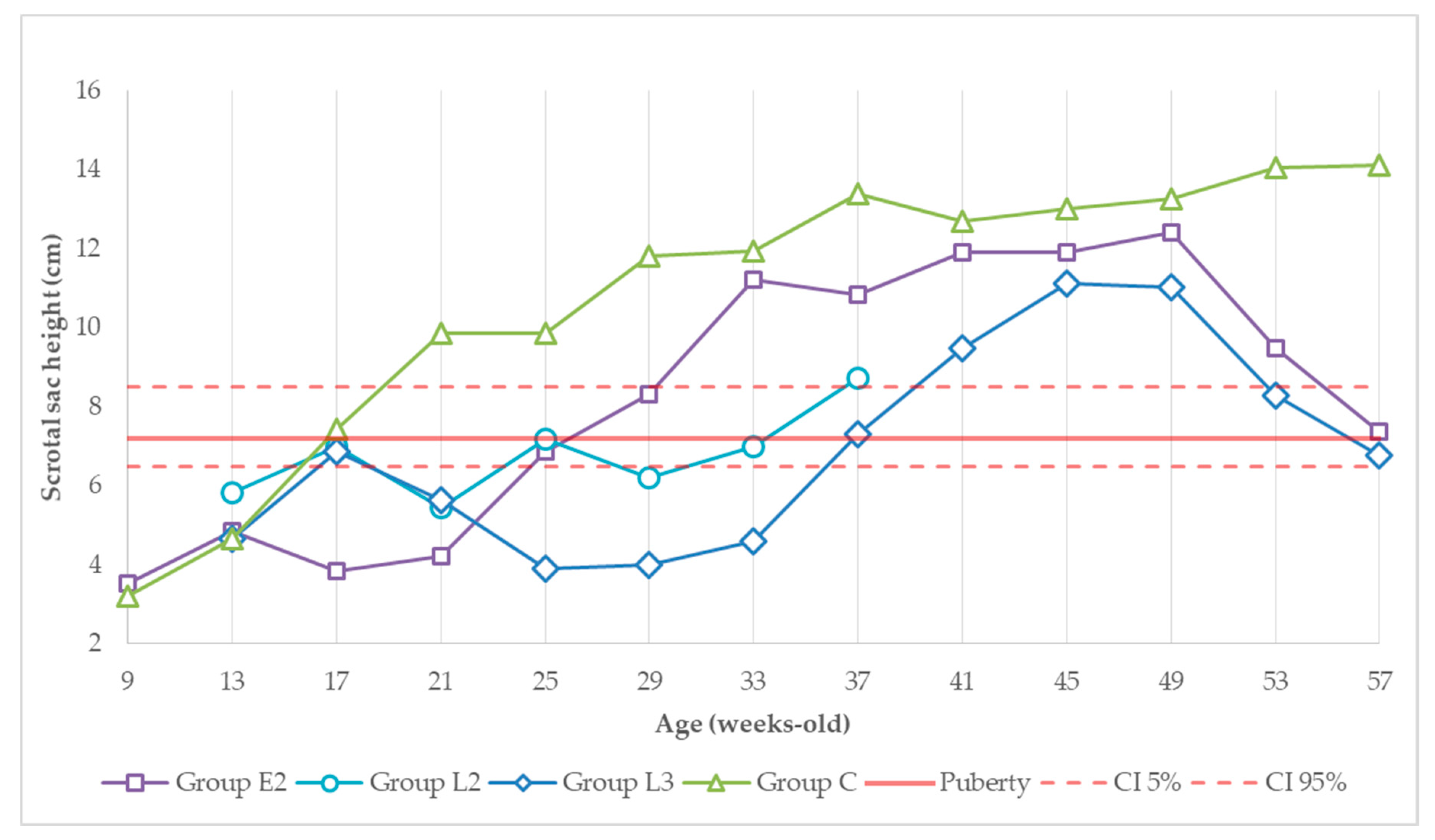
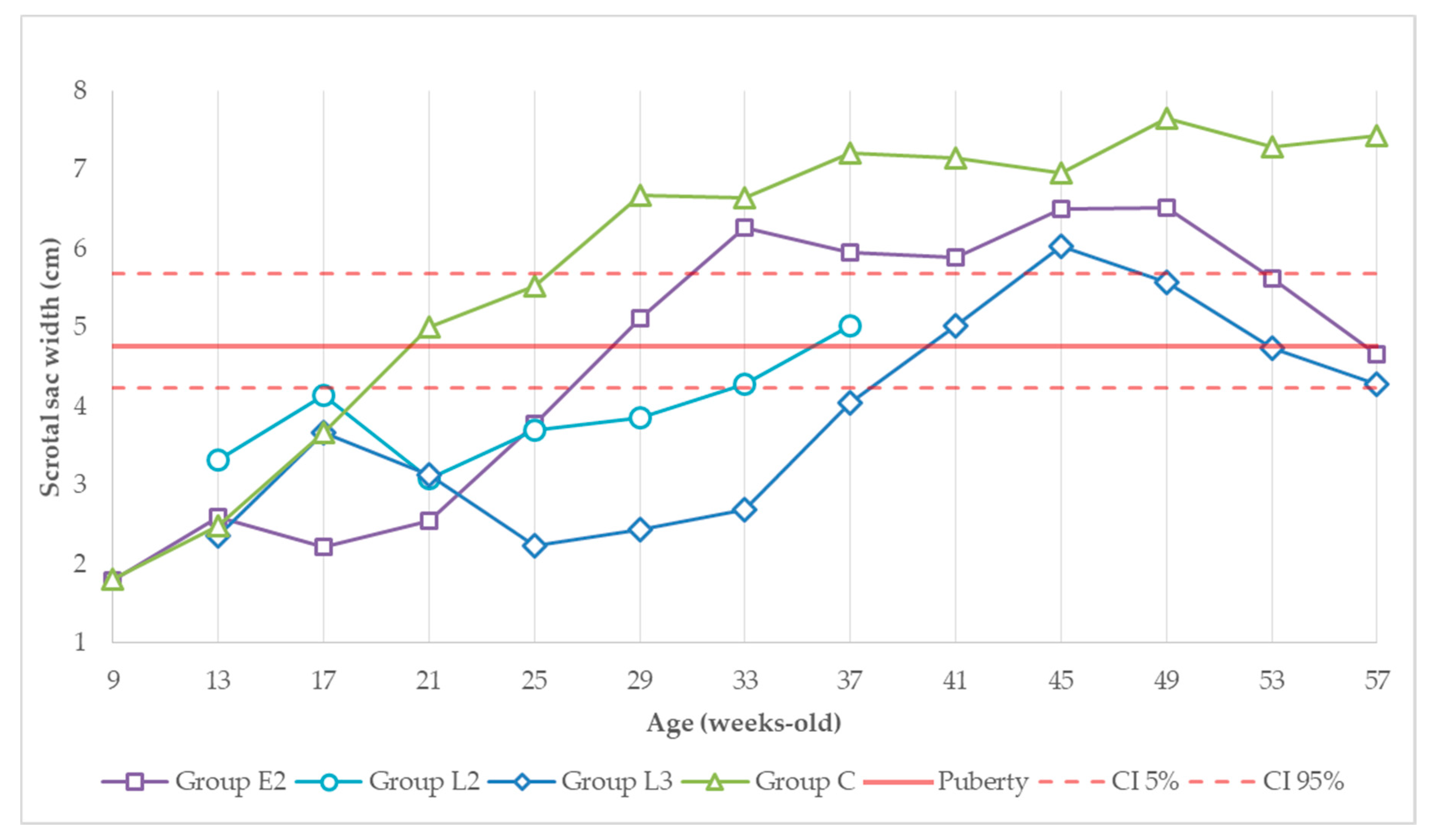
3.1. Absolute Dimensions of the Scrotal Sacs
3.2. Variation of Scrotal Dimensions
3.3. Post-Mortem Morphometry
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- European Comission. European Declaration on Alternatives to Surgical Castration of Pigs; Affairs Directorate G—Veterinary and International Affairs: Brussels, Belgium, 2010. [Google Scholar]
- De Briyne, N.; Berg, C.; Blaha, T.; Temple, D. Pig castration: Will the EU manage to ban pig castration by 2018? Porc. Health Manag. 2016, 2, 1–11. [Google Scholar] [CrossRef]
- Prunier, A.; Bonneau, M.; von Borell, E.H.; Cinotti, S.; Gunn, M.; Fredriksen, B.; Giersing, M.; Morton, D.B.; Tuyttens, F.A.M.; Velarde, A. A review of the welfare consequences of surgical castration in piglets and the evaluation of non-surgical methods. Anim. Welf. 2006, 15, 277–289. [Google Scholar]
- Brunius, C.; Zamaratskaia, G.; Andersson, K.; Chen, G.; Norrby, M.; Madej, A.; Lundstrom, K. Early immunocastration of male pigs with Improvac®—Effect on boar taint, hormones and reproductive organs. Vaccine 2011, 29, 9514–9520. [Google Scholar] [CrossRef] [PubMed]
- Von Borell, E.; Baumgartner, J.; Giersing, M.; Jaggin, N.; Prunier, A.; Tuyttens, F.A.; Edwards, S.A. Animal welfare implications of surgical castration and its alternatives in pigs. Animal 2009, 3, 1488–1496. [Google Scholar] [CrossRef]
- Fredriksen, B.; Furnols, M.F.I.; Lundstrom, K.; Migdal, W.; Prunier, A.; Tuyttens, F.A.M.; Bonneau, M. Practice on castration of piglets in europe. Animal 2009, 3, 1480–1487. [Google Scholar] [CrossRef]
- Tomasevic, I.; Bahelka, I.; Čandek-Potokar, M.; Čítek, J.; Djekić, I.; Djurkin Kušec, I.; Getya, A.; Guerrero, L.; Iordăchescu, G.; Ivanova, S.; et al. Attitudes and beliefs of eastern european consumers towards piglet castration and meat from castrated pigs. Meat Sci. 2020, 160. [Google Scholar] [CrossRef]
- Aluwe, M.; Tuyttens, F.A.; Millet, S. Field experience with surgical castration with anaesthesia, analgesia, immunocastration and production of entire male pigs: Performance, carcass traits and boar taint prevalence. Animal 2015, 9, 500–508. [Google Scholar] [CrossRef]
- Backus, G.; Støier, S.; Courat, M.; Bonneau, M.; Higuera, M. First Progress Report from the European Declaration on Alternatives to Surgical Castration of Pigs; Affairs Directorate G—Veterinary and International Affairs: Brussels, Belgium, 2014. [Google Scholar]
- Hennessy, D. Improvac® mode of action. In Technical Bulletin; Pfizer Animal Health: Parkvile, Australia, 2008. [Google Scholar]
- Zamaratskaia, G.; Rydhmer, L.; Andersson, H.K.; Chen, G.; Lowagie, S.; Andersson, K.; Lundström, K. Long-term effect of vaccination against gonadotropin-releasing hormone, using Improvac™, on hormonal profile and behaviour of male pigs. Anim. Reprod. Sci. 2008, 108, 37–48. [Google Scholar] [CrossRef]
- Zamaratskaia, G.; Andersson, H.K.; Chen, G.; Andersson, K.; Madej, A.; Lundstrom, K. Effect of a gonadotropin-releasing hormone vaccine (Improvac) on steroid hormones, boar taint compounds and performance in entire male pigs. Reprod. Domest. Anim. 2008, 43, 351–359. [Google Scholar] [CrossRef]
- Zamaratskaia, G.; Squires, E.J. Biochemical, nutritional and genetic effects on boar taint in entire male pigs. Animal 2009, 3, 1508–1521. [Google Scholar] [CrossRef]
- Han, X.; Meng, F.; Cao, X.; Du, X.; Bu, G.; Kong, F.; Huang, A.; Zeng, X. Fsh promotes fat accumulation by activating pparγ signaling in surgically castrated, but not immunocastrated, male pigs. Theriogenology 2021, 160, 10–17. [Google Scholar] [CrossRef]
- Hernández-García, F.I.; Izquierdo, M.; Del Rosario, A.I.; Montero, A.; Pérez, M.A.; García-Gudiño, J.; Garrido, N. Adaptation of immunocastration treatment to montanera system for male iberian pigs: Effects on reproductive organs and carcass traits. Arch. de Zootec. 2018, 67, 97–100. [Google Scholar] [CrossRef]
- Martinez-Macipe, M.; Rodríguez, P.; Izquierdo, M.; Gispert, M.; Manteca, X.; Mainau, E.; Hernández, F.I.; Claret, A.; Guerrero, L.; Dalmau, A. Comparison of meat quality parameters in surgical castrated versus vaccinated against gonadotrophin-releasing factor male and female iberian pigs reared in free-ranging conditions. Meat Sci. 2016, 111, 116–121. [Google Scholar] [CrossRef]
- Pasquale, J.D.; Vecchio, Y.; Martelli, G.; Sardi, L. Health risk perception, consumption intention, and willingness to pay for pig products obtained by immunocastration. Animals 2020, 10, 1548. [Google Scholar] [CrossRef] [PubMed]
- Vanhonacker, F.; Verbeke, W. Consumer response to the possible use of a vaccine method to control boar taint v. Physical piglet castration with anaesthesia: A quantitative study in four european countries. Animal 2011, 5, 1107–1118. [Google Scholar] [CrossRef]
- Daza, A.; Latorre, M.A.; Olivares, A.; Bote, C.J.L. The effects of male and female immunocastration on growth performances and carcass and meat quality of pigs intended for dry-cured ham production: A preliminary study. Livest. Sci. 2016, 190, 20–26. [Google Scholar] [CrossRef]
- Aluwe, M.; Degezelle, I.; Depuydt, L.; Fremaut, D.; Van den Broeke, A.; Millet, S. Immunocastrated male pigs: Effect of 4 v. 6 weeks time post second injection on performance, carcass quality and meat quality. Animal 2016, 10, 1466–1473. [Google Scholar] [CrossRef] [PubMed]
- Paixão, G.; Esteves, A.; Payan-Carreira, R. Characterization of a non-industrial pig production system: The case of bísaro breed. R. Bras. Zootec. 2018, 47. [Google Scholar] [CrossRef]
- Ugwu, S.O.C.; Onyimonyi, A.E.; Foleng, H. Testicular development and relationship between body weight, testis size and sperm output in tropical boars. Afr. J. Biotechnol. 2009, 8, 1165–1169. [Google Scholar]
- Bekaert, K.M.; Aluwé, M.; Millet, S.; Goethals, K.; Nijs, G.; Isebaert, S.; De Brabander, D.L.; Verheyden, K.; De Brabander, H.F.; Vanhaecke, L.; et al. Predicting the likelihood of developing boar taint: Early physical indicators in entire male pigs. Meat Sci. 2012, 92, 382–385. [Google Scholar] [CrossRef]
- Font-i-Furnols, M.; Carabus, A.; Munoz, I.; Candek-Potokar, M.; Gispert, M. Evolution of testes characteristics in entire and immunocastrated male pigs from 30 to 120 kg live weight as assessed by computed tomography with perspective on boar taint. Meat Sci. 2016, 116, 8–15. [Google Scholar] [CrossRef]
- Stojanovic, S.; Uscebrka, G.; Zikic, D.; Stukelj, M. Histological and morphometric examination of the testes of boars and male pigs immunocastrated with Improvac®. Acta Sci. Vet. 2017, 45. [Google Scholar] [CrossRef]
- Lugar, D.W.; Rhoads, M.L.; Clark-Deener, S.G.; Callahan, S.R.; Revercomb, A.K.; Prusa, K.J.; Estienne, M.J. Immunological castration temporarily reduces testis size and function without long-term effects on libido and sperm quality in boars. Animal 2017, 11, 643–649. [Google Scholar] [CrossRef] [PubMed]
- Gogic, M.; Radovic, C.; Candek-Potokar, M.; Petrovic, M.; Radojkovic, D.; Parunovic, N.; Savic, R. Effect of immunocastration on sex glands of male mangulica (swallow-bellied mangalitsa) pigs. Braz. J. Vet. Res. Anim. Sci. 2019, 48. [Google Scholar] [CrossRef]
- Mitjana, O.; Bonastre, C.; Tejedor, M.T.; Garza, L.; Latorre, M.A.; Moreno, B.; Falceto, M.V. Immuno-castration of female and male pigs with anti-gonadotrophin releasing hormone vaccine: Morphometric, histopathological and functional studies of the reproductive system. Anim. Reprod. Sci. 2020, 221, 106599. [Google Scholar] [CrossRef]
- Paixao, G.; Esteves, A.; Payan-Carreira, R.; Carolino, N. Demographic structure and genetic diversity of the endangered bísaro pig: Evolution and current status. Czech. J. Anim. Sci. 2018, 63, 452–461. [Google Scholar] [CrossRef]
- Paixão, G.; Martins, Â.; Esteves, A.; Payan-Carreira, R.; Carolino, N. Genetic parameters for reproductive, longevity and lifetime production traits in bísaro pigs. Livest. Sci. 2019, 225, 129–134. [Google Scholar] [CrossRef]
- Martins, J.; Fialho, R.; Albuquerque, A.; Neves, J.; Freitas, A.; Nunes, J.; Charneca, R. Portuguese local pig breeds: Genotype effects on meat and fat quality traits. Animals 2020, 10, 905. [Google Scholar] [CrossRef]
- Paixão, G.; Esteves, A.; Carolino, N.; dos Anjos Pires, M.; Payan-Carreira, R. Evaluation of gonadal macroscopic and microscopic morphometry reveals precocious puberty in bísaro pig. Reprod. Domest. Anim. 2020, 55, 1706–1713. [Google Scholar] [CrossRef] [PubMed]
- European Union. Council directive 2008/120/ec of 18 december 2008 laying down minimum standards for the protection of pigs. Off. J. Eur. Union 2009, 47, 5–13. [Google Scholar]
- Davis, D.L.; Hines, R.H. Scrotal measurements and visual scores of boar testicle size correlated with testicle weight. Kans. Agric. Exp. Stn. Res. Rep. 1977, 10, 44–45. [Google Scholar] [CrossRef]
- Paixão, G. Immunocastration protocol testing—In vivo and post-mortem measurements. Mendeley Data 2021. [Google Scholar] [CrossRef]
- Sladek, Z.; Prudikova, M.; Knoll, A.; Kulich, P.; Steinhauserova, I.; Borilova, G. Effect of early immunocastration on testicular histology in pigs. Vet. Med. 2018, 63, 18–27. [Google Scholar] [CrossRef]
- Hernández-García, F.; Duarte, J.L.; Pérez, M.A.; Raboso, C.; del Rosario, A.I.; Izquierdo, M. Successful long-term pre-pubertal immunocastration of purebred Iberian gilts reared in extensive systems. In Proceedings of the 8th International Symposium on the Mediterranean Pig, Ljubljana, Slovenia, 10–12 October 2013; pp. 123–126. [Google Scholar]
- Bailly-Chouriberry, L.; Loup, B.; Popot, M.-A.; Dreau, M.-L.; Garcia, P.; Bruyas, J.-F.; Bonnaire, Y. Two complementary methods to control gonadotropin-releasing hormone vaccination (Improvac®) misuse in horseracing: Enzyme-linked immunosorbent assay test in plasma and steroidomics in urine. Drug Test. Anal. 2017, 9, 1432–1440. [Google Scholar] [CrossRef] [PubMed]
| Weeks after FI | Scrotal Sac Height | Scrotal Sac Width | ||||||
|---|---|---|---|---|---|---|---|---|
| Grp E2 | Grp L2 | Grp L3 | p-Value | Grp E2 | Grp L2 | Grp L3 | p-Value | |
| 4 | 0.38 | 0.20 | 0.48 | 0.020 | 0.47 | 0.26 | 0.61 | 0.216 |
| 8 | −0.21 | −0.22 | −0.17 | 0.714 | −0.14 | −0.26 | −0.11 | 0.053 |
| 12 | 0.08 b | 0.33 c | −0.29 a | 0.001 | 0.13 a | 0.23 a | −0.26 b | <0.001 |
| 16 | 0.69 a | −0.13 b | 0.04 b | 0.005 | 0.53 | 0.04 | 0.11 | 0.009 |
| 20 | 0.26 | 0.16 | 0.24 | 0.674 | 0.37 | 0.09 | 0.17 | 0.073 |
| 28 | 0.51 | 0.21 | 1.20 | 0.020 | 0.35 | 0.19 | 1.00 | 0.039 |
| 32 | 0.07 | - | 0.18 | 0.186 | −0.06 | - | 0.21 | 0.001 |
| 36 | 0.00 | - | 0.00 | 0.710 | 0.11 | - | −0.07 | 0.001 |
| 38 | 0.04 | - | - | 0.00 | - | - | ||
| Second Vaccination Cycle | ||||||||
| 4 | −0.24 | - | −0.23 | 0.934 | −0.13 | - | −0.14 | 0.804 |
| 8 | −0.22 | - | −0.17 | 0.509 | −0.17 | - | −0.09 | 0.186 |
| Item | Group C | Group E2 | Group L2 | Group L3 | p-Value 1 |
|---|---|---|---|---|---|
| n = 11 (57w) | n = 8 (57w) | n = 6 (37w) | n = 11 (57w) | ||
| Carcass weight (kg) | 132.6 ± 4.7 | 123.1 ± 6.1 | 113.0 ± 4.4 | 117.2 ± 3.8 | 0.412 |
| Aggregated weight (g) | 829.7 ± 42.5 | 259.6 ± 45.9 | 375.7 ± 48.2 | 227.3 ± 38.3 | 0.103 |
| Testis weight (g) | 314.4 ± 17.1 | 85.6 ± 18.5 | 140.0 ± 19.5 | 77.0 ± 15.5 | 0.065 |
| Epididymal weight (g) | 10.5 ± 4.6 | 44.3 ± 4.9 | 42.6 ± 5.2 | 36.6 ± 4.1 | 0.741 |
| Testis length (cm) | 12.4 ± 0.4 | 8.0 ± 0.5 | 8.5 ± 0.5 | 7.8 ± 0.4 | 0.320 |
| Testis width (cm) | 7.7 ± 0.3 | 5.9 ± 0.3 | 4.7 ± 0.3 | 5.6 ± 0.2 | 0.132 |
| Epididimal length (cm) | 20.0 ± 0.7 | 14.2 ± 0.7 | 16.0 ± 0.7 | 13.7 ± 0.6 | 0.133 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Paixão, G.; Fontela, S.B.; Marques, J.; Esteves, A.; Charneca, R.; Payan-Carreira, R. Long-Term Immunocastration Protocols Successfully Reduce Testicles’ Size in Bísaro Pigs. Animals 2021, 11, 632. https://doi.org/10.3390/ani11030632
Paixão G, Fontela SB, Marques J, Esteves A, Charneca R, Payan-Carreira R. Long-Term Immunocastration Protocols Successfully Reduce Testicles’ Size in Bísaro Pigs. Animals. 2021; 11(3):632. https://doi.org/10.3390/ani11030632
Chicago/Turabian StylePaixão, Gustavo, Sofia Botelho Fontela, Jorge Marques, Alexandra Esteves, Rui Charneca, and Rita Payan-Carreira. 2021. "Long-Term Immunocastration Protocols Successfully Reduce Testicles’ Size in Bísaro Pigs" Animals 11, no. 3: 632. https://doi.org/10.3390/ani11030632
APA StylePaixão, G., Fontela, S. B., Marques, J., Esteves, A., Charneca, R., & Payan-Carreira, R. (2021). Long-Term Immunocastration Protocols Successfully Reduce Testicles’ Size in Bísaro Pigs. Animals, 11(3), 632. https://doi.org/10.3390/ani11030632







